Abstract
Coniferous species represent a valuable source of bioactive phytochemicals, including polyphenols, terpenoids, and alkaloids, with growing interest in their pharmacological potential. Algeria’s unique geography fosters exceptional biodiversity; however, knowledge about its endemic conifers remains limited. This study investigated the phytochemical composition and biological activity of leaf and cone extracts of Abies numidica. High-performance liquid chromatography–mass spectrometry (HPLC–MS) confirmed the presence of multiple phenolic constituents. Antioxidant assays demonstrated moderate activity in both extracts, as evidenced by oxygen radical absorbance capacity (ORAC) and ferric reducing antioxidant power (FRAP) values. Cytotoxicity experiments revealed selective cytotoxic potential: Cone extracts significantly reduced the viability of MCF-7 breast cancer cells but showed lower selectivity toward normal cells, whereas leaf extracts were more effective against the triple-negative cell line MDA-MB-231 at lower concentrations. In silico analyses indicated that the lead compounds complied with Lipinski’s rules, suggesting favorable pharmacological properties. Notably, ellagic acid presented a distinctive metabolic and toxicity profile. Computational predictions revealed its ability to increase the expression of genes related to cell adhesion, genomic stability, and tumor progression, while decreasing the expression of those associated with energy metabolism and inflammatory signaling. Overall, the findings highlight the pharmacological potential of A. numidica and underline the need for further preclinical evaluation of its bioactive compounds.
Introduction
Phytochemicals represent a diverse and abundant class of aromatic, non-nutritive compounds synthesized by plants. They play a crucial role in plant defense mechanisms, allowing resistance to biotic factors such as pathogens and abiotic stressors including ultraviolet radiation. 1 Owing to their conjugated ring structures and hydroxyl groups, phytochemicals function as antioxidants, quenching reactive oxygen species through multiple mechanisms. 2 Beyond their ecological role, these bioactive molecules possess therapeutic potential against degenerative diseases such as hypertension, cardiovascular disorders, diabetes, stroke, and cancer. Their ability to restore redox balance and regulate molecular pathways implicated in chronic pathogenesis further supports their biomedical relevance. 3 The hypothesis that free radicals induce oxidative damage to key biomolecules—including lipids, proteins, and DNA—has gained increasing scientific support.4–6
Coniferous plants, distributed across eight families, 70 genera, and approximately 330 species, constitute a significant global reservoir of bioactive phytochemicals, particularly polyphenols, terpenoids, and alkaloids. Extracts derived from different tree parts—bark, needles, cones, twigs, wood, and berries—have demonstrated a wide range of biological effects, including antioxidant, anti-inflammatory, anticancer, antimicrobial, analgesic, antidiabetic, and anticonvulsant activities. Some extracts have advanced commercial applications, highlighting their pharmacological and economic importance.7,8
Algeria’s unique geographical position fosters remarkable biodiversity, with a high prevalence of endemic species. However, despite this botanical richness, considerable gaps remain in the scientific knowledge of many taxa. One example is the Algerian fir (A. numidica de Lannoy ex Carrière), a conifer native to the Babor Mountains in northern Sétif, covering an estimated 2,300 hectares. Belonging to the Pinaceae family, this species is characterized by its distinct branching pattern with needles tightly arranged around the branches. 9 Traditionally, A. numidica. has been used in Algerian medicine as an anti-inflammatory agent, in the treatment of respiratory disorders, and for topical applications as poultices. However, the scientific literature on this species remains scarce, with most studies focusing on its antimicrobial and antibacterial activities.
The essential oil composition of Algerian fir needles was first described by Tlili Ait Kaki et al. (2012), who identified bornyl acetate, camphene, α-pinene, and β-pinene as the predominant constituents. Additional investigations reported both the chemical composition and antimicrobial potential of extracts obtained from the leaves of Algerian fir.8,10 Furthermore, ethyl acetate fractions demonstrated strong antibacterial and antioxidant properties, 11 while n-butanol extracts from needles exhibited comparable bioactivities. 12 More recent studies expanded this knowledge by analyzing the LC–MS composition and bioactivity of leaf fractions (n-butanol and ethyl acetate). These investigations combined in silico, in vitro, and in vivo approaches, highlighting not only antioxidant and antibacterial potential but also toxicity profiles and anti-tyrosinase activity. 13 Collectively, these findings broaden the understanding of the phytochemical profile of Algerian fir, underscoring its pharmacological promise and the necessity for further exploration of its therapeutic applications.
The present study aimed to investigate the phytochemical composition; hydromethanolic extracts of Algerian fir needles and cones using high-performance liquid chromatography (HPLC) analysis and antioxidant capacity (via oxygen radical absorbance capacity (ORAC) and ferric reducing antioxidant power (FRAP) assays). Furthermore, for the first time, this research explores the cytotoxic effects of these extracts through comprehensive in vitro and in silico analyses. The study also examines ADMET properties (absorption, distribution, metabolism, excretion, and toxicity) and assesses gene expression regulation. By providing the first thorough description of the bioactive properties of Algerian fir, this research underscores the tree’s potential as a natural source of antioxidants and potential anticancer agents, highlighting its significance in the search for novel therapeutic compounds.
Material and Methods
Plant Material
Samples of A. numidica were collected randomly during the month of September 2018 (Autumn season), from the Djebel El Ouehch forest, Constantine, Algeria. Needles and cones were dried for one month, pulverized, and stored in a dry and dark place. Secondary metabolites were extracted by cold maceration with 80% (v/v) methanol-water. Powders were macerated for 24 hours, solvents were evaporated in a rotary evaporator, and crude extracts were stored for later analysis. The leaves and cones were collected and dried (500 g of the leaves and 300 g of cones). The powder taken to extract secondary metabolites was 50 g for each sample. The yield of crude methanolic extract was 6.56% for leaves and 2.3% for cones.
Determination of Total Phenolic Compounds
The Folin–Ciocalteu reagent was used to determine the content of phenolic compounds. 14 An automated microplate reader was used to add 100 µl of Folin–Ciocalteu (1:10) to a volume of 80 µl of Na2CO3 that had been combined with 20 µl of the samples at various concentrations in a 96-well microplate. Following a one-hour dark incubation period, the absorbances were measured. Gallic acid’s calibration curve was applied. Gallic acid equivalent (GAE), or milligrams of phenolic compounds per milligram of extract, was the unit of measurement.
Determination of Individual Polyphenol Content by HPLC
The HPLC analysis was performed on a Shimadzu LC-10A system equipped with an SPD-M10A photodiode array detector. Separation was achieved on a reversed-phase C18 column (Kinetex, 250 × 4.6 mm, 5 µm particle size). The mobile phase consisted of water and acetonitrile acidified with 0.05% (v/v) trifluoroacetic acid, using a gradient elution from 0% to 35% acidified acetonitrile over 35 minutes. The flow rate was set at 1 mL•min⁻ 1 and the injection volume was 50 µL. The compounds were identified using authentic reference standards analyzed under identical chromatographic conditions (gallic acid, caffeic acid, ellagic acid, catechin, cyanidin, delphinidin, and chlorogenic).
Antioxidant Activity
Oxygen Radical-scavenging Capacity (ORAC) Assay
The ORAC assay was conducted as described by Peñalver et al. (2022) using 96-well opaque plates and a Synergy HT microplate reader (Biotek). Fluorescein working solution was prepared by serial dilution in PBS (pH 7.0). AAPH solution was obtained by dissolving 0.216 g AAPH in PBS.
FRAP Assay
The FRAP assay was performed according to Peñalver et al. (2022). The FRAP reagent consisted of 0.3 M acetate buffer (pH 3.6), 2 ml of 10 mM TPTZ, and 2 ml of 20 mmol/L FeCl₃•6H₂O. In a 96-well plate, 20 μL of sample dilutions were combined with 180 μL of FRAP reagent. Blanks contained reagent and water. The plates were incubated at 37 °C for ten minutes, and absorbance at 592 nm was recorded. Trolox was used as a standard, and results were expressed as μmol TE/g dry weight.
Cytotoxic Effect of Methanolic Extract of Leaves (MEL) and Methanolic Extract of Cones (MEC) from Algerian Fir in Cell Lines
Cell Culture
HaCaT keratinocytes, MCF-7 and MDA-MB-231 breast cancer cells, and SW480 and HT-29 colon adenocarcinoma cells were obtained from ATCC (Manassas, VA, US). Cells were cultured in DMEM (Gibco, US) supplemented with 10% FBS (Gibco) and 1% penicillin/streptomycin (Gibco) and maintained at 37°C in a humidified 5% CO₂ atmosphere. For experiments, 1 × 10⁴ cells/100 µL were seeded in 96-well plates in triplicate and incubated for 24 hours under standard conditions.
Cytotoxicity Assay
The cytotoxic activity was carried in HaCaT, MCF-7, MDA-MB-231, SW480, and HT-29 adherent cells treated separately with 500, 1,000, 1,500, 2,000, and 2,500 µg/ml of MEL and MEC extracts during 48 hours of exposure. Previous assessments carried out at lower concentrations did not show a significant effect on the variables analyzed; consequently, these results were not included for publication. 10 μL of MTT (0.5 mg/ml) were added, cultures were re-incubated at 37°C for 2 hours in darkness. Finally, their absorbance was determined at 570 nm in a multi-plate Varioskan reader (ThermoScientific). The C50 was calculated using GraphPad Prism version 8.0 (GraphPad Software, San Diego, CA, USA). Selectivity index (SI) was calculated by the ratio of IC50 values 48 hours in non-malignant HaCaT cells to IC50 of each cell line. All experiments were performed in triplicate.
Statistical Analysis
All data were represented as mean ± standard deviation (SD) or mean ± standard error (SE) from three separate experiments. Non-linear regression analysis was employed to determine IC50 values. ANOVA analysis was conducted using GraphPad Prism version 8.0 (GraphPad Software, San Diego, CA, USA) to identify statistical differences between treated and untreated control groups, and subsequent Tukey tests were employed to pinpoint specific differences.
In Silico Study
Determination of Canonical SMILES of Molecules
The PubChem web server was used to determine the canonical SMILES of each compound (accessed May 9, 2024). To predict the possible biological activities of molecules found in major and identified hydromethanolic extracts obtained from A. numidica needles and cones. These compounds were gallic acid, caffeic acid, ellagic acid, catechin, cyanidin, delphinidin, and chlorogenic acid.
Physicochemical Properties
Two-dimensional structures were generated with ChemDraw Professional 16.0. Physicochemical properties (size, polarity, lipophilicity, insolubility, flexibility, unsaturation) were predicted using SwissADME (accessed May 9, 2024). Criteria for drug-likeness included logP ≤ 5, high bioavailability, molecular flexibility, and polar surface area (PSA) < 140 Å, consistent with oral drug requirements. 15
Pharmacokinetics and ADMET Properties
ADMET profiles were predicted using admetSAR 2.0. 16 Parameters included human intestinal absorption (HIA), CaCO₂ permeability, BBB penetration, oral bioavailability (HOB), P-glycoprotein status, subcellular localization, CYP interactions, skin/eye toxicity, carcinogenicity, Ames mutagenesis, hepatotoxicity, respiratory, mitochondrial and nephrotoxicity, and acute oral toxicity (AOT).
Cytotoxicity of the Compound’s Prediction
Cytotoxicity of compounds from A. numidica hydro-methanolic extracts was predicted using CLC-Pred (
Gene Expression Profiles
In silico prediction of gene expression modulation by four compounds was conducted using DIGEP Pred (
Results
Phenolic Compounds and Antioxidant Ability
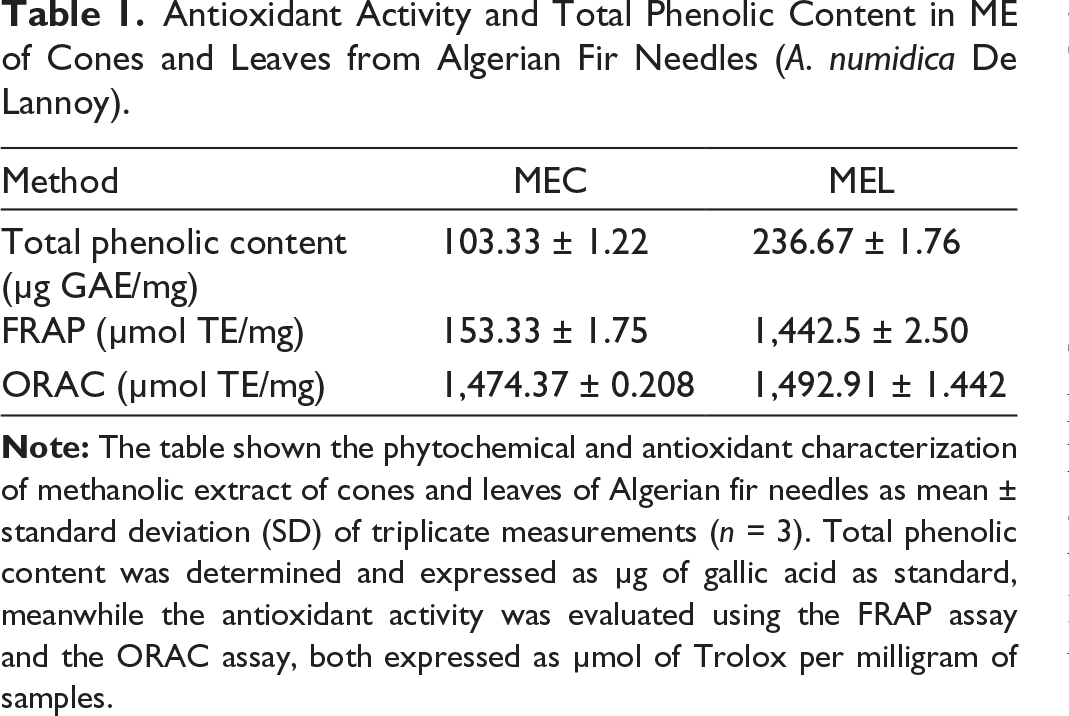
The present analysis assessed phenolic compounds and antioxidant activity of hydromethanolic extracts from cones and leaves of Algerian fir (Table 1). MEL contained the highest phenolic content (236.67 ± 1.76 μg GAE/mg), followed by MEC with 103.33 ± 1.22 μg GAE/mg. These results corroborate Benouchenne, who reported high phenolic (490.76 ± 6.65 mg GAE/g, 381.15 ± 22.70 μg GAE/ml) and flavonoid contents (49.79 ± 2.81 μg QE/ml, 143.19 ± 5.59 mg QE/g extract) in Algerian fir fractions. Similar findings were noted by Gupta et al. 18 for A. pindraw leaves, rich in polyphenols and flavonoids. Variability in metabolite composition depends on extraction solvent, climate, environmental factors, and soil nutrients.
Antioxidant Activity and Total Phenolic Content in ME of Cones and Leaves from Algerian Fir Needles (A. numidica De Lannoy).
HPLC Analysis
This study is the first to report the tannin composition of A. numidica MEL and MEC. HPLC analysis identified three phenolic compounds: cyanidin, delphinidin, and ellagic acid (EA) (Table 2). MEL contained cyanidin, delphinidin, and EA, whereas MEC included only cyanidin and delphinidin. Delphinidin was the predominant compound, with higher levels in MEC, followed by cyanidin, also more abundant in MEC. EA appeared exclusively in MEL. Other phenolics, such as gallic acid, caffeic acid, catechin, and chlorogenic acid, were absent. These results reveal distinct polyphenolic profiles between extracts, with MEC enriched in delphinidin and cyanidin.
LC–MS Analysis of Phenolic Compound Composition in Hydromethanolic Extracts from Algerian Fir (A. numidica) Leaves and Cones.
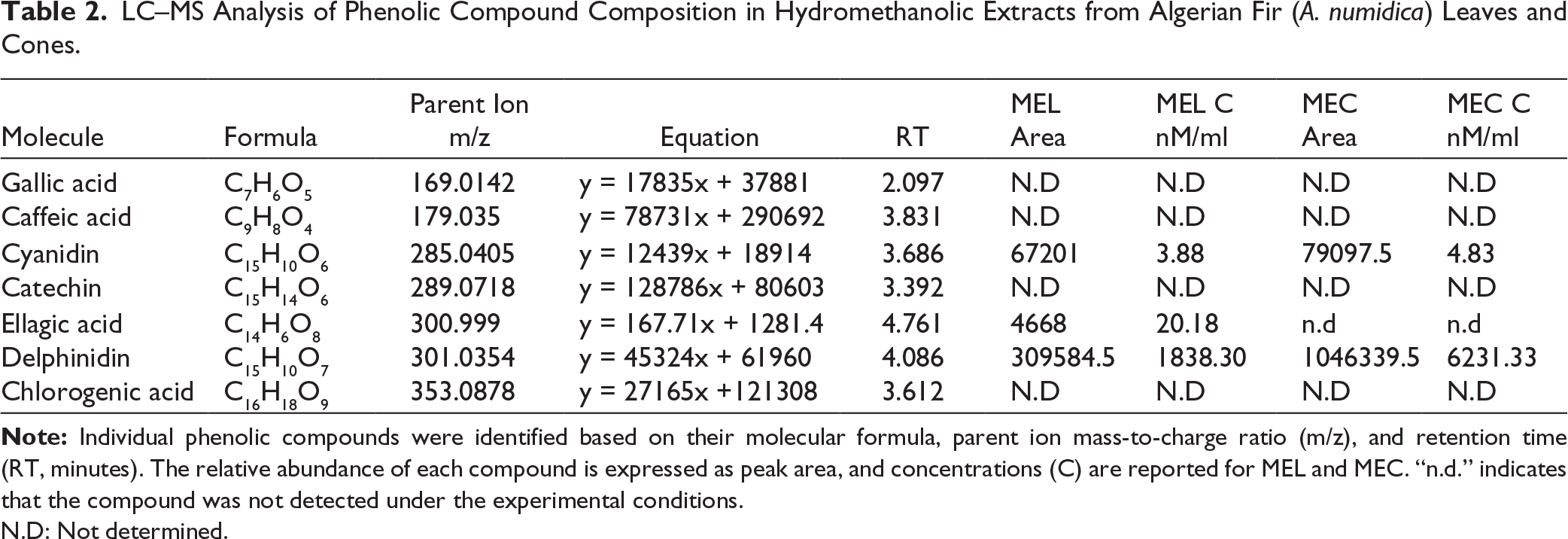
N.D: Not determined.
Studies indicate that the antioxidant properties of tannins depend on their polymerization, with radical-scavenging and metal-chelating capacity linked to hydroxyl groups and ring substituents. 19 Procyanidins exhibit even stronger activity, neutralizing free radicals 15–30 times more effectively than simple phenolics. 20 In A. numidica, Benouchenne et al. reported that the ethyl acetate fraction of needles was enriched in phenolic and flavonoid compounds, with LC–MS confirming high levels of quercetrin, hyperoside, and astragalin. Similarly, the n-butanol fraction also contained significant amounts of flavonoids and phenolics.
Cytotoxic Effect Extracts from Algerian Fir Leaves and Cones in Cancer Cell
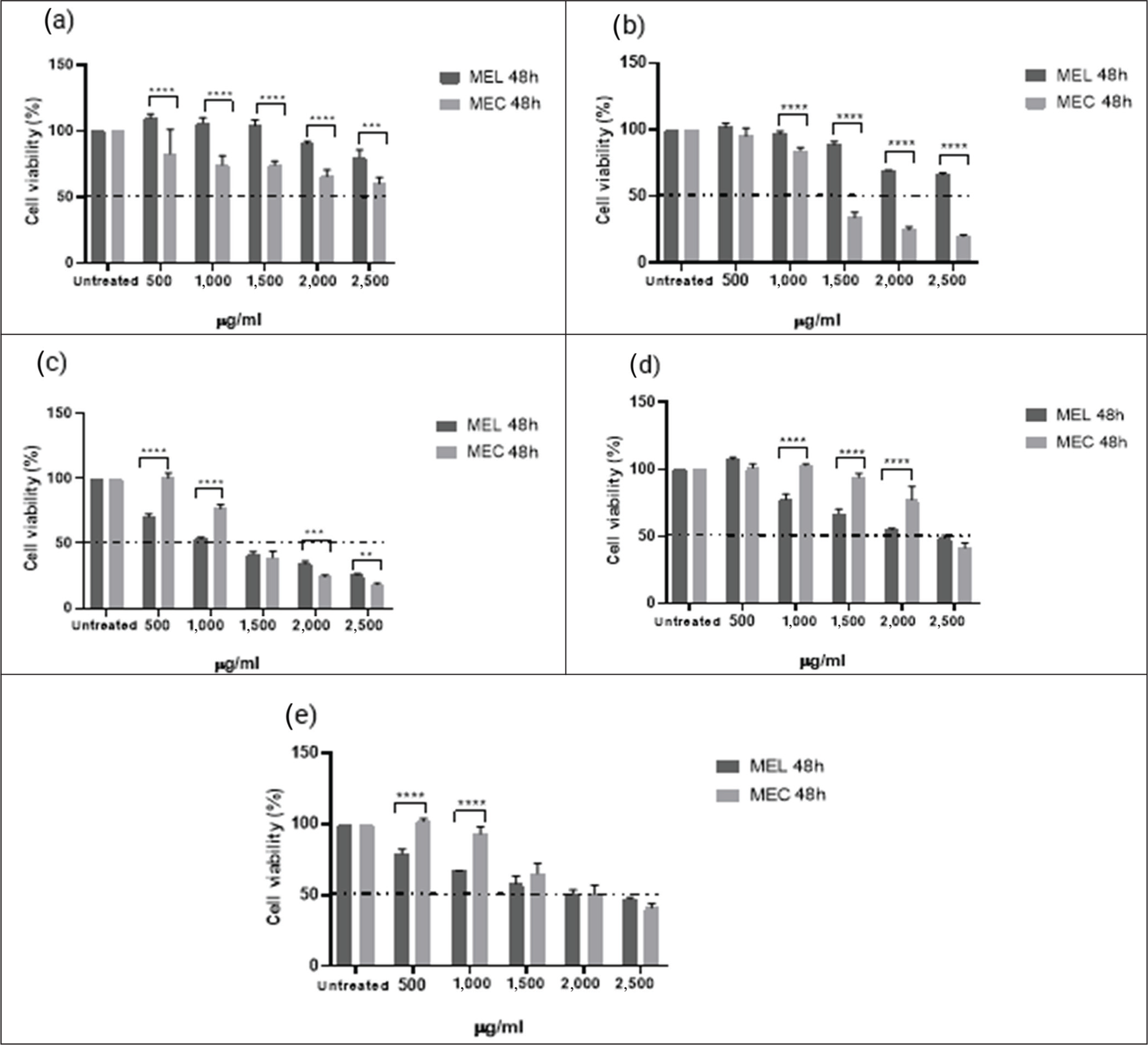
The MTT assay (48 hours, 500–2,500 µg/mL) evaluated MEL and MEC cytotoxicity in four cancer cell lines and HaCaT keratinocytes (Figure 1). The results obtained display a differential response to the evaluated extracts, varying on the cell type. In HaCaT keratinocytes, both extracts showed low cytotoxicity, particularly the MEL, suggesting a lower toxicity profile in non-tumor cells and supporting its potential safety margin.
Effect of MEL and MEC Pine Extracts on Cell Viability on Different Cell Lines Treated for 48 Hours. (a) HaCaT Human Epidermal Keratinocytes. (b) MCF7 Human Breast Cancer. (c) MDA-MB-231 Triple-negative Breast Cancer Cell (d) SW480 Colon Adenocarcinoma and (e) HT29 Colorectal Adenocarcinoma. Untreated Cells were Used as a Control Group. Values Represent Averages ± SD (Standard Deviation) of Triplicate Measurements (n = 3). Asterisks Represent Significant Differences by Tukey-Kramer test (p < .05) Between Extracts for Each Concentration.
The IC₅₀ values estimated after 48 hours of treatment denote that the pine extracts MEC and MEL exhibit cell type-dependent cytotoxic activity, with variations in potency and selectivity among the cell lines evaluated. In the normal HaCaT cell line, the MEL showed a higher IC₅₀ value than MEC, suggesting lower cytotoxicity against non-tumor cells and allowing its use as a reference for calculating the SI. In the MCF7 cell line, the MEC compared to MEL showed a lower IC₅₀ value, indicating bigger potency; however, the selectivity indices obtained for both extracts were similar. In the MDA-MB-231 cell line, the MEL showed lower IC₅₀ values and a higher SI compared to MEC, suggesting greater sensitivity of this cell line to MEL treatment. In the SW480 and HT29 colorectal cancer (CRC) cell lines, the MEL showed higher selectivity indices, while IC₅₀ values were comparable between both extracts and slightly lower for MEL. Taken together, these results suggest that both extracts possess cytotoxic activity in vitro, and that the MEL tends to exhibit lower cytotoxicity in normal cells and a more favorable selectivity profile in certain tumor cell lines.
In MCF-7 breast cancer cells, for both extracts a reasonable cytotoxic activity was observed, with distinguished potency for the MEC compared to MEL, while selectivity was equivalent between the two treatments. Considering that the MCF-7 cell line corresponds to a luminal subtype characterized by the expression of hormone receptors specifically ER and PR, these differences could be related to the hormone-dependent nature of this cell model, 21 in which the variation between extracts is mainly shown at the level of potency rather than selectivity.
On the other hand, MDA-MB-231 triple-negative breast cancer cells showed the greatest sensitivity to the A. numidica extracts, exhibiting a favorable response to both treatments. In particular, the MEL exhibited greater cytotoxic potency and a significantly higher SI, which is especially relevant considering the highly aggressive nature of this tumor subtype and the lack of effective targeted therapies. 22 This differential behavior suggests that the extracts could interact more efficiently with the predominant molecular pathways in triple-negative breast cancer cells.
EA exhibited anti-inflammatory activity and regulated signaling in TNF-α/IFN-γ–stimulated HaCaT, mitigating UVB-induced toxicity. 23 Thus, MEL, through its ellagic acid content, may offer therapeutic potential for UV-related skin disorders such as atopic dermatitis.
Ellagic acid exhibits anti-inflammatory and photoprotective effects in human epidermal keratinocytes (HECs). It modulates TNF-α/IFN-γ–induced inflammatory pathways, 24 attenuates UVB-induced toxicity by preventing collagen degradation and reducing inflammation, 25 and protects against oxidative stress by decreasing reactive oxygen species production and DNA damage. 26 Considering this report and findings we suggest a potential contribution of ellagic acid present in the MEL to the observed biological effects; though, the results should be preliminary. While MEL exhibited a higher selectivity index (SI) compared to MEC, showing greater selectivity toward cancer cells with minor effects on normal cells, further studies involving fractionation, compound isolation, and bioavailability assessment are required before any therapeutic implications, including applications related to inflammatory skin conditions, can be established. (Table 3; Figure 2; Supplementary data).
IC50 Values Estimated by Linear Regression of the Cytotoxic Effect and SI of Pine Extract MEC and MEL for 48 Hours of Treatment on Different Cell Lines.
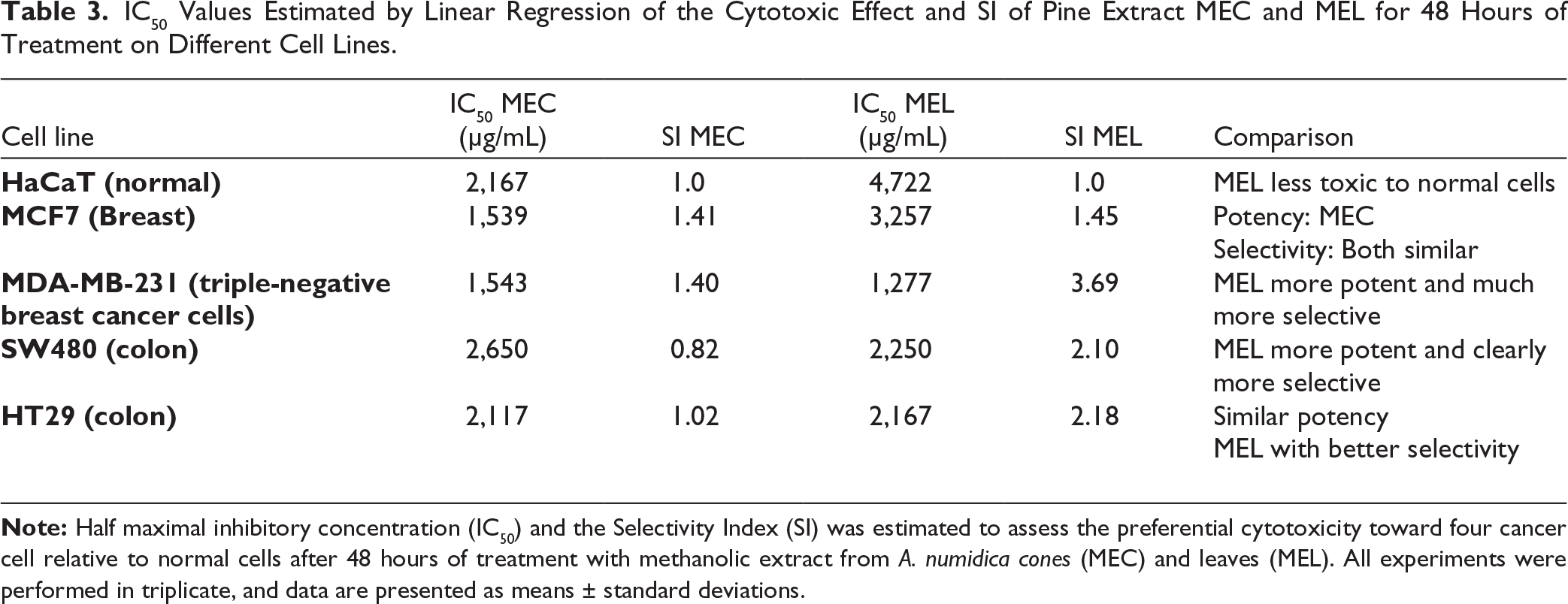
In MCF-7 breast carcinoma cells, MEL and MEC exhibited distinct efficacy profiles. MEL significantly reduced cell viability at 1,500 µg/mL in a dose-dependent manner, though higher concentrations were required compared to MEC, this showed stronger inhibition, with an IC₅₀ of 1.539 µg/mL versus 3.257 µg/mL for MEL. Despite this, MEL had a slightly higher selectivity index (SI = 1.45) than MEC (SI = 1.41), suggesting superior selectivity toward cancer cells over normal cells. The activity of MEL may be attributed to its phenolic content, particularly cyanidin, delphinidin, and EA. Cyanidin reduces glioma stem cell viability via Wnt signaling modulation, while delphinidin induces G2/M arrest in breast cancer cells by inhibiting cyclin B1 and Cdk1 and promoting mitochondrial apoptosis through ERK, NF-κB, and JNK regulation. 27 Collectively, these findings highlight the selective antiproliferative potential of MEL against MCF-7 cells.
In MDA-MB-231 triple-negative breast carcinoma cells, MEL and MEC extracts exhibited distinct antiproliferative activity. MEL showed superior efficacy with an IC₅₀ of 1.277 µg/mL compared to 1.543 µg/mL for MEC. Notably, demonstrated a markedly higher SI (SI = 3.70) than MEC (SI = 1.40), indicating enhanced specificity toward cancer cells while sparing normal cells. Although MEC required higher concentrations, its inhibitory capacity increased significantly at elevated doses, surpassing MEL efficacy at the highest concentrations tested.
Canonical SMILES Determination
The molecular characterization of the identified compounds in the methanolic extract of A. numidica provides valuable insights into their structural and chemical properties. The molecular formula and canonical SMILES of cyanidin (C₁₅H₁₁O₆⁺), ellagic acid (C₁₄H₆O₈), and delphinidin (C15H11O7) were determined using PubChem, as summarized in Table 4.
Canonical SMILES, Molecular Formula, and Structure Determination of Molecules Identified in Methanolic Extract Using PubChem.
Cyanidin and delphinidin, anthocyanidins with multiple hydroxyl (-OH) groups, possess strong antioxidant potential by scavenging free radicals, donating electrons, and chelating metal ions, thereby stabilizing reactive oxygen species. EA also exhibits potent antioxidant activity due to its conjugated system and abundant hydroxyl groups. These structural features underscore the bioactivity of these molecules, particularly in oxidative stress-related disorders. Their presence aligns with previous reports on flavonoid-rich Abies species.
Prediction of Physicochemical Properties of Molecules Using Lipinski Rules
Lipinski’s Rule of 5 defines criteria for drug-likeness: molecular weight ≤500 Da, ≤5 hydrogen bond donors, ≤10 acceptors, and log P slightly above 5, favoring high bioavailability. 28 As shown in Table 5, most examined molecules, particularly from ethyl acetate and n-butanol fractions, these parameters with molecular weights between 154 Da and 448 Da. Exceptions were hesperidin and rutin (610 Da), exceeding the recommended limit, thus potentially reducing their suitability as drug-like candidates. 29
Physicochemical Properties of Three Phenolic Compounds Determined by HPLC in MEL and MEC Hydromethanolic Extracts from Leaves.

All tested compounds showed logP <5, confirming hydrophilicity. Delphinidin exhibited highest solubility with very low logP. All maintained molecular weight <500 Da and <10 acceptors; however, delphinidin exceeded five hydrogen donors, while cyanidin presented exactly five.
Prediction of Cytotoxicity
Cytotoxicity of phenolic compounds from A. numidica MEL and MEC were evaluated using the CLC-Pred server (Table 6). EA was the only molecule predicted to exert cytotoxicity, specifically against the Hs 683 oligodendroglioma cell line, suggesting selective anticancer activity in gliomas, consistent with prior reports. Conversely, cyanidin and delphinidin showed no predicted cytotoxicity, underscoring the need for further experimental validation of their potential anticancer properties.
In Silico Cytotoxic Effect Prediction of Phenolic Compounds Determined in MEL and MEC from Algerian Fir.

Ellagic acid shows anticancer activity via oxidative damage, membrane alteration, and nucleic acid disruption. 30 It reduces glioblastoma proliferation, downregulates anti-apoptotic proteins, and induces apoptosis through S-phase arrest, supporting its therapeutic potential against glioma and other cancers. 31 In silico predictions of cytotoxicity and changes in gene expression provide valued preliminary information about potential biological effects, however, these predictions are fundamentally limited by the assumptions of the computational models and training dataset.
Therefore, predicted gene related cytotoxic effects should be interpreted with caution and considered hypothesis-generating rather than definitive evidence. Experimental confirmation with in vitro or in vivo studies is thus necessary to approve the biological relevance of these findings
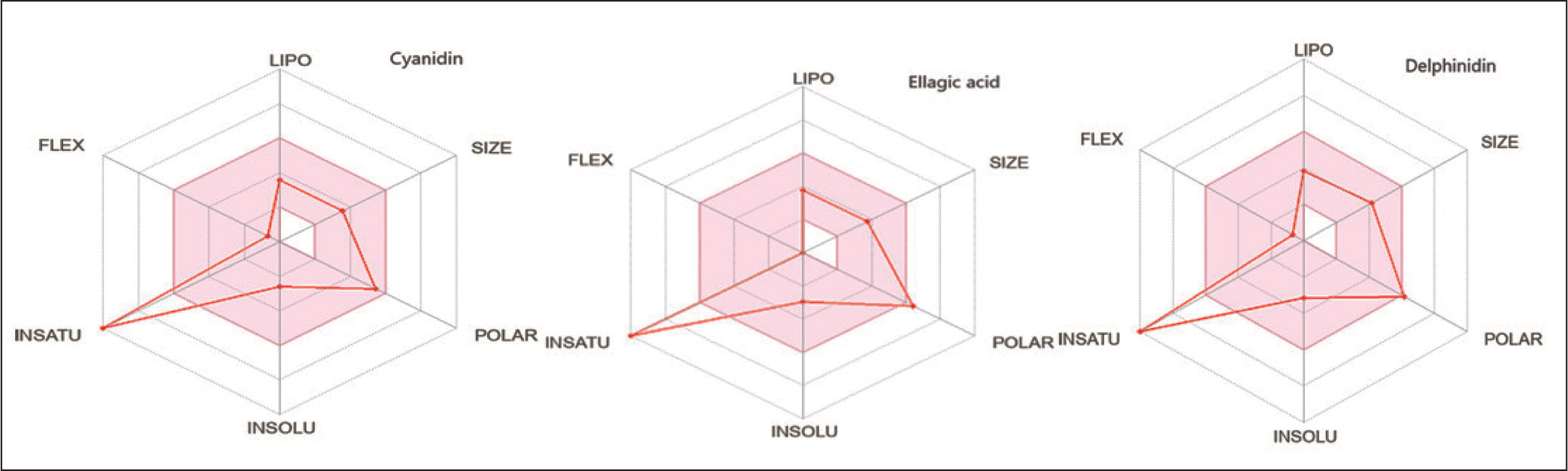
Figure 2 illustrates the physicochemical properties of the identified compounds, analyzed through the SwissADME web server. Six key parameters were evaluated: size, lipophilicity (LIPO), insolubility (INSOLU), unsaturation (INSATU), flexibility (FLEX), and polarity (POLAR). These characteristics strongly influence pharmacokinetics, bioavailability, and therapeutic potential. Polarity emerged as the most distinctive factor: Ellagic acid showed the highest polarity, favoring interactions with hydrophilic biomolecules and suggesting stronger therapeutic potential. Delphinidin displayed moderate polarity, whereas cyanidin was the least polar, implying higher lipophilicity and potentially enhanced membrane absorption. All compounds exhibited high unsaturation, consistent with antioxidant properties derived from conjugated double bonds. Cyanidin and delphinidin demonstrated greater molecular flexibility due to higher numbers of rotatable bonds, which may improve adaptability to biological targets, while EA remained rigid because of its polycyclic structure. Lipophilicity values were moderate and similar across compounds, highlighting the importance of achieving a balance between hydrophilicity and lipophilicity for optimal absorption. Insolubility analysis confirmed poor aqueous solubility in all compounds, consistent with polyphenols’ hydrophobic aromatic rings. 32
ADMET Pharmacokinetic Properties of Selected Molecules
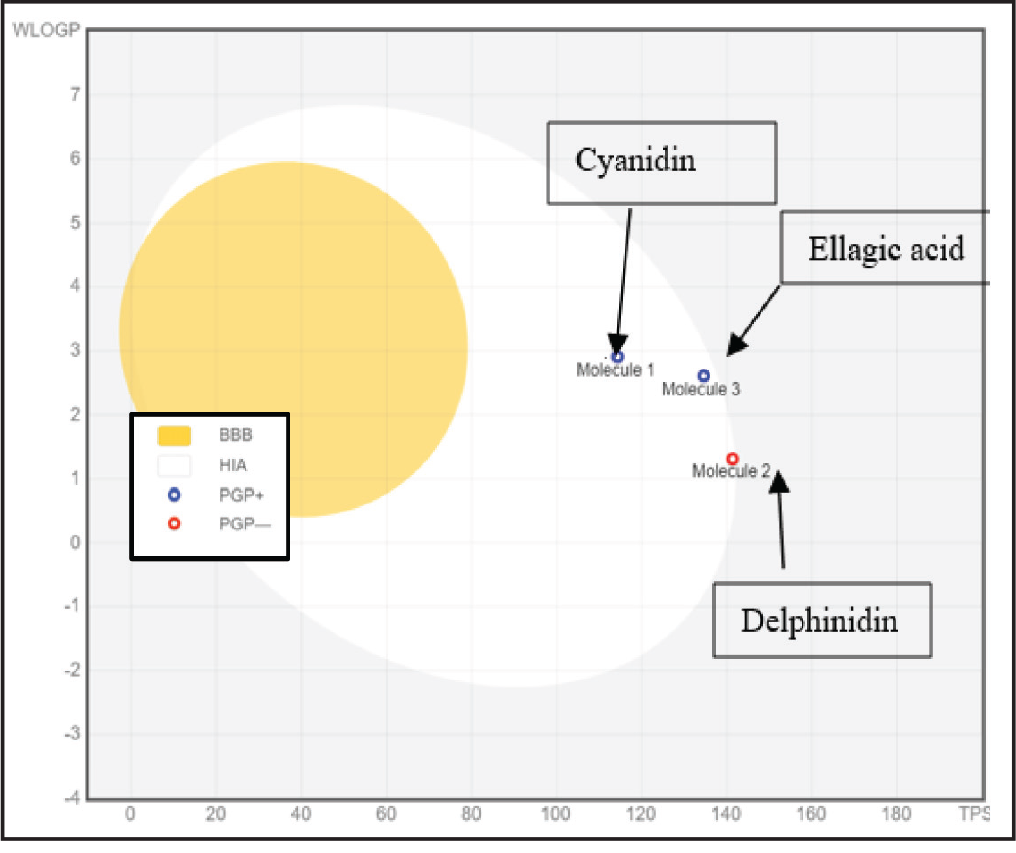
Table S7 (supplementary data) presents the Prediction of the ADMET properties and toxicity of the three tested compounds. Figure 3 shows absorption predictions: Cyanidin and delphinidin displayed positive intestinal absorption (HIA+), supporting favorable oral bioavailability. 33 EA showed negative absorption (HIA−), attributed to its polarity and rigidity. 34 None crossed the BBB, and only cyanidin/delphinidin exhibited positive Caco-2 permeability, reinforcing oral suitability.
Prediction of Absorption and Brain Penetration of Molecules Determined in MEL and MEC Using Swiss ADME Server. The Yellow Region Indicates the Physicochemical Space Associated with Blood–brain Barrier (BBB) Permeation, While the White region Corresponds to Compounds Predicted to Have High Human Intestinal Absorption (HIA), Associated Whether the Compounds are Predicted to be Substrates (PGP+) or Non-substrates/inhibitors (PGP–) of P-glycoprotein.
Distribution parameters describe a drug’s ability to cross membranes and reach tissues. Subcellular localization analysis revealed that cyanidin and delphinidin were predominantly found in the nucleus, whereas ellagic acid was localized in the mitochondria. Nuclear localization suggests that cyanidin and delphinidin may directly interact with DNA or nuclear proteins, potentially regulating gene expression, DNA repair, or apoptosis, mechanisms of high relevance in anticancer activity. 35 Conversely, ellagic acid’s mitochondrial localization indicates a role in cellular respiration, ROS regulation, and modulation of mitochondrial membrane potential, supporting involvement in intrinsic cell death pathways. 33
Metabolism determines drug stability, transformation, and elimination. 36 Cyanidin and delphinidin inhibited CYP3A4 and CYP1A2, enzymes essential for xenobiotic metabolism. CYP3A4 inhibition, relevant for ~50% of pharmaceuticals, may elevate drug plasma levels, altering pharmacokinetics and toxicity, while CYP1A2 inhibition could affect metabolism of aromatic compounds such as caffeine and theophylline. 37 EA showed no major CYP inhibition, suggesting alternative biotransformation via glucuronidation or sulfation, 38 consistent with its mitochondrial role in oxidative stress regulation. 39
Elimination is critical for drug clearance, all compounds inhibited hepatic OATP1B1 and OATP1B3, potentially prolonging systemic circulation and increasing drug–drug interaction risks. 40 Cyanidin additionally inhibited OATP2B1, affecting intestinal absorption and hepatic elimination. These inhibitions could impair biliary clearance, elevate plasma concentrations, and exacerbate adverse effects. 41 No renal transporter interactions were found, suggesting hepatic elimination predominance. Further studies should assess compensatory urinary excretion and long-term toxicity
Gene Expression Profiles
Table 7 shows that ellagic acid modulates mRNA expression of genes linked to tumor progression, DNA repair, cell adhesion, and metabolism. Overexpression of TCF12, IGFBP2, BLM, CEP72, and ITGA6 suggests effects on proliferation and genomic stability. These findings align with in vitro evidence, where A. numidica methanolic extracts, particularly MEC, reduced cancer cell viability, albeit with limited selectivity toward normal cells.
mRNA Based Prediction Results Probable Effects of the Ellagic Acid on the Expression of Some Genes Related to Cancer.
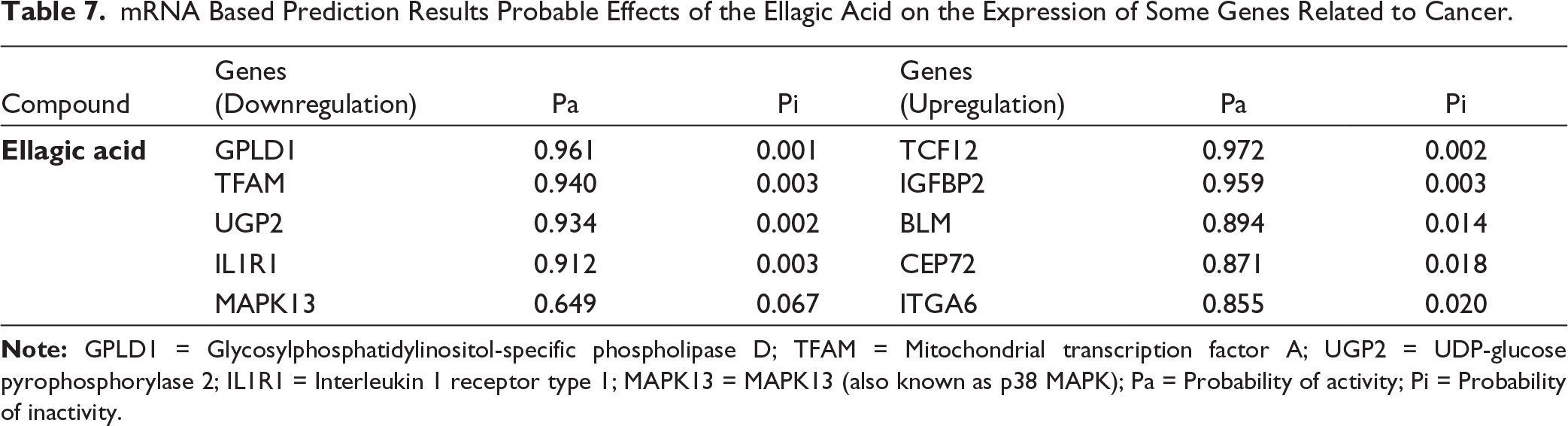
Increased expression of ITGA6, an integrin implicated in adhesion, stemness, metastasis, angiogenesis, and drug resistance, suggests that ellagic acid may modulate tumor–microenvironment interactions, possibly explaining differential responses to extracts. The lower selectivity of MEC toward normal cells could reflect ITGA6 regulation across cell types. ITGA6’s role in multiple cancers underscores its value as a prognostic/diagnostic marker and therapeutic target. 42
Overexpression of BLM, a helicase central to DNA repair, supports the hypothesis that EA induces genotoxic stress, impairing replication and triggering apoptosis. 43 This correlates with reduced cell viability in vitro. Similarly, CEP72 upregulation, a centrosomal protein required for chromosome segregation, may indicate alterations in cell cycle control, 44 contributing to growth inhibition. Conversely, EA downregulated GPLD1 (Pa = 0.961), TFAM (Pa = 0.940), UGP2 (Pa = 0.934), and IL1R1 (Pa = 0.912), with moderate MAPK13 reduction (Pa = 0.649). GPLD1 repression could disrupt signaling and tumor spread. TFAM depletion, a regulator of mitochondrial DNA transcription may compromise oxidative phosphorylation-dependent tumor survival, consistent with observed cytotoxicity. UGP2 downregulation, linked to glycogen metabolism suggests impaired energy homeostasis. Reduced IL1R1 expression indicates modulation of inflammatory and immune pathways, while MAPK13 suppression may interfere with survival signaling and proliferation. 45
Together, these gene expression changes may provide a molecular rationale for the antiproliferative activity observed in vitro. EA appears to act through DNA damage induction, cell cycle disruption, metabolic alterations, and modulation of adhesion and inflammatory signaling. The limited selectivity of MEC highlights the need for optimization before therapeutic use. Overall, ellagic acid emerges as a promising anticancer candidate, though preclinical validation is required to confirm efficacy and assess safety. 46
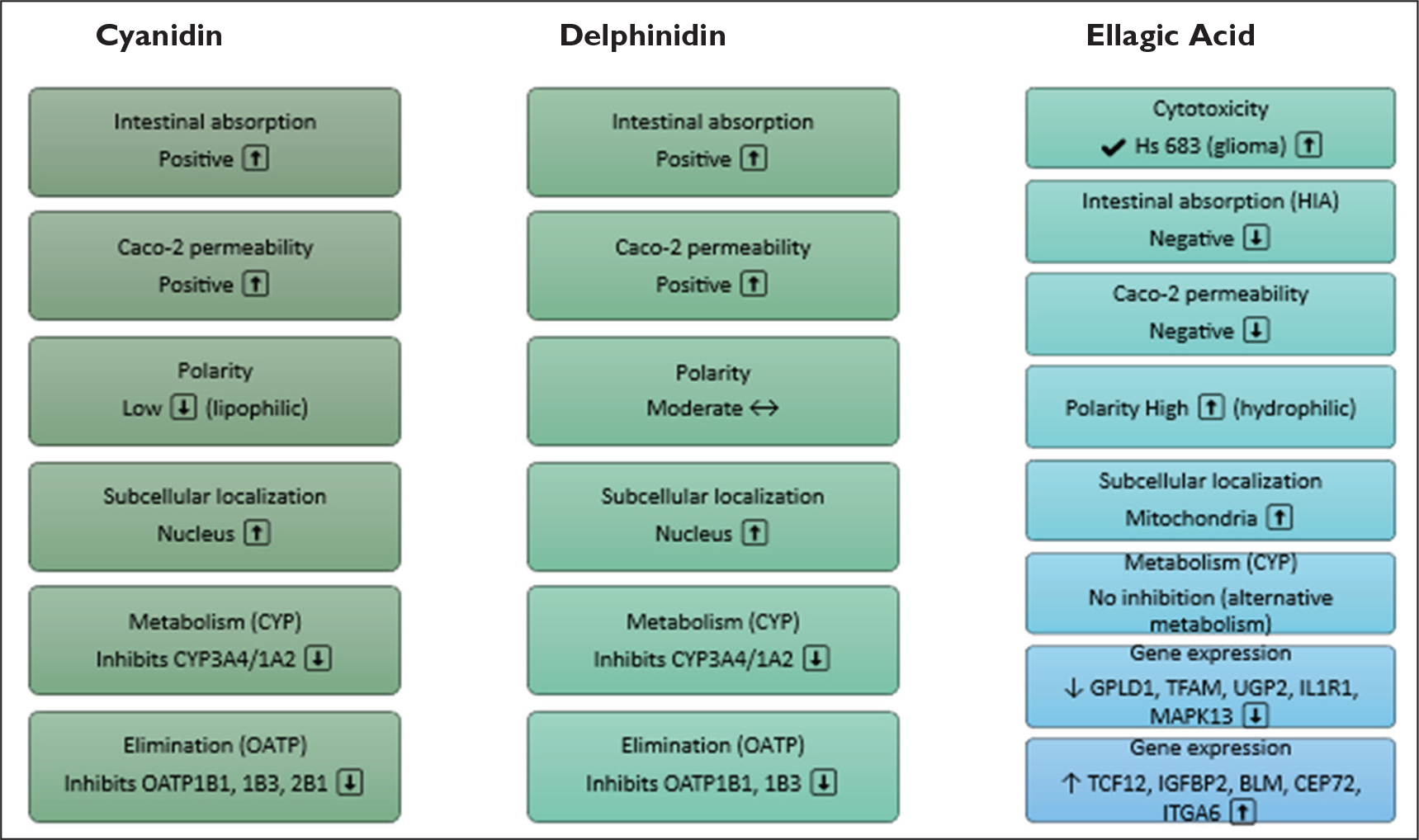
These findings presented in Figure 4. highlight the distinct pharmacokinetic and molecular profiles of cyanidin, delphinidin, and ellagic acid, underscoring their potential therapeutic relevance. While cyanidin and delphinidin exhibit favorable intestinal absorption, nuclear localization, and CYP inhibition, ellagic acid stands out for its cytotoxicity in glioma cells and its impact on gene expression, despite limited absorption and permeability. This comparative analysis provides valuable insights into their mechanisms of action and supports further investigation into their biomedical applications.
Comparative Summary of Biopharmacological and Molecular Properties of Cyanidin, Delphinidin, and Ellagic Acid Identified in Methanolic Extracts of Algerian Fir (A. numidica). In Silico Predictions Cover Intestinal Absorption, Caco-2 Permeability, Polarity, Subcellular Localization, Metabolic Interactions, Elimination Routes, and Cytotoxicity. Cyanidin and Delphinidin Show Good Absorption, Caco-2 Permeability, Nuclear Localization, and Inhibit CYP3A4, CYP1A2, and OATP1B1/1B3/2B1. Ellagic Acid is Cytotoxic to Hs 683 Glioma Cells, has Lower Absorption and Permeability, Higher Polarity, Mitochondrial Localization, and Follows CYP-independent Metabolic Pathways. Gene-expression Changes for Ellagic Acid are Shown with Arrows Indicating Up or Downregulation.
Discussion
Considering the scarcity of research conducted on coniferous species, the present study is particularly significant as it provides new information on its chemical characteristics and biological properties. This investigation contributes essentially to the existing knowledge concerning A. numidica along with their chemical characterization significantly contributes to advancing our knowledge of this plant species. The antioxidant capacity of hydroethanolic extracts from A. numidica cones (MEC) and leaves (MEL) was assessed by FRAP and ORAC assays, with MEL showing superior activity. These findings align with Benouchenne et al., supporting the genus as a source of natural antioxidants. Fractions of n-butanol and ethyl acetate displayed strong antioxidant effects, with IC₅₀ values of 3.07 ± 0.12 µg/mL and 5.28 ± 0.26 µg/mL, respectively, in the DPPH test. Activity is attributed to phenolics and flavonoids, whose aromatic rings and hydroxyl groups enable electron transfer, metal chelation, and free radical-scavenging, stabilizing reactive oxygen species. Such mechanisms highlight their protective role against oxidative stress and related pathologies.
In this context, the exploration of natural extracts for anticancer therapies is highly relevant. This study evaluated the cytotoxic effects of A. numidica extracts: MEL, containing ellagic acid, cyanidin, and delphinidin; and MEC, containing cyanidin and delphinidin. Recent advances in analytical and computational tools have improved the characterization of medicinal plants, enabling deeper insight into chemical structures and mechanisms. Predicting pharmacokinetic properties has become more feasible, whereas traditional methods remain laborious and time-consuming. Research often begins with compound extraction and analysis, but efficiency is now enhanced by in silico approaches. Network pharmacology has accelerated identification of complex drug–target interactions, while ADMET parameters—absorption, distribution, metabolism, elimination, and toxicity—are central to drug development.
In this context, anthocyanins are recognized for antioxidant and anti-inflammatory properties, with roles in inhibiting proliferation and inducing apoptosis. 47 EA, exclusively in MEL, is a potent antioxidant that modulates proliferation-related pathways. Golmohammadi reported that ellagic acid suppresses cell growth by G0/G1 arrest, inhibiting migration, invasion, and metastasis, and inducing apoptosis via TGF-β/Smad3 signaling. EA also inhibits CDK6, binds ACTN4 to reduce motility, 48 suppresses PI3K/AKT signaling and VEGFR-2 activity and enhances docetaxel cytotoxicity by overcoming drug resistance through MELK-FoxM1-ABCB1 inhibition. These findings position EA as a potential therapeutic candidate against triple-negative breast cancer. In CRC cells, MEL showed stronger selectivity than MEC. In SW480, MEL had an IC₅₀ of 2.250 µg/mL versus 2.650 µg/mL for MEC, with higher SI (2.10 vs. 0.82). Similarly, in HT-29 cells, MEL displayed superior selectivity (SI = 2.18 vs. 1.02), highlighting its greater therapeutic potential despite only slight differences in efficacy. These findings align with previous studies on EA, a phenolic compound extensively investigated for its anticancer potential, particularly in CRC. 49 EA inhibits CRC proliferation, induces cell cycle arrest, and promotes apoptosis, mainly via modulation of the Wnt/β-catenin pathway, has showed cytotoxic effects in HT-29 cells by promoting cycle arrest and apoptosis, reinforcing its therapeutic promise. 50
Cyanidin, delphinidin, and EA exhibited diverse toxicity profiles, cyanidin showed hepatotoxicity and nephrotoxicity, whereas all compounds displayed potential ocular, respiratory, and mitochondrial toxicity, along with acute oral toxicity. These results highlight both their therapeutic promise and the need for careful dose optimization to ensure safety before clinical application. EA-modulated genes are linked to tumor progression. TCF12 overexpression, a repressor of E-cadherin, suggests possible EMT regulation, influencing migration and invasion. 51 Increased IGFBP2, acting through IGF-dependent/independent pathways and importin-α expression, may disrupt resistance mechanisms via PTEN modulation, consistent with reduced proliferation and viability observed in vitro. 52
These findings highlight the therapeutic potential of cyanidin, chlorogenic acid, and delphinidin as antiproliferative agents, supporting natural product-based cancer therapy development. Further studies must isolate and characterize A. numidica compounds, with in vivo validation required to confirm mechanisms, efficacy, and clinical applicability. The results presented in this work show one of the first evidence over studying the cytotoxic activity of A. numidica in cancer cell models. In this regard, the results provide preliminary experimental evidence of a differential response to the evaluated extracts, with greater sensitivity observed in breast tumor cell lines. These findings afford relevance to the study and support the interest in continuing to characterize the biological activity of this coniferous species, particularly in the context of its potential antitumor activity. However, considering the limited exploration of the study and its in vitro cytotoxicity assays, additional research is essential, comprising cellular mechanistic studies and validation in other biological models, to determine the scope and meaning of the observed effects. While the IC₅₀ values obtained correspond to extracts and can be considered relatively high, the observed effects indicate significant biological potential. In this regard, the results support the need for further studies aimed at optimizing extraction methods and improving the potency and selectivity of the extracts.
Conclusion
Hydromethanolic extracts of A. numidica showed a phytochemical profile rich in phenolic compounds, including cyanidin, delphinidin, and ellagic acid. Both extracts displayed moderate antioxidant activity and differential cytotoxicity against tumor cell lines. MEL showed greater efficacy against MDA-MB-231 cells, while MEC was more active against MCF-7, with lower selectivity toward non-tumor cells. In silico studies confirmed pharmacokinetic characteristics compatible with bioactive molecules, and ellagic acid stood out for its ability to modulate genes involved in cell adhesion (ITGA6), genomic stability (BLM), energy metabolism (TFAM, UGP2), and inflammation (IL1R1). These findings support the potential for further functional studies.
Supplemental Material
Supplemental material for this article available online.
Footnotes
Acknowledgements
The authors would like to express their sincere gratitude to their respective institutions for providing the facilities and support necessary to carry out this research.
Authors’ Contribution
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability
The data set supporting the conclusions of this article are included within the article (and its additional file(s)). Project name: In silico prediction of ADMET properties and cytotoxicity of bioactive compounds from A. numidica. Other requirements: Internet access to use PubChem, SwissADME, admetSAR 2.0, CLC-Pred, and DIGEP Pred.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study does not involve experiments on animals or human subjects.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
Use of Artificial Intelligence-assisted Technology
The authors declare that they have not used artificial intelligence (AI)-tools for writing and editing of the manuscript, and no images were manipulated using AI.
