Abstract
Drug-related problems (DRPs) significantly impact patients’ medication therapy outcomes. These include adverse drug reactions, ineffective drug therapy, excessive or sub-therapeutic drug dosage, development of drug resistance, drug–drug interactions, medication errors, and medication non-adherence. A clinical decision support system (CDSS) helps mitigate DRPs by providing clinical suggestions and reminders that enable decision-making by pharmacists and clinicians toward improving the overall well-being of patients. CDSS, associated with pharmacy services, enable pharmacists to consider substitutes for specific medications and address specific health problems by providing medication therapy management (MTM), acute pain management and other related services. Our research focused on identifying the benefits of CDSS integration in the pharmacist workflow toward managing DRPs. We conducted a thorough search across major databases, Scopus, Embase, PubMed, and Google Scholar, to obtain appropriate articles published through November 2025 and selected articles that met the inclusion criteria. Some of the challenges in CDSS implementation identified were integration issues with existing electronic health records (EHRs), incompatibility with current workflows, costs, inadequate leadership, and poor data quality.
Keywords
Introduction
The past decade has witnessed significant evolution in medical science and health information technology (HIT). 1 Rise in medication management complexity and drug-related problems (DRPs), an increase in adverse outcomes reported during treatment. 2 Pharmacists, frontline medical professionals, can significantly mitigate DRPs. Through direct relationship with patients and by interdisciplinary collaboration with healthcare professionals (HCPs), pharmacists can help minimize and manage DRPs.3–8
A clinical decision support system (CDSS) provides clinical suggestions and reminders that enable pharmacists in decision-making to improve the overall well-being of patients. 9 “CDSS” refers to knowledge and information systems that aim to improve drug use and include both standalone innovations and integrated systems that support multiple electronic health record (EHR) activities. CDSS deploys essential patient information against dynamically updated medical knowledge and guidelines to assist HCPs in decisions.10,11 CDSS in pharmacy addresses different health delivery stages, including reminders, alerts, guidance, protocols, order sets, and care plan management. 12 Recently, CDSS was integrated into pharmacy services, enabling pharmacists to consider substitutes for specified medications and address specific health problems by medication therapy management (MTM), acute patient management, and wellness. 13
DRPs that significantly impact patients’ medication therapy outcomes 14 include adverse drug reactions, ineffective drug therapy, excessive or sub-therapeutic drug dosage, development of drug resistance, drug–drug interactions, medication errors, and medication non-adherence.15,16 DRPs can compromise quality of life due to health deterioration or injury arising from organ damage, disability, or even death. DRPs also increase healthcare utilization and costs for both patients and healthcare systems,17–19 owing to increased physician consultations, diagnostic tests, hospital stays, accessing the emergency room, and additional medications to manage DRP-related complications. DRPs also increase the workload of HCPs, decrease productivity, and strain healthcare facilities. 2 Interdisciplinary collaboration among HCPs is vital for the identification, prevention, and management of DRPs.
Although prior research shows the effectiveness of CDSS in significantly reducing DRPs, its clinical application remains marginal, necessitating the monitoring and validation of challenges in the usage and effectiveness of the current form of CDSS. 20 Real service ability problems need to be addressed to increase the CDSS utilization rate. 21
Many have analyzed the benefits of CDSS implementation and its role in ensuring medication safety, but they focus on benefits and ignore real challenges in CDSS adoption and consequent benefits from mitigating DRPs.11,12,22,23 Some reports on the benefits of CDSS have not addressed the real-world barriers and challenges in implementation.11,24–26 This narrative review critically evaluates the impact of CDSS in pharmacy practice toward minimizing and managing DRPs and identifies the real-world barriers that challenge CDSS implementation.
Methodology
We identified the population of interest as patients who require a pharmacist’s intervention in the context of implementing CDSS in a clinical setting. We considered experimental and observational study settings, including clinical practice.
We then thoroughly searched through Scopus, Embase, PubMed, and Google Scholar to obtain appropriate articles published until November 2025 and included articles that met the inclusion criteria (Table 1).
The Inclusion Criteria to Identify Appropriate Articles.

A total of 1,403 articles were identified from PubMed (n = 119), Scopus (n = 1,166), and Embase (n = 118). Further, a snowball search in Google and Google Scholar also identified a few articles of interest. A team of experts, comprising academicians and clinical pharmacists, selected the articles considered for the review based on DRPs, integration of CDSS in clinical settings, and the challenges and barriers in adoption.
Results
Drug-related Problems in Pharmacy Practice
DRPs, a major concern across medical disciplines, often arise from the complexity of pharmacotherapy, patient-related factors, and system issues. Pharmacists have a key role in optimizing medication by assessing the appropriateness, effectiveness, and safety of each regimen, as well as monitoring adherence and identifying unmet therapeutic needs.27–30
Unwanted or duplicate therapy, underuse or overuse of medications, and adverse events are DRPs that result in compromising the patient’s quality of life and increasing the chances of hospitalization, morbidity, and healthcare expenses.2,14,17–19
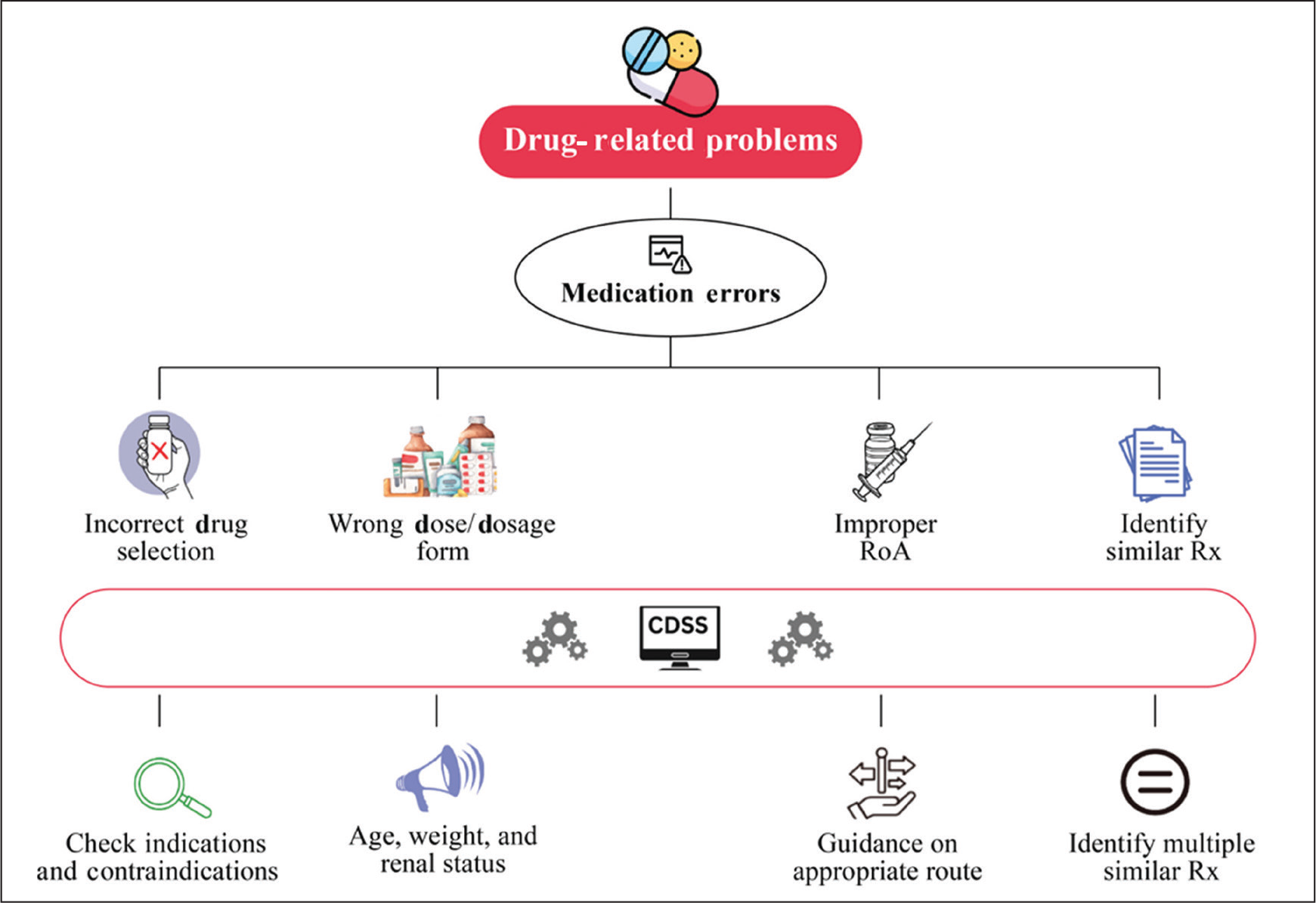
Incorrect dose, dosage form, duration, and frequency, prescribing errors, and ambiguous medication details are among the main factors that contribute to medication errors.31–33 The multifactorial causality of DRPs necessitates multidisciplinary collaborations of HCPs, including pharmacists and physicians.33,34 Open communication, knowledge sharing, and developing a plan to identify DRPs by a multidisciplinary team can minimize DRPs, improve outcomes, and patient safety.30,33,34
Impact of Drug-related Problems on Patients and the Healthcare System
Pharmacotherapy is complex, and DRPs are unavoidable. Identifying DRPs in early stages, its systematic management approach can prevent the majority of DRPs. Strategies include pharmacokinetic and pharmacodynamic monitoring, clinical pharmacists’ involvement in multidisciplinary teams, and MTM services.14,17,18
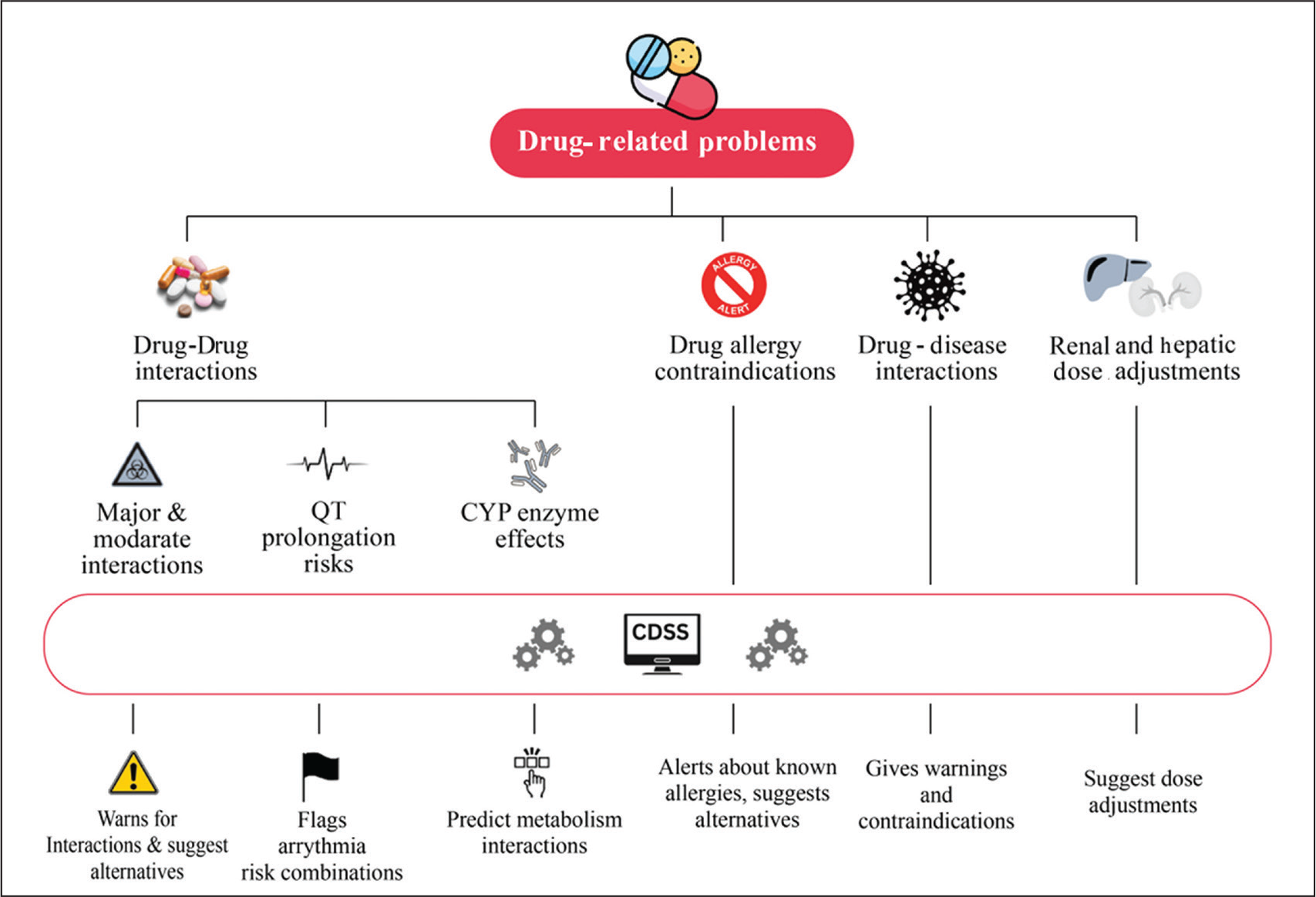
CDSSs integrated with EHRs have emerged as an important tool for pharmacists and clinicians to detect and resolve DRPs.22,23,35 For instance, CDSSs have helped clinical pharmacists efficiently identify a high proportion of DRPs in hospitalized patients and prioritize those requiring intervention. 35 Likewise, CDSS alerts for look-alike and sound-alike medications have reduced wrong-drug errors and prompted prescription modifications. 36 CDSSs also partly mitigate workforce constraints by supporting DRP detection and monitoring in settings with limited access to clinical pharmacists, provided EHR data (including drug allergies) are accurately and consistently updated.37,38
Integration of CDSS into Pharmacy Practice
Pharmacists provide medication management and pharmaceutical care services, mainly involving drug therapy decision-making. Services involve drug, patient, disease-related, population-specific, community, and pharmacy-related factors, and therapeutic goals.
The current adoption of drug therapy decisions predominantly supports checks for drug–drug interactions and other safety aspects in pharmacy dispensing applications. Such drug therapy decision supports the lack of personalized patient information, which could add excellent value to the decision-making process. Only EHRs can integrate personalized patient information with current population-specific knowledge.
EHRs play multiple roles in documentation and communication, clinical event recording, quality control process automation, CDSS, clinical process redesign, care coordination, payment bundling, and patient access to health records. Integrating the EHR with the CDSS changes the dispensing mode from a “push” to a “pull” model. In the push model, information received by individual staff members is shared with relevant team members, generally resulting in an excessive information load.
Benefits of CDSS Implementation
Implementing CDSS can optimally enable staff to participate in clinical management activities, despite certain patient groups being more susceptible to management-related DRPs. Pharmacy practice can become more proactive in clinical decision-making by implementing advanced HIT tools. CDSS can also enable evidence-based clinical advice, real-time alerts, and reminders, patient support services that improve outcomes and medication adherence, and administrative tools for workflow and resource management. Moreover, computerized surveillance and monitoring of prescribing and dispensing can detect pharmaceutical errors. Clinical dashboards displaying critical performance indicators empower data-driven decision-making. CDSS can enhance patient care by improving medication safety and medication management, avoiding adverse drug events and error prevention, in addition to DRP handling and interprofessional communication. Moreover, CDSS can lower healthcare costs and improve patient outcomes and satisfaction. By combining clinical expertise, patient-specific data, and computer algorithms, CDSS also provide clinicians with critical and accurate clinical information, evidence-based recommendations, and decision support alerts to aid patient care decision delivery. CDSS can also aid in evaluating treatment alternatives, implementing clinical recommendations into practice, and identifying potential drug interactions, drug allergies, and other patient variables that alter responses to medication. CDSS enhances communication between HCPs by exchanging patient data, treatment plans, and medication histories across healthcare settings. CDSS can enhance treatment continuity, lower medication errors, and improve patient outcomes.23,39,40 CDSS is a strategic instrument that can potentially transform pharmacy practice by integrating clinical knowledge, patient-specific data, digital technologies, and evidence-based medicine. 23
Recent incorporation of artificial intelligence (AI) with CDSS further improved DRP management by identifying complex drug-related patterns and facilitating individualized pharmacotherapy.36,41,42 AI-driven CDSS prototypes utilizing machine-learning (ML) algorithms improved the detection of high-risk prescriptions with fewer non-actionable alarms.35,36,42 The accuracy and efficiency of decision-making in pharmacy practice are possibly better with the application of advanced natural language and other AI models in conjunction with EHRs.41,42 AI integration with CDSS has transformed healthcare. 43 Deep learning-based CDSS performs better in early breast cancer detection. Large language models integrated with EHRs simplify and improve decision-making while lowering healthcare costs. 44
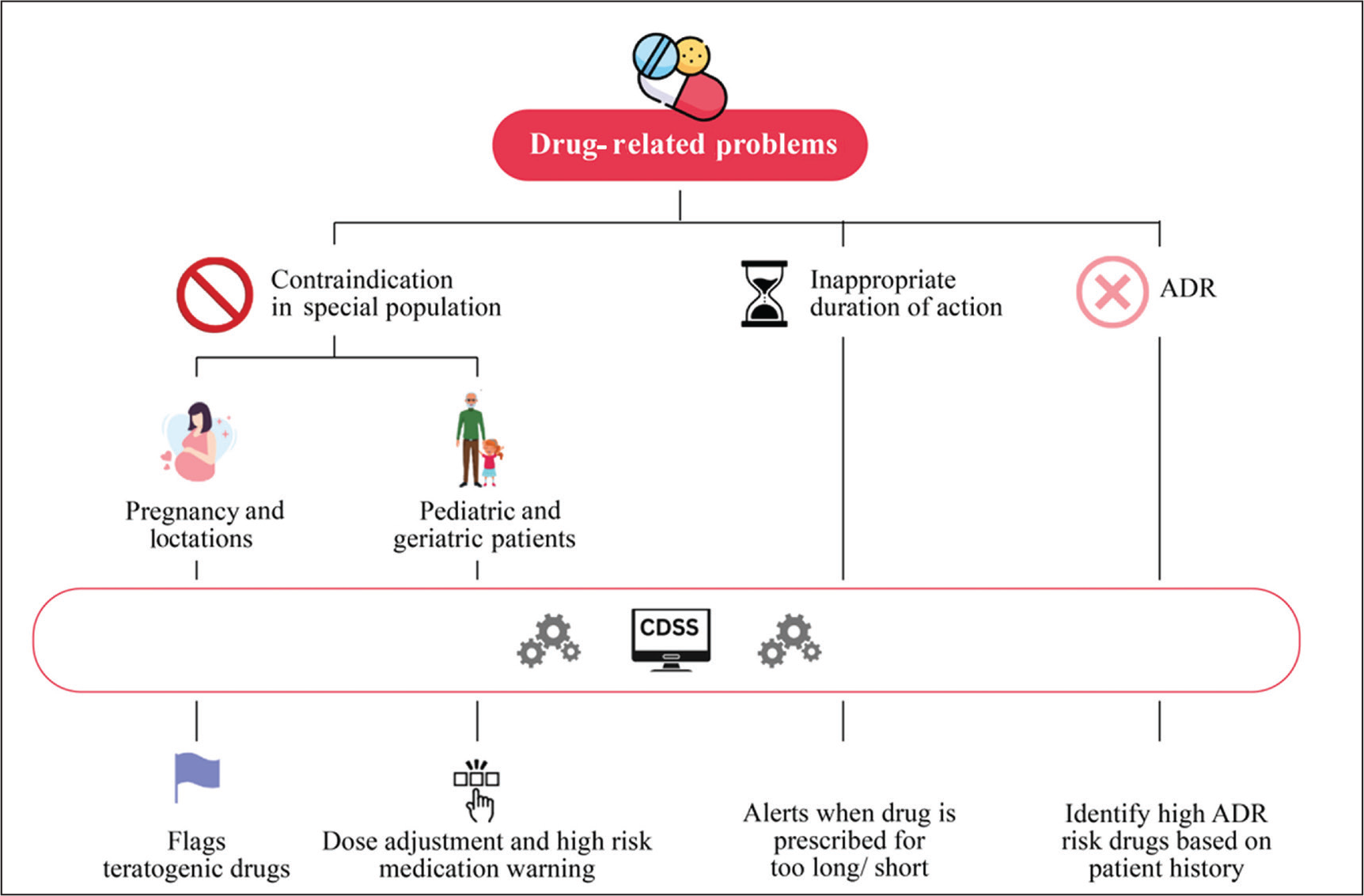
Advanced CDSS can improve management of DRPs in particular patient populations, enabling improved drug safety procedures and proactive clinical decisions on medicine management. A legitimate hybrid decision support technology by Levivien et al. combined ML and a rule-based expert system, which outperformed a traditional rule-based CDSS, by reducing severe DRPs, and determining the possible need for medicinal intervention depending on event severity, 45 enhancing patient diagnostics, ensuring correct medicine dispensing, and precise prescriptions. Additionally, an advanced CDSS can substantially improve the detection of possible drug interactions and may help to comply with laws and regulations pertaining to prescription orders.
Figures 1–3 highlight how the CDSS can enhance medication safety by mitigating key DRPs. The integration of CDSS reduces the frequency of medication errors and adverse drug events by real-time identification of high-risk circumstances and prompt medication safety alerts. CDSS also helps identify potential DRPs, such as drug–drug and drug allergy interactions, drug-disease contraindications, adverse drug reactions, and inappropriate therapy or duration, particularly in vulnerable populations.
In Routine Pharmacy Practice, Drug–Drug and Drug-disease Interactions, Drug Allergy Contraindications, and dose Adjustments in Renal/Hepatic Impairment are the Major DRPs. CDSS Helps Pharmacists Manage these by Providing Timely Alerts.
Challenges and Barriers to CDSS Adoption
A critical challenge in CDSS implementation in healthcare settings is the requirement for integrating an advanced EHR system with CDSS. 46 Undoubtedly, CDSSs are complex applications, and developing, adapting, and deploying them demands developer resources and expertise.
In some settings, critical patient-level data or information are stored in separate, non-interfaced databases or systems, hindering organizations’ ability to use them in CDSS. The most reported factors in implementation were compatibility of CDSS with workflows, practical use of CDSS output, CDSS technical dependencies, and design. Also, the credibility of users in the CDSS input data and evidence base, and the compatibility of the CDSS with the user’s role or clinical setting. 46 Other issues include data connectivity, updating guidelines, precision of AI-driven recommendations, and the possibility of losing human input in decision-making. 47 The main obstacles to adoption were simplified CDSS, limited clinical usefulness in multimorbidity, and inadequate workflow integration. 48 System failure, overreliance on the system, and inability to trust the system are possible threats to implementation. The closeness of the system and the ease of use of the interface may have helped in selecting the most effective CDSS. 49
Overall, while CDSSs can reduce selected DRPs, medication errors, and inappropriate prescribing, the magnitude and consistency of benefits differ across settings and systems. Many reports are single-center or pre–post designs with short follow-up, focusing on process indicators (for example, alert acceptance, guideline-concordant orders, or number of DRPs detected) rather than hard patient outcomes such as mortality or hospital readmissions.11,20,22,35,40,48,50 Differences in CDSS architecture, clinical context, user groups, and outcome definitions also make direct comparison difficult and limit the generalizability of results, especially to resource-constrained environments.11,20,22,35
Research on implementation further shows that the real-world impact of CDSSs is strongly influenced by data quality, workflow integration, usability, and user attitudes.20,23,46,48,50–52 Incomplete or inconsistently coded clinical data, poor interface design, and high volumes of non- actionable alerts can undermine trust and contribute to alert fatigue, reducing the likelihood of following clinically important recommendations.20,23,46,48,49,52 Limitations highlight that CDSSs should complement, rather than replace, the clinical judgment of pharmacists and other HCPs in preventing and managing DRPs.23,49,50
Implication for Pharmacy Professionals
Like other HITs, CDSS have important implications for delivering pharmacy practices in a well-organized and patient-centric manner.23,40,50 Best practice advisory alert (BPA) in CDSS helps pharmacists notify and address the most frequent and significant DRPs such as drug–drug and drug-disease interactions, allergies, dose-related issues, and deviations from globally approved protocols and clinical guidelines by providing evidence-based suggestions and thereby ensuring safe medication practices that prioritize patient safety.12,22,23,35,50 However, the individual role of pharmacists in managing DRPs remains unavoidable because their clinical role in decision making is very rigid and important in interpreting the BPA suggestions on benefits risk ratio by considering each patient on a case-by-case basis.23,30,45,50 This narrative review summarizes evidence guiding the appropriate implementation and use of CDSS in pharmacy practice, ensuring that these tools enhance rather than replace professional decision-making. CDSS can enhance the clinical standards of pharmacy practice, but it is not a substitute for pharmacy professionals in decision-making.
Conclusion
CDSS integration has a huge scope in pharmacy practice to mitigate the DRPs. While having promising positive outcome expectations, its implementation remains challenging based on various factors such as information technology infrastructure, system capabilities, institutional, and individual characteristics. Key determinants, such as backing, prior experience, personal disposition, and dedication, shape the deployment of CDSS. Support for real-time patient-centered decision-making matters for safe medication practice. Decreasing DRPs is a priority for all HCPs in community and hospital settings. Personalized and easy-to-understand pop-up alert set-ups are very important. Integration of CDSS into healthcare settings depends on collaboration and communication among all stakeholders. Incorporating tools is more important for facilitating the evolving role of pharmacists. This approach enables staff to view alerts as a shared responsibility in clinical management, thereby benefiting patients. However, the benefits of CDSS are evident, healthcare system must not ignore the value of pharmacists’ knowledge and experience as it transitions to a digital landscape. The future role and capabilities of CDSS highlight the need to integrate them into clinical workflows successfully.
Footnotes
Acknowledgements
The authors gratefully acknowledge Johns Hopkins Aramco Healthcare for covering the Article Processing Charge (APC) for this article.
Authors’ Contribution
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be authors as per the International Committee of Medical Journal Editors’ requirements/guidelines.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study does not involve experiments on animals or human subjects.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors, or the reviewers. This journal remains neutral about jurisdictional claims in published institutional affiliation.
Use of Artificial Intelligence-assisted Technology
The authors declare that they have not used AI-tools for writing and editing of the manuscript, and no images were manipulated using AI.
