Abstract
Background:
Multimorbidity in elderly patients is a major challenge for physicians, because of a high prevalence of and associations with many adverse outcomes. However, the mechanisms of progressing multimorbidity are not well-understood. The aim of our study was to determine if the progression of multimorbidity is influenced by health behaviour and social support and to analyse if the patients’ socio-economic status had an effect on these prognostic factors.
Methods:
The study was designed as prospective cohort study based on interviews of 158 GPs and 3189 patients randomly selected from GP records (response rate: 46.2%). Patients were aged 65–85 years at recruitment and observed in four waves of data collection (dropout rate: 41.5%). Statistical analyses of the ‘hot deck’ imputed data included multilevel mixed-effects linear regression allowing for random effects at the study centre and GP practice within study centre level.
Results:
Regarding cardiovascular and metabolic diseases, multimorbidity progressed more rapidly in patients who reported less physical activity (ß = −0.28; 95% confidence interval = −0.35 to −0.20), had more tobacco-related pack years (0.15; 0.07–0.22) and consumed less alcohol (−0.21; −0.31 to −0.12) at baseline. Multimorbidity related to psychiatric and pain-related disorders progressed more rapidly if the patients had less perceived social support (−0.31; −0.55 to −0.07) and reported less physical activity (−0.08; −0.15 to −0.02) at baseline. Education and income only slightly modified the effects of these variables.
Conclusion:
Depending on the multimorbidity cluster, different strategies should be used for slowing down the progression of multimorbidity. Changing lifestyle and increasing social support are beneficial for the entire group of elderly multimorbid patients – regardless of their socio-economic status.
Registration:
ISRCTN89818205
Introduction
Although there is no consensus on definition and operationalization, 1 most researchers agree that multimorbidity in elderly patients is a major challenge for attending physicians. 2,3 Many studies show prevalence rates of multimorbidity between 50% and more than 80% 1,4,5 as well as associations with adverse outcomes like mortality, 6–8 disability, 7–9 high use and high costs of healthcare. 10,11 Interrelations between disorders are complex 12 and clinical practice guidelines for single diseases usually do not consider co-morbidity appropriately. 13 However, in recent years, special guidelines for multimorbidity have been designed. 14
The incidence and progression of multimorbidity is influenced by age, sex, mental health disorders and socio-economic deprivation. 1,4,15 Among these factors, the socio-economic status plays a special role as it also seems to increase the risk for mental health disorders 15 whereby socially deprived patients have a higher risk for adverse outcomes even after adjusting for the disease burden. 16
So far, however, it has not been well-understood how the disease burden in patients with multimorbidity could be addressed by intervention studies. There are few longitudinal studies on this topic, information on disease severity is mostly lacking and possible risk factors, such as social support and health behaviour, which may influence the incidence or outcome of many chronic diseases, 17–20 have rarely been considered. 1,4
The aim of this study is therefore to determine if the 5-year progression of multimorbidity severity in elderly general practice patients is influenced by health behaviour and social support. In addition, we examine the effect of the socio-economic status of the patients on these prognostic factors.
Methods
Design
We analysed the data set of the MultiCare Cohort Study. The methods of this study have been described in trial registration ISRCTN89818205, the published protocol 21 and an additional paper. 22 In short, the study has been designed as a cohort study of multimorbid patients from general practice based on patient and GP interviews. The analyses presented here are based on data from baseline and three subsequent follow-up assessments between 21 July 2008 and 4 December 2013. The study was approved by the local Ethics Committee of the Medical Association of Hamburg in February 2008 and amended in November 2008 (approval no. 2881).
Patient recruitment
The patients were recruited from 158 practices in the German cities of Bonn, Düsseldorf, Frankfurt am Main, Hamburg, Jena, Leipzig, Mannheim and Munich. In every practice, we created a list of patients based on the electronic database of the GP. Patients were included in this list if they had been between 65 and 85 years old and had consulted the GP at least once within the last completed quarter (3-month period). Using random number tables, we randomly selected 50 eligible patients with multimorbidity per practice from this list and contacted them for their written informed consent. During patient recruitment, multimorbidity was defined as coexistence of at least 3 out of 29 disease categories. 21
Patients were excluded if they were not regular patients of the participating practice, if they were unable to participate in interviews (e.g. blindness or deafness) or if they were not able to speak or read German. Further exclusion criteria were residence in a nursing home, severe illness probably lethal within 3 months according to their GP, missing capacity to consent (e.g. dementia) and participation in other studies at the time of recruitment.
Study population
A total of 24,862 patients were checked for multimorbidity and the exclusion criteria described above. Of those, 7172 patients were eligible and contacted for participation; 3317 patients agreed to participate which corresponds to a response rate of 46.2%. Retrospectively, we had to exclude 128 patients, because they died or they met one or more exclusion criteria without the GP’s knowledge. Finally, 3189 patients were included in the study. Details of the sampling procedure were published in detail in another paper. 22
Participation rates and loss to follow-up are shown in Figure 1. Reasons for dropping out included that some patients did not want to be interviewed anymore or could not be contacted, for example, because they made a long holiday or had moved to another town. Additionally, several patients could not participate in interviews because of their bad health condition. Some patients dropped out only temporarily. However, there were also some patients who had died between the assessments. In total, 2746 patients (86.1% of the baseline population) completed the first, 2375 patients (74.5%) the second and 2070 patients (64.9%) the third follow-up assessment.

Participation rates in patient interviews and loss to follow-up.
Our dependent variable, the severity of multimorbidity, was assessed in GP interviews. For this reason, we could only consider patient data if GP interviews had been conducted. At baseline, all GP interviews had been concluded. However, at the first follow-up, we had to exclude 17 patients because of missing morbidity data, 199 patients were affected at the second and 205 patients at the third follow-up. Thus, the final sample size was 3189 patients at baseline, 2729 (85.6%) at the first, 2176 (68.2%) at the second and 1865 (58.5%) at the third follow-up.
Dependent variable
The dependent variable in our analyses was the patients’ morbidity assessed in GP interviews. In these interviews, we coded the morbidity in 46 diagnoses and diagnosis groups (below: ‘diagnoses’). The 46 diagnoses were selected according to prevalence and chronicity. Diagnoses were grouped together if diseases and syndromes had a close pathophysiological similarity and if ICD codes of related disorders were used ambiguously by coding physicians in clinical reality, respectively. However, seven diagnoses had not been part of the standardized GP questionnaire at baseline and four diagnoses had been missing in the standardized questionnaire at the first follow-up. These diagnoses were assessed with an open-ended interview question (‘Which additional diagnoses does this patient have?’) and standardized during data preparation.
Multimorbidity severity was assessed in a pragmatic way by a weighted disease count, which was calculated by adding the GPs’ severity rating for each diagnosis ranging from 0 = marginal to 4 = very severe. Based on results of a tetrachoric factor analysis published in a different paper, 23 we used three different disease sets for our analyses: (1) a complete set of all 46 recorded diagnoses; (2) a subset of cardiovascular and metabolic disorders; and (3) a subset of anxiety, depression, somatoform (ADSom) and pain-related disorders. The diagnoses included in each of the subsets are shown in Table 1.
Subsets of multimorbidity by sexa – results from tetrachoric factor analyses.

PAOD: peripheral arterial occlusive disease; COPD: chronic obstructive pulmonary disease; GERD: gastro-oesophageal reflux disease.
aSex-specific associations of diagnoses with a pattern are presented in italic letters.
Independent variables
The independent variables included in our analyses were collected in patient interviews. The socio-economic status was assessed by education and income. Education was described in three groups according to the international CASMIN classification: (1) inadequately completed general education, general elementary education or basic vocational qualification; (2) intermediate qualification or general maturity certificate; and (3) lower or higher tertiary education. 24 Income was reported as household net adjusted disposable income per month, which is calculated as household total net income per month divided by the equalized household size, which gives 1.0 to the householder, 0.5 to other household members aged 15 or over and 0.3 to each child aged less than 15 years. 21
Physical activities were assessed by the International Physical Activity Questionnaire (IPAQ-7) 25 and classified in three categories (i.e. low, medium and high). For lifetime tobacco smoking, we retrospectively calculated pack years. 26 Alcohol consumption was assessed by the Alcohol Use Disorders Identification Test for Subthreshold Consumption (AUDIT-C) 27 and perceived social support by the 14-item version of the ‘Fragebogen zur sozialen Unterstützung’ [questionnaire on social support] (F-SozU K14). 28 For these two instruments, a summary score was calculated.
Imputation of missing values
We performed ‘hot deck’ imputation based on Gower distance measures in order to avoid bias generated by deleting lists of cases with missing values from statistical analyses. This procedure has been described elsewhere in detail. 22 For the analyses presented here, missing values were imputed in the variables income (proportion of missing values: 12.4%), smoking behaviour (2.2%), physical activities (1.6%), social support (0.5%), alcohol consumption (0.3%) and the severity ratings of chronic diseases (proportion of missing values ranging between 0% and 6.7% with a mean rate of missing values of 1.2%). The other variables in our statistical models did not contain any missing values.
Statistical analyses
The progression of multimorbidity severity over time was depicted by the number of diagnosed diseases weighted by the severity of each disease. We analysed the association of the prognostic variables and the progression of multimorbidity severity by multilevel mixed-effects linear regression allowing for random effects at the study centre and GP practice within study centre level. We conducted separate analyses for the three disease sets described above and applied a logarithmic transformation of income before starting the analyses.
In order to determine the influence of socio-economic status on the prognostic variables, all analyses were conducted in two different statistical models, that is, (1) controlled for age, sex and the three follow-up assessments; (2) additionally adjusted for education and income. We determined by likelihood ratio test if there was a statistically significant increase in model fit (p ≤ 0.05) from model 1 to model 2.
We used complete data sets including imputed data for all inferential statistics. An α-level of 5% (i.e. p ≤ 0.05) was defined as statistically significant. All statistical tests were conducted using R version 3.2.3.
Results
On average, patients were 74.4 ± 5.2 years old at the time of recruitment and 59.3% of them were women. According to CASMIN, 62.3% of the patients had a low and 10.9% had a high education level. The patients had a mean household net adjusted disposable income of €1412 ± 704. A detailed description of the MultiCare Cohort at baseline can be found elsewhere. 22
The patients showed a high degree of perceived social support as 84.7% of the patients scored 3.5 points or higher on the five-point F-SozU K14 scale with 64.8% scoring 4.0 points or higher. The mean F-SozU score was 4.1 ± 0.7 points. Regarding health behaviour, the patients showed large differences in physical activity with a mean of 2173 ± 2468 metabolic equivalent task minutes per week. Using the IPAQ-7-categories, approximately one-third of the patients (33.0%) showed a low level and almost one-fourth (24.6%) showed a high level of physical activity.
Alcohol and tobacco consumption were prevalent in the cohort. The patients scored a mean of 2.2 ± 1.9 points on the 12-point AUDIT-C scale. Considering the thresholds of four points for men and three points for women, problem drinking was detected in 46.5% of the male and 27.8% of the female study population. Of the study population, 49.1% of the patients reported that they had never smoked. Considering the study population of 1587 smokers and ex-smokers, lifetime tobacco smoking resulted in a mean of 29.0 ± 27.4 pack years.
On average, GPs diagnosed 7.0 ± 2.5 chronic conditions per patient from our complete set of 46 diagnoses. In all waves of our data collection, the most prevalent chronic conditions were hypertension, lipid metabolism disorders and chronic lower back pain (cf. Table 2). Figure 2 shows the change in multimorbidity severity. Considering the complete set of all recorded diagnoses, the mean severity count was 11.2 at baseline and increased at every wave of our data collection. The two subsets of diseases presented a comparable change.
Prevalence of diagnosis groups by follow-up.

a Used for patient inclusion.
b Assessed with open-ended question.
c Exclusion criterion at baseline.

Change in multimorbidity severity scores over 5 years: means and standard deviations.
The results from multilevel mixed-effects linear regression are shown in Tables 3 to 5. The 5-year progression of multimorbidity severity was slowed down if patients had a higher level of physical activity, stronger perceived social support, less pack years and a higher score of problem drinking at baseline. The introduction of education and income in model 2 led to a significant increase in model fit. Although both socio-economic variables had a negative association with the progression of multimorbidity severity, they did not change the effect of age and they had only a slight effect on the coefficients of sex and the prognostic variables. Statistical significance of the independent variables of model 1 also remained unchanged.
Prognostic variables for the progression of multimorbidity severity (all diagnosis groups included).
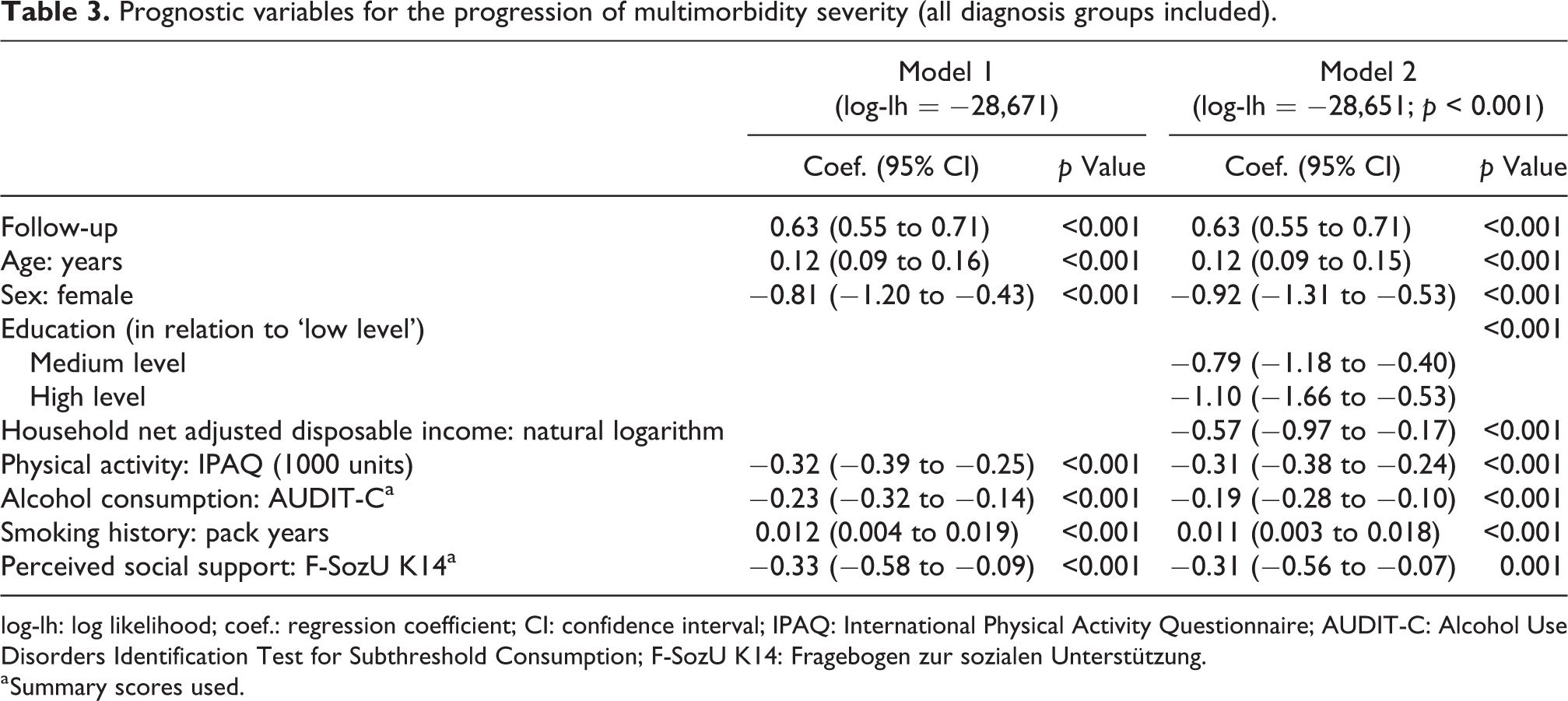
log-lh: log likelihood; coef.: regression coefficient; CI: confidence interval; IPAQ: International Physical Activity Questionnaire; AUDIT-C: Alcohol Use Disorders Identification Test for Subthreshold Consumption; F-SozU K14: Fragebogen zur sozialen Unterstützung.
a Summary scores used.
Prognostic variables for the progression of multimorbidity severity (cardiovascular and metabolic disorders).

log-lh: log likelihood; coef.: regression coefficient; CI: confidence interval; IPAQ: International Physical Activity Questionnaire; AUDIT-C: Alcohol Use Disorders Identification Test for Subthreshold Consumption; F-SozU K14: Fragebogen zur sozialen Unterstützung.
a Summary scores used.
Prognostic variables for the progression of multimorbidity severity (anxiety, depression, somatoform disorders and pain-related disorders).

log-lh: log likelihood; coef.: regression coefficient; CI: confidence interval; IPAQ: International Physical Activity Questionnaire; AUDIT-C: Alcohol Use Disorders Identification Test for Subthreshold Consumption; F-SozU K14: Fragebogen zur sozialen Unterstützung.
a Summary scores used.
The progression of multimorbidity severity within the multimorbidity cluster of cardiovascular and metabolic disorders showed similar associations. However, social support did not have a significant association with the progression of multimorbidity severity within this cluster. In contrast, the progression of multimorbidity severity of ADSom and pain-related disorders showed a significant association with perceived social support. It was also linked with physical activity, but to a much lower extent as in the other multimorbidity cluster. Smoking history and alcohol consumption did not show any effects. Additionally, education and income had no effect on the progression of multimorbidity severity in the ADSom and pain-related disorder cluster and they did not change coefficients or statistical significance of the independent variables of model 1.
Discussion
The severity of multimorbidity increased over time. Depending on the multimorbidity cluster, age, sex, perceived social support and health behaviour were associated with the change in severity. Although socio-economic status had an influence on the progression of multimorbidity in two of the three disease sets, the association of health behaviour and perceived social support with the progression of multimorbidity severity seemed to be at least partially independent of the socio-economic status.
Strengths and weaknesses
The response rate was 46.2%. A non-responder analysis showed a small selection bias concerning age and 2 of 29 diseases used for patient inclusion. 22 We excluded patients with dementia and patients residing in nursing homes. It was also not possible to interview patients who were blind, deaf or not able to speak and read German. The recruitment only took place in larger German cities, so that rural areas were not included in our study.
The results are based on a 5-year observation period. However, independent variables were represented in our statistical models at baseline only, so that changes in health behaviour or social support after baseline could not be considered. Furthermore, at the third follow-up, 41.5% of the participants had dropped out of the study. Although patients with censored data were included in our statistical models, there might be effects of selective survival.
Our measure of morbidity is based on the GPs’ severity ratings, which is a pragmatic approach and represents the patients’ health condition better than coded diagnoses. Additional strengths consist of multivariate analyses, multilevel models allowing for cluster effects and an advanced treatment of missing values.
Comparison with existing literature
During the 5-year observation period, physical activity at baseline proved to be a protective factor regarding the progression of multimorbidity severity. Other studies showed mixed evidence. While some studies found a strong inverse association of these two factors, 29 –31 others did not find any correlation. 32 –34 However, physical activity seems to be an effective strategy for the prevention and treatment of many chronic conditions. 17,35 –38
In our study, a higher degree of alcohol consumption showed to have a positive effect on the progression of cardiovascular and metabolic disorders. Positive effects of alcohol consumption have also been documented elsewhere. 39,40 However, literature shows that consumption of a higher amount of alcohol is related to increasing the risk of stroke incidence and mortality. 39 Furthermore, alcohol consumption is linked to the occurrence of chronic diseases, such as cancer, and a higher rate of overall mortality. 19,41 Currently, the protective effects of low-volume alcohol use are being discussed (again) regarding the methodology of the underlying studies. 42,43
Our results show a higher number of tobacco-related pack years to be a minor risk factor for the progression of multimorbidity severity of cardiovascular and metabolic disorders. Literature states far higher risks. Jha and Peto, for example, stated that tobacco smoking was ‘the biggest external cause of non-communicable diseases’ 44 and many authors claim that smoking cessation can lead to substantial health benefits by avoiding premature death and chronic disease incidence. 18,44 –46 The difference to our study might be explained by the high age of our study participants who can loosely be referred to as the ‘healthy survivors’. Other studies also found a positive association between the amount of tobacco smoking and the degree of multimorbidity, 33,47 but the effect was much smaller in the older age group than in younger patients. 47
Social support can be measured as subjectively perceived or as de facto received support. 48 In our study, a better perception of social support showed to be beneficial against progressing severity of ADSom and pain-related disorders, but we found no relationship with the progression of multimorbidity severity linked to cardiovascular and metabolic disorders. This is in contrast to literature showing an inverse relationship between social support and mental 49,50 as well as physical health. 20,51 In multimorbidity research, several studies showed that a large social network seemed to be a protective factor against multimorbidity. 47,52,53
Concerning the effects of socio-economic status, education and income were inversely linked to the progression of cardiovascular and metabolic disorders. Although some studies showed the socio-economic status to be inconsistently related to multimorbidity, 54,55 our findings were in line with many other multimorbidity studies. 1,4,15
Implications for practice
Multimorbidity is prevalent and associated with adverse outcomes. But multimorbidity is also a complex entity and useful strategies in patients with single diseases may not be appropriate for patients with multimorbidity. Therefore, evidence on the treatment of single diseases needs to be reassessed for patients with multimorbidity. The approach of our study was to reduce the complexity of multimorbidity by introducing two prevalent clusters of diseases.
Our results indicate that, depending on the multimorbidity cluster, different strategies should be used for slowing down the progression of multimorbidity. Regarding cardiovascular and metabolic disorders, physical activity and smoking cessation are the most promising approaches. Regarding ADSom and pain-related disorders, strengthening the perceived social support is the most promising approach identified in our study. Surprisingly – and in contrast to other studies – the social support did not influence the disease severity in the multimorbidity cluster of cardiovascular and metabolic disorders.
The social status had only a minor influence on the effects of these strategies. This is good news, as health behaviour and social support seem to be more influenceable than the socio-economic status. For daily practice, this means that changing lifestyle and increasing social support are worthwhile factors in elderly multimorbid patients – regardless of their socio-economic status.
Footnotes
Authors’ note
This article was written on behalf of the MultiCare Cohort Study Group, which consists of Attila Altiner, Horst Bickel, Wolfgang Blank, Monika Bullinger, Hendrik van den Bussche (principal investigator), Anne Dahlhaus, Lena Ehreke, Michael Freitag, Angela Fuchs, Jochen Gensichen, Ferdinand Gerlach, Heike Hansen, Sven Heinrich, Susanne Höfels, Olaf von dem Knesebeck, Hans-Helmut König, Norbert Krause, Hanna Leicht, Margrit Löbner, Melanie Luppa, Wolfgang Maier, Manfred Mayer, Christine Mellert, Anna Nützel, Thomas Paschke, Juliana Petersen, Jana Prokein, Steffi Riedel-Heller, Heinz-Peter Romberg, Ingmar Schäfer, Martin Scherer (principal investigator), Gerhard Schön, Susanne Steinmann, Sven Schulz, Karl Wegscheider, Klaus Weckbecker, Jochen Werle, Siegfried Weyerer and Birgitt Wiese.
Acknowledgements
We are grateful to the general practitioners in Bonn, Dusseldorf, Frankfurt am Main, Hamburg, Jena, Leipzig, Mannheim and Munich who supplied the clinical information on their patients, namely Claudia Adrian, Theodor Alfen, Martina Amm, Bernhard Andrea, Katrin Ascher, Philipp Ascher, Heinz-Michael Assmann, Hubertus Axthelm, Leonhard Badmann, Angelika Bakro, Horst Bauer, Veit-Harold Bauer, Sylvia Baumbach, Brigitte Behrend-Berdin, Kathrin Beimes, Rainer Bents, Peter Berdin, Werner Besier, Liv Betge, Arno Bewig, Maria Beykirch, Hannes Blankenfeld, Harald Bohnau, Gisela Böhnisch, Claudia Böhnke, Ulrike Börgerding, Gundula Bormann, Martin Braun, Inge Bürfent, Klaus Busch, Jürgen Claus, Peter Dick, Heide Dickenbrok, Wolfgang Dörr, Nadejda Dörrler-Naidenoff, Ralf Dumjahn, Norbert Eckhardt, Richard Ellersdorfer, Farahnaz Fathollahi-Halm, Doris Fischer-Radizi, Martin Fleckenstein, Anna Frangoulis, Daniela Freise, Denise Fricke, Roland Friedmann, Nicola Fritz, Irina Fuchs, Sabine Füllgraf-Horst, Angelika Gabriel-Müller, Rainer Gareis, Benno Gelshorn, Kilian Glänzer, Maria Göbel-Schlatholt, Manuela Godorr, Jutta Goertz, Cornelia Gold, Peter Claus Göttlich, Stefanie Grabs, Hartmut Grella, Peter Gülle, Elisabeth Gummersbach, Heinz Gürster, Eva Hager, Wolfgang-Christoph Hager, Henning Harder, Matthias Harms, Dagmar Harnisch, Bernd Harzer, Martina Hase, Marie-Luise von der Heide, Katharina Hein, Ludger Helm, Silvia Helm, Freya Hildebrandt, Udo Hilsmann, Claus W Hinrichs, Bernhard Hoff, Karl-Friedrich Holtz, Wolf-Dietrich Honig, Christian Hottas, Helmut Ilstadt, Detmar Jobst, Anke Jodl, Markus Jungwirth, Gunter Kässner, Volker Kielstein, Gabriele Kirsch, Thomas Kochems, Martina Koch-Preißer, Andreas Koeppel, Ulrike Kopp, Almut Körner, Gabriele Krause, Jens Krautheim, Nicolas Kreff, Daniela Kreuzer, Franz Kreuzer, Reinhard Kuhlmann, Judith Künstler, Christiane Kunz, Doris Kurzeja-Hüsch, Felizitas Leitner, Peter Lelling, Holger Liebermann, Ina Lipp, Thomas Lipp, Bernd Löbbert, Klaus Löblein, Meiken Löffler, Guido Marx, Vera Matthäus, Stefan Maydl, Manfred Mayer, Stefan-Wolfgang Meier, Jürgen Meissner, Anne Meister, Silvely Menz, Ruth Möhrke, Christian Mörchen, Andrea Moritz, Ute Mühlmann, Gabi Müller, Sabine Müller, Karl-Christian Münter, Helga Nowak, Erwin Ottahal, Kerstin Övermann, Christina Panzer, Guido Pascha, Thomas Paschke, Ulrike Pehnke, Helmut Perleberg, Jörg Pieper, Eberhard Prechtel, Hubertus Protz, Sandra Quantz, Eva-Maria Rappen-Cremer, Thomas Reckers, Elke Reichert, Bettina Relke, Ingolf Resa, Birgitt Richter-Polynice, Anke Riester, Ute Rippel-Lau, Franz Roegele, Joachim Rogalli, Heinz-Peter Romberg, Anette Rommel, Michael Rothe, Uwe Rumbach, Michael Schilp, Franz Schlensog, Ina Schmalbruch, Angela Schmid, Birgit Schmidt, Holger Schmidt, Gabriele Schmidt-Wolf, Christina Schmitt, Lothar Schmittdiel, Matthias Schneider, Ulrich Schott, Sabine Schuchardt, Gerhard Schulze, Heribert Schützendorf, Susann Schwindt, Harald Siegmund, Gerd Specht, Jürgen Specovius, Karsten Sperling, Meingard Staude, Hans-Günter Stieglitz, Martin Strickfaden, Hans-Christian Taut, Golo Teßmann, Stefan Teweleit, Johann Thaller, Uwe Thürmer, Ljudmila Titova, Michael Traub, Martin Tschoke, Maya Tügel, Christian Uhle, Kristina Vogel, Florian Vorderwülbecke, Hella Voß, Christoph Weber, Klaus Weckbecker, Sebastian Weichert, Sabine Weidnitzer, Brigitte Weingärtner, Karl-Michael Werner, Hartmut Wetzel, Edgar Widmann, Alexander Winkler, Ulrike Wirsching, Otto-Peter Witt, Martin Wolfrum, Rudolf Wolter, Armin Wunder and Steffi Wünsch.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the German Federal Ministry of Education and Research (Grant Nos 01ET0725-31 and 01ET1006A-K). The funding body had no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
