Abstract
Flurbiprofen is a non-steroidal anti-inflammatory drug (NSAID) with anti-inflammatory, antipyretic, and analgesic properties. NSAIDs are commonly used to treat conditions such as gout, arthritis, muscle pain, pyrexia, dysmenorrhea, and migraines; however, their toxicity may lead to hepatotoxicity, renal damage, and hypertension. In this study, a reversed-phase high-performance liquid chromatography (RP-HPLC) method was developed using a central composite design (CCD). The developed method was validated for linearity, robustness, limit of detection (LOD), limit of quantification (LOQ), precision, and accuracy, and was also applied to analyze flurbiprofen nanostructured lipid carriers (NLCs) and marketed formulations, including topical gel and eye drops. The calibration curve for flurbiprofen was linear over the concentration range of 0.125–50 µg/mL. The percentage recovery at sample concentrations of 400, 500, and 600 ng was 97.79%, 100.26%, and 100.61%, respectively, indicating good accuracy. The percentage relative standard deviation (%RSD) was less than 2%, confirming the precision of the method. Robustness was demonstrated by %RSD values below 2% following deliberate variations in flow rate, injection volume, and mobile phase pH. The LOD and LOQ were determined to be 50 ng/mL and 125 ng/mL, respectively. Using the optimized method, flurbiprofen was successfully identified and quantified in NLCs as well as in marketed topical gel and eye drop formulations, producing well-resolved peaks with high accuracy, precision, and sensitivity. Comprehensive testing and validation confirmed that the developed method is suitable for routine analytical applications.
Keywords
Introduction
An important non-steroidal anti-inflammatory drug (NSAID) that is used to treat rheumatoid arthritis is flurbiprofen. When treating rheumatoid arthritis, flurbiprofen is just as effective as other NSAIDs, such as diclofenac, indomethacin, and ibuprofen.1–4 Flurbiprofen is used to treat herpetic stromal keratitis and ocular gingivitis. 5 Research on the pharmacokinetics, tolerability, and safety of flurbiprofen in healthy elderly individuals revealed that it had a good safety profile and had dose-dependent central nervous system penetration. Due to its growing application in the pharmaceutical and clinical efficacy fields, a sensitive, accurate, and more practically relevant analytical method for flurbiprofen determination is required.6–8 Bagde et al. (2022) analyzed flurbiprofen at a wavelength of 247 nm using a C18 column (5 µm, 4.6 × 250 mm). The mobile phase was composed of 70:30 (v/v) acetonitrile (ACN) and water (pH 6). Injection volume of 50 μl, retention time of 2.5 minutes, flow rate of 0.5 mL/min. A linear calibration curve with a correlation value of R2 = 0.99. The limit of detection (LOD) was 1.31 μg/mL, and the limit of quantification (LOQ) was 3.97 μg/mL. 9
Akhlaq et al. (2011) used a high-performance liquid chromatography (HPLC) system that had a quaternary pump, an automated sampler, and a photodiode array detector. The wavelength was fixed at 247 nm. A Gemini C18 column (5 μm; 4.6 mm × 250 mm) was utilized to do the chromatographic separation. The mobile phase was made in a 50:50 v/v of ACN and disodium hydrogen phosphate (30 mM) at a pH of 7.0. 1 mL/min was the flow rate. The injector was rinsed using a 50:50 v/v ACN: Water mixture, and the injection volume was 5 mL. Validation was done once the method was developed. The calibration curves were linear in the 5–50 μg/mL range. The three samples’ intra-day and inter-day precision ranged from 0.004 to 0.030% and 0.001 to 0.027%, respectively. The LOD was 0.173 μg/mL, and the LOQ was 0.578 μg/mL, respectively. 10 In this research, we have developed an HPLC method for the quantification of flurbiprofen using design of experiment (DoE) software. Compared to conventional trial-and-error methods, DoE software helps to predict the optimized batch. It quickly identifies which HPLC parameters have major effects on the desired outcomes. It also reduces variation, minimizes defects, and makes processes more reliable and consistent.11,12 After the method development, we have validated the method. Then, the flurbiprofen was identified using the optimized method in nanostructured lipid carrier (NLCs), a marketed topical gel, and eye drops. The quantitative estimation revealed that the drug had remarkable peaks and achieved great sensitivity, precision, and accuracy. The main novelty of the work is that no HPLC method is developed for flurbiprofen using the DoE software, and also no authors have compared the developed method with the NLCs and the marketed topical gel and eye drops.
Materials and Methods
Chemicals
Flurbiprofen was purchased from Yarrow Chem, Maharashtra, India. Methanol and ACN (HPLC grade) were purchased from Finar, Ahmedabad, Gujarat, India. Triethanolamine and orthophosphoric acid were purchased from Loba, Mumbai, Maharashtra, India. High-quality pure water was taken from a Millipore purification system.
Equipment
For liquid chromatography estimations, an Agilent Infinity 1260 model was used with a variable wavelength detector, and OpenLab software was used. A C18 column (Phenomenex→, 250 mm length, 4.6 mm diameter, 5 µm particle size) was used for the analysis of flurbiprofen.
Preparation of Stock Solution and Working Standard Solution
One milligram of flurbiprofen was accurately weighed and dissolved in 1 mL of methanol to get 1,000 μg/mL. Then, from the 1,000 μg/mL stock, 100 μl was taken and diluted with 900 μl of methanol to get the second stock of 100 μg/mL. From the 100 μg/mL stock, 100 μl was taken and diluted with 900 μl of methanol to get a third stock solution of 10 μg/mL. To prepare the calibration curve, the working solutions of flurbiprofen were serially diluted with methanol to get the concentrations of 0.125–50 μg/mL. These stock solutions and working standard solutions were protected from light and stored at room temperature.
Preliminary Method Development Studies
Flurbiprofen’s solubility in organic solvents, specifically methanol and ACN, was evaluated. The purpose of this assessment was to determine their maximal solubility profiles. The method development involved combining the organic and aqueous phases (90:10). Triethanolamine and orthophosphoric acid were used to adjust the pH of the aqueous phase. All possible mobile phase ratios (50:50, 60:40, 70:30, 80:20) at different pH values were investigated to enhance chromatographic estimations. 13
Optimization of Analytical Method Using Design of Experiment
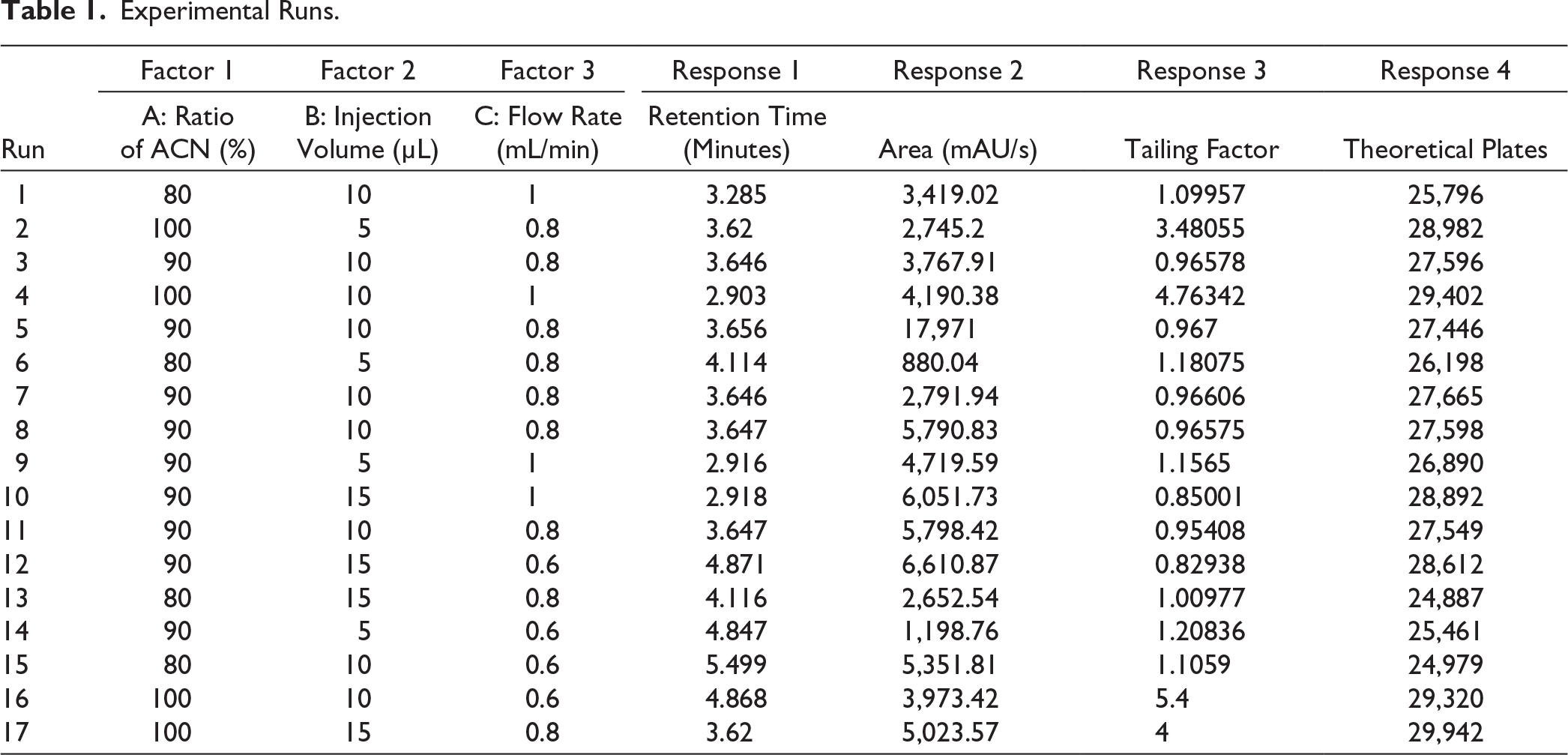
In this research work, to optimize the parameters, the central composite design (CCD) was employed. This design was performed in Design Expert software version 13. To estimate quadratic surfaces, CCD is a sequential response surface methodology that expands upon factorial designs. CCD tests extreme factor values (axial points), in contrast to Box–Behnken. Although it requires more runs than Box–Behnken design or screening-focused Taguchi methods, it is more effective for optimization than three-level factorials. 14 The ratio of ACN (80%–100 % range), injection volume (5–15 µL range), and flow rate (0.6–1 mL/min range) were selected as independent variables. Area, retention time, theoretical plates, and tailing factor were selected as dependent variables. The DoE experimental runs are given in Table 1.
Experimental Runs.
Development of a Reversed-phase-HPLC Method
To optimize the analytical method, we have used DoE (CCD) software. From the CCD, we got the optimized batch. The mobile phase used in the study was ACN and water (water pH was adjusted to 3.5), at a ratio of 90:10 (v/v), wavelength of 247 nm, injection volume of 15 μl, flow rate of 0.6 mL/min, samples were kept in room temperature, and column oven not used; analyses performed at ambient temperature 25 ± 2°C. Open lab software was used for data collection and analysis. 15
Validation of the Method
Linearity
The different concentrations of flurbiprofen aliquots were prepared by using methanol as a solvent with a concentration range of 0.125–50 μg/mL to test the method’s linearity. Each aliquot was prepared in triplicate. To obtain the calibration curve, the peak area was plotted against concentration. 16
Accuracy
The percentage mean recovery of the sample was the basis for the outcomes of recovery experiments. For the purpose of flurbiprofen estimation, three distinct concentrations, namely 400 ng, 500 ng, and 600 ng, were taken in sextuplicate. Recovery percentages were recorded once the samples were examined. The acceptable range for percentage recovery was 98%–102%, according to International Council for Harmonisation (ICH) guidelines. 17
Precision
To establish intraday and interday precision, flurbiprofen concentrations of 400, 500, and 600 ng/mL were analyzed at three distinct times in sextuplicate. On the same day, samples were analyzed with a four-hour interval, which is called intraday, and the next day same samples were analyzed, which is called interday. Percentage relative standard deviation (%RSD) is a measure of precision that expresses the standard deviation as a percentage of the mean value. Acceptable limit for the %RSD of area was less than two. 17
Robustness
By introducing small changes to the method’s state, such as flow rate (0.55 mL/min, 0.6 mL/min, and 0.65 mL/min), injection volume (14 μl, 15 μl, and 16 μl), and the pH (3.4, 3.5, 3.6) of the mobile phase composition, the robustness of the method was determined. Less than two was the acceptable limit for the %RSD of peak area. 18
Limit of Detection and Limit of Quantification
LODs are the lowest drug concentrations that can be reliably recognized and distinguished from the background. LOQs are the lowest concentrations at which they can be quantified. LOD is always when the signal-to-noise (S/N) ratio is 3:1. LOQ is always when the S/N ratio is 10:1. 19
Application of the Optimized Method in the Flurbiprofen-loaded NLC’s and in the Marketed Pharmaceutical Formulations
The flurbiprofen-loaded NLCs were prepared and analyzed by using a developed HPLC method for the estimation of flurbiprofen present in the prepared formulation. Flurbiprofen topical gel and eye drop was procured from the local pharmacy store. Flurbiprofen topical gel and eye drop samples for HPLC analysis were prepared in methanol and analyzed using a validated chromatographic method. But for the topical gel, we had made a further dilution to get a clear, non-viscous solution. The unknown concentration of the drug in these two marketed formulations was calculated from a linear equation, which we got from linearity. 20
Greenness Analysis of HPLC Method
To assess the environmental impact of an HPLC method by Modified Green Analytical Procedure Index (MoGAPI), Analytical GREEnness Metric Approach and Software (AGREE), Blue Applicability Grade Index (BAGI), it is necessary to provide procedural details such as solvent volume/mass, energy consumption, waste, process steps, reagents hazards, and sample throughput into their respective free online calculators or Excel software. MOGAPI:
Results and Discussion
Optimization of Analytical Method Using Design of Experiment
Retention Time
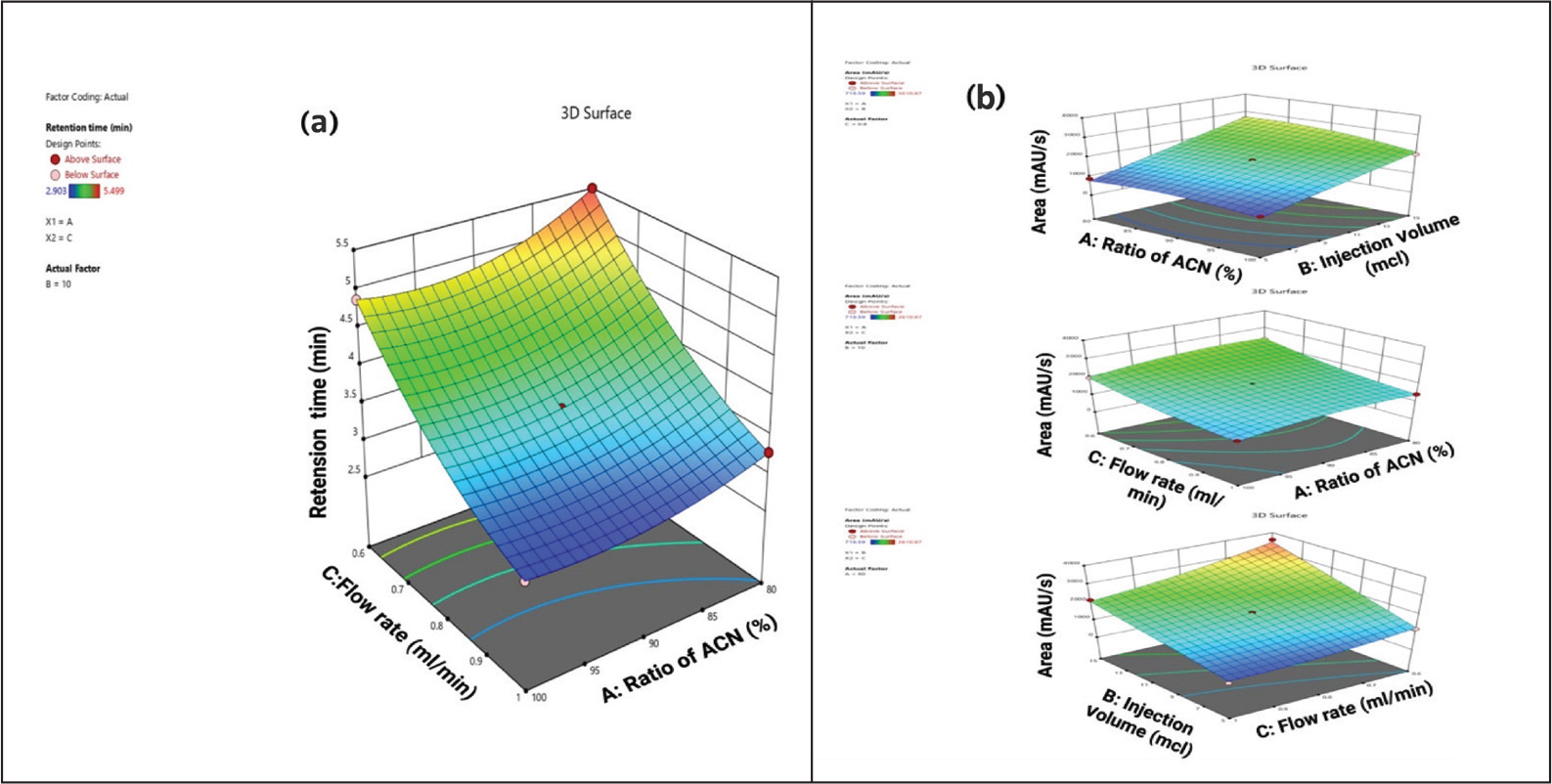
As per the polynomial equation, the ratio of ACN has a negative impact on the retention time, and the flow rate has a positive impact on the retention time. The 3D surface plot (Figure 1a) depicts that, when there is a decrease in flow rate, there will be an increase in retention time. A decrease in the ratio of ACN leads to an increase in retention time because it reduces their interaction with the stationary phase. According to the Analysis of Variance (ANOVA) table, the model’s F-value of 27,394.25 indicates that it is significant. The lack of fit F-value of 3.91 indicates that, relative to the pure error, the lack of fit is not significant.
(a) 3D Surface Plot of Retention Time, (b) 3D Surface Plot of Area.
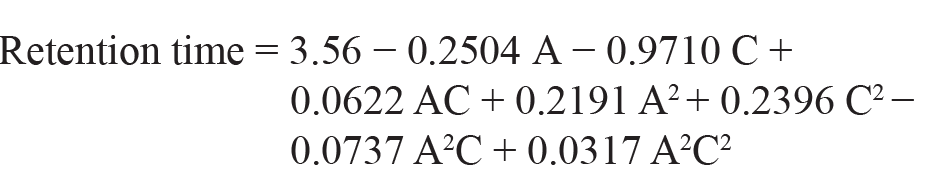
Area
As per the polynomial equation, the ratio of ACN and flow rate has a negative impact on area, and injection volume has a positive impact on area. The 3D surface plot depicts (Figure 1b) that, when there is a decrease in the ratio of ACN, there is an increase in area. When there is an increase in injection volume, there will be an increase in area. When there is a decrease in flow rate, there will be an increase in area, whereas when there is a decrease in the ratio of ACN, there will be an increase in area. According to the ANOVA table, the model’s F-value of 73.78 indicates that it is significant.
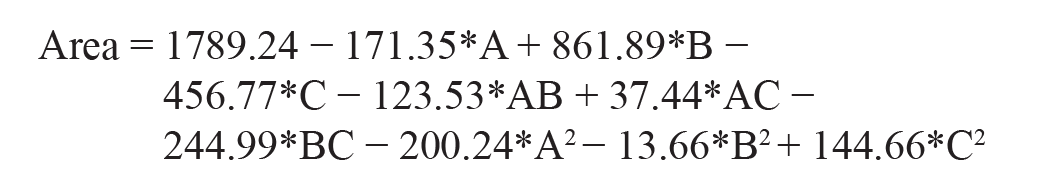
Tailing Factor
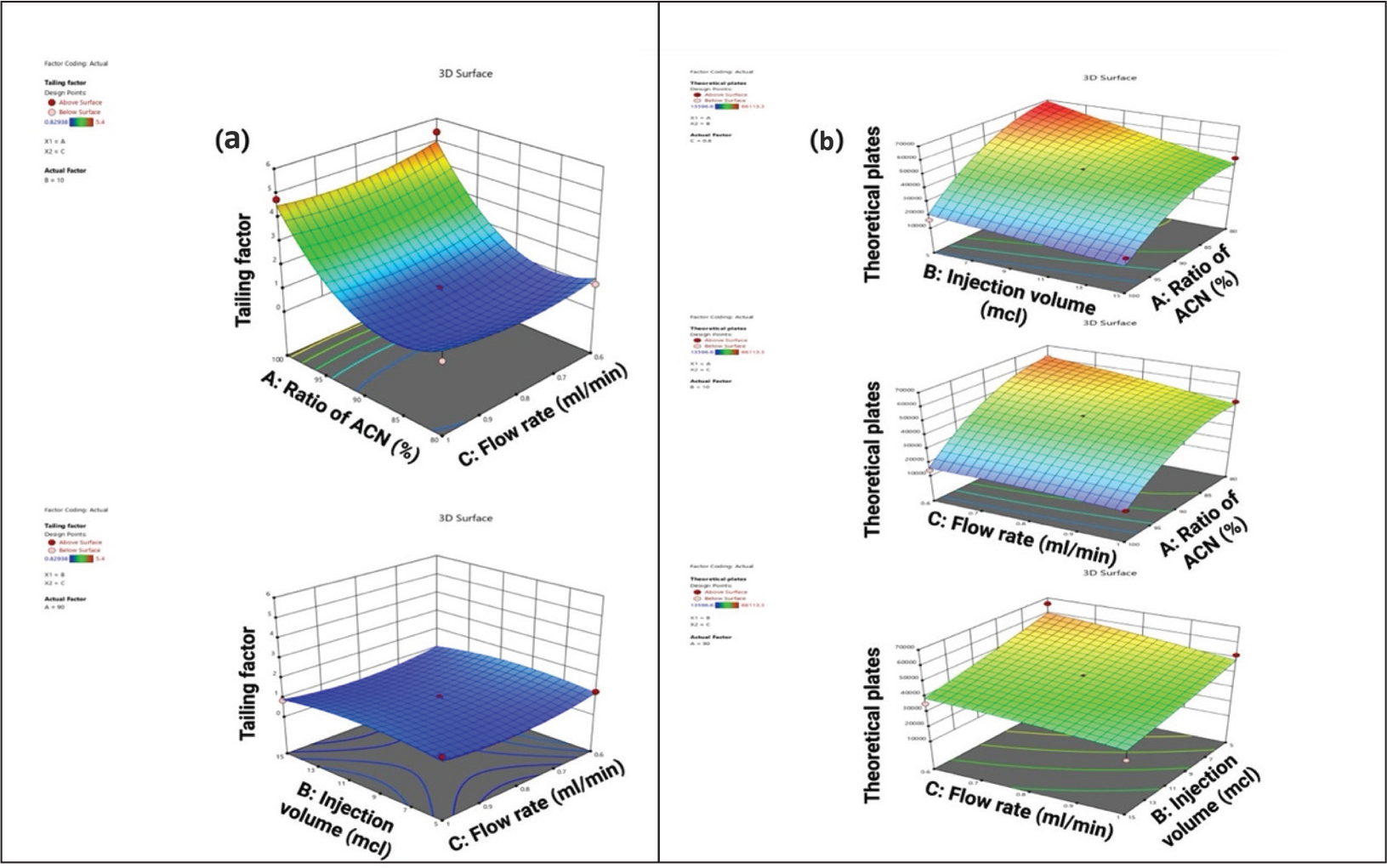
As per the polynomial equation, the ratio of ACN has a positive impact on the tailing factor, whereas injection volume and flow rate have a negative impact on area. As per the 3D surface area plot (Figure 2a), when there is a decrease in flow rate, there will be an increase in retention time. When there is an increase in the ratio of ACN, there is an increase in the tailing factor. When there is a decrease in the ratio of ACN, there is a decrease in retention time. When there is an increase in the ratio of ACN, there is an increase in the tailing factor. When there is a decrease in flow rate, there is a decrease in the tailing factor. The ANOVA suggested that the model F-value of 26.46 implies the model is significant.
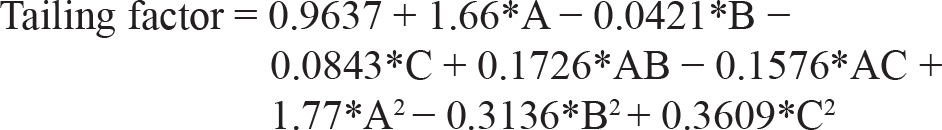
Theoretical Plates
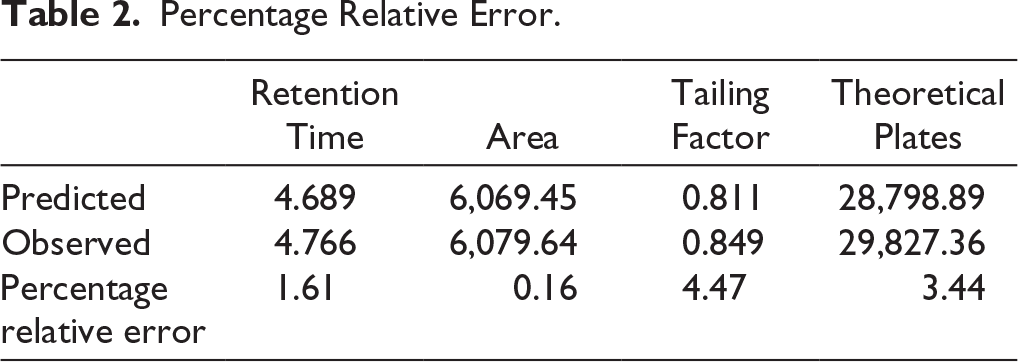
As per the polynomial equation, the ratio of ACN has a positive impact on theoretical plates, whereas injection volume and flow rate have a negative impact on theoretical plates. As per the 3D surface area plot (Figure 2b), when there is a decrease in injection volume, there will be a decrease in theoretical plates. A decreased ratio of ACN will increase the theoretical plates. Decreased flow rate will decrease the theoretical plates. The ANOVA table suggested that the model F-value of 16.75 implies the model is significant. The ANOVA table for particle size and Polydispersity Index is given in Supplementary Table 1. The ANOVA table for zeta potential and percentage entrapment efficiency is given in Supplementary Table 2. The predicted values versus observed values with the percentage relative error are given in Table 2.
Percentage Relative Error.
(a) 3D Surface Plot of Tailing Factor, (b) 3D Surface Plot of Theoretical Plate.
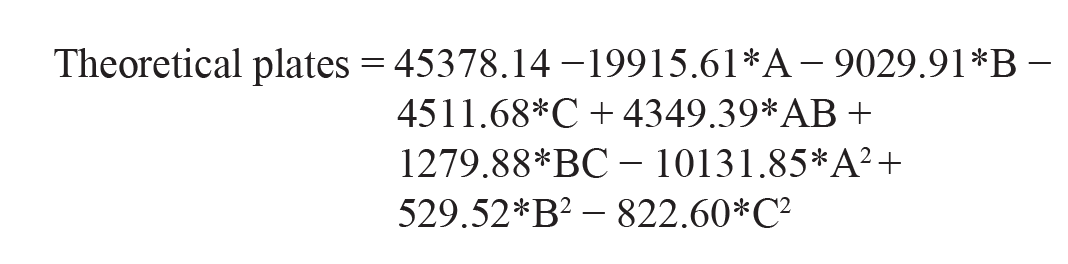
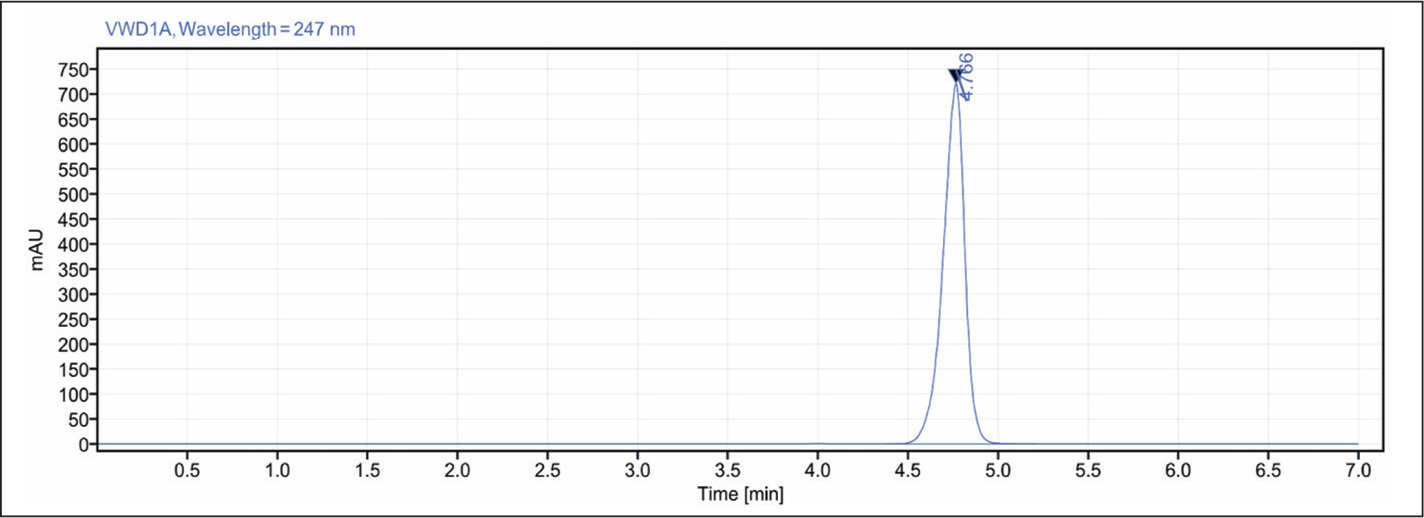
Development of a Reversed-phase-HPLC Method
ACN and water (pH adjusted to 3.5) were the given set of mobile phases for the HPLC chromatogram. The ratio was 90:10 (v/v), the wavelength was 247 nm, the flow rate was 0.6 mL/min, the injection volume was 15 μl, the run time was 7 minutes, the samples were kept at room temperature, and the column oven was not used; analyses performed at ambient temperature 25 ± 2°C. Then we got the HPLC chromatogram of flurbiprofen, which was depicted in Figure 3.
HPLC Chromatogram of Flurbiprofen.
Validation of the Method
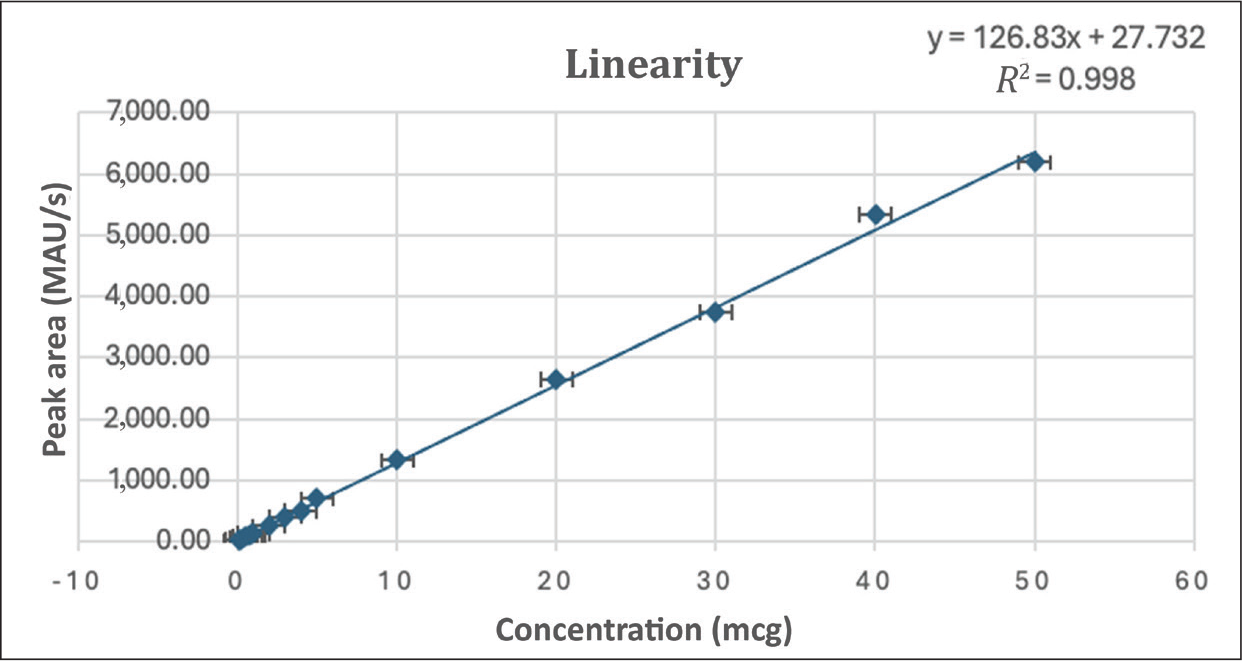
Linearity
Flurbiprofen’s calibration curve was linear over the 0.125–50 μg/mL concentration range, as seen in Figure 4. When the graph was plotted with peak area versus concentration, the regression equation for the calibration curve was typically determined to be y = 126.83 × + 27.732 with a 0.998 correlation coefficient.
Linearity of Flurbiprofen.
Accuracy
Analyses of the various flurbiprofen concentrations (400, 500, and 600 ng/mL) were conducted in sextuplicate. In Table 3, the percentage recovery was given.
The Percentage Recovery of the Samples.

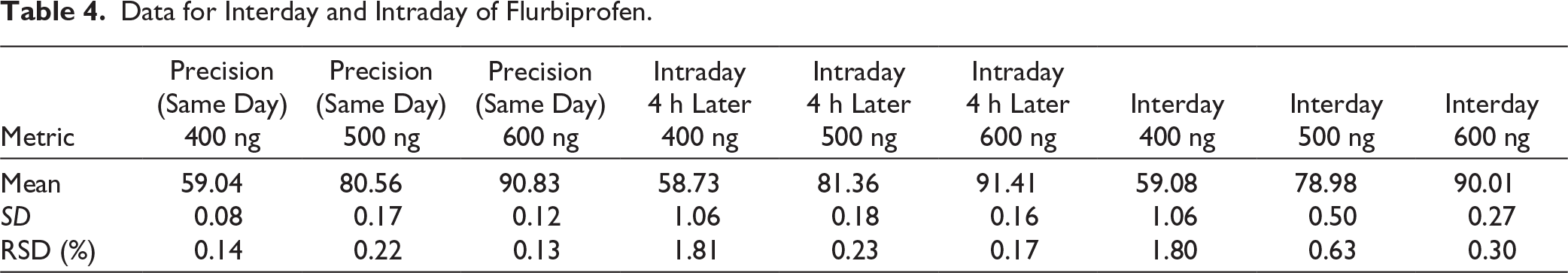
Precision
Analyses of the various flurbiprofen concentrations (400, 500, and 600 ng/mL) were conducted in sextuplicate at different times. In Table 4, precision, interday precision, and intraday precision were given. The %RSD was found to be less than two, which means that the established method was precise.
Data for Interday and Intraday of Flurbiprofen.
Robustness
For robustness studies, 3 μg/mL solution of flurbiprofen was used. Small changes to the method state, such as flow rate (0.55 mL/min, 0.6 mL/min, 0.65 mL/min), injection volume (14 μl, 15 μl, 16 μl), and the pH (3.4, 3.5, 3.6) of the mobile phase composition, were used to assess the method’s robustness. The %RSD of peak area was found to be less than two by change in flow rate, injection volume, and the pH of the mobile phase composition (Table 5).
Data for Robustness.
LOD and LOQ
Assuming a linear detector response in the low concentration range and stable baseline noise, the S/N approach was used to estimate the LOD and LOQ. LOD and LOQ were assessed at approximate S/N ratios of 3 and 10, respectively, in compliance with International Organization for Standardization (ISO) 11843, Eurachem guidelines, and ICH Q2(R2). Based on baseline noise measurement, the chromatographic data acquisition software automatically calculated the S/N ratios. In the present study, a flurbiprofen concentration of 50 ng/mL produced an S/N ratio of 3.9 and was therefore considered as the LOD, whereas a concentration of 125 ng/mL yielded an S/N ratio of 16 and was considered as the LOQ. 25
Application of the Optimized Method in the Flurbiprofen-loaded NLCs and in the Marketed Pharmaceutical Formulations
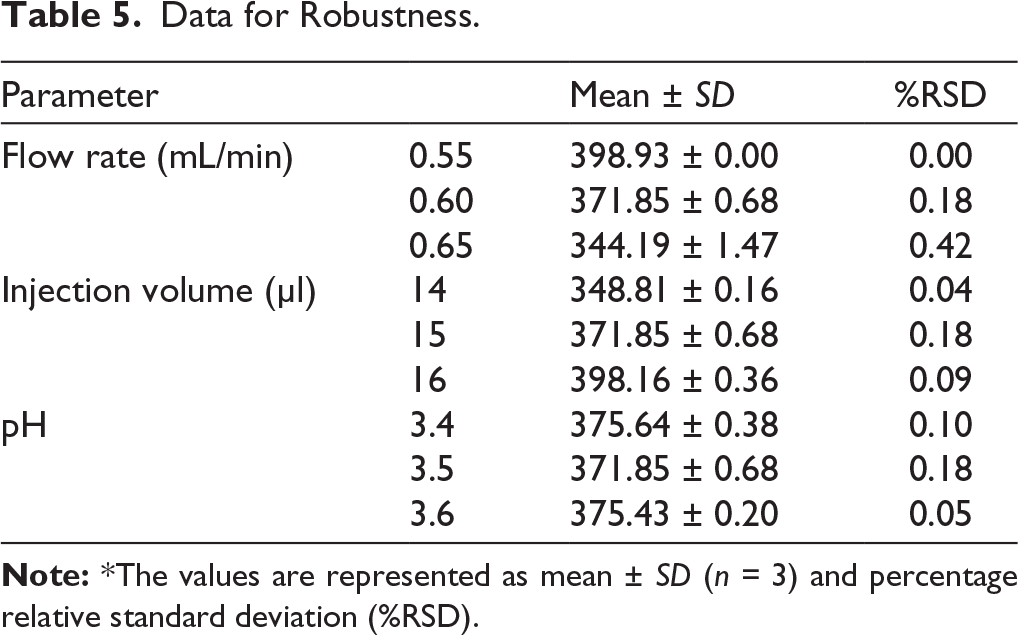
The amount of drug recovered from the prepared NLC formulation was found to be 96.05%. Flurbiprofen in commercially available topical gel and eye drops was effectively identified using the optimized method. The developed method was confirmed to be suitable for routine analysis. Figure 5a, Figure 5b, and Figure 5c show the HPLC chromatograms for the flurbiprofen NLCs, topical gel, and eye drop, respectively.
HPLC Chromatogram for (a) Flurbiprofen NLC, (b) Flurbiprofen Topical Gel, and (c) Flurbiprofen Eye Drop.
Greenness Analysis of HPLC Method
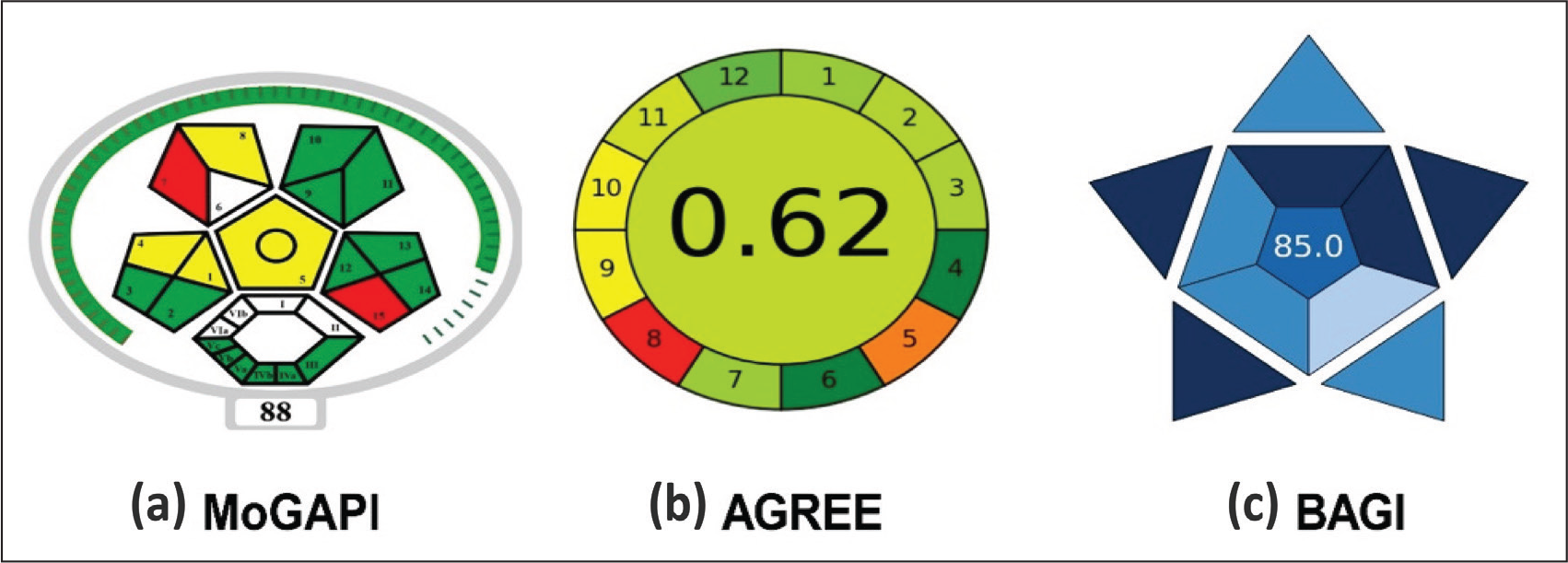
The HPLC method scores a moderate level of greenness according to MoGAPI (score 88), indicated by mixed green/yellow/red pictogram segments that evaluate 15 aspects of the procedure, such as solvents and waste. AGREE gave it a higher rating of 62%, indicating that the method was environmentally sustainable, based on 12 green chemistry principles, but still has some room for improvement in terms of energy or reagents. BAGI gave 85; thus, despite the moderate eco impact, the method was very practical, robust, and applicable for routine use. In general, the method was sufficiently green (50–75 range) and highly practical for pharmaceutical analysis, with the aim of further optimization of greener solvents. The results of the greenness assessment of the developed HPLC method are depicted in Figure 6.
Greenness Assessment of the Developed HPLC Method.
Conclusion
The developed reversed-phase-HPLC method for the estimation of flurbiprofen has been thoroughly tested and validated. The method exhibited excellent linearity over a broad concentration range of 0.125–50 µg/mL, demonstrating its suitability for detecting both low and high levels of the drug. Accuracy studies showed percentage recoveries close to 100% at multiple concentration levels, confirming that the method provides reliable and unbiased quantification of flurbiprofen. In the precision study, various flurbiprofen concentrations at different times were analyzed. The %RSD was found to be less than two, which means that the established method was precise. Robustness tests involving deliberate variations in flow rate, injection volume, and mobile phase pH resulted in minimal impact on the method’s performance, reflecting its stability under small procedural changes. Sensitivity parameters were also favorable, with the LOD at 50 ng/mL and the LOQ at 125 ng/mL, supporting the method’s capability to detect and quantify trace amounts of flurbiprofen. When applied to real pharmaceutical samples, including flurbiprofen-loaded NLCs, marketed topical gels, and eye drops, the method successfully identified and quantified the drug with distinct chromatographic peaks and consistent results. This confirms the method’s practical applicability in the routine analysis of complex flurbiprofen formulations.
Supplemental Material
Supplemental material for this article is available online.
Footnotes
Acknowledgements
The authors express heartfelt gratitude toward the Manipal College of Pharmaceutical Sciences, Manipal Academy of Higher Education, Manipal, for providing all the obligatory facilities for the completion of this research work.
Authors’ Contribution
All authors made substantial contributions to conception and design, acquisition of data, or analysis, and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be authors as per the International Committee of Medical Journal Editors’ requirements/guidelines.
Consent for Participate
Not applicable.
Consent to Publication
Not applicable.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
This study does not involve experiments on animals or human subjects.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable.
Patent Consent
An Indian patent has been filed for this research work. Patent application number is 202541071588.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors, or the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
Use of Artificial Intelligence-assisted Technology
The authors declare that they have not used artificial intelligence (AI) tools for writing and editing of the manuscript, and no images were manipulated using AI.
