Abstract
Posaconazole (PSC), a potent broad-spectrum triazole antifungal agent, is widely used in managing invasive fungal infections. Accurate measurement of PSC in pharmaceutical formulations is essential to ensure consistent therapeutic efficacy. Although high-performance liquid chromatography (HPLC) is the gold standard for detecting trace pharmaceutical concentrations, a notable gap remains due to the limited availability of validated HPLC methods specifically for PSC quantification. In this study, a sustainable and validated HPLC method was developed using PSC bulk drug and subsequently applied to quantify it in pharmaceutical formulations. The optimal chromatographic conditions consisted of a mobile phase composed of acetonitrile and water acidified with 0.5% acetic acid (55:45, v/v), a flow rate of 1 mL/min, and an injection volume of 20 µL. The method demonstrated excellent linearity within the concentration range of 0.5–32 µg/mL (R² = 0.9999), with a limit of detection (LOD) and limit of quantification (LOQ) of 0.16 and 0.50 µg/mL, respectively. Accuracy and precision results were within acceptable limits. Sustainability assessments using AGREE and GAPI tools confirmed the eco-friendly nature of the developed method. The validated HPLC method was successfully employed to estimate PSC content in a self-nanoemulsifying drug delivery system (SNEDDS) and during in vitro dissolution studies. Overall, the proposed HPLC method is analytically robust, reproducible, and environmentally sustainable, making it suitable for routine quantification of PSC in both bulk and formulation samples.
Introduction
Fungi are abundant in the environment, but only a few fungal species have been recognised as pathogenic in humans. Many of these fungi naturally coexist in the microbiomes of humans and animals. Fungal infections exhibit a broad spectrum of severity, ranging from mild, superficial manifestations to deeply invasive and potentially fatal conditions, particularly in immunocompromised individuals. 1 Recent estimates indicate that approximately 1.9 million individuals develop acute invasive fungal infections (IFIs) annually, while about 3 million people worldwide are affected by chronic severe fungal infections. These infections are often life-threatening, contributing to more than 1.6 million deaths each year globally. 2 However, the clinical diagnosis of IFIs remains challenging. The variations in the antifungal activity, dosage form, pharmacokinetic and pharmacodynamic properties, and adverse drug reactions necessitate a thorough understanding of each drug class. Even with the discovery of new drugs, there is still a great need for novel antifungal drugs to treat IFIs, which can be fatal.3,4 Presently, five azole antifungal drugs are used for systemic fungal infections. They are ketoconazole (an imidazole drug) and four triazole drugs: voriconazole, itraconazole, fluconazole, and the recent addition, posaconazole (PSC). Azoles are chosen over amphotericin B (AmB) because they are less toxic and suitable for oral administration. 5 The growing global incidence of multidrug-resistant fungal species poses a serious threat, leading to poor treatment outcomes and increased mortality rates. Several fungal pathogens have developed resistance across all four major antifungal drug classes, polyenes, azoles, echinocandins, and the pyrimidine analogue 5-flucytosine, while some strains exhibit intrinsic resistance and high tolerance to these agents. Given the limited availability of systemically active antifungal drugs, the management of IFIs remains a significant clinical challenge. 4
PSC (C37H42F2N8O4) is an N-aryl piperazine that consists of piperazine with two 4-substituted phenyl groups at positions 1 and 4, and is the first-line treatment for oropharyngeal candidiasis 6 (Figure 1). It is also a preventive medicine for acute myeloid leukaemia (AML) patients undergoing induction chemotherapy and recipients receiving immunosuppressive treatment after haematopoietic stem cell transplantation (HSCT). It is also prescribed to patients with myelodysplastic syndrome (MDS) who are at risk of IFI due to persistent neutropenia. 7 PSC is highly recommended for managing mucormycosis in combination with AmB for treating refractory illnesses or salvage therapy.
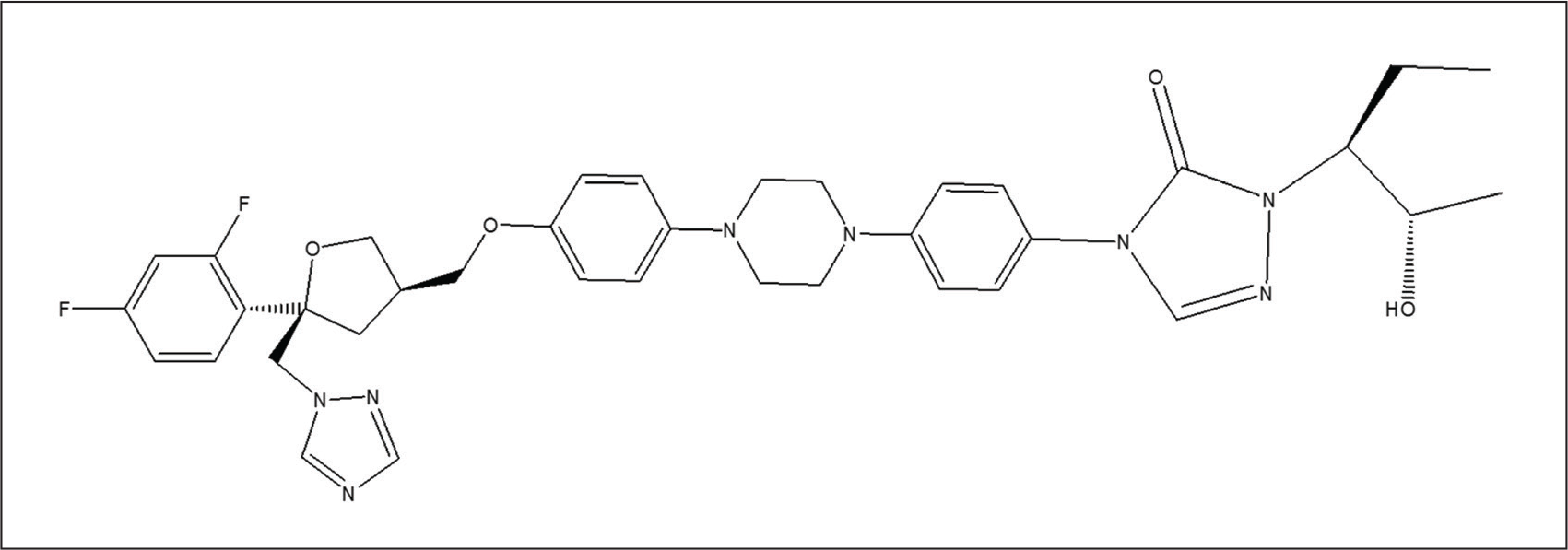
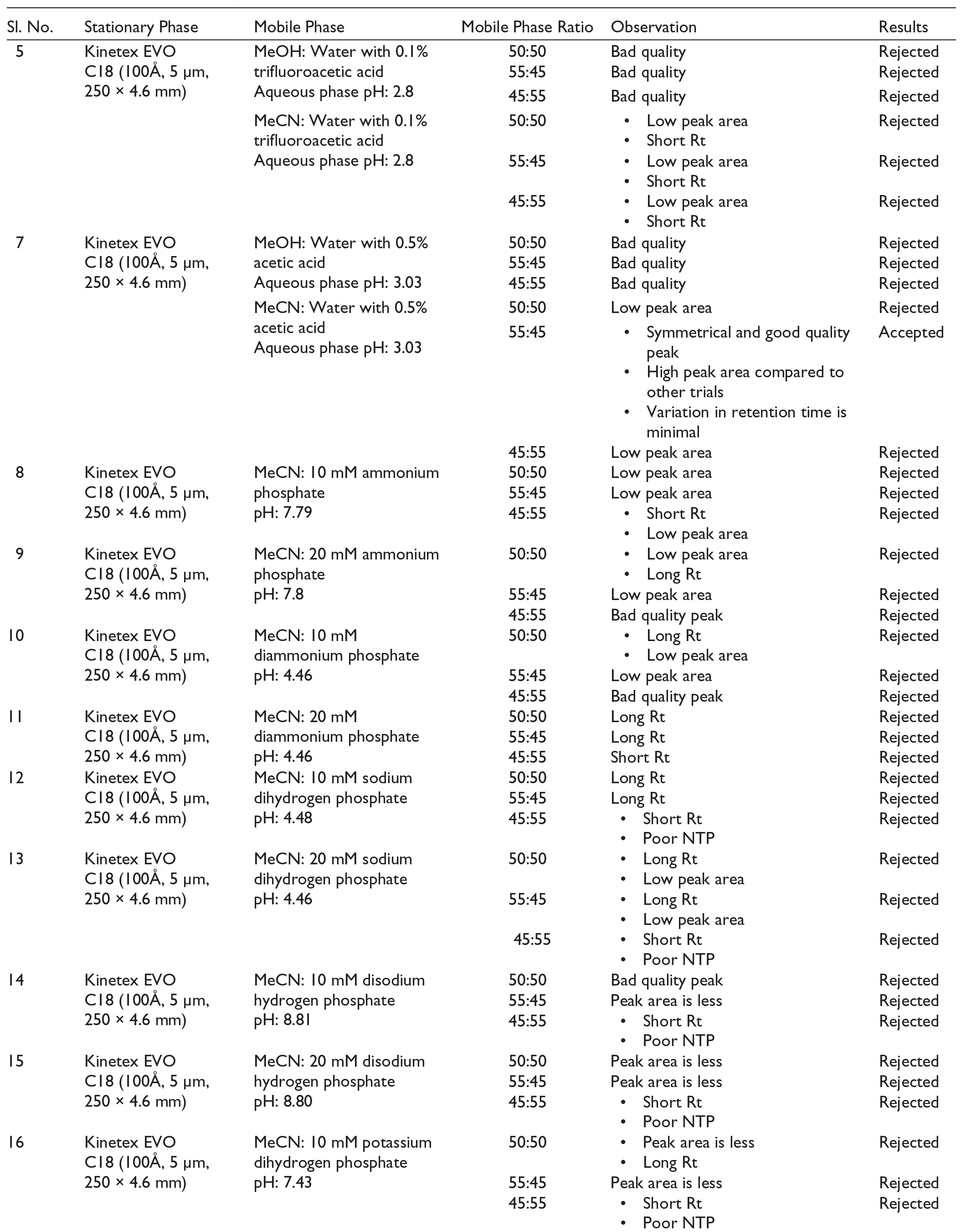
The mechanism of action of PSC involves the inhibition of lanosterol 14α-demethylase, an enzyme essential for the synthesis of ergosterol, a vital component of fungal cell membranes. This inhibition leads to the accumulation of precursor sterols and a reduction in ergosterol content, thereby disrupting the structure and function of the fungal cell membrane and resulting in potent antifungal activity. PSC, although it demonstrates potent in vitro activity against a wide range of Candida species, is not approved for the treatment of invasive or other systemic Candida infections beyond oropharyngeal candidiasis and prophylaxis. This limitation is primarily due to the lack of large, well-controlled clinical trials establishing its efficacy and safety in invasive candidiasis. 8
PSC is a weakly basic compound with high lipophilicity, closely resembling itraconazole in this respect. 9 PSC exhibits comparable in vitro antifungal activity to itraconazole, but with a lower minimum inhibitory concentration (MIC₅₀), and has demonstrated clinical utility in cases involving itraconazole-resistant fungal strains. Like itraconazole, PSC displays relatively poor oral bioavailability (approximately 8%–47%); however, this limitation can be mitigated through the use of lipid-based formulations and administration with a high-fat meal. Notably, administration of single doses exceeding 400 mg does not further elevate the maximum plasma concentration, and daily doses above 800 mg fail to enhance systemic absorption. 10
Despite significant advancements in pharmaceutical technologies that enhance the formulation characteristics of drug molecules, the development and validation of robust analytical methodologies remain essential for accurately evaluating nanoformulations. The rigour and quality of these analytical approaches exert a critical influence on the overall success of formulation development. In the case of PSC, precise and reproducible quantification within pharmaceutical formulations is essential to ensure accurate dosage delivery, therapeutic efficacy, quality assurance, and reliable pharmacokinetic profiling. 11
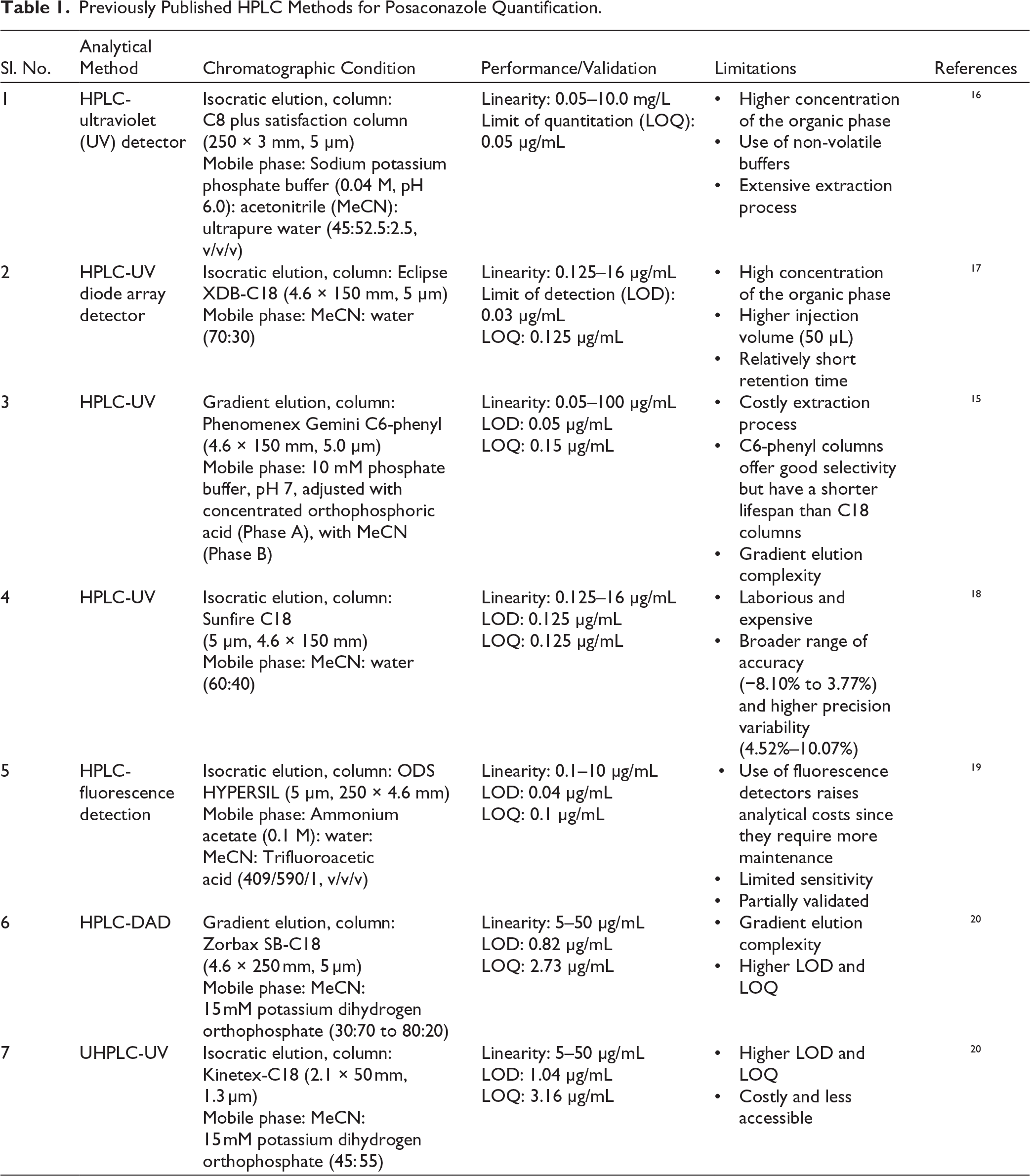
This study was primarily undertaken to develop and validate a UV-assisted high-performance liquid chromatography (HPLC) method, characterised by high speed, sensitivity, selectivity, and precision, for the accurate quantification of PSC in pharmaceutical formulations. The method’s applicability was demonstrated by quantifying PSC in an optimised self-nanoemulsifying drug delivery system (SNEDDS), and it is readily extendable to other PSC-loaded formulations. A few HPLC methodologies have been reported for the accurate quantification of PSC, with even fewer subjected to comprehensive scrutiny and validation. While these techniques provide exceptional sensitivity, specificity, and reduced run times, their complexity and substantial cost render them inaccessible to numerous laboratories. Comparatively higher concentrations of organic solvents in the mobile phase of the proposed chromatographic methods raise high costs and are incompatible with green chemistry.12–15 Table 1 summarises previously reported HPLC analytical methods for PSC quantification, including their respective chromatographic parameters.
Previously Published HPLC Methods for Posaconazole Quantification.
A trial-and-error or one-factor-at-a-time (OFAT) strategy is frequently used to develop HPLC methods. This approach changes one chromatographic parameter in successive runs until a satisfactory resolution between chromatographic peaks is reached. 21 A thorough assessment of existing analytical methods served as the basis for developing a sustainable HPLC method for the quantification of PSC, with literature-based insights directing the choice of a strategy that achieves a compromise between cost-effectiveness and technical precision. Important insights into common analytical difficulties, effective separation techniques, critical method parameters, and potential areas for innovation were obtained from the methodical review of published research. 22
Extensive literature exploration was carried out across various scientific databases, including Scopus, Web of Science, and Google Scholar. Following further refinement of these publications, articles were selected for comprehensive parameter evaluation based on the practicality of the analytical methodologies. The systematic review conducted a thorough analysis of key HPLC method parameters. Initially, the mobile phase characteristics were extensively examined, including the optimisation of the organic phase composition, selection of suitable aqueous phases, and meticulous adjustment of aqueous phase pH to improve separation efficiency.
Green analytical chemistry (GAC) is instrumental in substituting or reducing the volume of harmful organic solvents while maintaining the chromatographic efficiency in pharmaceutical quality control (QC) via liquid chromatography (LC). Furthermore, environmentally friendly mobile phases meet the criteria established by the US Environmental Protection Agency, intended to limit the adverse environmental impacts related to analytical waste. 23 Rooted in the 12 principles of green chemistry, the Analytical Greenness (AGREE) calculator serves as a robust, flexible, and transparent tool for evaluating the environmental sustainability of analytical methodologies. Additionally, the Green Analytical Procedure Index (GAPI) tool evaluates the environmental impact of developed methods. GAPI uses a visual representation to classify the eco-friendliness of specific stages within an analytical method, adopting a colour-coded scale with two or three degrees of assessment for every step. 24 The previously published methods for estimating PSC did not consider environmental concerns while developing analytical methods.
Considering the preceding findings and observations, this study focused on developing and validating a green, stability-indicating RP-HPLC method for PSC quantification. The method was systematically optimised by modifying key chromatographic parameters, including mobile phase composition, pH levels, and phase ratio variations. Adhering to the International Conference on Harmonisation (ICH) Q2 (R1) guidelines, 25 the validation process ensured accuracy, precision, and specificity of the developed method. It was successfully employed for the quantification of PSC in both its bulk substance and formulated pharmaceutical dosage forms.
Materials
Method development was performed using an HPLC system, Shimadzu LC-2010CHT equipped with a dual-wavelength UV detector (SPD-M20A prominence). Subsequent analysis of the chromatograms was performed using LC Solution software (version 5.57). The Kinetex EVO C18 (100Å, 5 µm, 250 × 4.6 mm) LC column was used as the stationary phase. Filtration of the mobile phase was accomplished using a Millipore glass filter with a 0.22-micron pore-size nylon membrane filter paper, connected to a glass vacuum filtration unit. Degassing of the mobile phase was done by sonicating for 10 min using a GT Sonic Professional Ultrasonic Cleaner (GT-2013QTS, GT Sonic, China). pH measurement of the Mobile phase was conducted using a pH meter (Systronics, India). The study utilised Milli Q Direct-Q 3 UV Water Purification System (Merck, United States), Analytical Weighing Balance (BSA224S-CW model, Sartorius in Germany), and UV spectrophotometer (UV 1800, Shimadzu, Japan).
PSC (CAS Number: 171228-49-2) was obtained as a complimentary sample from Lupin Healthcare Limited. HPLC grade acetonitrile (CAS Number: 75-05-8, purity ≥ 99%), acetic acid (CAS Number: 64-19-7, purity ≥ 99%), HPLC grade methanol (CAS Number: 67-56-1), formic acid, orthophosphoric acid, trifluoroacetic acid, ammonium phosphate, diammonium phosphate, sodium phosphate, disodium phosphate, potassium phosphate, dipotassium phosphate, potassium acetate, and dipotassium acetate, were obtained from Merck Ltd. (Mumbai, India). A nylon membrane filter (0.45 μm) was procured from HiMedia Pvt. Ltd (Mumbai, India).
Methodology
Selection of Wavelength
The stock solution was prepared by precisely weighing PSC (10 mg) and dissolving it in MeOH (10 mL). The UV spectrophotometer identified the absorbance maxima across a concentration range of 0.5–30 μg/mL, which were subsequently utilised to establish the HPLC analytical method. 26
Systematic Investigation of Optimised Chromatographic Conditions
The methodology involved evaluating different mobile phases in various ratios, with varying drug concentrations and pH values. Optimisation was based on chromatographic parameters such as peak area, retention time (Rt), number of theoretical plates (NTP), and tailing factor (Tf). The organic phases included MeOH and MeCN, while the aqueous phases consisted of acidified water (0.1% formic acid, 0.1% trifluoroacetic acid, 0.1% orthophosphoric acid, 0.5% acetic acid),27,28 phosphate buffers, Ammonium dihydrogen phosphate (10 mM, 20 mM), Diammonium hydrogen phosphate (10 mM, 20 mM), Sodium dihydrogen phosphate (10 mM, 20 mM), Disodium hydrogen phosphate (10 mM, 20 mM), and acetate buffers,29–31 pH values were adjusted between 2.8 and 8.9.
Chromatographic Conditions
The chromatographic condition utilised a stationary phase of Kinetex EVO C18 (100Å, 5 µm, 250 × 4.6 mm). Mobile phase consisting of MeCN and water acidified with 0.5% acetic acid mixed in a 55:45 volume ratio, with the resulting aqueous phase pH measured at 3.03. An isocratic elution technique was implemented, maintaining a flow rate of 1 mL/min for a 10-minute run period. The injection volume was set to 20 μL, with the column temperature sustained at 25°C. Detection of the analyte was carried out at a wavelength of 262 nm.
A precisely measured 10 mg of PSC was introduced into a 10 mL volumetric flask and solubilised in MeCN to produce a stock solution. A final concentration of 1 µg/mL was then prepared by appropriate dilution of the stock solution. The analysis was performed using an isocratic elution method.
Validation of Developed HPLC Analytical Method
Analytical test method validation aims to confirm that a certain analytical method, like HPLC, is reliable, accurate, and consistent within a certain range when analysing a specific analyte. 32 Validation assures the method’s reliability under typical application conditions while also establishing a structure for carrying out such validation. It is sometimes referred to as the procedure for giving written proof that the method achieves its intended results. This feature receives significant consideration in both the literature and regulatory standards. Guidelines from the Food and Drug Administration (FDA), United States Pharmacopoeia (USP), and the ICH offer a structured approach for validating HPLC methods in the pharmaceutical field.25,33
As per the requirements of the ICH Q2 (R1), the performance of the optimised analytical method was assessed in this study through a sequence of validation experiments.33–35
Specificity
The term ‘specificity’ is an analytical method’s ability to precisely detect the target analyte within a sample, even in the presence of other excipients, thereby ensuring minimal interference from extraneous substances. 36 Specificity was effectively demonstrated by comparing the chromatograms of the pure drug and the marketed formulation for the presence of any additional peaks or overlapping signals at the Rt of PSC. Three samples, a blank solution, PSC solution at a concentration of 1 µg/mL, and a commercial formulation of PSC equivalent to 1 µg/mL concentration, were analysed on the instrument to determine the specificity of the optimised analytical method. The chromatograms of all samples were examined to detect any possible interference caused by excipients or diluents at the Rt of PSC. 26
Linearity
Validating linearity requires analysing independently produced sample solutions. The linearity evaluation of the optimised HPLC analytical method involved generating a wide concentration series of standard PSC solutions ranging from 0.5 to 32 µg/mL. This broad range was selected to ensure the method could accurately quantify both low and high concentrations of the PSC in pharmaceutical formulations across a wide range of possible concentrations. 37
Each concentration was subjected to six injections, and the corresponding peak areas (mV-min) were systematically plotted against their respective concentrations (µg/mL) to construct a regression model. The intercept and slope of this plot were calculated using a linear regression equation.
Accuracy
Accuracy in an analytical method refers to the extent to which the measured value aligns with a reference standard or an accepted true value. The accuracy of the developed method was evaluated by analysing standard PSC working solutions at four concentration levels, limit of quantification (LOQ), 2, 4, and 8 µg/mL. Each level was injected six times, and accuracy was expressed as the mean percentage recovery. This ensures that the method’s ability to provide precise and reliable measurements is evaluated across low, medium, and high points within the quantifiable range, thereby validating its suitability for accurate determination of PSC in formulations.
Intra-day accuracy was evaluated by injecting each concentration six times at two distinct intervals within the same day (10:00 and 20:00 hr), while inter-day accuracy was determined by analysing six injections of each concentration across two consecutive days. The accuracy of the method was determined by calculating the mean percentage recovery at each concentration level. 26
Sensitivity
Sensitivity is characterised by either the limit of detection (LOD) or the LOQ.
38
LOD is defined as the smallest amount of analyte (or measurand) in a sample that may be reliably detected with a specific probability. LOQ is defined as the smallest quantity of a measurement in a sample that can be quantitatively determined with accuracy and precision. The LOQ and LOD are calculated using the formula
39
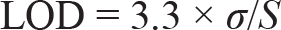
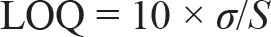
Where, σ = residual standard deviation of the regression line; S = slope of the regression line
Precision
Precision is defined as the reproducibility of an analytical measurement, reflecting the method’s ability to consistently generate concordant results under identical experimental conditions. The precision of the developed method was evaluated using four QC levels. QC concentrations were prepared from the stock solution of PSC within the validated linearity range of 0.5–32 µg/mL to evaluate the accuracy, precision, and reproducibility of the developed HPLC method. The low QC (LQC), medium QC (MQC), and high QC (HQC) levels were selected to represent the lower, middle, and upper portions of the calibration curve, respectively. The LQC was chosen near the lower end of the validated range to confirm method performance at low analyte concentrations. The MQC corresponded to the midpoint of the range, and the HQC represented the upper limit.26,40 This selection of QC concentrations ensures that reproducibility is thoroughly assessed across the entire analytical range, encompassing concentrations near the detection limit, the mid-range, and approaching the upper limit, thereby validating the suitability of the method for routine quantification of PSC in formulations.
Six injections of each QC concentration were analysed twice in a day to ensure intra-day precision. For two days in a row, two injections of each QC concentration were examined for inter-day precision. 41 The peak area for each concentration and the percentage relative standard deviation (RSD%) were calculated.
Robustness
Robustness assesses the methodological reliability and stability, confirming that slight deviations in operational conditions do not adversely impact performance or analytical accuracy. The robustness of the developed method was assessed by three injections of a 1 µg/mL drug concentration while varying parameters such as the mobile phase ratio (%), column oven temperature (°C), wavelength (nm), flow rate (mL/min), injection volume (μL), and pH of the mobile phase. The peak area and Rt corresponding to each condition were analysed to calculate the percentage relative standard deviation, ensuring the consistency and reliability of the measurement. 42
System Suitability
The performance and dependability of an Analytical method must be evaluated for system suitability to determine whether the system is suitable for the intended analysis. The system suitability for the optimised method was determined by carefully analysing the peak areas and Rts that resulted from six injections of a drug solution at a concentration of 1 µg/mL in this investigation. An essential statistic for assessing the overall effectiveness and consistency of the analytical process was the calculation of the RSD% for chromatographic parameters. 26
Forced Degradation Studies
Forced degradation studies are an essential component of stability-indicating HPLC method development, providing critical insight into the chemical behaviour and stability profile of the drug under various stress conditions. It helps demonstrate that the analytical method can reliably separate the drug from its potential degradation products (DP), ensuring accurate quantification and supporting regulatory requirements for quality assurance. The study was performed in accordance with ICH Q1A (R2) guidelines, 43 exposing PSC to various stress conditions, including acidic, basic, oxidative, photolytic, and thermal degradation.
Acidic hydrolysis was carried out using 1 N of HCl, and base hydrolysis with 1 N of NaOH. Oxidative degradation was induced using 3% hydrogen peroxide (H2O2) at 80°C. To the standard solution (500 μL of 10 μg/mL), 500 μL of each degradation solution (1 N of HCl, 1 N of NaOH, and 3% H2O2) was added, and the mixture was maintained at 80°C. At predetermined intervals (24, 48, 72 hr), 100 μL aliquots were withdrawn, neutralised, diluted and analysed. The PSC sample (10 μg/mL) was subjected to photodegradation by exposure to natural UV light and thermal degradation at 80°C. For analysis, the PSC samples (100 μL) were withdrawn at 24, 48, and 72 hr diluted with the mobile phase. 27
Assessment of the Eco-friendliness of the Developed Analytical Method
Recently, there has been a growing trend among researchers to adopt the ideas of GAC. This strategy aims to achieve molecular-level sustainability while minimising or reducing the adverse impacts of chemical procedures on the environment. Paul Anastas and John Warner authored the first green chemistry manual in the 1990s. It contained a list of 12 recommended green chemistry principles that focused on avoiding or ceasing the use of hazardous solvents in chemical procedures and assessment, as well as preventing the generation of residue from these methods. The advancement of analytical techniques stands as one of the most dynamic areas of research and development within green chemistry. 44 The current investigation used two types of greenness assessment tools, AGREE 45 and GAPI, 46 in compliance with the 12 principles of green chemistry.
Analytical Greenness
AGREE is an inclusive, adaptable, simple evaluation tool that gives easily interpretable outcomes. The assessment criteria of AGREE are derived from the 12 principles (SIGNIFICANCE) of GAC, which are then converted into a scale ranging from 0 to 1. The SIGNIFICANCE principles are used to compute the final score. The result is represented in a traffic light colour scheme, which encompasses greenness standards represented by the green colour, analytical effectiveness indicated by the red colour and productivity indicated by the blue colour, offering a visual interpretation of both the final score and the performance of the analytical method across each criterion. 47
Green Analytical Procedure Index
The GAPI assesses the overall environmental sustainability of an analytical method, encompassing every stage from sample collection to the final evaluation. GAPI combines pictograms and eco-scale to provide both broad information on analytical methods and qualitative information. GAPI uses a symbol with five pentagrams to assess the impact on the environment of each analytical step. Green, yellow, and red represent low, medium, and high impacts, respectively. 48 The GAPI tool is mainly useful for comparing analytical methods. 49
Method Applicability
The validated HPLC method was applied to accurately quantify PSC in the developed SNEDDS. The method was employed to determine the drug content in the formulation and to evaluate the in vitro drug release profile during dissolution studies, thereby demonstrating its suitability as a stability-indicating and formulation-assessing analytical method.
Formulation of SNEDDS of PSC
A surfactant combination (Smix) was made by mixing 25% of Tween 20 and 62.38% polyethylene glycol 200 (PEG 200). In a glass vial, 125 mg of precisely weighed PSC was combined with 12.62% of the oil phase, and the mixture was gently stirred on a magnetic stirrer at 25°C ± 2°C until it was completely dissolved. While being continuously stirred at 300 rpm, the Smix was gently added to the drug-oil solution. After homogenising the mixture for 10 min at 500 rpm using a vortex mixer, it was ultrasonically sonicated for an additional 10 min in a temperature-controlled bath sonicator (GT Sonic Professional, GT-2013QTS) to ensure even dispersion.
Determination of Total Drug Content in PSC-loaded SNEDDS
Approximately 100 μL of the developed PSC-loaded SNEDDS was transferred into an Eppendorf tube, followed by the addition of 900 μL of methanol. After thorough mixing, the mixture was centrifuged at 20,000 rpm and 4°C for 15 min. The resulting supernatant was carefully diluted, and the drug content was determined using a validated HPLC analytical method.
In Vitro Drug Release Study
The dialysis bag method was employed to assess in vitro drug release, utilising a semi-permeable membrane that retains excipients, carriers, and formulation components within the bag while allowing only the free (solubilised or unbound) drug to diffuse into the release medium. The release behaviour of pure PSC and optimised PSC-loaded SNEDDS was systematically evaluated using this technique. For each experiment, an equivalent of 35 mg PSC was accurately placed into a pre-hydrated dialysis membrane (molecular weight cut-off: 12–14 kDa) and sealed carefully to eliminate air entrapment. The study design incorporated a sequential media transition: simulated gastric fluid (SGF, pH 1.2) for the first 0–2 h, followed by simulated intestinal fluid (SIF, pH 6.8) containing bile salts for 3–24 h. Experimental conditions were maintained at 37°C ± 0.5°C with continuous agitation at 100 ± 2 rpm. Samples (2 mL) were withdrawn at predetermined intervals (15, 30, 60, 90, and 120 min in SGF; 180, 240, 300, 360, 420, 480 min, and 24 h in SIF), with immediate replacement by equal volumes of fresh medium to maintain sink conditions. The collected samples were analysed using a validated HPLC method.
Results and Discussion
Systematic Investigation of Optimised Chromatographic Conditions
Quantitative distribution patterns from the comprehensive review of various published HPLC methods for PSC quantification provided numerous important new insights. The concentration of the organic phase plays a crucial role, as selectivity in HPLC is commonly enhanced by modifying the mobile phase composition. 50 The type of the organic phase is just as significant as the concentration. Using different organic phases has been reported to change the mechanism of retention. For instance, the retention mechanism is more partition-like for combinations of water and MeOH and more adsorption-like for mixtures of water and MeCN. 51 Additionally, the type of organic phase affects the thermodynamic behaviour of the retention process. This discrepancy is thought to result from either the superior capacity of MeCN to penetrate the stationary phase or its stronger propensity to aggregate around solutes in comparison to MeOH. 52 MeCN emerged as the preferred organic solvent (~80%) over MeOH (~20%), attributed to its advantageous chromatographic properties, including reduced viscosity and improved separation efficiency.
The evaluation of aqueous phase composition revealed a clear stratification, with water being the primary constituent, followed by phosphate buffer as the most employed alternative, while formate, acetate, and bicarbonate buffers were utilised to a lesser extent. 17 pH distribution ranged from 2 to 8, with a notable predominance of acidic conditions (pH 2–4) in many reported methods.
Relevant data regarding HPLC analytical methods and the chromatographic behaviour of PSC were obtained through a comprehensive review of the literature. The OFAT method was chosen due to the exploratory nature of the work, where the main goal was to identify suitable excipients and approximate concentration ranges before implementing complex multivariate designs. The OFAT approach allowed systematic assessment of individual variables under controlled conditions, ensuring reproducibility and straightforward interpretation of their effects. Additionally, given the limited number of critical factors and resource constraints, OFAT provided a practical and efficient way to generate robust preliminary data. Although experimental design or analytical quality by design approaches offer considerable advantages for multifactorial optimisation, the insights gained from OFAT in this study establish a solid basis for subsequent DoE-based refinement. The optimised chromatographic conditions were then determined by conducting various trials that varied the mobile phase, its composition, and the concentration of samples. An exploration of different solvent systems as mobile phases was performed to develop an environmentally friendly HPLC method, aligned with green chemistry principles. The conclusions from these preliminary optimisation trials are summarised in Table 2.
List of Preliminary Trials for HPLC Method Development.

Following the optimisation process, the chromatographic conditions outlined in Section ‘Chromatographic Conditions’ were used for the optimised method.
Validation of Developed HPLC Method
Specificity
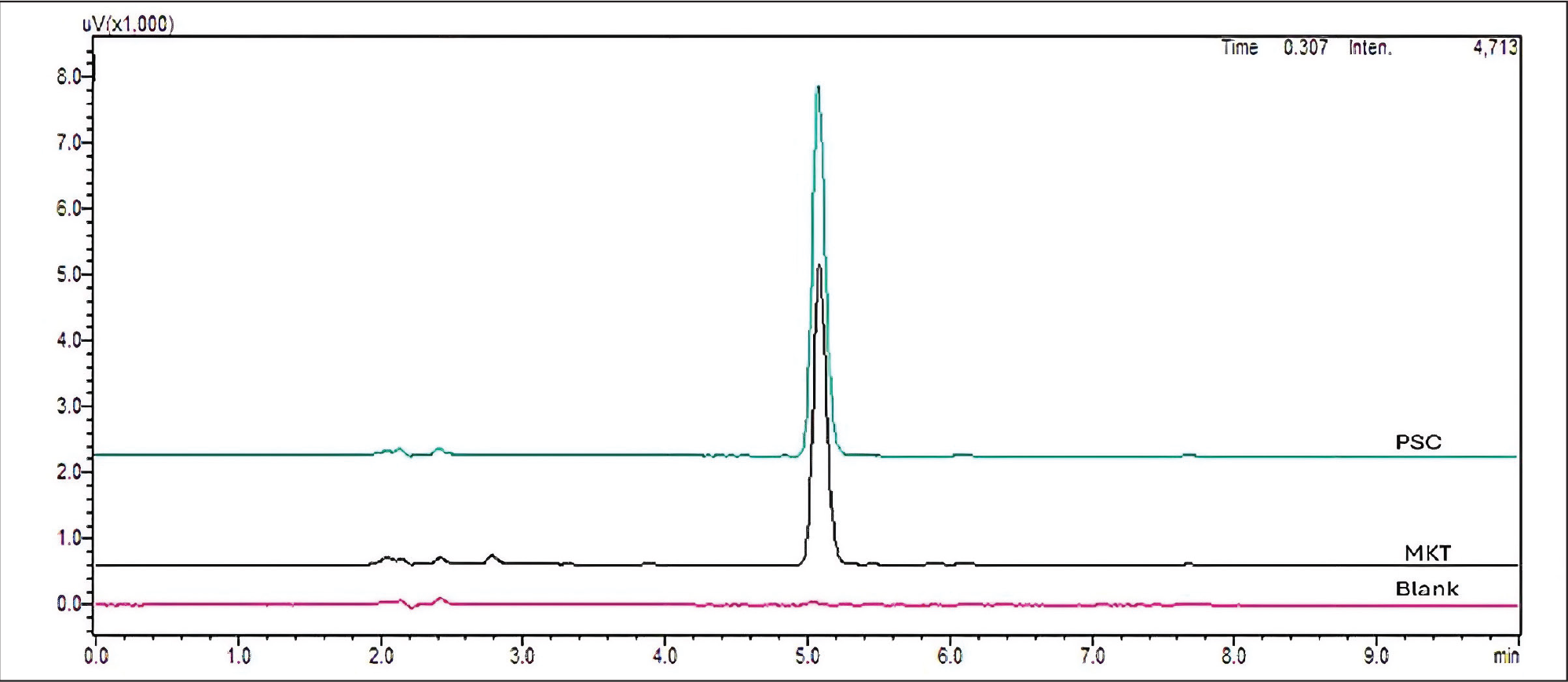
The specificity of the developed method was assessed by comparing the chromatograms of the blank, standard, and marketed formulation (1 µg/mL) to assess the specificity of each one. The criterion for acceptance was defined as no co-eluting peaks with the response peak. The HPLC system was loaded with 20 µL each of the blank, standard PSC solution, and marketed formulation. Figure 2 depicts the chromatogram findings. The optimised HPLC method demonstrated high specificity in the detection of PSC, as evidenced by the lack of co-eluting peaks at the Rt of 5.089 min for pure PSC and 5.085 min for the commercially available PSC formulation. Chromatographic comparison of PSC and marketed formulation demonstrating method specificity is provided in Table S1. This observation implies that the approach is capable of reliably quantifying PSC regardless of the presence of coexisting excipients.
Linearity and Construction of Calibration Curve
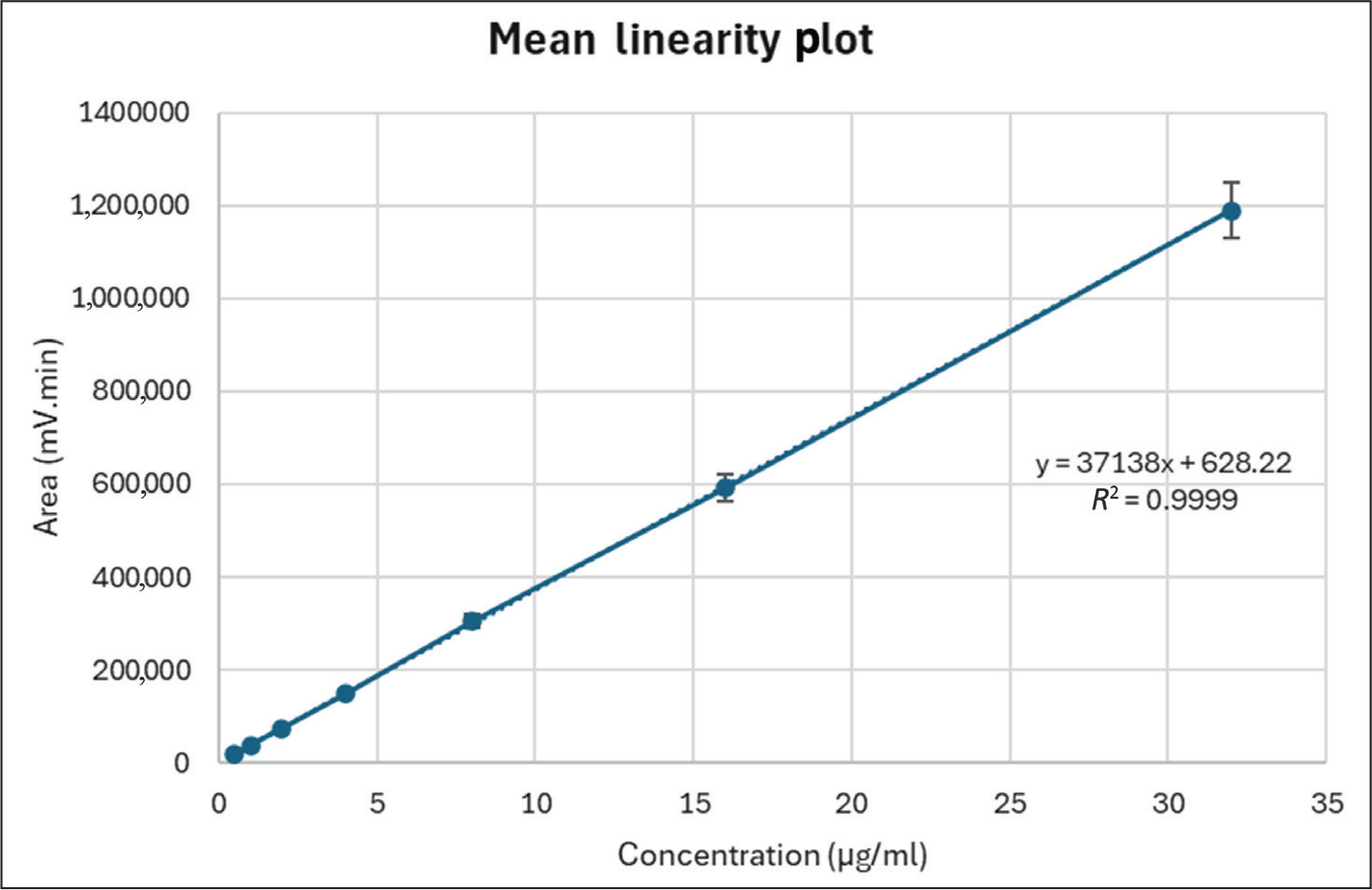
A series of standard PSC solutions at concentrations ranging from 0.5 to 32 µg/mL was prepared, and six injections of each concentration were injected into the HPLC system. A mean linear regression plot was constructed. The linear regression analysis generated the equation y = 37138× + 628.22 for PSC, with a coefficient of determination (R² = 0.9999), signifying an exceptional linear correlation between analyte concentration and peak area. The calibration plot exhibited a well-defined linear response over a wide concentration range. The correlation coefficient was quite significant. The ordinate intercept indicated no deviation from linearity. Figure 3 represents the mean linearity plot of the optimised HPLC method. Comprehensive linearity data across the full concentration range, including regression parameters and recoveries, are presented in Table S2.
Mean Linearity Plot of Posaconazole (PSC) by the Validated HPLC Method Over the Concentration Range of 0.5–32 µg/mL. The Calibration Curve Exhibited Excellent Linearity with the Regression Equation y = 37138× + 628.22 and Correlation Coefficient (R² = 0.9999).
Accuracy
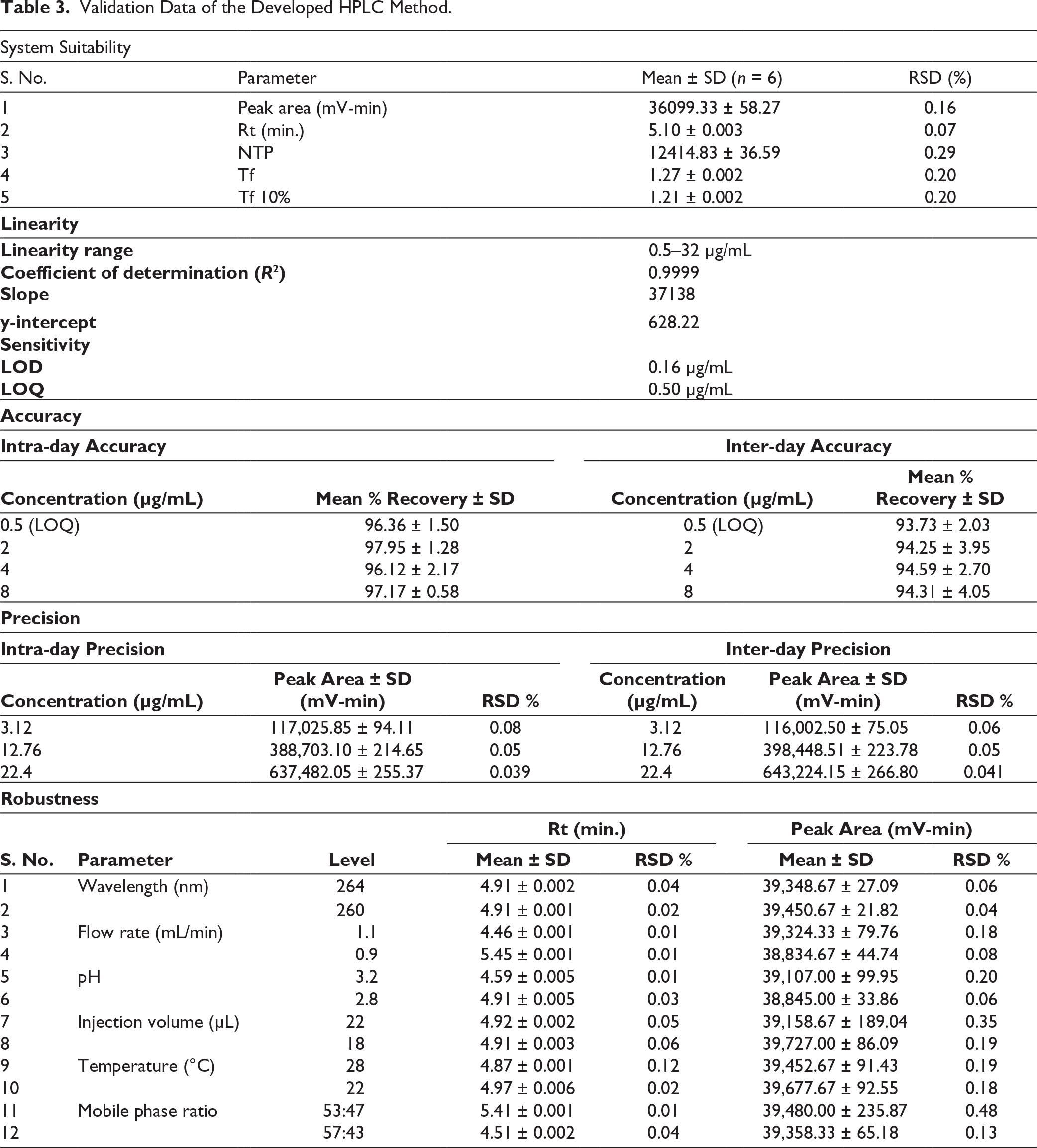
The accuracy of the refined HPLC analytical method for PSC quantification was systematically examined through a series of intra-day and inter-day validation experiments. In the intra-day assessment, PSC was injected six times across concentrations LOQ, 2, 4, and 8 µg/mL, while the inter-day analysis involved two injections at the same concentrations. In the intra-day investigation, the mean percentage recoveries were found to be 97.95% ± 1.28% for 2 µg/mL, 96.12% ± 2.17 % for 4 µg/mL, and 97.17% ± 0.58% for 8 µg/mL. Similarly, at these concentrations, the mean percentage recoveries for the inter-day study were 94.25% ± 3.95%, 94.59% ± 2.70%, and 94.31% ± 4.05%, respectively. Additionally, the method demonstrated excellent accuracy, with intra-day % recovery of 96.36% ± 1.50% and inter-day % recovery of 93.73% ± 2.03% at the LOQ concentration of 0.5 µg/mL. Representative chromatograms corresponding to each linearity concentration level are included in Figure S1. The findings indicated that the mean recovery percentages for both intra-day and inter-day analyses of PSC remained within the acceptable range of 85%–115%, affirming the accuracy and suitability of the optimised method for precise PSC quantification. The data shown in Table 3 demonstrates the accuracy of the developed method.
Validation Data of the Developed HPLC Method.
Sensitivity
The LOD and limit of quantitation (LOQ) for the optimised method were calculated using the residual standard deviation and the slope of the regression line. The LOD and LOQ were determined to be 0.16 µg/mL and 0.50 µg/mL, respectively. These values highlight the high sensitivity of the developed HPLC method, allowing for the precise detection of PSC even at minimal concentrations.
Precision
The results indicate that the RSD% of the peak area remained below 2% for all evaluated QC concentrations, underscoring the high reproducibility and consistency of the analytical method. Figure S2 represents the precision chromatogram overlays for LQC, MQC, and HQC. Such a low RSD% value signifies minimal variability across repeated measurements, thereby demonstrating the exceptional precision. This rigorous precision assessment substantiates the reliability of the optimised methodology, confirming its suitability for the accurate and consistent quantification of PSC. Table 3 demonstrates the precision of the optimised method.
Robustness
The results of robustness studies elucidated the stability and consistency of the developed method under varying operational conditions; the mean RSD% of peak area and Rt remained below 2%. Consequently, notwithstanding minor fluctuations in operational parameters such as mobile phase ratio (%), column oven temperature (°C), wavelength (nm), flow rate (mL/min), injection volume (μL), and pH of the mobile phase, the optimised methodology guarantees precise quantification of PSC. The actual Rt under optimised conditions was 5.089 min, and the peak area was consistently reproducible. Minor changes in Rt were observed with variations in flow rate, pH, temperature, and mobile phase ratio, ranging from 4.46 to 5.45 min. These shifts are expected because Rt is inherently sensitive to changes in chromatographic conditions; for example, increasing the flow rate shortened Rt, while decreasing the flow rate lengthened it. Despite these variations, the RSD% values for both Rt (0.01%–0.12%) and peak area (0.04%–0.48%) were very low, indicating that the method reliably quantifies PSC even under slightly altered conditions. Chromatographic profiles obtained under various robustness conditions are presented in Figure S5. Overall, these results confirm that the method is robust, precise, and suitable for routine analysis. The data in Table 3 demonstrate the robustness of the developed HPLC method.
System Suitability
The system suitability study was conducted to evaluate the performance and reliability of the analytical method in accurately quantifying PSC. A standard drug solution, prepared at a concentration of 1 µg/mL, was injected six consecutive times under identical chromatographic conditions. For each injection, the peak area and Rt were recorded and analysed. A low RSD% value (<2%), indicating minimal variation between successive injections, confirms the robustness of the system. Representative system-suitability chromatograms from six replicate injections of PSC are shown in Figure S4. The consistency of the measured Rts and peak areas across all trials further validates the capability of the method to produce precise and reliable results, ensuring its suitability for the accurate quantification of PSC. Table 3 demonstrates the system’s suitability for the method.
Forced Degradation Study
The forced degradation studies were performed under rigorous stress conditions, encompassing acidic, basic, neutral, oxidative, thermal, and photolytic environments. In most cases, forced degradation testing involves testing a few stress conditions to cover a range of possible degradation paths. Therefore, the objective was to investigate different stress scenarios. Furthermore, forced degradation conditions are intentionally severe, often exceeding the molecule’s typical operational limits. These conditions are inherently challenging to regulate and replicate precisely. 53 In this study, the forced degradation study was qualitative, centring on the detection of the presence of degradant peaks rather than optimising their quantification.
Chromatograms of PSC subjected to 1N of HCl acid degradation showed no additional peaks apart from the optimised analyte peak (Figure 4A). Assay results confirmed that samples exposed to 1N of HCl exhibited 100% recovery under all tested conditions. According to this, the PSC did not degrade and remained chemically stable in acidic environments. A complete recovery of PSC also confirmed that there was no degradation.
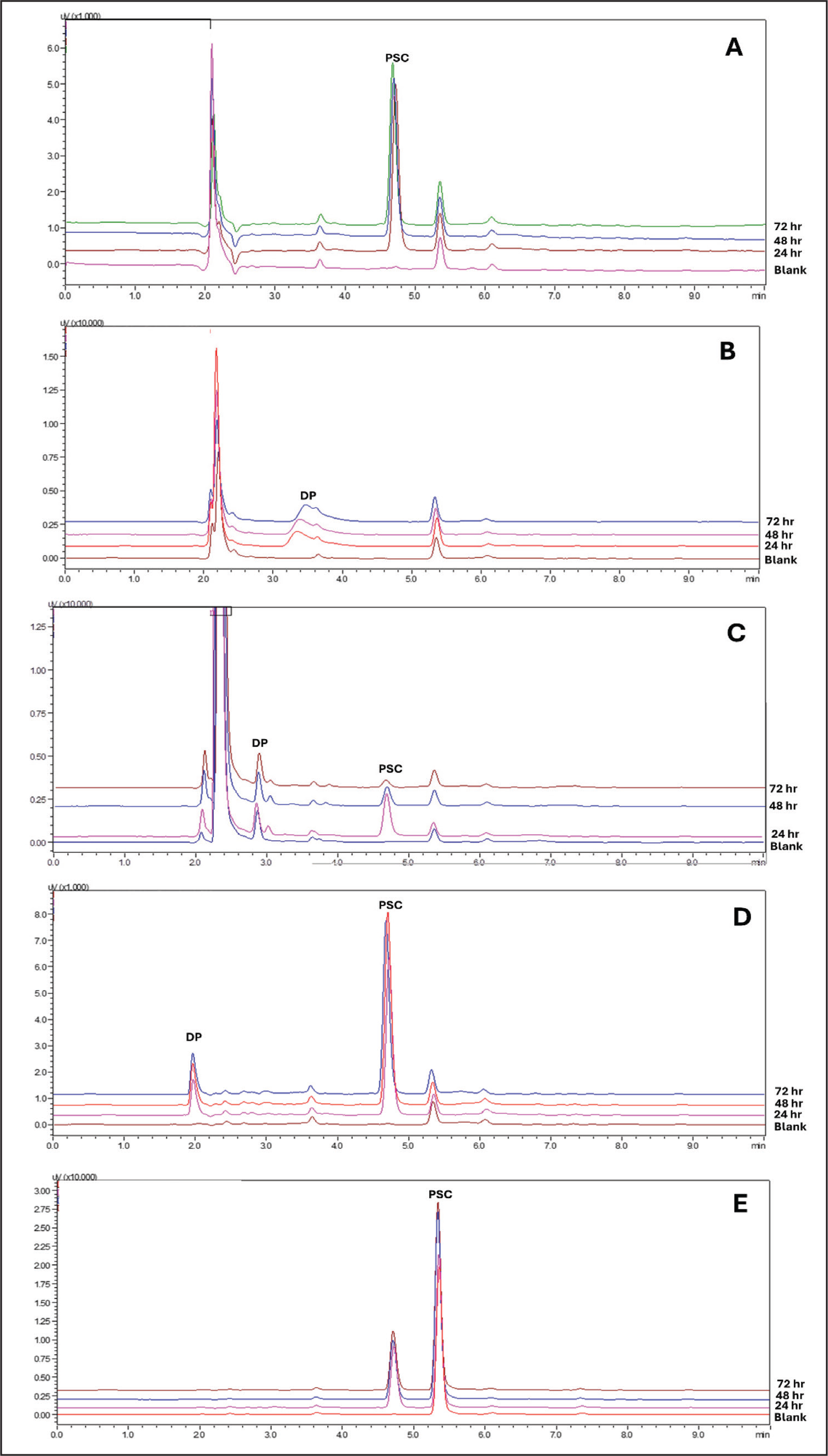
The absence of detectable drug peaks in the HPLC chromatogram following alkali hydrolysis of PSC suggests that degradation was extensive, potentially leading to multiple DPs, none detected under the applied chromatographic conditions. This raises the possibility that the DPs formed were either non-chromophoric at the detection wavelength or highly unstable, further degrading into smaller, undetectable fragments (Figure 4B).
Stability of PSC in both acidic and alkaline environments is essential to comprehending its chemical behaviour, especially concerning formulation and storage issues. However, literature reports on the stability of PSC in acid and base conditions are conflicting. Some studies have suggested that the drug is stable even under severe hydrolytic conditions, with no significant reduction in drug area or spectral purity changes, while others have demonstrated piperazine ring cleavage upon treatment with strong acids and bases. This result contradicts some earlier research that claimed PSC was susceptible to hydrolysis in both basic and acidic conditions, especially at the piperazine ring. Our findings imply that PSC is resistant to acidic hydrolysis, at least in the studied conditions. In contrast, PSC exhibited total degradation in alkaline environments. Given the elevated pH, the observed degradation is probably the result of molecular breakdown followed by nucleophilic attack on the piperazine ring or other labile functional groups. This is consistent with previous studies that showed instability of PSC in alkaline conditions. 27 The disparity in degradation profiles may be attributed to intrinsic molecular characteristics of PSC and extrinsic factors such as impurities, catalysts, residual solvents, and variations in synthetic routes. These factors can significantly impact the degradation pathways and stability of complex molecules like PSC. 54
PSC appeared more vulnerable to oxidative degradation than acidic or alkaline conditions. After 24 hr, approximately 11.46% of PSC degraded, resulting in the formation of a single DP eluting at Rt = 3.0 min, relative to the parent compound (Rt = 4.67 min) (Figure 4C). The parent compound showed a Rt of 5.089–5.101 min in system suitability and specificity runs, whereas it appeared at 4.677 min in the oxidative stress sample. This slight forward shift (0.4 min) is within acceptable chromatographic variation and can be attributed to differences in the sample composition under oxidative conditions. The presence of oxidative degradants or quenching residues can alter the polarity, pH, or ionic strength of the analyte, thereby affecting the analyte-stationary phase interaction and resulting in minor Rt changes. Although PDA spectral or matrix-effect evaluations were not performed, the consistent chromatographic profile and absence of additional interfering peaks suggest that the observed change arises from altered chromatographic conditions rather than the formation of a new entity. Hence, the 4.677 min peak is reasonably assigned to the parent compound under oxidative stress conditions.
The oxidative degradation progressed over time, reaching 62.37% after 48 hr and 87.30% after 72 hr, indicating a continuous oxidative breakdown of PSC. Our results are consistent with the research by Garcia et al., who found an oxidative DP at Rt = 2.90 min. 55 Sophisticated analytical methods like LC-MS or NMR could provide further insights into the structural information of the oxidative DPs and their impact on the therapeutic activity of PSC.
Thermal stress studies conducted at 80°C demonstrated that approximately 0.8% of PSC underwent degradation after 24 hr, yielding a distinct DP with a Rt of 1.9 min, which became more prominent after 72 hr (Figure 4D). These findings highlight the minimal impact of heat on drug integrity. Notably, our results are in close agreement with those reported by Yang et al., who observed a comparable degradation of 0.4% after 24 hr at 85°C, further confirming the thermal sensitivity of PSC. 27
Exposure to photodegradation stress had no discernible impact on the chromatographic behaviour of PSC, as no additional peaks were observed in Figure 4E. Additionally, the assay demonstrated 100% recovery of PSC after 72 hr, indicating good photostability of PSC.
Due to resource and scope limitations, advanced structural elucidation techniques such as liquid chromatography-Tandem Mass Spectrometry (LC-MS) or Nuclear Magnetic Resonance (NMR) Spectroscopy were not performed for DP identification. Similarly, Photodiode Array spectral purity assessment, although valuable, was not included in this work. Instead, chromatographic separation of the parent drug and degradation peaks with acceptable resolution and peak symmetry was considered adequate to establish specificity. The reported degradation profiles provide sufficient evidence that the developed method is stability-indicating for the intended analytical application. Complete chromatographic profiles of forced degradation studies are given in Tables S4–S7.
Assessment of the Eco-friendliness of the Developed Analytical Method
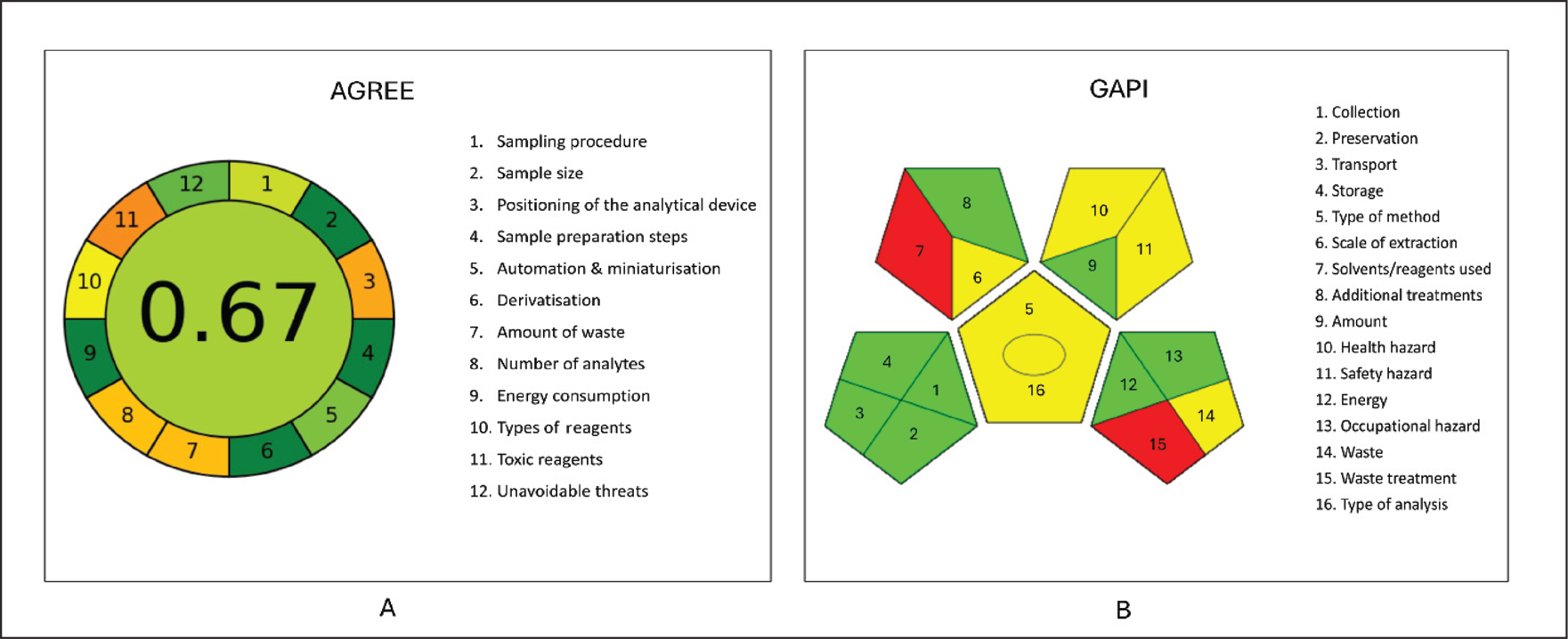
The AGREE tool represents a clock layout with a total score shown in the centre. Scores nearing 1 are displayed in a dark green colour. Each section illustrates how the process adheres to SIGNIFICANCE. Green indicates the least environmental harm, and red indicates the most severe impact. 47 There are 15 pentagrams in the GAPI, and each one represents a distinct aspect of the analytical process, including sampling, the chemicals and solvents used, sample processing, instruments, and waste produced. 48
AGREE and GAPI were the two assessment tools used to evaluate the environmental sustainability of the optimised method. The developed HPLC method for PSC had a higher overall AGREE score (0.67), indicating that it is more environmentally friendly (Figure 5A). The proposed HPLC procedure in the GAPI tool displays eight green, five yellow, and two red, indicating the level of environmental sustainability attained (Figure 5B).
Greenness Assessment of the Developed RP-HPLC Method for Posaconazole Using (A) the AGREE Metric (Overall Score = 0.67) and (B) the GAPI. The Results Confirm That the Method Is Environmentally Acceptable, With High Compliance With Green Analytical Chemistry Principles.
The complete AGREE and GAPI green assessment, including all scoring components and justification, is provided in Tables S8 and S9.
Method Applicability
Determination of Total Drug Content in SNEDDS of PSC
One of the fundamental evaluation parameters in characterising any new formulation is determining the total drug content. A simple methodology is used, with the drug being extracted from the developed formulation and then analysed using an appropriate analytical technique. This study used a validated HPLC method to analyse a precise quantity of PSC in SNEDDS. The results showed a peak area of 231192 from which the total drug content was determined to be 93.21% ± 3.21%.
In Vitro Drug Release Study of SNEDDS of PSC
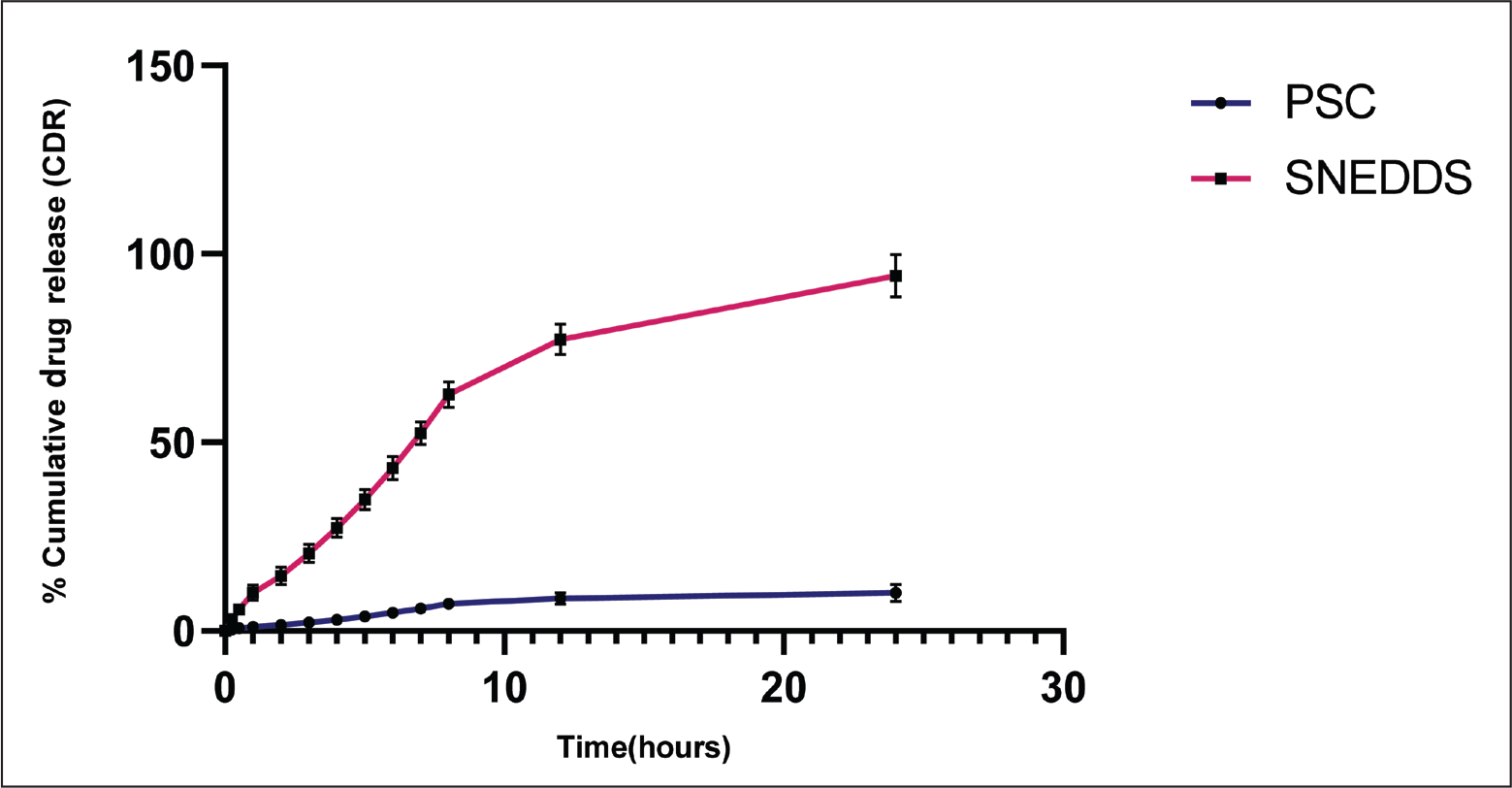
SNEDDS of PSC exhibited a minimal drug release in SGF, protecting the formulation through the stomach, while a marked increase in release was observed in SIF, achieving a percentage cumulative drug release (% CDR) of 94.19 ± 5.62 within 24 hr (Figure 6). In contrast, the pure drug exhibited only 10.04% ± 2.31% release within 24 hr. PSC exhibited minimal release throughout the study period, with only a small fraction of the drug solubilised even at extended time points. This poor dissolution behaviour can be attributed to the crystalline nature and inherent hydrophobicity of PSC, which restricts its wettability and aqueous solubility. In contrast, the SNEDDS formulation demonstrated a rapid and significantly higher release, with a steep initial increase followed by a sustained release phase. The enhanced dissolution profile is a consequence of the spontaneous formation of nanoemulsion droplets upon dispersion in the medium, which provides a large surface area for drug release and maintains PSC in a solubilised state. The presence of surfactant and co-surfactant further reduced interfacial tension and improved wettability, facilitating faster drug release compared to the pure drug. These findings clearly establish that the SNEDDS system effectively overcomes the solubility limitations of PSC, offering a more predictable release profile that supports its potential for improved oral bioavailability.
In Vitro Cumulative Drug Release Profile of Bulk PSC and PSC-loaded SNEDDS Over Time (n = 3, Mean ± SD). The SNEDDS Formulation Exhibited Significantly Higher and Faster Release Compared to Pure PSC, Demonstrating Enhanced Solubilisation and Dissolution Behaviour.
The unpaired t-test analysis demonstrated that the SNEDDS formulation achieved a significantly higher drug release compared to PSC. While PSC showed a mean release of only 3.8%, the SNEDDS system enhanced the release to 34.3%, indicating a nearly 10-fold improvement. The large difference in mean values, supported by a narrow confidence interval, confirms that this enhancement is statistically reliable. Moreover, the highly significant p value (<.01) further emphasises the superiority of SNEDDS in improving solubility and dissolution.
The applicability of the method is not confined to SNEDDS alone; rather, due to its demonstrated specificity, sensitivity, precision, and robustness, it can be effectively extended to the analysis of PSC in a wide range of formulations, including other lipid-based delivery systems, solid dosage forms, and conventional preparations. This versatility makes the method highly suitable not only for routine QC testing but also for comparative formulation studies and various stages of formulation development.
Conclusion
The developed RP-HPLC method for PSC quantification exhibited excellent analytical performance, characterised by high simplicity, selectivity, accuracy, precision, robustness, sensitivity, and stability. This approach was thus used to assay PSC in pharmaceutical dosage forms. The approach is favourable due to its high sensitivity, use of a sustained mobile phase, simple extraction processes, short retention durations, and lack of an internal standard. These characteristics constitute a superior alternative for the routine analysis of PSC. The developed HPLC method involves the substitution of commonly employed hazardous solvents and chemicals with harmless, less toxic alternatives, offering environmentally compatible materials to the more perilous compounds and approaches in drug analysis. The established RP-HPLC method was employed for the quantification of PSC in the SNEDDS of PSC and for investigating the in vitro drug release profile of the formulation.
Supplemental Material
Supplemental material for this article available online.
Footnotes
Acknowledgements
The authors are grateful to the Manipal Academy of Higher Education, India, for providing the funding and facilities for this study through its Intra-Mural Fund.
Authors’ Contribution
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be authors as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability
All the data is available with the authors and shall be provided upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethics Approval
This study does not involve experiments on animals or human subjects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Manipal Academy of Higher Education, India, through its Intra-Mural Fund.
Informed Consent
Not applicable.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
Use of Artificial Intelligence-assisted Technology
The authors declare that they have not used artificial intelligence (AI) tools for writing and editing of the manuscript, and no images were manipulated using AI.
