Abstract
Medicinal plants have served as a source of medicine since human existence. Cannabis sativa, also known as cannabis, or hemp, is one of the most widely known medicinal plants used for the management of ailments, including muscular and/or joint pains, and their associated injuries. Cannabis contains valuable secondary metabolites, terpenes (α-pinene, β-caryophyllene, limonene, and myrcene), terpenoids (camphor, linalool, and terpineol), phenolics (stilbene, spiroindan, and cannflavin), and phytocannabinoids [cannabidiol (CBD), delta-9-tetrahydrocannabinol (Δ9-THC), cannabinol (CBN), cannabigerol (CBG), and their acidic forms]. These classes of compounds are known to contribute to the analgesic, anti-inflammatory, anti-arthritis, and other muscle relaxant-related properties of cannabis. Structurally, phytocannabinoids with an extended side chain at position C3 can activate cannabinoid receptors, promoting inhibition of signaling pathways MAPK and cAMP associated with pain and inflammation. This review discusses C. sativa phytocompounds with muscle relaxant-related properties (inflammation, pain, and arthritis) and their structure-activity relationship, with a view to identifying natural muscle relaxants as possible drug candidates.
Introduction
The musculoskeletal system, in simple terms, refers to the structural components of the body, such as the muscles, bones, and joints, as well as the adjoining connective tissues (tendons and ligaments). 1 Musculoskeletal conditions (MSCs) such as rheumatoid arthritis, fibromyalgia, and low back pain are characterized by progressive chronic inflammation of the musculoskeletal system, resulting in cartilage and bone damage, whose deterioration can lead to disability. 2 The MSCs affect bones, muscles, ligaments, and joints, causing severe pain, inflammation, and impaired physical function.3,4 These conditions impact all aspects of life by affecting dexterity and mobility, and by limiting activities of daily living.4,5 MSCs often lead to a decline in quality of life and bear the risk of cumulative comorbidities.2,6 They are one of the leading causes of disability worldwide, with low back pain being the major contributor.6,7 In 2019, low back pain accounted for approximately 568.4 million prevalent cases, 223.5 million incident cases globally, and according to literature, the burden of low back pain in some countries is still growing. 7
Musculoskeletal disorders such as arthritis and low back pain affect more than 1.7 billion people worldwide and have the 4th greatest influence on the complete fitness of the world population, causing both disability and death.8,9
The management guidelines for MSCs recommend non-drug therapies such as exercise as first-line treatment. 10 Second-line therapy is medication, whereby non-steroidal anti-inflammatory drugs, analgesics, and muscle relaxants are widely used in treating low back pain and arthritis. 10 However, these management strategies are imperfect and associated with adverse effects, and long-term usage is limited due to adverse gastrointestinal and renal effects. 11 Recently, various bodies advocated for opioid analgesics for treating chronic musculoskeletal disorders when other treatments were ineffective, resulting in widespread long-term use among a large percentage of people with chronic musculoskeletal disorders. Opioids confer a substantially greater risk of serious harm, addiction and death.10,12 All in all, the current treatment therapy produces short-term improvement in relieving the pain and cannot be used frequently. 13 This is prompting a search for effective alternative treatments to fill this gap. 14
Cannabis plants have served mankind as a source of medicine since their earliest beginnings, and their therapeutic applications have been increasing across the globe.15,16 Although cannabis is known for its psychoactive effect due to its high content of Δ9-tetrahydrocannabinol (Δ9-THC), the scientific community has shown high interest in harnessing fiber-type cannabis commonly known as hemp for pharmacological purposes.17,18 Non-psychoactive compounds found in cannabis are associated with fewer side effects and can be of use in several industrial applications. 19 The trace amounts of psychoactive components in cannabis attract the pharmaceutical interest of researchers to venture largely into hemp to obtain the non-psychoactive compound cannabidiol (CBD), for its therapeutic value in numerous diseases. 20 Various phytocannabinoids have been identified from hemp, 21 but there remains so much to learn about the biological activities of the different phytochemical components and their structure-activity relationships (SAR). This review article focuses on Cannabis sativa (cannabis) phytocompounds, their muscle relaxant-related properties (inflammation, pain, arthritis), and SAR.
Methodology
The databases, Google Scholar, Science Direct, and PubChem, were used to obtain published articles on C. sativa. The various search keywords as inclusive criteria were MSCs, C. sativa compounds, biosynthesis of C. sativa, cannabis, hemp, SAR of cannabis anti-inflammatory compounds, low back pain, and arthritis. Exclusion data were on the phytocompounds other than cannabis and their SAR. Literature data was collected till September 2025.
Botanical Description
C. sativa Linn. is a dioecious annual flowering plant belonging to the family Cannabaceae, which has multiple genera including Humulus, Celtis, and Cannabis. 22 C. sativa L. is characterized by palmate leaves with a venation pattern and broadly spread branches, whereby 3–13 leaves are present on the branch. 23 The female plant is leafier and long-lived. Its leaves have an elongated form with indented edges, pale green in color with a lighter back. 24 Its inflorescences contain several individual branches of flowers, whereby a large cluster on the upper torso and various small clusters are present in each branch (Figure 1). The plant parts are covered by trichome glands containing resin rich in secondary metabolites responsible for its aroma, defense, and interaction.19,25 C. sativa grows to reach a height of 1–5 meters depending on the environment, and its vegetative period lasts for 3–4 months.23,26 It has deep fibrous tap roots that grow well in fertile and well-drained land with neutral to slightly alkaline soil (Figure 1).
Cannabis sativa Female Inflorescences and Roots.

Cannabis has been classified into three species, namely:
These three species differ fundamentally in terms of their height. 25 C. sativa has a low content of Δ9-THC below the legal limit of 0.2%–0.3% and is rich in non-psychostimulating cannabinoids. 27
Origin and Geographic Distribution
C. sativa is one of the most ancient plants used by humans for multiple purposes, such as medicine, food, and fiber. It has been referred to as the oldest known cultivated fiber plant.
25
The discovery of the cannabis plant is attributed to Chinese research dating back to 10,000–12,000
Ethnomedicinal Uses
The beginning of the use of cannabis by humans is untraceable due to its long history of cultivation and consumption, even before the appearance of writing. 30 Ancient documents, such as the US Pharmacopoeia, described the use of cannabis inflorescence as an analgesic agent, 28 and William O’Shaughnessy, a physician, described the medicinal uses of cannabis apart from the intoxicant effect, whereby analgesic and muscle relaxant properties form part of the medicinal uses of the cannabis extract. 31 Martinenghi et al. 34 highlighted that cannabis has been associated with various pharmacological properties, including alleviating chronic pain and muscle spasm; as a result, it has been recommended for use in arthritis-related cases. The roots of the cannabis plant have been indicated to help manage inflammation, infections, and arthritis. 32 The Roman historian Pliny the Elder recounted the use of C. sativa roots for easing pain.25,35 Cannabis seeds have a historical record of being utilized as an analgesic remedy. 36 C. sativa wild leaf has been reported to be used as a topical paste for joint pain in Kupwara of the Jammu and Kashmir district of India. 37 Cannabis inflorescence is used traditionally to treat acute pain and wounds, while the stem bark is a remedy for physical injury and strangury.19,38
Chemistry of Cannabis
C. sativa contains more than 500 identified chemical compounds. Among them are 125 cannabinoids, 45 phenolic compounds, 120 terpenes and terpenoids, 34 flavonoids, and 2 alkaloids. 36 Cannabinoids are a unique and the largest group of compounds in cannabis. 31 Cannabinoids are meroterpenoids, specifically terpeno-phenolic compounds obtained from the alkylation of an alkyl resorcinol with a mono-terpene unit.27,39 The naturally occurring cannabinoids produced by C. sativa are classified as phytocannabinoids, mainly synthesized in the secretory cells of glandular trichomes, abundant in female inflorescences.22,27,40,41 Trichomes are also present in the leaves and bracts. 42 However, the roots’ surface and tissue do not produce phytocannabinoids. 20 The precursors of phytocannabinoids originate from two distinct biosynthetic pathways: (a) the polyketide pathway, giving rise to olivetolic acid (OLA), and (b) the plastidal 2-C-methyl-D-erythritol-4-phosphate (MEP) pathway, leading to the synthesis of geranyl diphosphate (GPP). 43 Hexanoyl-CoA is condensed with three molecules of malonyl-CoA by the action of a polyketide synthase (PKS) enzyme. The cyclization is catalyzed by the olivetolic acid cyclase (OAC), and the loss of coenzyme A leads to the formation of OLA. 15 Alkylation of OLA with geranyl pyrophosphate by a geranyl pyrophosphate: olivetolate geranyl transferase produces cannabigerolic acid (CBGA) (Figure 2). Cannabigerolic acid can be converted into either ∆9-tetrahydrocannabinolic acid (∆9-THCA), cannabidiolic acid (CBDA), or cannabichromenic acid (CBCA) by the action of specific synthase enzymes.20,25 Cannabinoids are synthesized and accumulated as cannabinoid acids, which are converted to their neutral forms by decarboxylation induced by environmental factors such as exposure to light, heat, and or aging.44–47
Schematic Diagram of the Biosynthetic Pathway Leading to Cannabis sativa Major Phytocannabinoids (THCA, CBDA, CBCA) and Terpenes. MEP = Methylerythritol Phosphate; PKS = Polyketide Synthase; OAC = OLA Cyclase; DMAPP = Dimethylallyl Diphosphate; IPP = Isopentenyl Diphosphate; GOT = Olivetolate Geranyl Transferase.
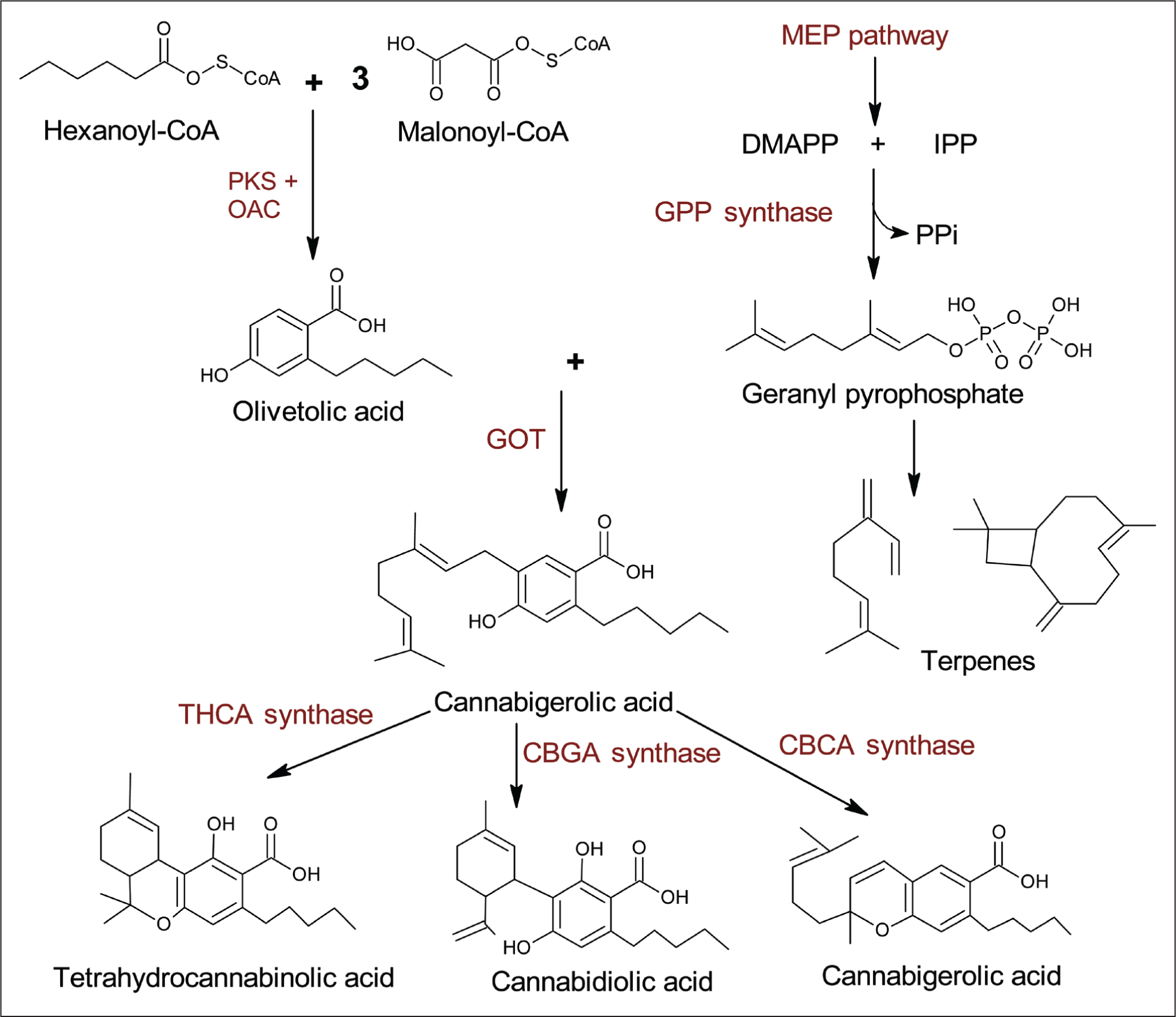
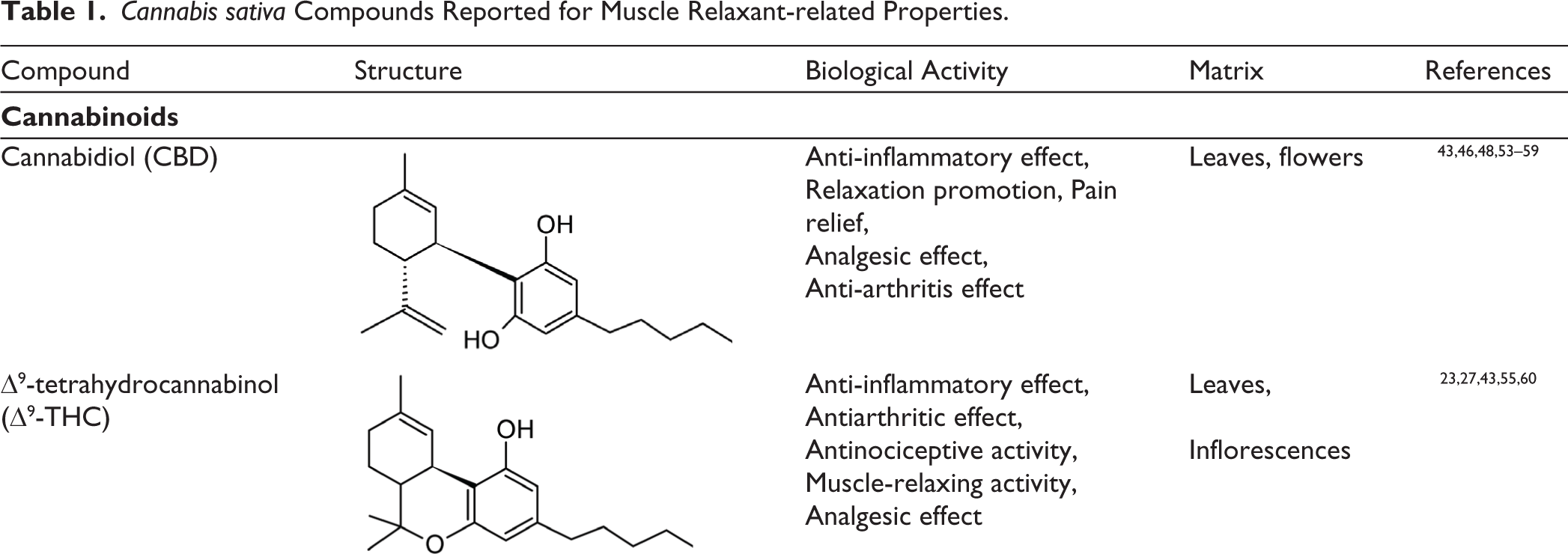
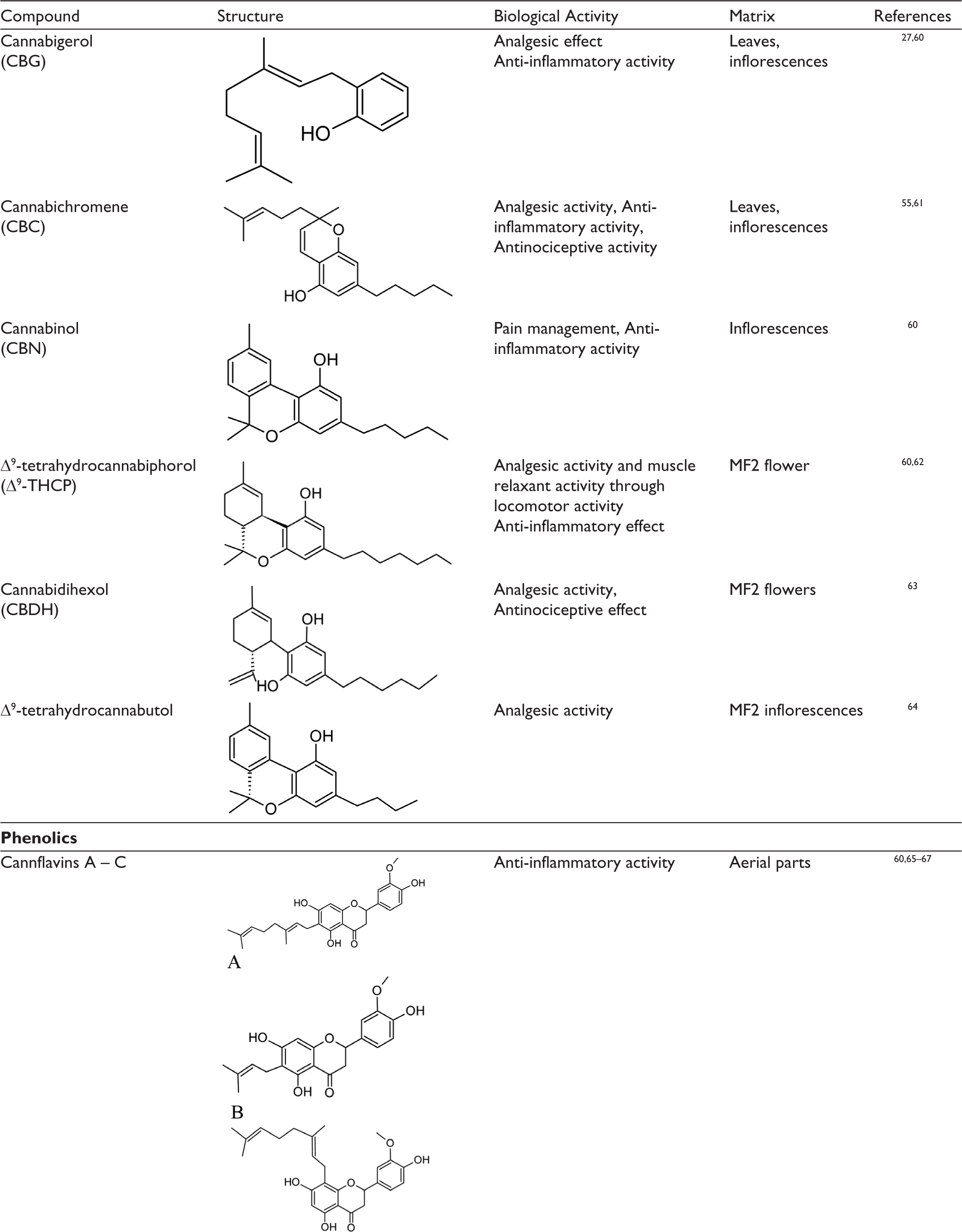
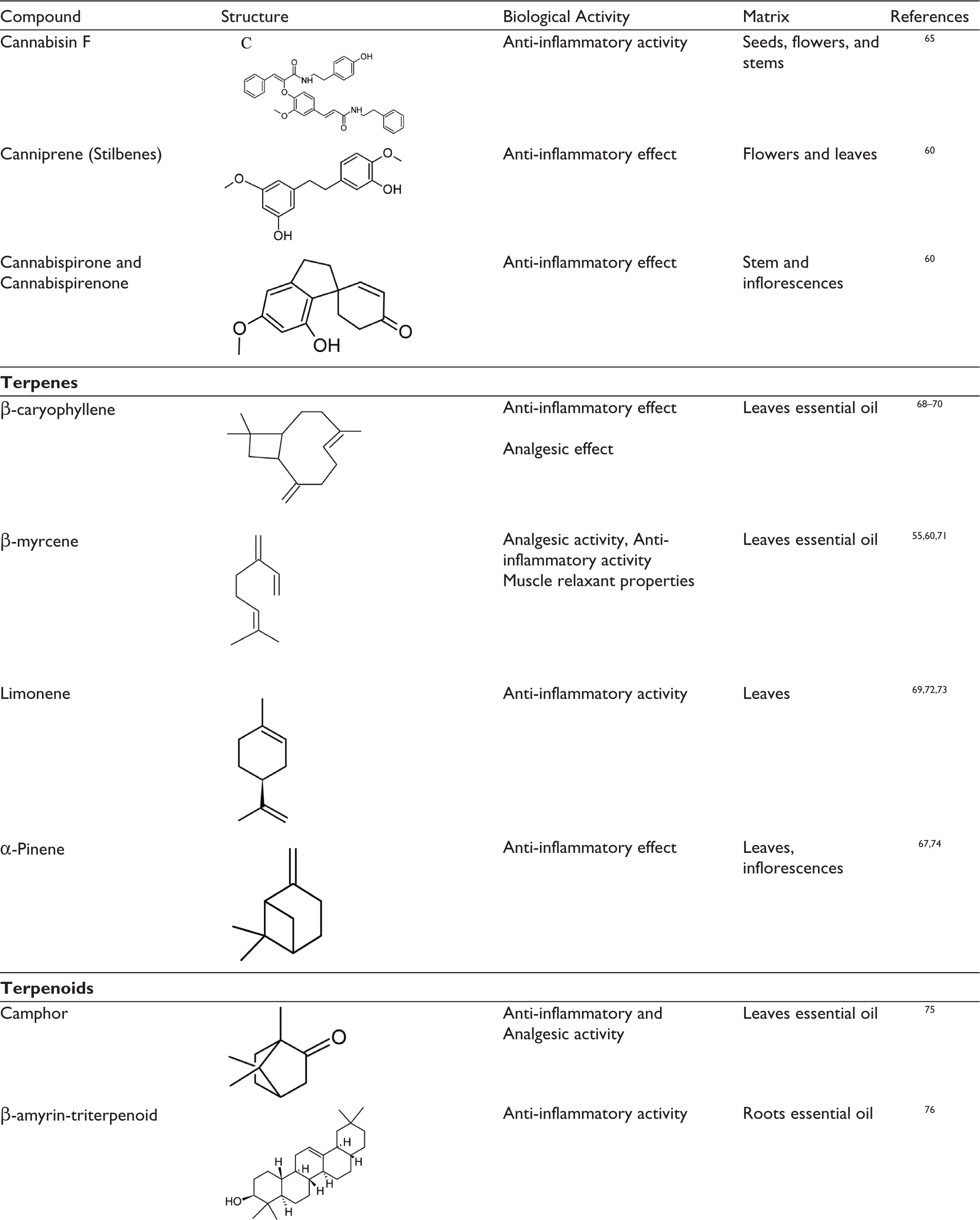
The well-known predominant compounds produced by cannabis are the psycho-stimulant Δ9-THC and a non-psychoactive CBD, followed by a noticeable amount of cannabichromene (CBC), cannabigerol (CBG), and cannabinol (CBN), which are of considerable biological interest.23,30,34,43,48,49 Previous Studies have revealed that these phytocannabinoids are linked to the modulation of the endocannabinoid system, giving feedback responses able to regulate pain, muscle relaxation, etc.50–52 Hemp predominantly produces bioactive compounds in the resin glands with low ∆9-THC effect. 25 These phytocannabinoids have been the focus of studies in the pharmaceutical industry. 34 Table 1 outlines various classes of cannabis compounds reported for muscle relaxant-related properties.
Cannabis sativa Compounds Reported for Muscle Relaxant-related Properties.
Endocannabinoid System
The biological effects of phytocannabinoids in the human body are thought to be mediated through interactions with cannabinoid receptors.78,79 The endocannabinoid system consists of the major known cannabinoid receptors, namely CB1 and CB2, categorized within the membrane protein family G-protein-coupled receptors (GPCR).43,60,80 A third G-protein-coupled cannabinoid receptor 55 was later suggested as CB3, expressed in the brain.60,81,82 Cannabinoid receptors are distributed in different tissues and organs in the human body.83,84 The CB1 is a central receptor that is dominant in the brain and spinal cord, while CB2 is a peripheral receptor located in immune cells and peripheral organs.83,85 This system also has endogenous cannabinoid ligands known as anandamide AEA and 2-arachidonoylglycerol 2-AG produced by the body in various tissues and organs on demand.86,87 They are the agonists for CB1 and CB2 receptors, with higher binding affinity to CB1 receptors.52,88 Phytocannabinoids can mimic endogenous ligands of the Endocannabinoid system. 60 Just like endogenous cannabinoids anandamide and 2-arachidonoylglycerol released in postsynaptic neurons, phytocannabinoids (∆9-THC) interact with and cause activation of G protein-coupled cannabinoid receptors CB1 and CB2.79,89 At the presynaptic neuron (Figure 3), the binding of phytocannabinoids and G protein-coupled cannabinoid receptors CB1 leads to the activation of the CB1 receptor in neuronal tissue. Secondary to the activation of CB1 receptors is the inhibition of neurotransmitter release and signaling pathways, cAMP, and MAPK involved in inflammation. 89 Activation of the CB2 receptor suppresses pro-inflammatory cytokines and immune cell response. 90
Mechanism of Action for the Endocannabinoid System and Phytocannabinoids. cAMP = Cyclic Adenosine Monophosphate; AC = Adenylate Cyclase; ATP = Adenosine Triphosphate; MAPK = Mitogen-activated Protein Kinase.
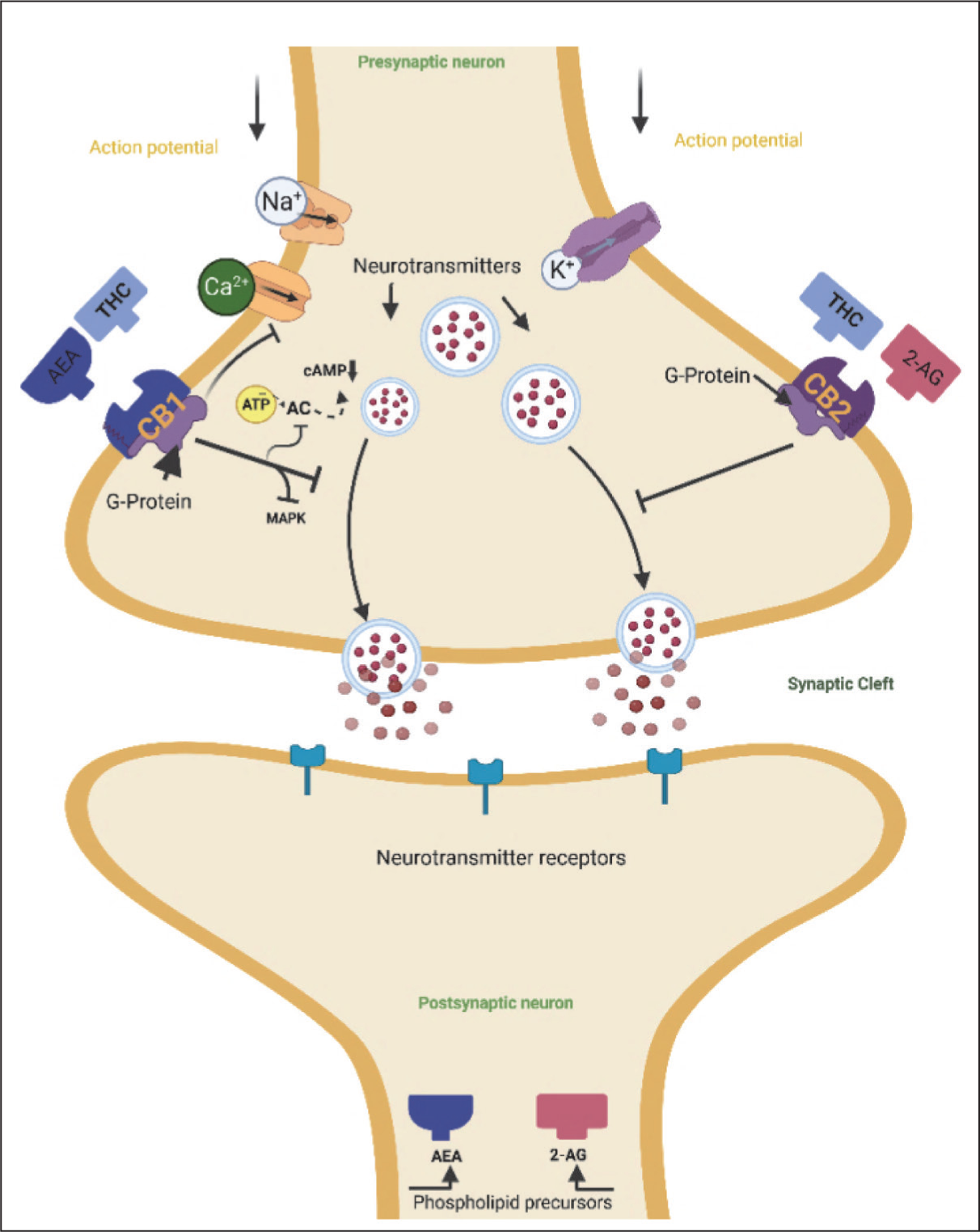
Previous studies have revealed that ∆9-THC and CBD are able to interact with the CB1 and CB2 receptors.50,91 Δ9-THC compound has an equal binding affinity and is an agonist to both CB1 and CB2 receptors,52,60,78 making the ∆9-THC compound psychoactive and analgesic. CBD binds with the CB2 receptor as an inverse agonist and other receptors related to the immune system. 89
SAR of Notable Cannabis Compounds with Muscle Relaxant-related Properties
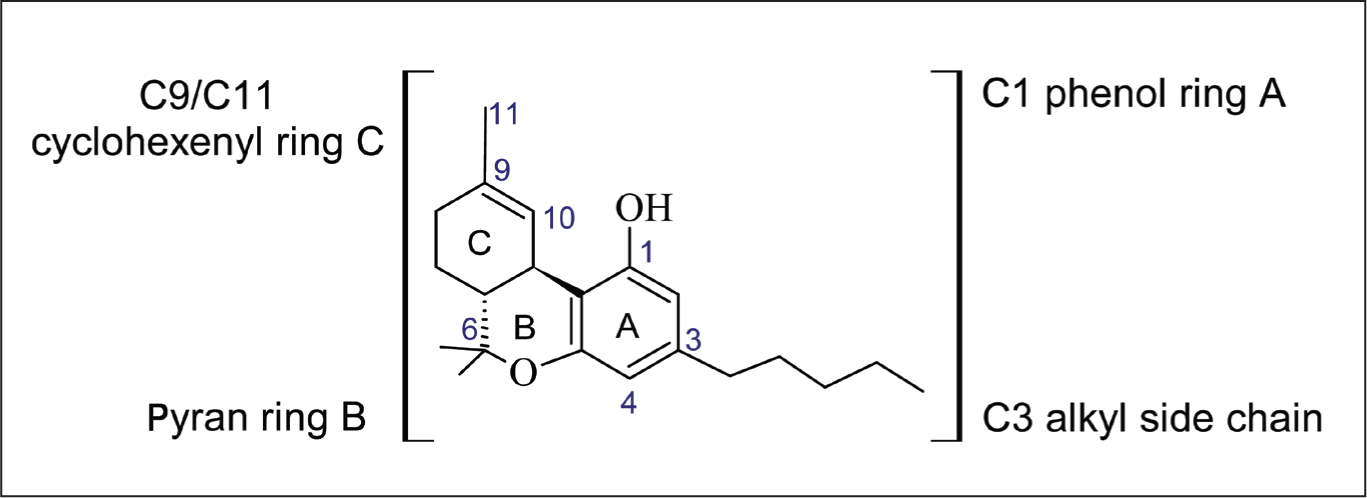
The SAR of phytocannabinoids showing a potential effect in treating musculoskeletal-related disorders is discussed. Previous SAR studies on isolated phytocannabinoids involved modification of the C3 alkyl side chain on the resorcinyl moiety.62–64,92 The length of the C3 side chain of THC directly correlates with CB1 and CB2 binding affinity 92 (Figure 4).
∆9-THC Major Structural Features: C3 Side Chain, C1 Phenolic Hydroxyl Ring A, Pyran Ring B, and C9/C11 Cyclohexenyl Ring C.
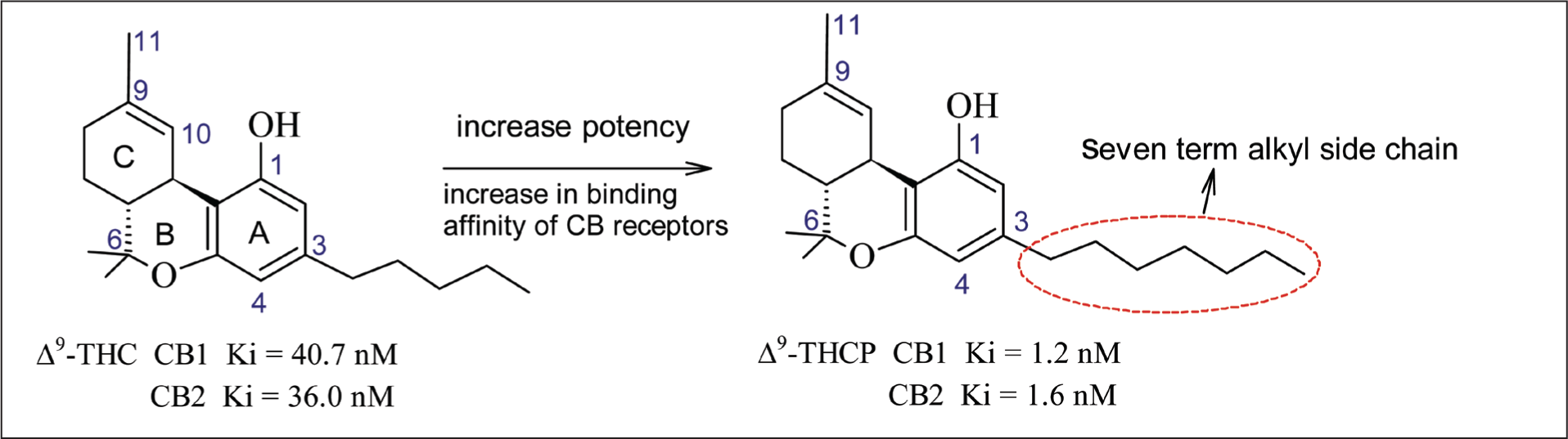
Citti et al. conducted an in vivo hot plate test of a newly isolated cannabinoid compound with an extended side chain, ∆9-Tetrahydrocannabiphorol (∆9-THCP) in mice. At a dose of 10–5 mg/kg, ∆9-THCP showed a significant antinociceptive effect. This newly identified compound is a homolog of ∆9-THC with a longer length of the C3 alkyl side chain (seven-carbon alkyl side chain). 62 Studies have shown that the length of the alkyl side chain on the resorcinyl moiety has proven to be the key pharmacophoric driving force for the biological activity exerted by Δ9-THC.52,92 (-)-trans-Δ9-THCP binds with higher affinity to the human CB1 and CB2 receptors with a Ki of 1.2 and 6.2 nM, respectively, proving that exhibited approximately 33-fold higher binding affinity to CB1 receptors compared to Δ9-THC (Ki = 40 nM) against the CB1 receptor and 10-times more active against the CB2 receptor (Ki = 36 nM) consequently a full agonist of CB1 and CB2 receptors 62 (Figure 5).
Structure-activity Relationship of Two Phytocannabinoids, Δ9-THC and Δ9-THCP.
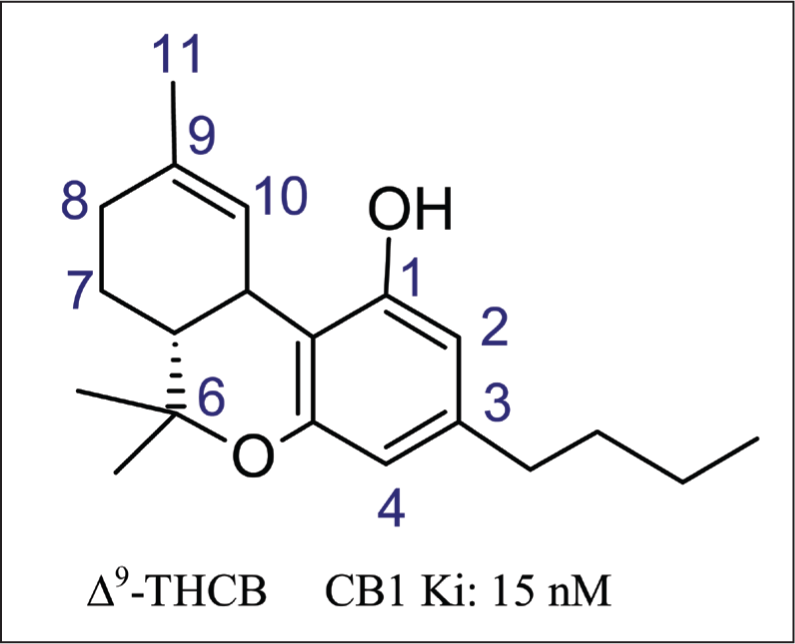
Linciano et al. reported a successful isolation of ∆9-tetrahydrocannabutol (∆9-THCB), a phytocannabinoid compound with similar structural features to the famous ∆9-THC. This compound has a butyl side chain in the resorcinyl moiety and is suggested to be a partial agonist for the CB1 receptor. 64 ∆9-THCB showed analgesic effect in mice at 3 mg/kg by reducing the first and second phases of the formalin test.
Linciano and co-workers identified yet another phytocannabinoid compound, cannabidihexol (CBDH), a CBD homolog with different lengths of the alkyl side chain. 63 Here, the analgesic effect of the CBDH compound was evaluated using the formalin test in mice. The result showed that at doses of 1 and 2 mg/kg, CBDH significantly reduced the late-phase formalin-induced nocifensive behavior, showing interesting analgesic activity. 63 An increase in C-(3) side chain length by the addition of a methyl group was attributed to the observable increase in its affinity and binding ability to CB2 receptors. 52 This CBD homolog is likely to demonstrate relatively low affinity to the CB1 cannabinoid receptors along with low psychoactivity. 93 CBDH is speculated to possibly activate receptors facilitating pain and nociception, such as TRPV1 or other channels, at doses of 1 and 2 mg/kg. 63
Conclusions and Future Perspectives
This study highlights the potential use of C. sativa as an agent for the management of chronic pain associated with MSCs. The rich in phytocannabinoids, terpenes, terpenoids, and phenolics from cannabis exhibiting muscle relaxant-related properties (anti-inflammatory, analgesic, and anti-arthritis activities) have been discussed. The study brings to the fore the muscle relaxant potential of Δ9-THCP, ∆9-THCB, and CBDH. Based on SAR studies, modification of the C3 alkyl side chain on the resorcinyl moiety of the phytocannabinoids, as demonstrated in the conversion of Δ9-THC to Δ9-THCP, has been shown to enhance the binding affinities to the cannabinoid receptors, CB1 and CB2, involved in musculoskeletal pain.
Future research should consider modifying the cannabinoid backbone for more bioactive derivatives. It would be worthwhile to obtain toxicological data and determine the analgesic, anti-inflammatory, and anti-arthritis activities of cannabis extracts, their putative compounds, including Δ9-THCP, ∆9-THCB, and CBDH, as well as their derivatives, in pre-clinical and clinical settings.
Footnotes
Acknowledgements
The authors are grateful to the Research Directorates of Walter Sisulu University (African Medicinal Flora and Fauna Niche Area and PDRFs) and NRF Rated Researcher Incentive for their financial support.
Authors’ Contribution
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be authors as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
Consent for Participate
Not applicable.
Consent to Publication
Not applicable.
Data Availability Statement
All data generated in this study have been incorporated into this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study does not involve experiments on animals or human subjects.
Informed Consent
This review has not involved studies on human participants.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
Use of Artificial Intelligence-assisted Technology
The authors declare that they have not used artificial intelligence (AI)-tools for writing and editing of the manuscript, and no images were manipulated using AI.
