Abstract
This systematic review provides a high-quality, comprehensive summary of recommendations on hypertension (HT) and type 2 diabetes mellitus (T2DM), accentuating patient blood pressure, HbA1c levels, patterns of drug treatment, management, and screening of these diseases. The overall objective of the review is to support adapting existing clinical practice guidelines in Indonesia, Vietnam, and Myanmar. The database PubMed and the web search engines Google and Google Scholar were searched from October to December 2019 for evidence-based guidelines covering the overall disease management in Europe, the United States of America, and low and middle-income countries (Indonesia, Vietnam, and Myanmar—IVM later on). Nine studies were selected for the review, seven concerning HT and five T2DM. Guidelines in IVM and Europe identified HT as increased blood pressure (BP; ≥140/90 mmHg). IVM guidelines also recommended commencing drug treatment if lifestyle interventions were not successful. Four international HT guidelines recommended monitoring BP every few months, and the other three guidelines gave recommendations based on the patient’s current BP levels. All five T2DM guidelines recommended target HbA1c levels below 7%–6.5%, but only IVM guidelines included re-examination every 3–6 months. Metformin was recommended as the first choice of medical treatment, if not contraindicated. Amid the guidelines’ recommendations, there were no major variations in the Class of recommendation and Level of evidence (except IVM guidelines where COR and LOE were missing). Revision and completion of IVM guidelines by this grading system would enhance evidence-based and informed decisions in clinical care.
Introduction
Non-communicable diseases (NCDs) present a major burden on health, healthcare systems, and development globally, as they cause premature deaths, thus economically affecting nations and their individuals.1,2 These diseases, particularly cardiovascular diseases, chronic respiratory diseases, diabetes mellitus, and cancer, are leading causes of mortality in the South-East Asia Region, with an estimated 9 million deaths each year. 3 Key elements in decreasing the occurrence of NCDs are early detection, screening, and treatment. 2 The most effective way to manage non-communicable diseases is to reduce the risk factors largely attributable to them, such as hypertension, diabetes mellitus, and stroke.4,5
The review was developed as a part of the EU Horizon 2020 SUNI-SEA project, which is being implemented in Indonesia, Vietnam, and Myanmar (IVM). Scaling-Up Noncommunicable Diseases Interventions in South-East Asia (SUNI-SEA) is an international project with the goal of verifying effective approaches to non-communicable diseases, extending evidence-based HT and T2DM prevention and treatment programs, and strengthening the provision for HT and T2DM prevention and management services. The project aims to provide evidence to achieve sustainable development goals, all based on experiences in Southeast Asian countries. 6
Hypertension (HT) is highly prevalent throughout the world, with the scale of increase of NCDs particularly observable in Southeast Asia. 7 Studies currently report the prevalence of HT to be around 35% and is accountable for approximately 1.5 million deaths annually.8,9 A much higher percentage is attributed to the lack of awareness and control of raised blood pressure for all people in the Southeast Asia Region, being nearly 50%. 7 In population-based studies, the prevalence of HT in Malaysia in 2011 was estimated at 43.5% (≥30 years), in Myanmar in 2009 at 30.1% (15–64 years), Sri Lanka between 2005 and 2006 at 23.7% (≥18 years) and in Vietnam from 2002 to 2008 at 25.1% (≥25 years). The prevalence of HT in Indonesia has not been thoroughly explored due to the lack of data on adults (15 years and older). 7
The Southeast Asia Region faces a diabetes epidemic, too. 10 Of the diabetic population, more than 85% suffer from type 2 diabetes mellitus (T2DM). 11 The prevalence of diabetes in South Asians is four times higher than in any other ethnic group owing to the individual susceptibility to T2DM. 12 Nevertheless, roughly 57% of the cases are undiagnosed. 13 The estimates of the prevalence of diabetes in adults range from 4.0% in Nepal to 8.8% in India. An increasing proportion of obese and overweight children and adolescents have led to an increased risk of T2DM. 14 Of particular concern is the additional number of deaths and the economic burden due to diabetes. Based on the latest statistics, patients pay between 43 and 870 USD to treat the disease and its complications. 11
Both HT and T2DM are preventable conditions and are commonly associated with an unhealthy lifestyle, a lack of physical activity, and the harmful use of tobacco and alcohol. 15 Therefore, it is imperative to produce and use evidence-based recommendations based on the best local and international evidence. The adaptation of recognized international recommendations from clinical practice guidelines for the specific environment in communities in Southeast Asian countries is addressed by international teams of the project SUNI-SEA with the aim of developing interventions tackling NCDs with the necessity of adjusting to local needs and requirements.
The overall objective of this systematic review is to propose modifications in existing guidelines in Indonesia, Vietnam, and Myanmar based on identified differences in the diagnosis, management, and treatment of HT and T2DM from current international evidence-based guidelines’ recommendations for these non-communicable diseases. The systematic review was conducted as a part of an EU-funded SUNI-SEA project. No ethical constraints were identified.
Methodology
A computerized literature search was initiated on publications from January 1, 2010 to December 31, 2019 and executed primarily within the PubMed database. This database offers free full-text biomedical and life sciences journals. The search was conducted from October to December 2019, using a combination of keywords and relevant subject headings related to HT, T2DM, guidelines, and recommendations. The search was complemented with browsing the web for determining pertinent publications (Table 1). The guidelines were searched in close collaboration with all SUNI-SEA project partners.
Search algorithms and key words.
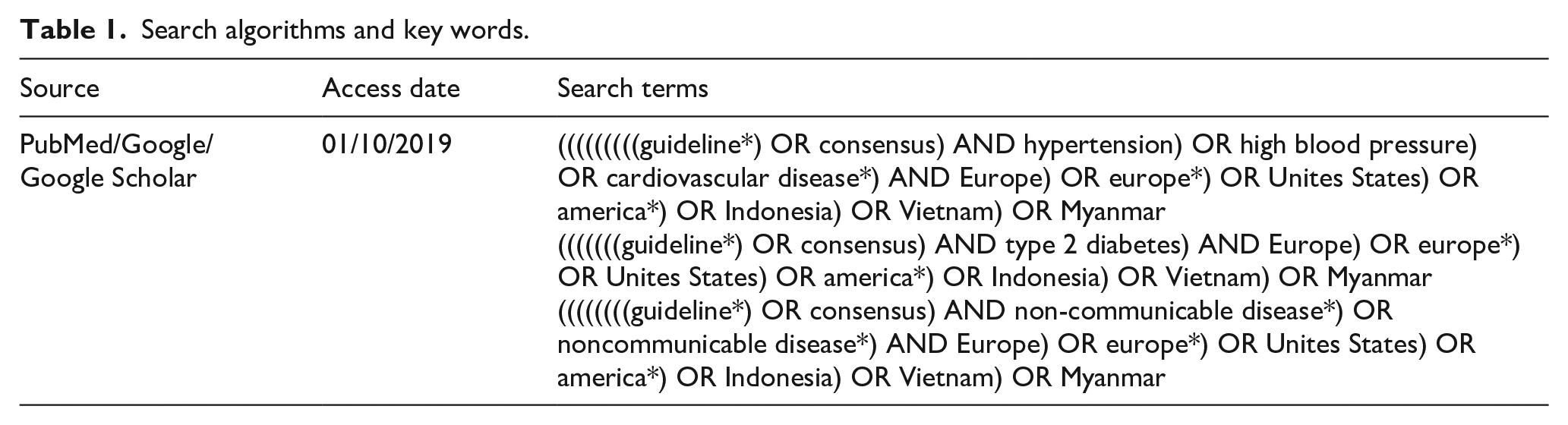
Selection criteria for the systematic review included guidelines for two non-communicable diseases—HT and T2DM—containing the Class of recommendations and Level of evidence, and that the publication originated either from Europe, the United States of America, or low and middle-income countries (focusing on IVM). Attempts were made to recognize and translate non-English language papers. Guideline publications only qualified for the systematic review if they had been fully published and not older than 10 years.
The authors of this article individually selected suitable publications based on the criteria and extracted all necessary data. All discrepancies were discussed and solved. The total number of selected guidelines concerning non-communicable disease was 9 (HT—7; T2DM—5), with some of the included guidelines covering both topics. The oldest guideline included in this review was published in 2012. In the results, we present the recommendations for HT and T2DM separately, thus guidelines 1–5 and 8–9 for HT, and guidelines 1 and 4–7 for T2DM. Table 2 presents the characteristics of the reviewed studies.
Characteristics of reviewed guidelines.

Subsequently, the following information, targeted at the prevention and treatment of HT and T2DM, was obtained from each guideline: guideline authors, year of publication, the Class of recommendations, and Level of evidence.
A total of 367 publications, including guidelines, recommendations, or their reports, were identified. After removing duplicates, 279 papers were screened, and from those, 258 were excluded. Overall, 21 publications were screened for eligibility, with 9 included in the qualitative synthesis (Figure 1).

Systematic reviews and meta-analyses (PRISMA) flowchart of the study selection process.
A consensus was reached after the discussion of the proper method used to compare findings. For this purpose, the guidelines were group stratified by the type of procedure: treatment process, pharmacotherapy, management, and screening. Recommendations targeted at NCDs in children and adolescents (<18 years), pregnant women, other ethnic groups, therapeutic strategies, and secondary prevention for associated comorbidities were excluded from the systematic review.
The study used administratively collected secondary data and no ethics committee approval was required.
Results
The onset of HT in the European guidelines and the guidelines used in Southeast Asian countries was classified as BP ≥140/90 mmHg. In the American guidelines, it was classified at lower levels (BP ≥130/80 mmHg). The beginning of a patient’s drug treatment in the first grade of HT was recommended mostly when lifestyle interventions had failed using combination therapy. American and European guidelines also strongly recommended that the start of medication should be based on the overall cardiovascular risk of the patient. Four guidelines advised to initiate hypertension treatment with one drug, but each guideline differed by the severity of hypertension. Recommendations on systolic blood pressure targets and blood pressure (BP) monitoring differed in these guidelines, as a few of them used age stratification or severity of HT as target criteria. The measurement of BP was recommended from 1 month to 5 years, depending on the guideline and patient’s condition (Table 3).
Comparison of guidelines recommendations—hypertension.

ASCVD: atherosclerotic cardiovascular disease; DBP: diastolic blood pressure; HMOD: hypertension-mediated organ damage; SBP: systolic blood pressure; ameasurements should be made over a period of several months to achieve an acceptable definition of an individual’s “usual” BP and to decide about initiating drug treatment.
Most guidelines recommended target HbA1c levels of <7%, but only a few altered these levels by the age of the patient (Guidelines 6 and 7). The recommended blood pressure target was estimated at <140/90 mmHg in Vietnamese and Indonesian guidelines, at <140/80 mmHg in European guidelines, and the estimation based on overall CVD risk was stated in the American and WHO guidelines (used in Myanmar). Metformin, if not contraindicated, was recommended as the first choice of treatment in all guidelines. If Metformin was not tolerated, Sulfonylurea was specifically recommended only in two guidelines. Six out of the seven guidelines recommended statin therapy to reduce CV risk (Table 4).
Comparison of treatment process and pharmacotherapy—type 2 diabetes mellitus.

CVD: cardiovascular disease; HbA1c: glycated hemoglobin.
Although retinopathy, neuropathy, and increased albuminuria and eGFR levels have been identified as diabetes-specific risk enhancers that are independent of other risk factors in T2DM, in most guidelines, the frequency of examinations for these risk enhancers was not specified (Table 5).
Comparison of management and screening—type 2 diabetes mellitus.

CVD: cardiovascular disease; eGFR: estimated glomerular filtration rate; HbA1c: glycated hemoglobin.
Example considerations for addressing social determinants of health to help prevent ASCVD events.
Discussion
The use of systematic reviews for clinical practice guidelines is essential to classify, analyze, and summarize evidence-based guidance on certain topics. Conducting systematic reviews has been proven to be beneficial in various clinical areas, ranging from preclinical testing, 25 single diseases, 26 and risk factors, 27 to complex non-communicable diseases. 28 In our systematic review, aimed at describing and comparing guideline recommendations on HT and T2DM, we emphasized targeted patients’ BP and HbA1c levels, patterns of drug treatment and management, and the screening of these diseases.
Selected American guidelines classified onset of HT as BP ≥130/80 mmHg, and to initiate a patient’s drug treatment, guidelines first recommended the assessment of the patient’s overall ASCVD risk. Guidelines used in IVM and Europe identified HT at a higher BP in comparison (≥140/90 mmHg), but IVM guidelines recommended initiating drug treatment only when lifestyle interventions had failed. Only four guidelines differentiated their recommendations on the number of drugs used to initiate treatment by a patient’s degree of HT (17, 18, 23, and 24), and the pill therapy to treat HT (single-pill combination therapy) was not specified in four guidelines (16, 18, 19, and 24). Systolic BP targets were differentiated by age in only two guidelines, with higher levels for older people, and the lowest recommended diastolic BP levels were 70 mmHg. Out of the seven guidelines, four guidelines recommended BP measurements/monitoring every few months, and recommendations in three guidelines were based on a patient’s current BP levels.
All five guidelines recommended target HbA1c levels below 7%–6.5%, but only IVM guidelines included the re-examination of these levels every 3–6 months. Two guidelines had differentiated these levels by age groups. Metformin was recommended as the first choice of medical treatment, if not contraindicated, in all guidelines; however, if metformin was not tolerated, Sulfonylurea was specifically recommended as the first choice of medical treatment by only two guidelines (20 and 22). Statin therapy, to reduce CVD risk, was recommended in all guidelines except 1 (22), and blood glucose self-monitoring was recommended only in two guidelines. Screening recommendations varied remarkably among the guidelines, most recommending annual kidney disease screenings (four guidelines) and annual foot examinations (three guidelines). Patient prevention against certain communicable diseases was recommended only in two guidelines.
Among the recommendations, there were no major differences between the guidelines with regards to the Class of recommendation and Level of evidence (among the ones which included such grading). The absence of essential quality rating systems in guidelines implies the necessity of their improvement to support evidence-informed decision-making in healthcare provision. 29 The systematic review—Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach to develop a common and transparent system for grading the quality of evidence and the strength of recommendations 30 —should be used in referral guidelines for all basic services. 31 As derivation from existing guidelines and adaptation to local context may lack transparency, it is crucial to set a working group with expertise in medical content and methods, but also in the values and preferences of communities. 32 Adjustments and implementations of guidelines, on a national level, can be feasibly achieved through proceeding within methodological standards, feasible frameworks, and guides such as the GRADE Handbook.30,33,34 Pragmatic approaches are required in settings where resources are limited; thus, purposeful clinicians’ workgroups are needed to make sustained efforts for guideline improvement.35,36
As stated, the systematic review was conducted in order to propose amendments to currently used clinical practice guidelines concerning overall management of HT and T2DM in Indonesia, Vietnam, and Myanmar based on the findings; however, the possibility of their implementation is severely limited. The current situation in Myanmar precludes the implementation of the review’s findings, as the military seized power in February 2021. The country is in an unstable political environment, and the military is attempting to maintain power by shutting down all communication, whether via the internet or mobile data. Both Indonesia and Vietnam have centralized authority, which means that the government has a greater proportion of decision-making power. As a result, any modification or usage of the guidelines is strictly monitored by this authority.
Promoting and assisting the modification and implementation of these guidelines remains one of the primary goals of the SUNI-SEA project; therefore, the project team is considering a new policy with this purpose in view. Furthermore, project activities include developing a strategy for measuring the use of existing guidelines in primary practice in Vietnam.
Limitations
Although we proceeded in our systematic review according to the PRISMA statement, we were unable to uphold all items in the PRISMA checklist. Due to the heterogeneity among the American, European, and IVM guidelines, we could not properly assess the risk of bias within or across the recommendations. Five out of nine guidelines did not include the Class of recommendations, and four out of nine guidelines did not include the Level of evidence. Moreover, some of the selected recommendations were identified in the text of a guideline but these recommendations were only supported by additional studies.
Some identified guidelines in a few sections referred to additional guidelines, which were not included in this review. This content could include additional recommendations not stated in a selected guideline resulting in a biased not-applicable (NA) for certain recommendations.
As the guidelines are often robust and comprehensive in their topics, we were forced to exclude many further complications and comorbidities of HT and T2DM to retain the aimed intention of this review.
Conclusions and recommendations
We observed significant differences among guidelines being used in countries of Southeast Asia and those from Europe and the USA. Most of those evaluated did not mention important attributes, such as the Class of recommendation or the Level of evidence. Without those, the use of recommendations is feasible, but users might be less willing to use them for clinical decisions in their clinical practice. It is recommended that these guidelines are revised and completed to be used as credible sources in the manner of evidence-based medicine.
Footnotes
Author contributions
MR, JM, and PS conceptualization; JM and PS, manuscript writing and analysis; JM, PS, and PPT, methodology; RM, PPT, and MM, review and editing. All authors contributed significantly to the study and read and approved the final manuscript version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work presented received funding for APC within the Scaling-Up Noncommunicable Diseases Interventions in South-East Asia (SUNI-SEA) project funded by the European Union’s Horizon 2020 research and innovation program under grant agreement No. 825026.
Significance for public health
To support the adaptation of clinical practice guidelines in Southeast Asian countries, this systematic review offers a high-quality, thorough summary of recommendations on the management of hypertension and type 2 diabetes mellitus disease. The revision and completion of those guidelines are essential for improving their quality and usage, as it was discovered during the review that the majority of the evaluated guidelines omitted key elements like the Class of recommendation or the Level of evidence. By including these attributes, they will become a reliable source in the area of evidence-based medicine.
Availability of data and materials
The database used during the current study are available upon reasonable request from the corresponding author.
