Abstract
Stability-indicating analytical methods are essential for accurately monitoring pharmaceutical residues and their transformation products in complex matrices, particularly when photo-degradation may compromise analyte integrity. Ultra-performance liquid chromatography (UPLC) was employed to effectively separate and quantify hydrochlorothiazide (HCTZ) alongside its three primary photo-degradation products in river water samples. The linearity range for quantification was established between 0.1 and 10 µg/mL. To properly prepare samples for analysis, the solid-phase extraction (SPE) method has been developed and is widely utilized. Even when up to 90% of the main photodegradable products are present, the suggested approach can still assess HCTZ. After a thorough validation process that followed the parameters set by the International Council for Harmonisation (ICH-Q2B), river water samples were treated using SPE. This study presents a novel UPLC method for detecting the target pharmaceutical alongside its primary photo-degradates in river water. The presented method demonstrates sufficient sensitivity to effectively monitor the level of environmental contamination resulting from the pharmaceutical under investigation.
Keywords
Introduction
The uncontrolled utilization of pharmaceuticals has escalated significantly in recent decades, with some being classified as emerging contaminants. Multiple investigations validate the existence of a diverse array of these pollutants in various water environments (both surface and underground). Diuretics, particularly hydrochlorothiazide (HCTZ), a thiazide-type diuretic, are prevalent in surface water at significant concentrations.
HCTZ, which has the chemical structure 6-chloro-3,4-dihydro-2H-1,2,4-benzothiadiazine-7-sulfonamide-1,1-dioxide, works by preventing the renal tubules from reabsorbing electrolytes.
It is extensively utilized, either alone or in conjunction with other medications, for managing hypertension and edema, in addition to being used for other conditions such as diabetes insipidus, hypoparathyroidism, or hypercalciuria. This chemical undergoes minimal metabolism, with at least 61% excreted unchanged in urine and partially in feces, thereby re-entering the water cycle. 1 The Scientific Committee on Toxicity, Ecotoxicity, and the Environment has advised that degradation and reaction by-products of drugs warrant consideration. 2
The wide-ranging use of diuretics, particularly HCTZ, in the last few years has considerably jeopardized human life because of the increased probability of their release into the surrounding ecosystems. Potential human health concerns associated with long-term, low-level exposure to HCTZ in river-derived water sources include the possibility of subtle physiological effects, particularly related to electrolyte and fluid balance, as well as increased susceptibility in sensitive populations such as infants, the elderly, and individuals with renal or cardiovascular conditions. Although environmental concentrations are far below therapeutic doses, continuous exposure, combined with mixture effects from other co-occurring pharmaceuticals and the formation of transformation products with uncertain toxicity, may contribute to cumulative health risks.3–5 Therefore, it is essential to utilize highly sensitive and effective techniques for monitoring HCTZ levels in aquatic settings. These methods must accurately detect HCTZ, even in the presence of compounds that have a similar structure, especially its primary photo-degraded products that probably occur in surface waters such as rivers due to HCTZ’s high susceptibility to photo-degradation. It is crucial to monitor the concentrations of the tested drug in the surrounding environment, especially in surface water bodies such as rivers. The analytical method employed for quantifying HCTZ must exhibit sufficient selectivity to distinguish between the drug and its primary photo-degradation products.
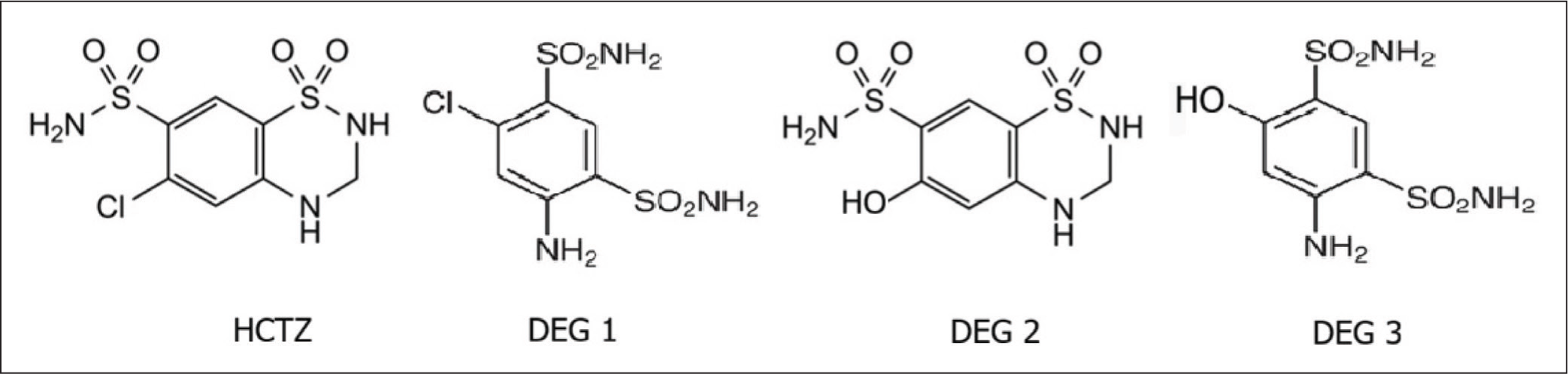
HCTZ is acknowledged as a photosensitive drug; its degradation in water was examined after 200 hours under a 150 W sun simulator. 2 The principal photo-degradation products were 4-amino-6-chloro-1,3-benzenedisulfonamide (DEG 1), 6-hydroxy-3,4-dihydro-2H-1,2,4-benzothiadazine-7-sulfonamido-1,1-dioxide (DEG 2), and 4-amino-6-hydroxy-1,3-benzenedisulfonamide (DEG 3). DEG 1 and DEG 2 exhibited yields over 10%. Figure 1 illustrates the structures of HCTZ and its principal photo-degraded products.
Chemical Structure of HCTZ and Its Main Photo-degradation Products. 2
To quantify the drug under study, either alone or in the presence of other drugs in various sample forms, analytical techniques such as spectroscopy, 6 high-performance liquid chromatography (HPLC),7–9 capillary electrophoresis,10,11 liquid chromatography–tandem mass spectroscopy (LC–MS/MS),12,13 and electrochemistry14–16 were employed. On the other hand, LC–MS/MS 17 and electrochemistry 18 were used to quantify HCTZ in water samples.
A range of analytical techniques was employed to evaluate the stability of HCTZ, both in separate and in combination with other pharmaceuticals, under a variety of stress conditions such as acidic and alkaline hydrolysis, oxidation, photolysis, thermal degradation, and the presence of different impurities. These methodologies encompass chemometric analysis, 19 HPTLC, 20 HPLC,21–24 and LC–MS/MS, 25 capable of quantifying the investigated medication across various sample types, including bulk materials, biological fluids, and dosage forms.
Ultra-performance liquid chromatography (UPLC) is a chromatographic technique that employs a mix of materials with a reversed-phase (RP) packing with a particle size of 1.7 μm, and works at pressures ranging from 6,000 to 15,000 psi. Among the many advantages this method provides over traditional HPLC is its greater sensitivity. The reason behind this is the reduction in zone broadening. Its enhanced chromatographic peak resolution and faster analytical speed are particularly noteworthy. 26
A thorough examination of the current literature revealed the absence of published research addressing the selective quantification of HCTZ amidst its primary photo-degradation products, specifically DEG 1, DEG 2, and DEG 3, in river water samples. This sample type is selected because of the significant likelihood of photo-deterioration of HCTZ, as river water is subjected to prolonged sunlight exposure and HCTZ exhibits high photo-sensitivity to degradation.
The main focus of this study is to selectively quantify HCTZ amid its principal photo-degradates (DEG 1, DEG 2, and DEG 3) in river water samples.
From a pharmaceutical science perspective, HCTZ is a well-established active pharmaceutical ingredient for which photostability and impurity profiling are critical quality attributes during drug development and post-approval lifecycle management. The ability to selectively quantify intact HCTZ in the presence of its major photo-degradation products is essential for stability-indicating method development, forced degradation studies, and regulatory compliance with ICH guidelines. In this context, the application of the proposed method to river water represents a stress-exposed aqueous matrix that mimics extreme photolytic conditions, thereby extending conventional pharmaceutical stability assessment into a real-world system.
Experimental
Instrumentation
The UPLC attached to a diode array detector (DAD) consisted of an Agilent 1200 series (Agilent Technologies, Santa Clara, CA, USA) (quaternary pump, vacuum degasser, and diode array and multiple wavelength detector G1315 C/D and G1365 C/D) connected to a computer loaded with Agilent ChemStation software. A ZORBAX RRHD Eclipse Plus column packed with C18 particles with a size of 1.8 µm, and the column dimensions were 100 × 2.1 mm.
Materials and Reference Solutions
The reference substances HCTZ, DEG 1, DEG 2, and DEG 3 were supplied by a company located in Spain (Cymit Quimica). The certificate indicates that HCTZ, DEG 1, DEG 2, and DEG 3 exhibited purities of 100.65%, 99.73%, 99.86%, and 101.14%, respectively. The solvents utilized were of superior grade, while the chemicals used were of acceptable purity. Methanol, acetic acid, sodium hydroxide, and acetonitrile were supplied by Merck (Darmstadt, Germany). The distilled water needed for this study was obtained using the Milli-Q Plus system, which is manufactured by Millipore in Bedford, Massachusetts, in the United States. The samples were processed using Oasis HLB cartridges from WatersTM in Milford, MA, USA. Nylon membranes were used to filter the water samples from the river.
To prepare stock solutions (100 µg/mL) of each chemical, the appropriate quantity was dissolved in methanol. Methanol was used to dilute the stock solution to make working standard solutions (10 μg/mL). The prepared solutions were kept in a light-protected location.
River Water Samples’ Collection
Three different sites along the River Nile in Egypt were sampled for water free of HCTZ, and one sample suspected to contain HCTZ was taken from another site. To eliminate any traces of microscopic particles, the samples were filtered through a nylon filter. To protect the samples from photo-degradation, opaque glass vials were used to store the filtered solutions.
Method Development
To achieve a superior separation profile for HCTZ and its primary photo-degradation products, the chromatographic parameters were adjusted. Multiple attempts were made to optimally align the stationary phase with the mobile phase for superior separation.
Method Validation
Method validation was performed in accordance with ICH Q2(B) requirements, 27 consistent with regulatory expectations for stability-indicating analytical procedures applied to active pharmaceutical ingredients and their related impurities.
Linearity
Different quantities of the medication being studied at 25°C, varying from 1 to 100 µg, were individually placed into distinct 10 mL volumetric flasks. This was followed by the addition of methanol into every flask until the desired concentration of 0.1–10 µg/mL was achieved.
ZORBAX RRHD Eclipse Plus column (100 × 2.1 mm, 1.8 µm) was used as a stationary phase to perform the separation. The temperature of the column was 25°C. Water, methanol, and acetonitrile were combined with 1% v/v acetic acid in a volumetric ratio of 5:4:1 to form the developing system. The flow rate was established at 0.8 mL/min. A DAD at 272 nm was used for the detection.
Assessment of Accuracy
It can be defined as the ratio of the obtained analyte to the total quantity analyzed. 27 Data from nine samples, each containing 1, 3, and 5 µg/mL of the medication being investigated, were handled in accordance with the conditions of linearity.
Assessment of Precision
Measuring the percentage relative standard deviation across a range of statistically significant samples allows us to measure precision, which may be applied intra- and inter-day. Triplicate tests were conducted on all three drug concentrations (1, 3, and 5 µg/mL), either within one day (intra-day) or over the course of three days (inter-day).
Detection and Quantification Limits
The quantification limit is the lowest concentration of a substance that can be accurately and consistently measured, whereas the detection limit is the lowest concentration that can be distinguished from background noise. The purchase of these commodities was carried out in conformity with the rules set forth by the US Pharmacopoeia. 28 Choosing concentrations that produce peaks ten times taller than the baseline noise and three times taller than that, respectively, establishes the limit of quantification (LOQ) and the limit of detection (LOD). The S/N ratio was measured by the UPLC software according to the analyte standard in solvent and matrix.
Assessment of Robustness
It was evaluated by analyzing the effect of small changes on its performance. The robustness of the method can be assessed by a minor variation in the methanol concentration in the mobile phase (±2% absolute) or a slight alteration in the flow rate (0.8 ± 0.1 mL/min).
Method Application
HCTZ Quantification Amidst Its Photo-degradates
The laboratory-prepared mixes were made using a range of concentrations of intact HCTZ (from 90% to 10%) and its major photo-degradants (from 10% to 90%). The linearity procedure was employed to obtain the peak area associated with the analyzed medication, and the HCTZ concentration was calculated using the regression equation.
Determination of HCTZ in River Water Samples
For the solid-phase extraction (SPE) procedure, 200 mg, 6 mL Oasis HLB (Waters, USA) cartridges were utilized. The cartridge was activated by adding 1 mL of methanol three times, and it was conditioned by adding 1 mL of water three times. The initial pH of the river water samples was 6.5–8.5. Using 1 M NaOH, the pH of 500 mL of the river water sample was adjusted to 9.0 to optimize extraction efficiency. After that, any suspended particles were removed from the sample by filtering it. A precise quantity of the drug under investigation (0.5 µg) was added to the HCTZ-free sample before it was put into the cartridge that had already been conditioned. Two rinses of the cartridge were performed using 2.0 mL of a 9:1 water:methanol combination. After that, 3.0 mL of methanol was used to elute it. A rotating evaporator was used to evaporate the eluate under vacuum at a temperature of 40–45°C. After that, 0.5 mL of the mobile phase was added to reconstitute it. An enrichment factor of 1,000 is obtained during the pre-concentration procedure. The optimized UPLC process was then used.
The river water sample suspected to contain HCTZ (unspiked) was treated by the same pre-treatment procedures except for the spiking of the sample with HCTZ, then it was chromatographed under the same conditions. The standard addition technique was applied by spiking different known concentrations of HCTZ to the real river water sample suspected to contain HCTZ, then the procedure was completed as mentioned before.
Calibration in matrix was carried out by spiking five blank HCTZ-free river water samples with different HCTZ amounts to get concentrations 0.1 × 10−3–10 × 10−3 µg/mL (0.1–10 µg/L.) The prepared samples are subjected to the same SPE procedure mentioned before. The processed samples are injected after reconstitution, and a calibration curve using the initial, unconcentrated concentrations against the final peak areas is plotted.
A matrix blank was prepared by using 500 mL of the unspiked HCTZ-free river water sample, which was subjected to the SPE procedure mentioned before. The processed blank was injected, and the chromatogram was examined to ensure that there was no HCTZ.
Results and Discussion
Environmental pollution needs to be thoroughly investigated to protect human health. Due to the gradual entry of drug residues into the body and their potential catastrophic impact, their existence in domestic water or agricultural products may constitute a significant threat to human health.
The detection of HCTZ in river water raises significant concerns because of its possible effects on human health, particularly in areas where rivers are used as sources for drinking water or for irrigation and recreation. Although HCTZ is usually found in low concentrations, its ongoing release into river environments could lead to prolonged, low-level exposure for humans through drinking water collection, agricultural applications, and the food supply. Long-term exposure to pharmaceuticals at sub-therapeutic doses has been linked to mild physiological changes, such as imbalances in electrolytes and impacts on kidney function, especially in vulnerable groups such as children, the elderly, and those with existing kidney or heart conditions. Furthermore, HCTZ may undergo changes when exposed to sunlight in surface waters, creating degradation products that may have unknown properties or could potentially present greater risks. 29
This work established the viability of quantifying HCTZ amidst its primary photo-degradation products in river water samples. This was accomplished by the utilization of a sensitive, rapid, and precise UPLC method.
The main goal was to track the environmental concentrations of the medicine under investigation in the presence of its main photo-degradates, which are frequently detected in river water because of the water’s extended exposure to sunlight and HCTZ’s photo-liability.
Method Development
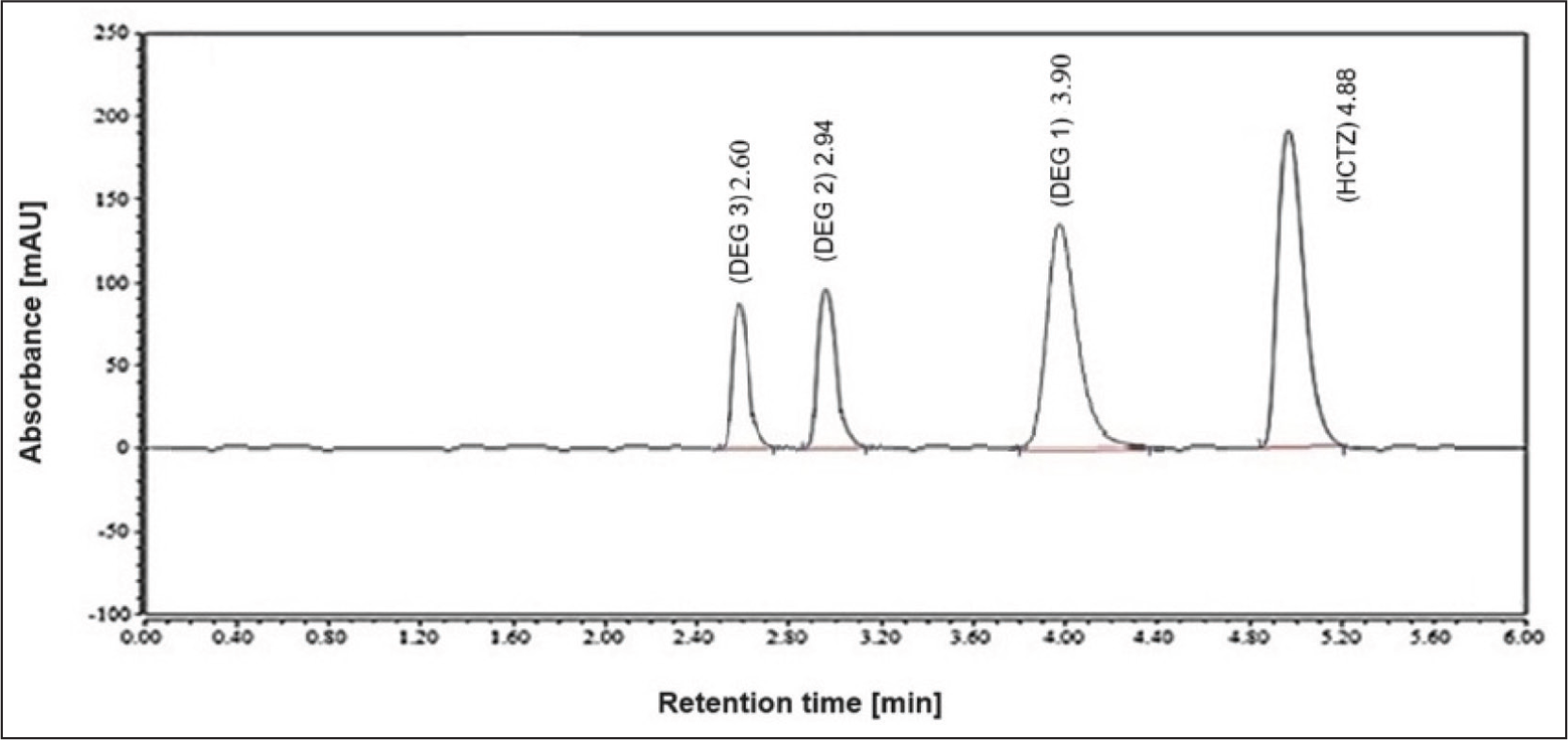
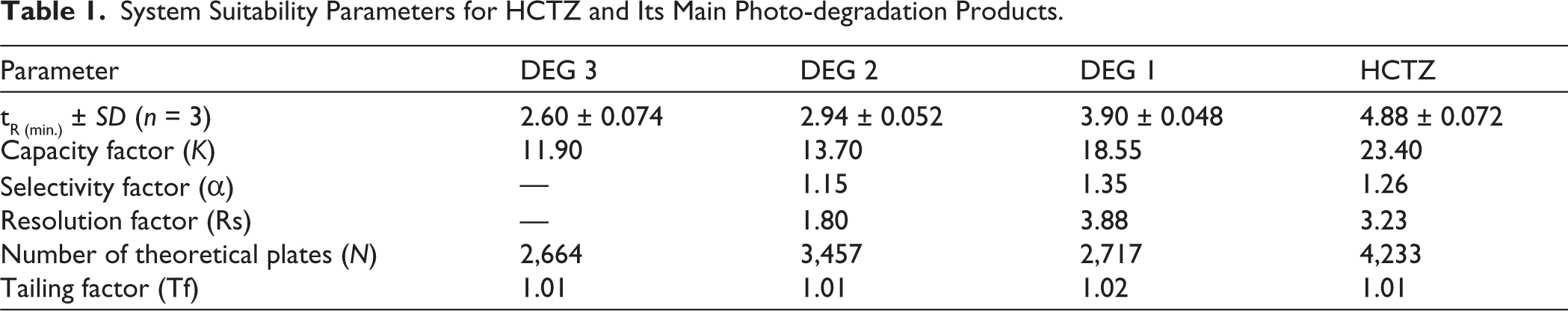
Optimizing chromatographic parameters is essential for achieving an optimal separation pattern with appropriate retention times, particularly when there is structure similarity between the analytes. Both C8 and C18 RP stationary phases were evaluated. The utilization of a 100 × 2.1 mm C18 RP ZORBAX RRHD Eclipse Plus column with 1.8 µm particles resulted in satisfactory separation. Various solvent mixtures were evaluated as the mobile phase to achieve optimal compatibility with the stationary phase and to provide the best possible separation profile with acceptable retention times without employing gradient elution. The ideal mobile phase composition was a blend of water, methanol, and acetonitrile, each incorporating acetic acid (1%, v/v) in a volumetric ratio of 5:4:1. The flow rate was adjusted to 0.8 mL/min. The DAD was used to confirm analyte peak purity. Figure 2 illustrates the ideal separation pattern between HCTZ and its main compounds that are produced due to photo-degradation in a ratio that mirrors that of an HCTZ photo-degraded sample. 2 The system suitability characteristics presented in Table 1 validate the high resolution of the separated peaks and the superior peak symmetry.
A Chromatogram Representing the Separation Pattern of HCTZ and Its Main Photo-degradation Products in a Ratio Resembling their Relative Percentage Yields After 200 Hours of Photo-degradation Using Solar Simulator. 2
System Suitability Parameters for HCTZ and Its Main Photo-degradation Products.
Validation of the Proposed Method
According to ICH-Q2B guidelines, the research was carried out. 27 By graphing the peak area against the concentrations of the medication under study, which fell within the range of 0.1–10 µg/mL, the suggested approach proved to be linear. The correlation between HCTZ concentrations and peak regions was demonstrated using the regression equation.
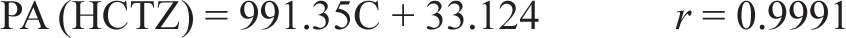
in which the correlation coefficient (r), concentration (C) (µg/mL), and peak area (PA) are defined.
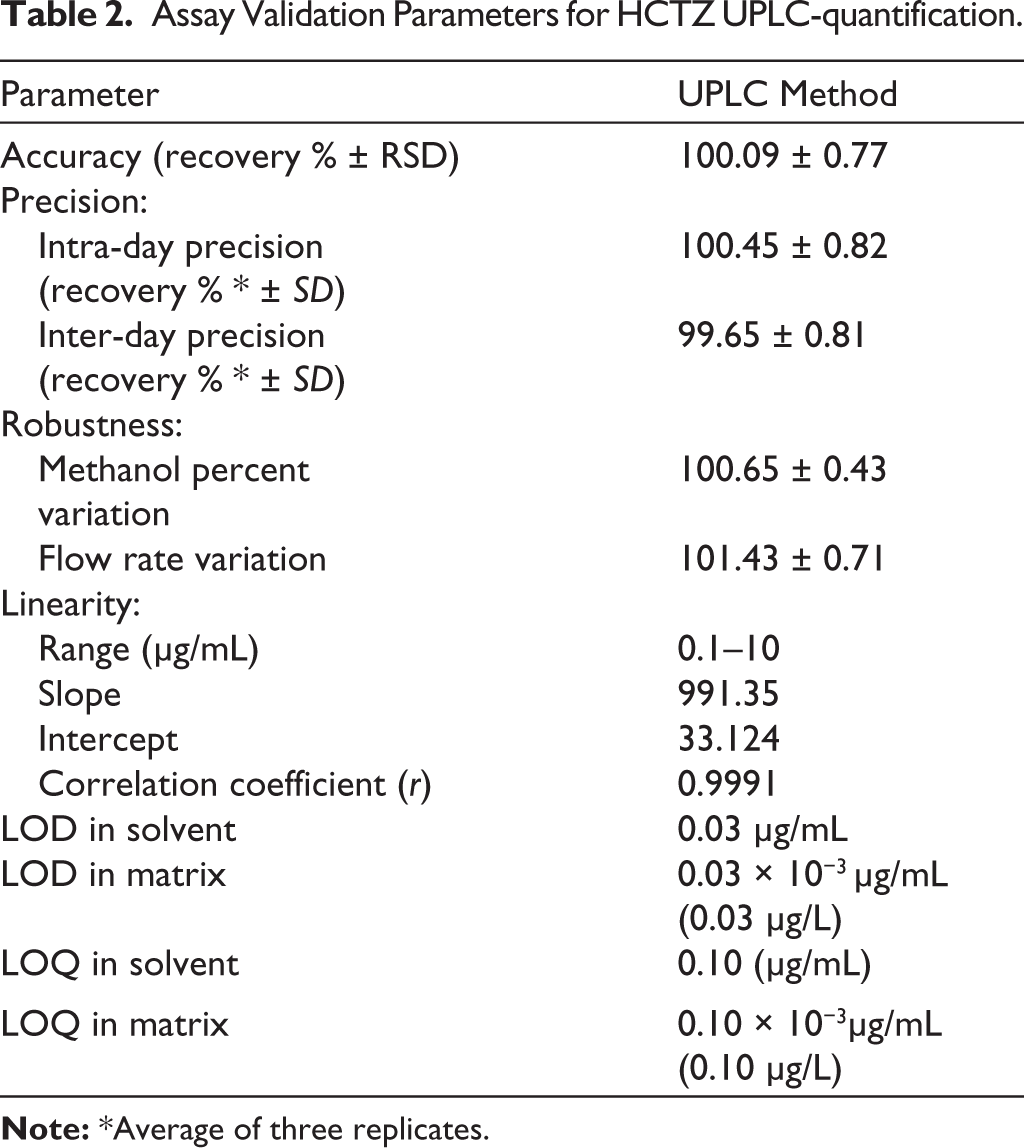
The procedures were evaluated for accuracy by calculating the recovery percentage following three runs with three different dosages of the relevant medicines. Table 2: Low RSD values attest to the methods’ remarkable accuracy. The accuracy of measurements was tested in the study, both within and between days. The results, which are displayed in Table 2, show a high degree of accuracy. The authors tested the method’s robustness by tweaking the assay parameters, such as changing the mobile phase composition and flow rate. According to the findings, the suggested approaches were unaffected by these little adjustments. Table 2 displays the results showing the LOQ and LOD, which are determined by the analyte concentrations, as peaks with heights that are 10 and three times larger than the baseline noise, respectively. The sensitivity of the approach is confirmed by the outcomes.
Assay Validation Parameters for HCTZ UPLC-quantification.
Application of the Proposed Procedures
Quantification of HCTZ Amidst Its Photo-degradant
Method selectivity for the determination of HCTZ in the presence of varying quantities of its main photo-degradant (10%–90%) was evaluated. To attain this objective, rigorous UPLC methods were employed to evaluate many laboratory-prepared combinations with differing concentrations of HCTZ and its major photo-degradant. The assay findings in Table 3 demonstrate that the specified techniques may accurately measure the investigated medication even when 90% of its primary photo-degradates are present.
Results Obtained for the Analysis of Laboratory-prepared Mixtures Containing Different Ratios of Intact HCTZ and its Main Photo-degradants by the Proposed Method.
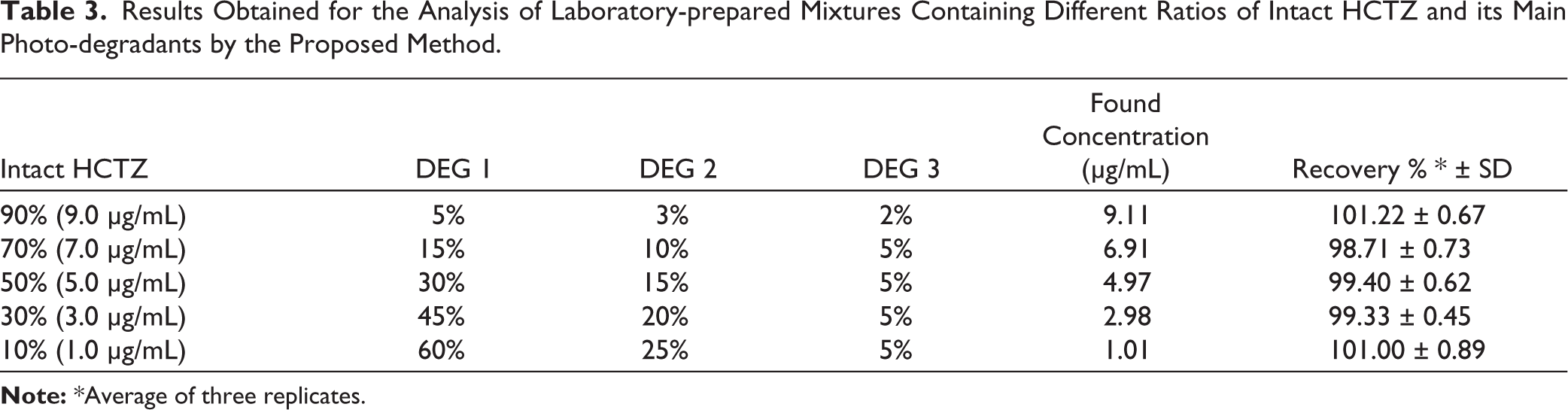
The successful quantification of the studied drug in the presence of up to 90% of its photo-degradation products demonstrates the suitability of the proposed method for impurity profiling and stability testing scenarios commonly encountered in pharmaceutical quality control laboratories.
Quantification of HCTZ in the River Water Samples
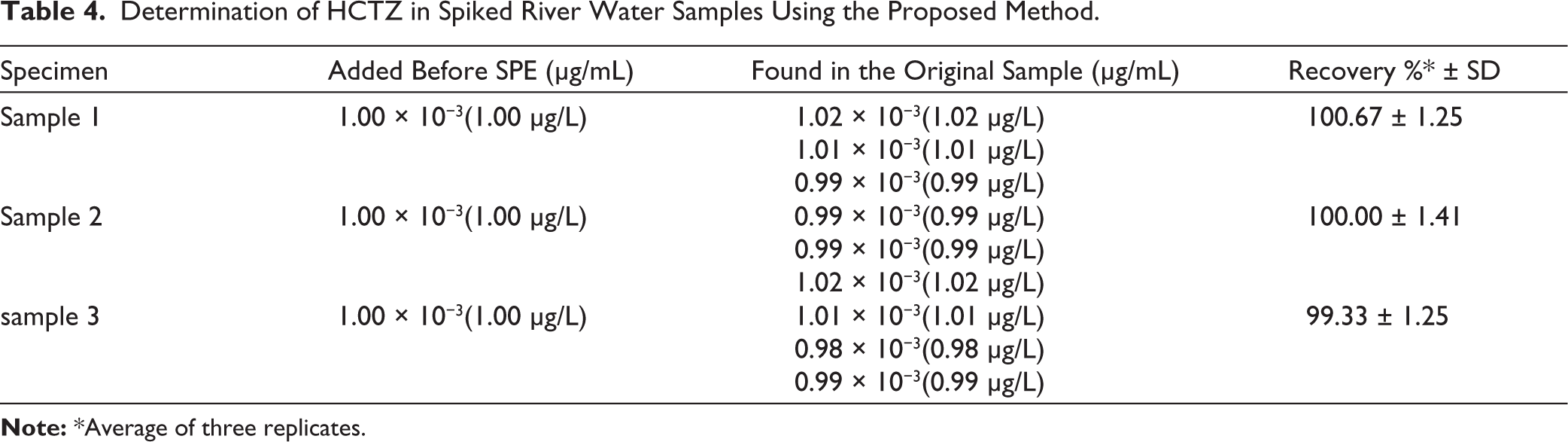
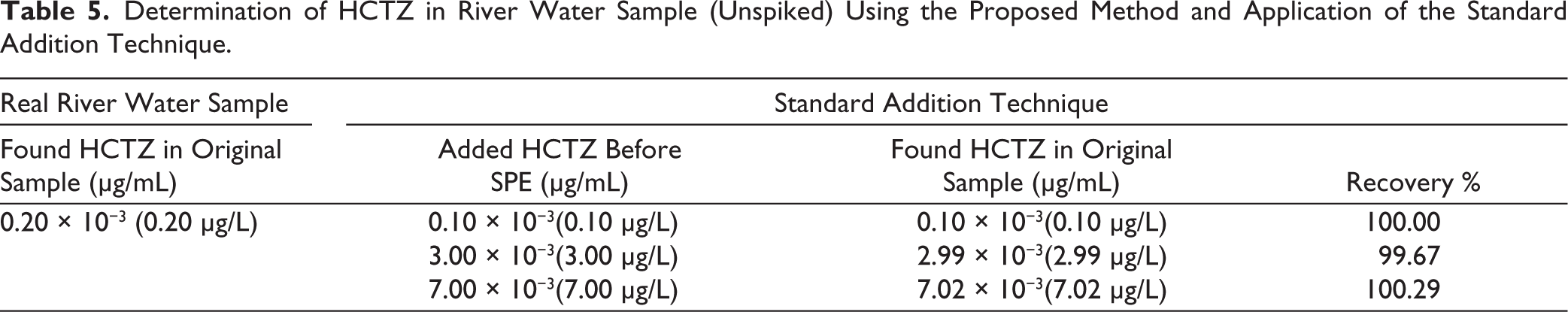
Utilizing a technique such as UPLC, known for its reliability and broad applicability, often necessitates a carefully chosen sample pre-treatment process because a wide range of substances can interfere with the analysis. Advantages over other procedures, such as protein precipitation and liquid-liquid extraction, led to the selection of SPE. SPE is a faster and more efficient purification method than protein precipitation. Additionally, the samples can be concentrated via SPE, which results in a suitable level of sensitivity for the application in environmental analysis. The moderate polarity of the Oasis HLB cartridges was the deciding factor in their selection for sample pre-treatment. LiChrolut RP-18 (Merck) and LC-18 (Supelco) were also evaluated for sample pre-treatment; however, the most favorable results were achieved with the use of Oasis HLB cartridges. At the outset, the preliminary washing stages utilized solely water. The use of methanol improved selectivity, allowed for thorough sample purification, and maintained excellent extraction yields. The recommended steps were carried out following the optimization of the sample pre-treatment process. The applied SPE procedure provided an enrichment factor of 1,000, resulting in an effective environmental LOQ of 0.10 µg/L for HCTZ in river water. This level of sensitivity is adequate for environmental monitoring purposes, as previous LC–MS/MS-based studies have reported HCTZ concentrations in wastewater-impacted rivers within the sub-µg/L to low-µg/L range, with concentrations approaching 1 µg/L depending on local anthropogenic influence.17,29 In the present study, the proposed method successfully quantified HCTZ in a real river water sample at a concentration of 0.2 µg/L, confirming the versatility of the method for the determination of environmentally relevant levels.
The data that has been collected after analyzing the spiked river water samples is presented in Table 4. On the other hand, the obtained results after analyzing the river water sample suspected to contain HCTZ and those after applying the standard addition technique are presented in Table 5.
Determination of HCTZ in Spiked River Water Samples Using the Proposed Method.
Determination of HCTZ in River Water Sample (Unspiked) Using the Proposed Method and Application of the Standard Addition Technique.
Comparing the Proposed Method with a Reference One
Table 6 shows a statistical comparison of the findings from the assay of pure HCTZ using the proposed technique with those from the analysis of the compound using a reference LC–MS/MS method. 15 No statistically significant difference in accuracy or precision was seen between the reference method and the suggested strategy, since both the calculated and tabulated t and F values are lower.
Statistical Comparison Between the Determination of Pure HCTZ by the Proposed Method and the Reference-reported Method.
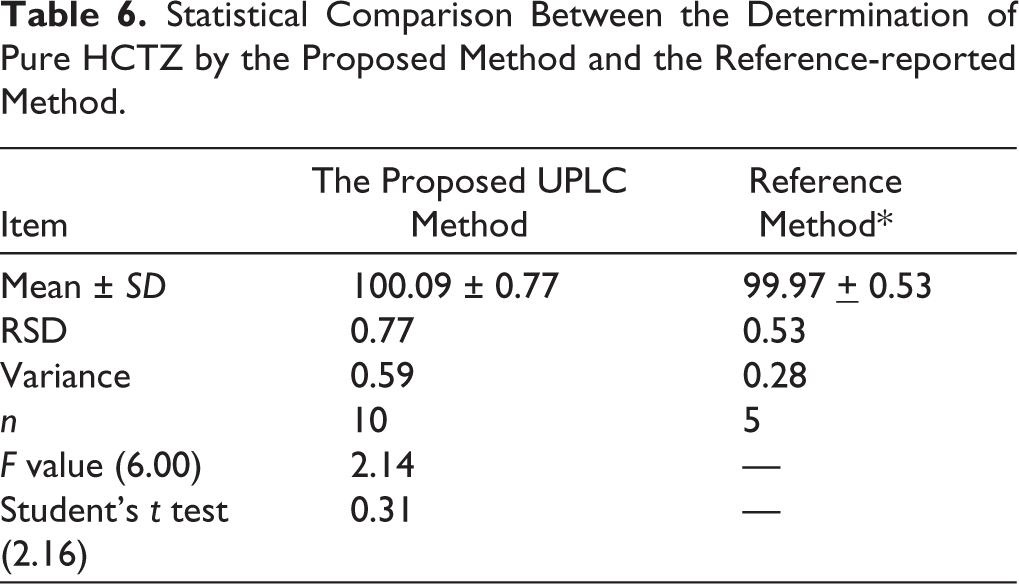
Unlike most reported LC–MS/MS river water methods, which focus mainly on ultra-trace quantification of the parent compound, the proposed method emphasizes stability-indicating selectivity by resolving HCTZ from its three major photo-degradation products under environmentally relevant conditions in a sensitivity level consistent with the environmental analysis purpose. This complementary analytical capability is particularly relevant for surface waters exposed to sunlight, where transformation products are likely to coexist with the intact drug.
Conclusions
This study involved the accurate quantification of HCTZ with the existence of its main products resulting from photo-degradation in samples obtained from river water, utilizing UPLC methodology. For maximum sensitivity, precision, and selectivity, the process described above has been fine-tuned. To ensure optimal purification and maximal extraction efficiency, SPE was applied as a method for sample preparation. In comparison to other extraction techniques, the suggested pre-treatment procedure offers a more effective approach to pre-treating the sample, necessitating less time. The proposed method shows good recoveries and verifies the best methods for extracting samples from river water. The proposed study presents a unique contribution to its field, as there are currently no published articles that focus on the measurement of HCTZ amidst its primary photo-degradation products in river water utilizing UPLC. Beyond its application to environmental samples, the proposed UPLC method fulfills key pharmaceutical analytical requirements by providing a validated, stability-indicating approach for HCTZ capable of resolving major photo-degradation products, thereby supporting API stability assessment, impurity control, and regulatory-compliant quality evaluation.
Footnotes
Acknowledgements
Not applicable.
Authors’ Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis, and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be authors as per the International Committee of Medical Journal Editors’ requirements/guidelines.
Consent for Participate
Not applicable.
Consent to Publication
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study does not involve experiments on animals or human subjects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors extend their appreciation to Prince Sattam bin Abdulaziz University for funding this research work through the project number (PSAU/2025/03/34679).
Informed Consent
Not applicable.
Use of Artificial Intelligence-assisted Technology
The authors declare that they have not used artificial intelligence (AI) tools for writing and editing of the manuscript, and no images were manipulated using AI.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors, or the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
