Abstract
Blumea lanceolaria (Roxb.) is a medicinal shrub traditionally used to treat infections, inflammation and gastric ulcers. This study investigated its antimicrobial and anti-ulcer potential. Antimicrobial phytochemicals were extracted and evaluated using disc diffusion and broth dilution methods against Pseudomonas aeruginosa, Proteus mirabilis, Enterobacter aerogenes and Escherichia coli. Bioactive compounds were characterised by LC-HRMS profiling, and their anti-ulcer potential was further examined through molecular docking studies against the human gastric proton pump. Sulfuric acid extracts showed the strongest antibacterial activity, with inhibition zones ranging from 8.9 to 14.5 mm. Acid neutralising capacity assays indicated that phosphate buffer leaf extracts had significant activity (280 mEq/g) compared to a standard antacid, suggesting potential anti-ulcer effects. LC-HRMS profiling identified 87 peaks, out of which 30 major bioactive compounds were followed by molecular docking against the human gastric proton pump (PDB ID: 3PGL). N2-Aristololactam-I-guanine and estrone showed high binding affinities (−9.3 and −8.6 kcal/mol) and favourable ADME properties, highlighting their potential as anti-ulcer drug candidates.
Introduction
A significant worldwide health concern is the sharp increase in multidrug-resistant (MDR) bacterial infections, which calls for the creation of alternative treatment approaches in addition to traditional antibiotics.1,2 Among developing techniques, antimicrobial phytochemicals (APs) along with antimicrobial peptides (AMPs) have attracted great attention due to their broad-spectrum activity, fast mode of action, and lower likelihood of resistance formation. 3 Plants are regarded as a viable source of antimicrobial compounds because they generate structurally varied bioactive phytochemicals that are essential to their defence against microbial diseases.4–6 Plant-derived APs and AMPs can be divided into many groups according to their chemical structure, functional groups, and biosynthetic classes. These groups frequently exhibit a variety of structural traits and biological activities that are similar to those of other naturally occurring antimicrobial substances.7–13 Thus, studies on various plant species have resulted in the development of tailored AMPs extraction methods from APs that consider the unique characteristics of plant tissues. 14 AMPs are small molecules with molecular weights ranging from 2 to 10 kDa. They are usually positively charged, amphiphilic, and exhibit a basic pH, which contributes to their antimicrobial activity.11,12,15–17 These phytochemicals, along with peptides, can be isolated from the stems, roots, seeds, flowers and leaves of plants.12,18
Blumea lanceolaria (Roxb.) is a medicinal plant widely used in traditional medicine to treat infections, inflammation and gastric problems. Despite its extensive use, it remains underexplored for bioactive peptides.19–21 Peptic ulcer disease (PUD) continues to be a major health issue, especially in developing areas, with current treatments often causing side effects, recurrence, and resistance. 22 Therefore, this study evaluates the extraction methods of AP-rich fractions from B. lanceolaria to develop a dual antimicrobial and anti-ulcer framework. The study also highlights potential bioactive compounds for therapeutic application and connects the traditional use of plants towards targeted drug discovery for MDR infections and peptic ulcers.
Material and Methods
Collection, Identification and Extract Preparation of Plant Sample
Leaves of B. lanceolaria were gathered from Debargaon village in Kokrajhar district, Assam, India (26°24′16.1964″ N, 90°16′23.1672″ E). The plant was identified and confirmed at the Department of Botany, Bodoland University. Mature, fully expanded, healthy leaves were selected, thoroughly washed with distilled water and oven-dried for 5 days. Once dried, the leaves were ground into a fine powder and kept in airtight containers for extraction.
To prepare the aqueous extract, 10 g of the leaf powder was blended with 100 mL of distilled water and heated in a water bath at 60°C for 3 hours, stirring intermittently. 23 The mixture was filtered through Whatman No. 1 filter paper and then lyophilised.
For the methanolic extract, 10 g of leaf powder was soaked in 100 mL of methanol and kept at room temperature for 3–4 days, with occasional stirring. 24 The mixture was filtered through Whatman No. 1 paper, and the filtrate was concentrated by evaporating the solvent. The dried extracts were stored at −20°C until needed.
Extraction of APs
Multiple extraction solvents were used to maximise the recovery of APs from plant leaves by leveraging differences in solubility, polarity, and pH sensitivity. Given that APs from plants exist in various forms, no single solvent can efficiently extract all bioactive fractions. Acidic solvents, including acetic acid (1 M) and sulfuric acid (50 mM), are utilised to break down cellular structures and promote the release of bioactive substances while reducing deterioration. Under weakly buffered conditions, polar APs can be extracted with the use of sodium acetate buffer (50 mM). Water-soluble phytochemicals are extracted while retaining their structural integrity using phosphate buffer (PB) (20 mM, pH 7.2, 0.1 M NaCl). Terpenoids and other lipophilic phytoconstituents that may contribute to antibacterial activity are extracted using dichloromethane (DCM), a non-polar solvent.16,25–27 To minimise enzymatic degradation and improve extraction efficiency, all extractions were carried out at 4°C with constant stirring. Comprehensive profiling of the plant material’s polar and non-polar APs was made possible by this combined solvent technique.
Syrup Preparation
Simple syrup was prepared by dissolving 66.7 g of sucrose in 1 L of water, heated with occasional stirring until fully dissolved. 50 g of B. lanceolaria leaf powder was boiled with 400 mL of water until the volume reduced to one-fourth. After cooling, the mixture was filtered to obtain the herbal syrup. One part of the decoction was mixed with five parts of simple syrup (1:5).28–30
Protein Quantification
The protein content of various chemical extracts used was determined using the Lowry assay method. This involved preparing an alkaline copper solution (Reagent C) by mixing 50 mL of 2% sodium carbonate in 0.1 N sodium hydroxide (Reagent A) with 1 mL of 0.5% copper sulfate in 1% potassium sodium tartrate (Reagent B). A diluted Folin’s reagent (Reagent D) was made by combining Folin-Ciocalteau reagent with an equal volume of 0.1 N NaOH. A standard curve was created using bovine serum albumin (BSA) with concentrations from 40 to 200 µg (Figure 1). The samples and standards were treated with 5 mL of Reagent C, incubated for 10 minutes, and then 0.5 mL of Reagent D was added. After 30 minutes of incubation in the dark, the absorbance was measured at 660 nm, and the protein concentration was calculated using the standard curve.31,32
Standard Curve for Protein Estimation.
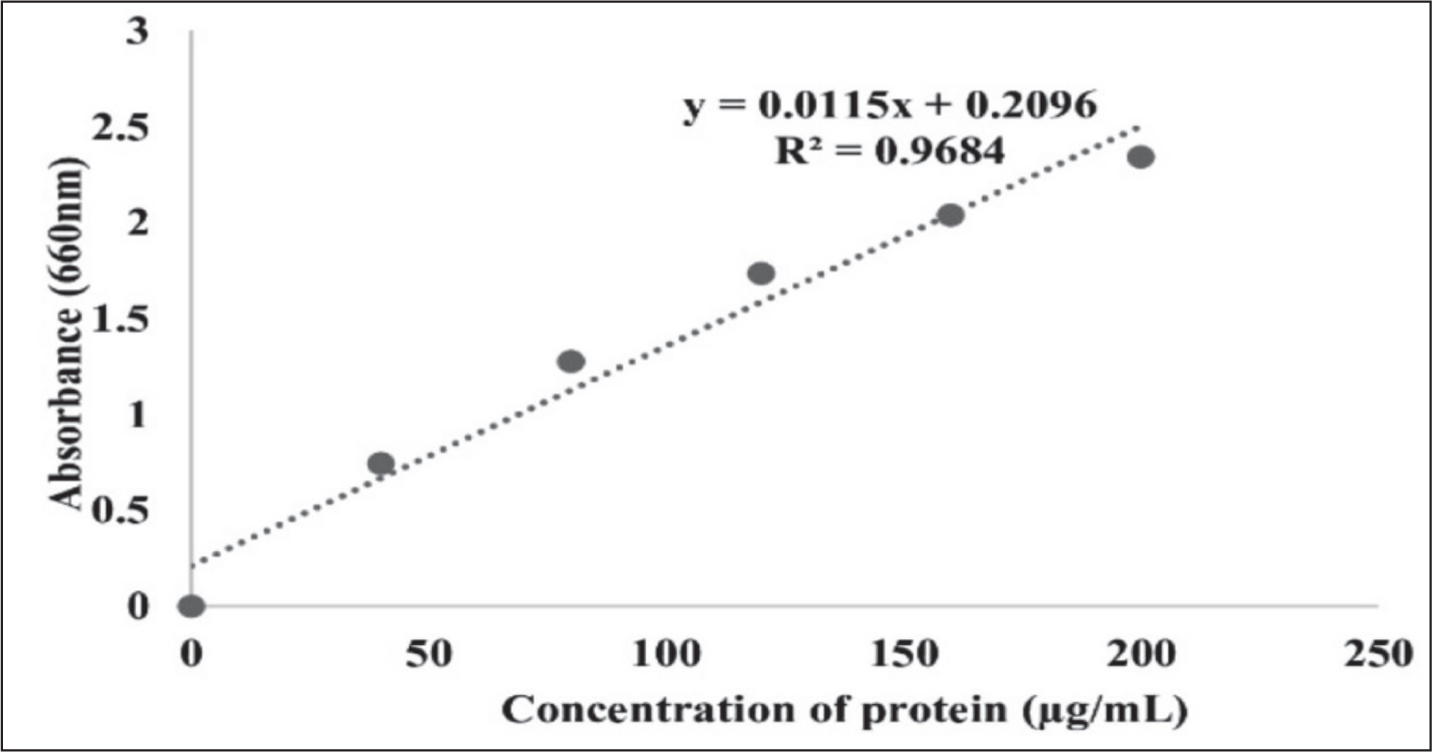
Microbial Strains
Pseudomonas aeruginosa (MTCC 1688), Escherichia coli (MTCC 443), Enterobacter aerogenes (MTCC 2822), and Proteus mirabilis (MTCC 1439) were obtained from Microbial Type Culture Collection (MTCC), Chandigarh, India.
Antimicrobial Activity Tests
Disc Diffusion Method
The antimicrobial activity of the extracts was determined using the disc diffusion method. 33 Microbial cultures were swabbed on nutrient agar plates, and discs of 6 mm diameter were placed on each plate. 10 µL of the plant extracts and syrup was added to the discs, which were incubated at 37°C for 24 hours. The development of the zone was noticed, and its diameter was measured. The experiment was repeated three times to confirm the accuracy of the result. A positive control (standard commercial Ciprofloxacin) and a negative control (sterile distilled water) were taken along with the different extracts. The standard Ciprofloxacin 5 µg antibiotic disc was tested against P. aeruginosa, E. coli, E. aerogenes, and P. mirabilis.
Broth Dilution Method
The antimicrobial activity of B. lanceolaria extracts was verified using the modified broth dilution method. 34 Glassware and media were sterilised by autoclaving at 121°C for 15 minutes. Nutrient Agar (2.3 g) and Nutrient Broth (8 g) were prepared and sterilised in 1 L of distilled water. E. coli, P. mirabilis, P. aeruginosa, and E. aerogenes were incubated at 37°C for 24 hours. A loopful of each culture was suspended in sterile normal saline to create a uniform inoculum. Extracts were prepared at concentrations of 100, 50, 25, 12.5, and 6.25 mg/mL, with Ciprofloxacin (1 mg/mL) as the positive control and distilled water as the negative control. Different extract concentrations were added to the Nutrient Broth and inoculated with 0.1 mL of bacterial suspension. After incubation at 37°C for 24 hours, antimicrobial activity was assessed by turbidity, and the lowest concentration with no visible growth was recorded as the Minimum Inhibitory Concentration (MIC).
Acid Neutralising Capacity
The acid neutralising capacity (ANC) per gram of antacid was determined by dividing the moles of HCl neutralised by the grams of extract. The ANC of plant extracts at concentrations of 100 mg/mL and herbal syrup was compared to a standard antacid by mixing each extract and syrup with water, adding 30 mL of 1N HCl, stirring, and titrating the excess HCl with 0.5 N sodium hydroxide, with neutralisation calculated as35,36
where,
V1 = Volume of HCl
N1 = Normality of HCl
V2 = Volume of NaOH
N2 = Normality of NaOH
ANC per gram of antacid = moles of HCl neutralised/grams of sample.
LC-HRMS Analysis
The liquid chromatography-high resolution mass spectrometry (LC-HRMS) analysis was performed using a Waters Alliance 2695 HPLC system coupled to a MassLynx 4.1 software-controlled mass spectrometer. Separation was achieved on an ACCUCORE C18 column (150×4.6mm, 2.6µm particle size) at a flow rate of 1.0mL/min, following a 40-minute gradient. The mobile phases consisted of 5mM ammonium acetate in 5% acetonitrile/water (A) and acetonitrile (B), with B increasing from 5% to 80% over 32 minutes, then returning to initial conditions. A 10µL injection volume was used, with the autosampler kept at 20°C. Detection was performed in both positive (ES+) and negative (ES−) electrospray ionisation modes, scanning from 100 to 750m/z in enhanced mode. The desolvation temperature was set to 350°C, with gas flows of 650L/hr (desolvation) and 50L/hr (cone gas). The Waters 2998 diode array detector monitored absorbance between 200–450nm, including channels at 220, 240, 280, 350, and 410nm, sampling at 20 points/sec. Data processing involved spike removal and smoothing.
In Silico Studies
The absorption, distribution, metabolism, and excretion (ADME) and toxicity study of the compounds was carried out using the SwissADME server (
Target Protein Structure Preparation
The 3D structure of the human gastric proton pump (PDB ID: 3PGL) was obtained from the Protein Data Bank (PDB) website. To prepare the structure for further analysis, the BIOVIA Discovery Studio Visualiser was employed to eliminate water molecules, heteroatoms, and other solvent-related entities from the model. Subsequently, AutoDock Tools (ADT) version 1.5.7 was used to prepare the receptor protein and to remove any bound ligands. Hydrogens were added to the structure, along with Kollman charges for the polar atoms and Gasteiger charges for all atoms. The system also involved detecting flexible torsions, and adjustments were made to the number of torsions as necessary. Both polar and non-polar hydrogens were merged appropriately. Finally, the docking input file, formatted as a PDBQT file, was generated using ADTs. Importantly, the chosen protein was docked without any prior relaxation, treating it as a rigid model structure throughout the docking process.
Ligand Structure Preparation
The ligands for this docking study were 30 bioactive compounds selected from LC-HRMS analysis of B. lanceolaria leaves. Structures were downloaded from the PUBCHEM database for 3D optimisation. Using Open Babel 3.1.1, SDF files containing all hydrogens were converted to PDB format. For ligands not found in PUBCHEM, 3D structures were created with Marvin Chemaxon using canonical SMILES. Gasteiger charges were added, non-polar hydrogens were merged, and aromatic carbons were prepared for docking in ADT, allowing for torsional flexibility and a wide range of conformational options.
Active Site and Grid Generation
A grid box for blind docking of the gastric proton pump was created with dimensions of 60, 62, and 76 along the X, Y, and Z axes. Standard docking reporting uses: Centre: X = 19.6373, Y = 10.904, Z = 4.837.
Molecular Docking
The AutoDock Vina tool was used to predict binding poses between 30 selected phytochemicals and the gastric proton pump. The docking procedure employed a local search global optimiser and the Fletcher Goldfarb-Shanno (BFGS) method for optimisation, serving as a positive control. With a docking exhaustiveness of 8, the software generated nine protein-ligand complexes, which were ranked by binding affinity.
Analysis and Visualisation of Docking Simulation Results
The conformation with the lowest binding affinity was chosen for further study after ranking the docking postures. The BIOVIA Discovery Studio Visualiser was used to display the binding poses, orientation, interacting residues, and bond details.
Drug Likeness and ADME Analysis
The pharmacokinetic profiles of the top binding affinity drugs were predicted using the Swiss ADME predictor. Ligand screening considered physicochemical properties, such as lipophilicity and water solubility, along with pharmacokinetics based on the Lipinski, Veber, Egan, and Muegge rules. Medicinal chemistry was also evaluated for lead likeness compliance.
Result and Discussion
Protein Quantification
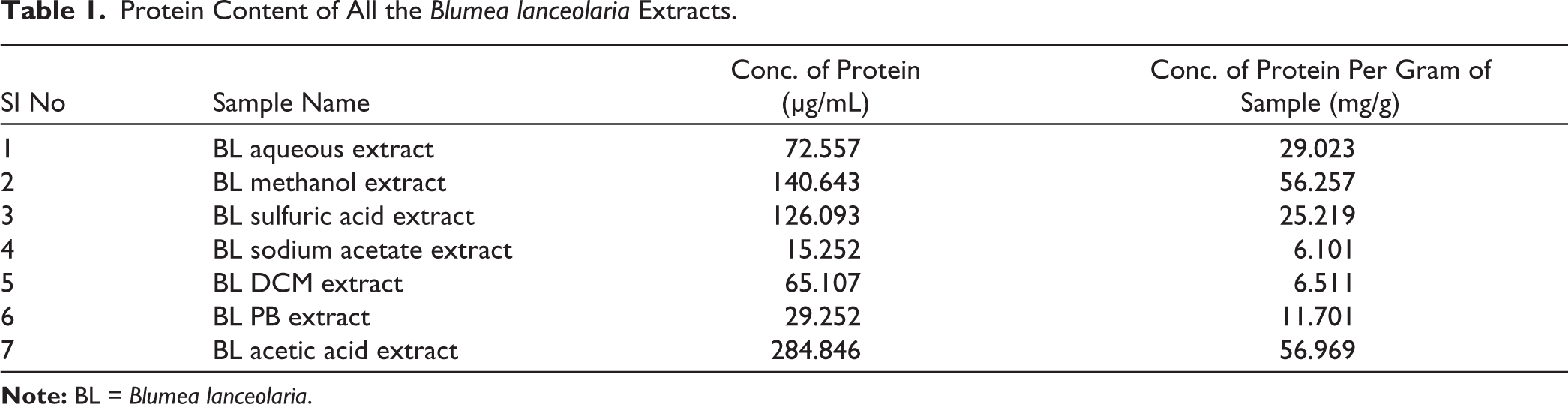
The protein content of various B. lanceolaria extracts is shown in Table 1 and Figure 2. The acetic acid extract exhibited the highest protein concentration at 284.846 µg/mL, corresponding to 56.969 mg/g of the sample, closely followed by the methanol extract, which had a concentration of 140.643 µg/mL (56.257 mg/g). The aqueous extract demonstrated moderate protein content with a concentration of 72.557 µg/mL (29.023 mg/g), while the sulfuric acid extract yielded 126.093 µg/mL (25.219 mg/g). Lower protein yields were recorded for sodium acetate (15.252 µg/mL; 6.101 mg/g), DCM (65.107 µg/mL; 6.511 mg/g), and PB extracts (29.252 µg/mL; 11.701 mg/g). Overall, acidic and polar solvents, particularly acetic acid and methanol, proved to be more effective in extracting proteinaceous components from B. lanceolaria compared to the less polar solvents. A similar finding was made by Ben Brahim et al. (2022), where proteins were detected in all leaf and root extracts of Anthyllis sericea and Astragalus armatus, with significant variations observed among the solvents used. Specifically, acetic acid extraction produced a higher protein content compared to sodium acetate. 16
Protein Content of All the Blumea lanceolaria Extracts.
Protein Content of Various Blumea lanceolaria Extracts.
Antimicrobial Activity
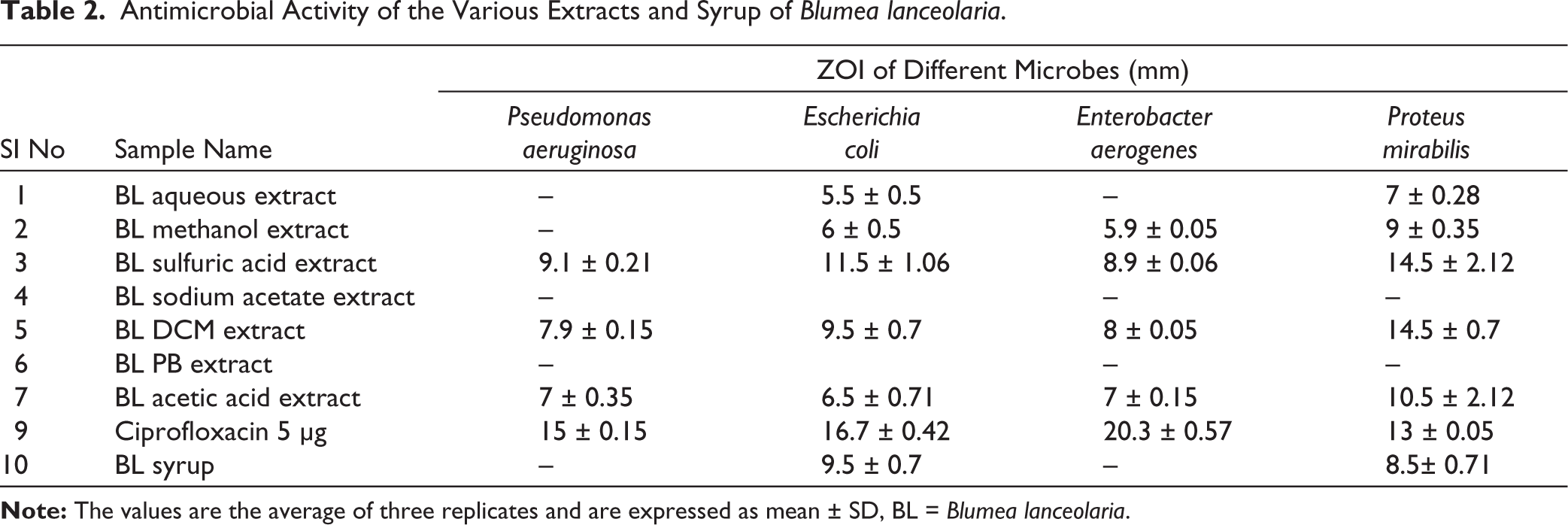
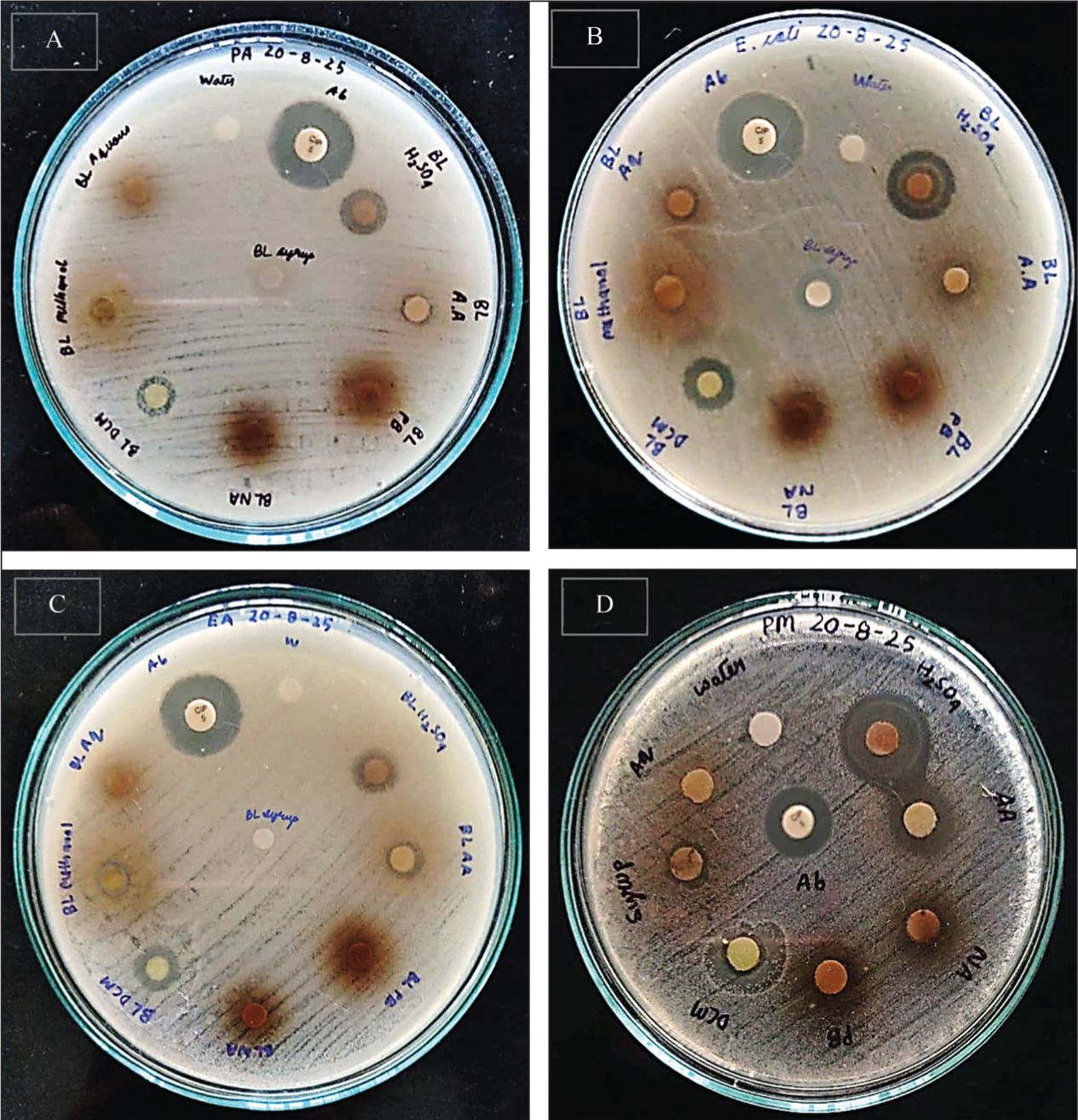
The antimicrobial activity of various B. lanceolaria extracts and syrup against P. aeruginosa, E. coli, E. aerogenes and P. mirabilis was assessed using disc diffusion and broth dilution methods. The sulfuric acid extract showed the highest antibacterial efficacy, with zones of inhibition (ZOI) of 9.1 mm (P. aeruginosa), 11.5 mm (E. coli), 8.9 mm (E. aerogenes), and 14.5 mm (P. mirabilis) (Table 2). The syrup exhibited moderate inhibition (9.5 mm for E. coli and 8.5 mm for P. mirabilis) (Table 2 and Figure 3). In the broth dilution assay, the MIC was the lowest effective concentration after 24 hours at 37°C, as shown in Table 3. The aqueous extract and syrup inhibited E. coli and P. mirabilis at 100 mg/mL but were ineffective against P. aeruginosa and E. aerogenes. The methanol extract worked against E. coli, E. aerogenes, and P. mirabilis at 100 mg/mL. The sulfuric acid extract was the most potent, with MIC values of 50 mg/mL against E. coli and P. mirabilis, and 100 mg/mL against P. aeruginosa and E. aerogenes. The DCM extract inhibited P. mirabilis at 50 mg/mL and was effective against the other strains at 100 mg/mL. Overall, the sulfuric acid extract demonstrated the strongest antibacterial activity, having the largest ZOI and the lowest MIC.
Antimicrobial Activity of the Various Extracts and Syrup of Blumea lanceolaria.
The Antimicrobial Activity of the Various Extracts of Blumea lanceolaria Against (A) Pseudomonas aeruginosa, (B) Escherichia coli, (C) Enterobacter aerogenes and (D). Proteus mirabilis.
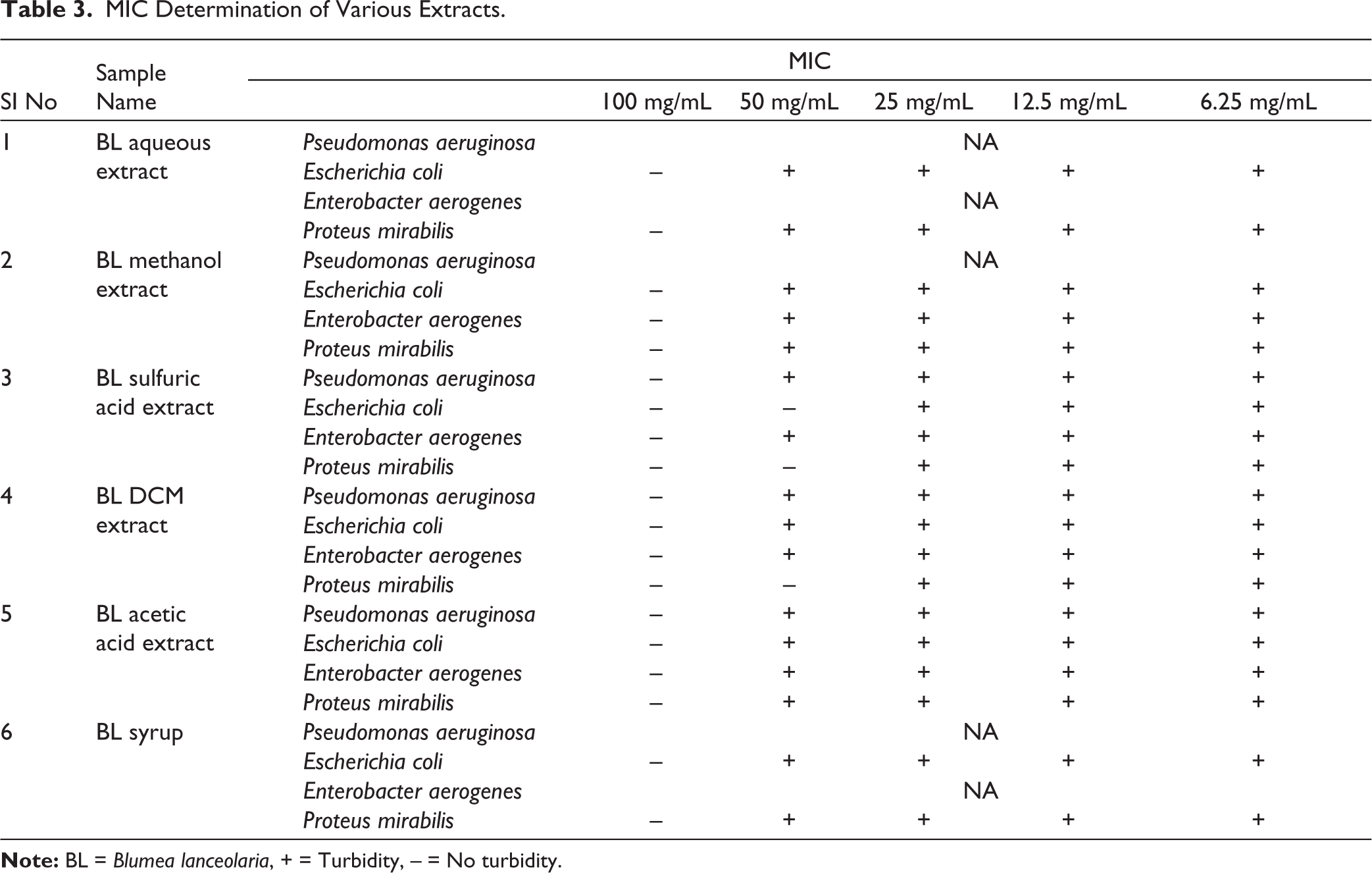
MIC Determination of Various Extracts.
Other studies done on leaf extracts of A. sericea, obtained using acetic acid and sodium acetate, demonstrated broad-spectrum activity against both gram-positive (Staphylococcus aureus, Bacillus subtilis, B. pumilus) and gram-negative (E. coli, Salmonella enterica) bacteria, with inhibition zones ranging from 9 to 22.5 mm, indicating a strong antibacterial efficacy. 16
Acid Neutralising Capacity
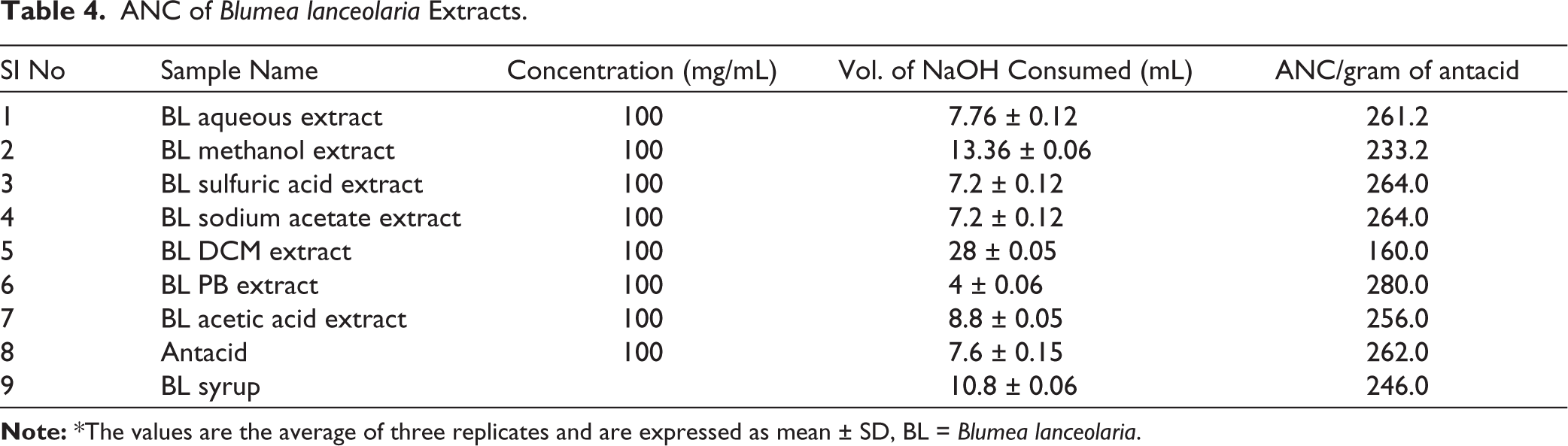
The ANC of B. lanceolaria extracts revealed significant antacid potential, with most extracts showing activity comparable to or higher than the commercial standard (262.0 mEq/g) presented in Table 4. Among the extracts, the PB extract exhibited the highest ANC (280 mEq/g), followed closely by sulfuric acid and sodium acetate extracts (264 mEq/g each), aqueous extract (261.2 mEq/g), and acetic acid extract (256.0 mEq/g), all of which were nearly equivalent or superior to the marketed antacid. The B. lanceolaria syrup (246.0 mEq/g) also showed moderate efficacy. At the same time, the methanol (233.2 mEq/g) and DCM (160.0 mEq/g) extracts were less effective, suggesting that polar phytoconstituents contribute more significantly to the antacid effect than non-polar ones.
ANC of Blumea lanceolaria Extracts.
Studies tested Anogeissus latifolia Roxb. for its ANC at doses from 100 to 1500 mg. They found that 1500 mg was more effective than 500 mg of aluminium hydroxide. 38 Another study showed that the aqueous extract of Ficus thonningii with a pH of 8.32 and an ANC of 7.4 mEq, exceeding the FDA’s minimum of 5 mEq, indicates its potential as a natural antacid. 39 Overall, these findings suggest that B. lanceolaria could be a promising natural alternative to antacids.
Liquid Chromatography-high Resolution Mass Spectrometry
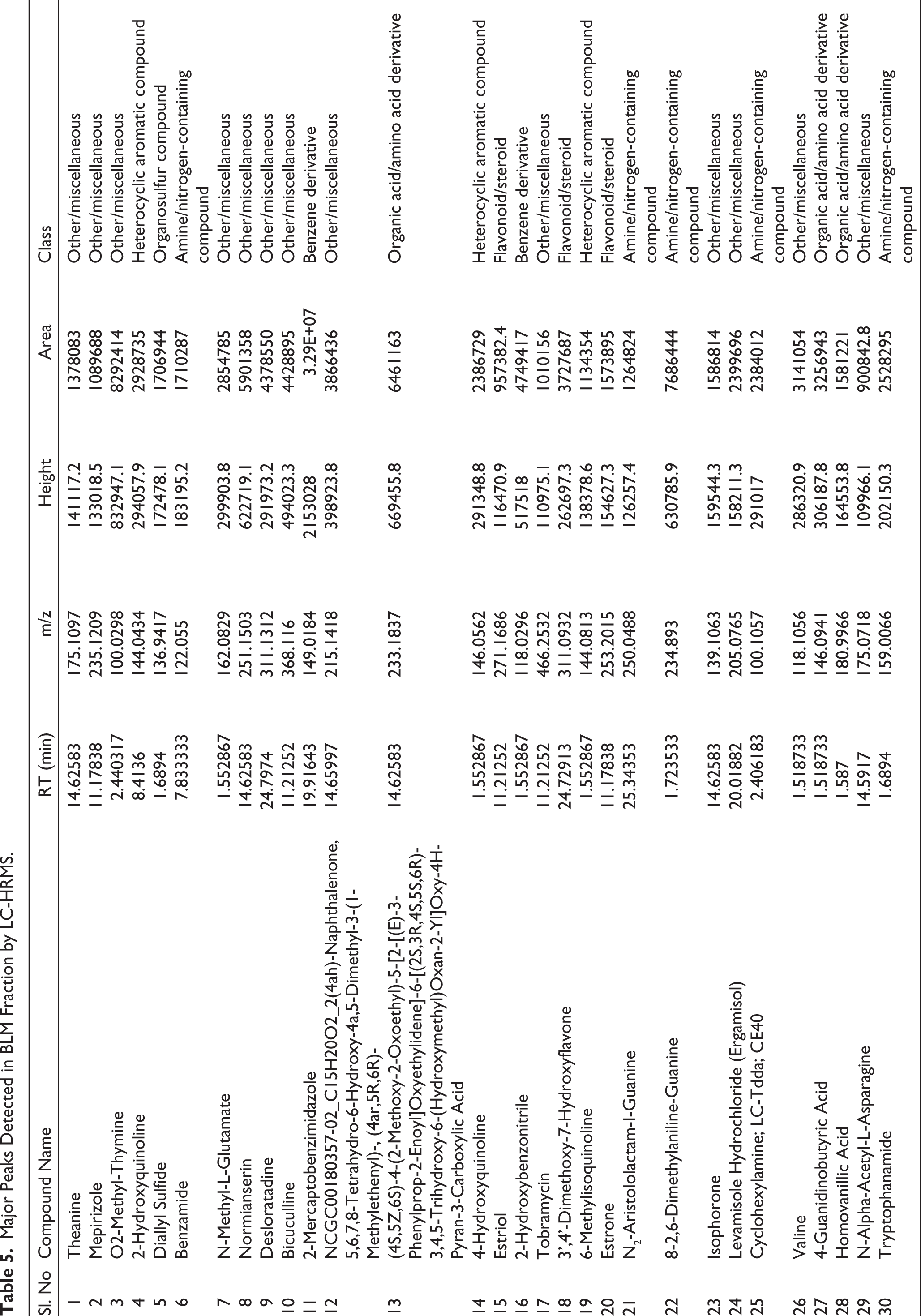
LC-HRMS analysis of the BLM (B. lanceolaria methanolic extract) identified a complex chromatographic profile with retention times from 1.5 to 15 minutes and m/z values ranging from 100 to over 1,000. A total of 87 peaks were detected in positive ionisation mode, most of which showed appropriate signal-to-noise ratios (S/N > 10) and a Gaussian distribution. The analysis revealed diverse metabolites, including amino acids, heterocyclic compounds, organosulfur compounds, flavonoids, and drug-like molecules. Tentative identification was performed by comparing mass values with reference databases. 30 high-intensity metabolites were selected as major components based on their area, height, and spectral quality, as summarised in Table 5.
Major Peaks Detected in BLM Fraction by LC-HRMS.
Molecular Docking
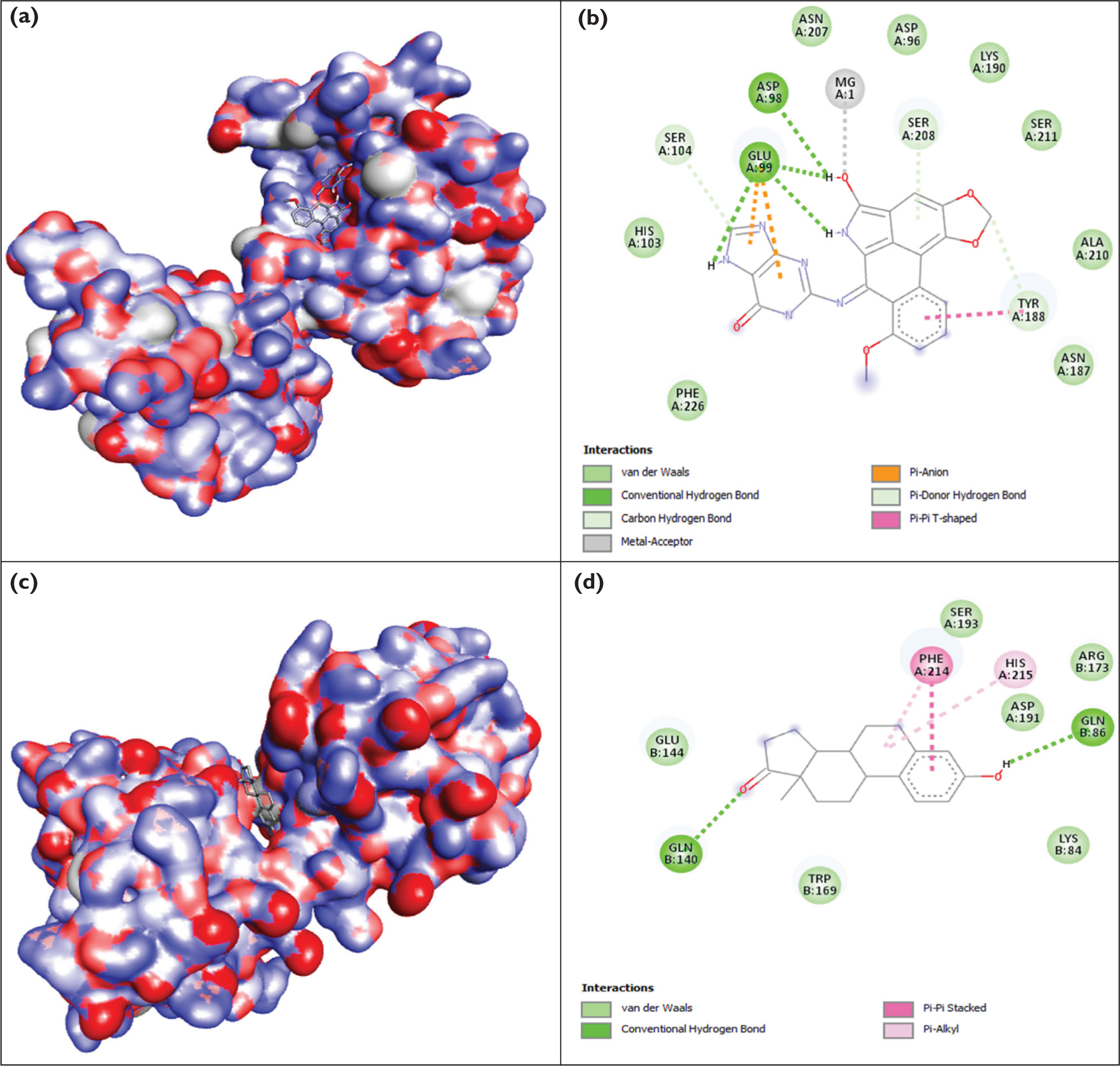
The inhibitory potential of certain phytocompounds against the target protein 3PGL, which is linked to the pathophysiology of peptic ulcers, was assessed using molecular docking analysis. With docking scores of −9.3 kcal/mol and −8.6 kcal/mol, respectively, N2-Aristololactam-I-guanine (N2-Aristololactam-I-guanine | C22H14N6O5 | CID 169449418 - PubChem) and Estrone (Estrone | C18H22O2 | CID 5870 - PubChem) showed the greatest binding affinities among the 30 ligands evaluated using AutoDock Vina (Table 6). Strong binding potential of both compounds is shown by docking scores below −7.0 kcal/mol, which are often regarded as suggestive of positive ligand-protein interactions. Using Chimera 1.17.1 and BIOVIA Discovery Studio visualizer software, the interaction between the complex of the 3PGL and both compounds was examined (Figure 4). Numerous stabilising interactions, including conventional hydrogen bonds, π-anion, π-donor hydrogen bonds, π-π T-shaped interactions, and metal-acceptor coordination involving mg 2 ⁺, were produced by N2-Aristololactam-I-guanine. In docking studies of ulcer-related proteins, important interaction residues, including ASP, GLU, HIS, TYR, and ASN, are commonly identified. These residues are linked to improved binding stability and an anticipated inhibitory effect.39,40 The current findings are relevant since quinazolinone derivatives and other small-molecule inhibitors that target enzymes linked to stomach ulcers have been shown to have similar binding energies (≤ −9.0 kcal/mol). 40 Additionally, estrone showed stable binding through hydrophobic interactions, such as π-alkyl contacts with aromatic residues and π-π stacking, as well as hydrogen bonding. Similar binding energies (−7.0 to −9.0 kcal/mol) have been reported in previous docking investigations on steroidal and plant-derived drugs that target Helicobacter pylori and ulcer-related proteins, suggesting physiologically significant interactions.41,42 Overall, the docking data point to estrone and N2-Aristololactam-I-guanine as possible 3PGL inhibitors.
Docking Scores of 30 Ligands and Their Interactions with the 3PGL Protein.

Molecular Docking Analysis (A) Complex 3PGL and N2-Aristololactam-I-guanine, (B) 2D Depiction of the Molecular Docking Interaction Between 3PGL and N2-Aristololactam-I-guanine, (C) Complex 3PGL and Estrone and (D) 2D Depiction of the Molecular Docking Interaction Between 3PGL and Estrone.
Swiss ADME Analysis
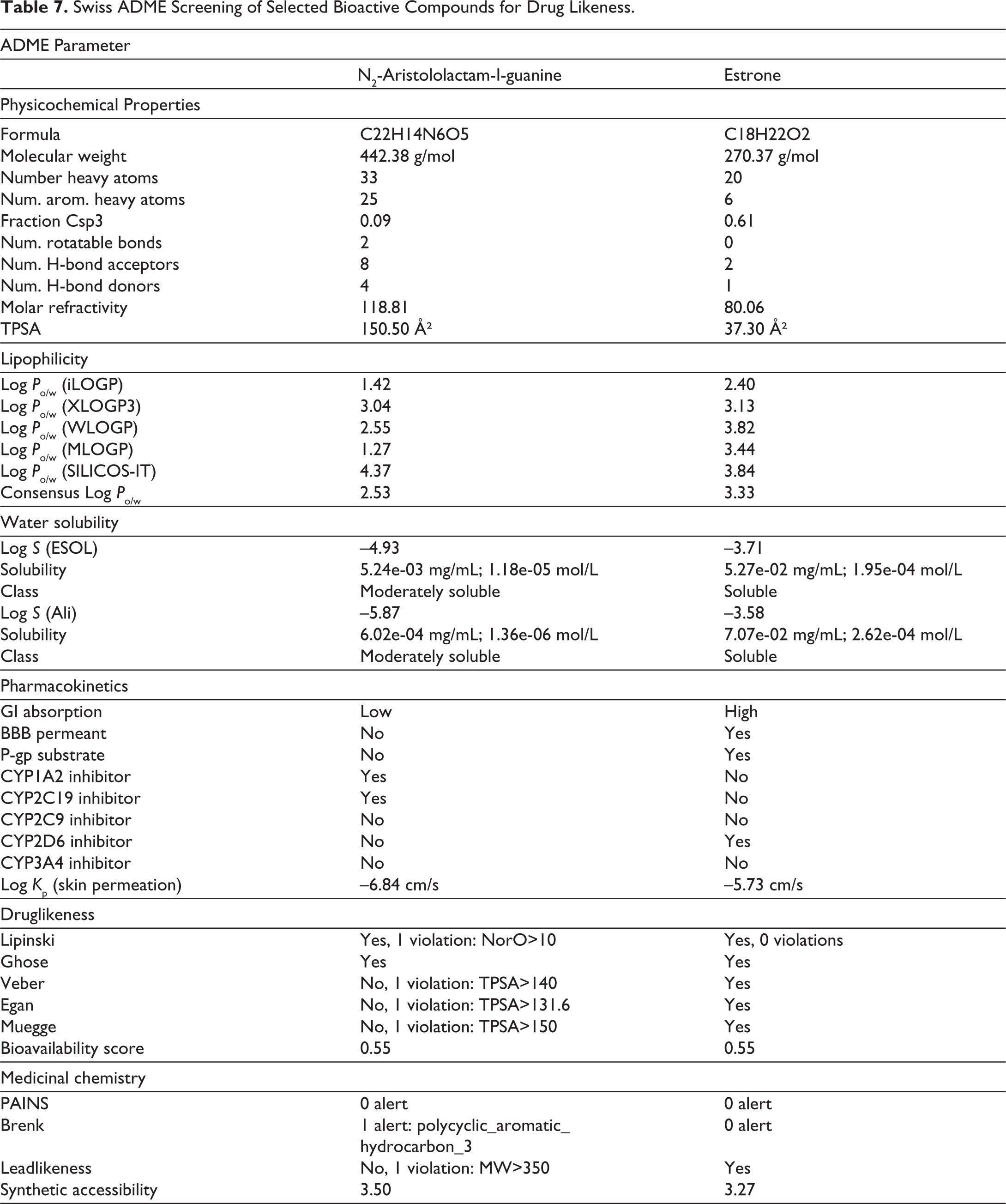
Compounds with the highest scores were selected for ADME analysis and presented in Table 6. The Swiss ADME tool was utilised to evaluate the pharmacokinetic properties of these compounds. Out of 30 ligands, 10 had favourable docking scores. Both compounds with high docking scores satisfy drug likeness criteria (Table 7). N2-Aristololactam-I-guanine showed high intestinal absorption, good bioavailability, and non-permeability of the blood-brain barrier. With a bioavailability score of 0.55. Additional research is needed to confirm its toxicology, pharmacology, and experimental bioavailability.
Swiss ADME Screening of Selected Bioactive Compounds for Drug Likeness.
Conclusion
The sulfuric acid extract of B. lanceolaria leaf demonstrated strong antibacterial activity against gram-negative bacteria, representing the first study of AMPs in this plant and indicating its anti-ulcerogenic potential. In silico studies revealed N2-Aristololactam-I-guanine and estrone as potential candidates for the treatment of peptic ulcer. These findings enhance understanding of natural interventions for MDR and peptic ulcers, supporting future research on effective treatments.
Footnotes
Acknowledgements
The authors kindly acknowledge the financial support as a studentship to Mr Shubam Sinha from the Department of Biotechnology (DBT), Government of India. Also, we would like to express our gratitude to the Department of Biotechnology, Government of India, Sponsored Bioinformatics Infrastructure Facility, Bodoland University and DST-FIST, Bodoland University, for providing the required resources and technical support and the Central Instrumental Facility, Assam Downtown University, for allowing us to utilise their instrumentation facility.
Author’s Contribution
All authors made substantial contributions to conception and design, acquisition of data or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
Consent for Participate
Not applicable.
Consent to Publication
Not applicable.
Data Availability
All the data is available with the authors and shall be provided upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics Approvals
This work does not contain any studies involving human and animal subjects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The first and second authors, Shubam Sinha and Pallavi Rabha, respectively, received financial support in the form of a PG-Studentship stipend from the Department of Biotechnology (DBT), Government of India, during the course of this research work. No specific funding was received for the research, authorship or publication of this article.
Informed Consent
Not applicable.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
Use of Artificial Intelligence-assisted Technology
The authors declare that they have not used artificial intelligence (AI)-tools for writing and editing of the manuscript, and no images were manipulated using AI.

