Abstract
Aims and Objectives:
Cytotoxicity assays are one of the methods used to assess the biocompatibility of dental materials. The objective of this study was to determine if surface area/volume ratios designated by the International Organization for Standardization (ISO) for biological evaluation of medical devices affects cytotoxicity and calcium release in an endodontic pulp-capping materials.
Material and Methods:
Human periodontal ligament fibroblasts (PDLFb) were treated with extractables derived from the calcium silicate pulp-capping material TheraCal LC at pellet surface area/culture medium volume ratios of 0.2, 0.5, and 1.0 cm2/mL. A colorimetric cytotoxicity assay (MTT) and phase-contrast microscopy was used to assess biocompatibility against the fibroblasts. The concentration of calcium leached into culture medium from TheraCal LC at pellet surface area/culture medium volume ratios of 0.5, 1.0, 2.0, 3.0, 4.0, 5.0, and 6.0 cm2/mL after 24 hours was determined by colorimetric assay.
Results:
Both MTT assays and microscopy demonstrate that at a pellet surface area/culture medium volume ratio of 1.0 cm2/mL TheraCal LC extractable challenge to fibroblasts results in a significant decrease in cell viability (P < 0.001) compared to control. TheraCal LC calcium release into culture medium varied with the surface area/volume ratio, with concentrations ranging from 2 to 8 times the calcium concentration in culture medium.
Conclusion:
Pellet surface area/culture medium volume ratios impacted parameters of TheraCal LC used in in vitro studies to determine biocompatibility with oral tissues. The ISO guidelines should be followed for all in vitro cytotoxicity studies of dental materials.
Introduction
In vitro cytotoxicity assays are one of the methods used to assess the biocompatibility of dental materials, 1 including endodontic pulp-capping materials. 2 The International Organization for Standardization (ISO) is a worldwide federation of national standards bodies. 3 The document ISO 10993–5 Biological Evaluation of Medical Devices, Part 5: “Tests For in vitro Cytotoxicity” describes three test methods to assess in vitro cytotoxicity. The document details the (a) extract, (b) direct contact, and (c) indirect contact methods for using cultured cells to assay for biocompatibility of components of medical and dental devices.
In the extract method test, materials in solid form are incubated in cell culture medium under standard conditions, typically 24 hours at 37ºC, in order to obtain “extractables,” soluble components of the solid material that leach into the medium. Extractables in liquid form are subsequently used to treat cultured cells, again typically for 24 hours at 37ºC. Cytotoxicity assays are often accomplished using the mouse fibroblast L929 cell line4,5 because of the ease in maintaining viable cultures; however technically any cell type can be used. With the availability of oral-derived fibroblasts, it can be argued that it is more clinically relevant to use such cells in lieu of the mouse fibroblast line. The colorimetric MTT assay is widely used to evaluate biocompatibility by monitoring mitochondrial dehydrogenase activity as this was shown to be the most sensitive in detecting cytotoxic events compared to LDH leakage or protein assay. 6
For medical and dental solid materials, ISO guidelines recommend a surface area to volume ratio of 0.5–6.0 cm2/mL to generate extractables for testing on cultured cells.7,8 The guidelines also delineate 70 per cent as minimum in vitro cell viability compared to control for a dental material to be considered non-cytotoxic and biocompatible with in vivo oral cells and tissues.
TheraCal LC is a light-cured, resin-modified calcium silicate-filled liner designed for use in direct and indirect pulp-capping procedures. 9 In a comparison, the ppm calcium released into water after three hours and 1, 3, 7, 14, and 28 days was significantly higher with TheraCal compared to Dycal. 9 The cytotoxic properties of TheraCal2,10,11 have been reported. However, a review of the methods used to determine calcium release and biocompatibility in published reports reveal inconsistent application of the ISO guidelines for determining biological parameters of these two dental materials. For example, surface area/volume ratios may not be stated, and studies generally do not test for cytotoxicity using a range of surface area/volume ratios.
The objective of this in vitro study is to determine if and how surface area to volume ratios as set forth in the ISO standards for biological evaluation of medical devices affect determinations of cytotoxicity and calcium release in an endodontic pulp-capping material. It is hypothesized that cytotoxicity and calcium release of TheraCal will vary depending on the surface area/volume ratio analyzed.
Methods
This in vitro study did not require human subject volunteers, data, or tissues; therefore no institutional review board approval was necessary. Additionally, the study did not employ live animals.
Preparation of Pulp-capping Material
TheraCal LC, a calcium silicate-based material (Bisco, Schaumburg, IL), was prepared according to manufacturer’s instructions. A customized mold with a diameter of 6 mm and a height of 1 mm was designed to create pellets of defined and consistent size. A height of 1 mm was used as it has been reported that TheraCal has a cure depth of 1.7 mm. 9 TheraCal LC was dispensed directly from the syringe as a single paste into the custom molds and light-cured from the visible surface with an LED unit for 20 seconds. During the preparation of the TheraCal pellets care was taken to create a flat surface on the free side of the mold following introduction of the materials into the mold itself. Pellets were used within one day of preparation. Using the formula 2πrh + 2πr2, the surface area of individual TheraCal LC pellets was calculated at 0.75 cm2.
Cell Culture
Human PDLFb were purchased at passage one (ScienCell Research Laboratories, Carlsbad, CA). PDLFb were cultured in fibroblast medium according to manufacturer’s directions under standard conditions of 37ºC and 5 per cent CO2. Components of the medium include “fibroblast growth medium” which contains essential and non-essential amino acids, vitamins, organic and inorganic compounds, hormones, growth factors, and trace minerals. Additional components are 2 per cent fetal bovine serum and 1 per cent penicillin-streptomycin solution. The manufacturer stated that the calcium concentration of the medium, as calcium chloride, is 2 mM. Confluent cells in T-75 flasks were passaged by standard methods of trypsinization, plated into wells of a 24-well plate for assays, and cultured to confluence for assays. Cells were used within two to three passages.
MTT Cytotoxicity Assay
In separate sterile tubes, one cured and UV-sterilized TheraCal pellet was placed in one of three volumes of culture medium (3.75, 1.5, or 0.75 mL) to produce pellet surface area/culture medium volume ratios of 0.2, 0.5, and 1.0 cm2/mL respectively. Sufficient replicates of each were prepared to produce a total of 6 mL of each SA/Vol ratio. Pellets were incubated in culture medium for 24 hours to produce extractables. Culture medium containing extractables was filtered through 0.2 μm syringe filters to remove any particulate matter before use. One mL of culture medium containing extractables was then added to each of the 6 wells of a 24-well plate containing confluent PDLFb. As negative control PDLFb were cultured in medium only and as positive control cells were cultured in medium containing 0.01 per cent chlorhexidine, a concentration that is cytotoxic to cells in culture.12,13
Extractables were cultured with the PDLFb for 24 hours at 37ºC and 5 per cent CO2. Prior to initiation of the MTT cytotoxicity assay, 14 cell adhesion and morphology were documented using phase-contrast light microscopy. For the cytotoxicity assessment 100 μL MTT solution (3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyl tetrazolium bromide, Cell Growth Determination Kit MTT Based; Sigma-Aldrich, St Louis, MO) was added to each well. As per kit instructions, MTT is to be added directly to the culture medium at a 1:10 ratio, thus 100 μL was added to 1 mL of culture medium per well. Cells were incubated with MTT for four hours. The resulting formazan crystals were dissolved by removing the culture medium, adding 1 mL “cell dissolution solvent” as defined by the assay kit, to each well, and stirring the plate for 10 minutes to dissolve the crystals. The absorbance at 570 nm (A570 nm) was determined using a SpectraMax M3 microplate reader (Molecular Devices, Sunnyvale, CA, USA). The assay was repeated three times with similar results.
The mean and standard error of the mean for each treatment and control was calculated from the six replicates. The mean for the negative control was arbitrarily set to 100 per cent and absorbance values were used to calculate percentage viability against the absorbance values of the negative control. Statistical analysis was performed using GraphPad software (Prism, Chicago, IL) with one-way analysis of variance (ANOVA) and Tukey multiple comparison post-test with significance of P < 0.05.
Calcium Assay
In separate sterile tubes, one cured and UV-sterilized TheraCal pellet was placed in culture medium (1.5, 0.75, 0.38, 0.25, 0.19, 0.15, or 0.13 mL) to create pellet surface area to culture medium volume ratios (SA/Vol) of 0.5, 1, 2, 3, 4, 5, or 6 cm2/mL respectively. After 24 hours, incubation at 37ºC samples of the culture medium were assayed for calcium concentration using a commercial colorimetric system (ScienCell Calcium Assay). A standard curve ranging from 0.3 to 20 mM was prepared by serial dilution of the calcium standard provided with the kit. 10 μL of calcium standards or TheraCal LC extractables were aliquoted, in duplicate, into wells of a 48-well plate. According to the kit protocol, 200 μL each of AMP buffer and color reagent (mixed as working reagent) were added to each well and incubated at room temperature for 15 minutes. The absorbance at 570 nm was determined by using a SpectraMax M3 (Molecular Devices, Sunnyvale, CA, USA) plate reader. The standard curve of 0.313 to 20 mM was used to calculate the calcium concentration in the TheraCal LC samples. All samples were within the assay range. The assay was repeated twice with similar results. Data are reported as means after subtraction of the calcium concentration in the control samples. Statistical analysis was performed using GraphPad software (Prism, Chicago, IL) with ANOVA and Tukey multiple comparison post-test with significance of P < 0.05.
Results
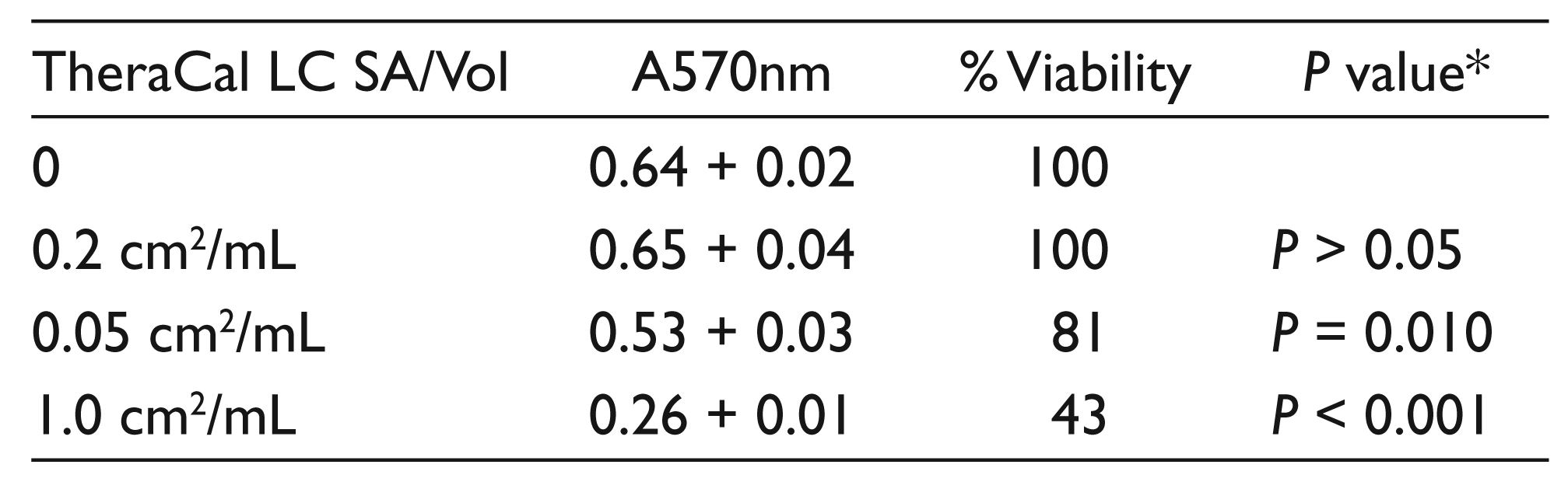
Human PDLFb were used in this study because they are an oral cavity-derived cell type. PDLFb were challenged with three pellet surface area/culture medium volume (SA/Vol) ratios of TheraCal LC extractables for 24 hours. Compared to untreated control, extractables derived from pellets at 0.2 cm2/mL showed high biocompatibility, at 100 per cent of control (Table 1). Note, however, that this SA/Vol is out of the recommended ISO range of 0.5–6.0 cm2/mL. At 0.5 cm2/mL, the low end of the ISO range, cell viability was reduced to 81 per cent, statistically lower than control (P = 0.010) but still above the 70 per cent cutoff for biocompatibility recommended by ISO guidelines. In contrast, at 1.0 cm2/mL cell viability was reduced to 43 per cent, both statistically lower compared to control (P < 0.001) and below the ISO percentage for biocompatibility. MTT assays at SA/Vol between 2.0 and 6.0 cm2/mL resulted in essentially total cell detachment and death (results not shown).
Phase-contrast microscopy, additionally, shows evidence that SA/Vol is critical in determining cytotoxicity (Figure 1). Comparing to control (Figure 1A), in increasing SA/Vol TheraCal extractables from 0.2 cm2/mL (Figure 1D) to 0.5 cm2/mL (Figure 1E) to 1 cm2/mL (Figure 1F), increasingly more detachment and loss of PDLFb can be observed. Similarly, to the MTT assay results, at SA/Vol between 1.0 and 6.0 cm2/mL essentially total cell detachment and death of PDLFb was observed (results not shown).
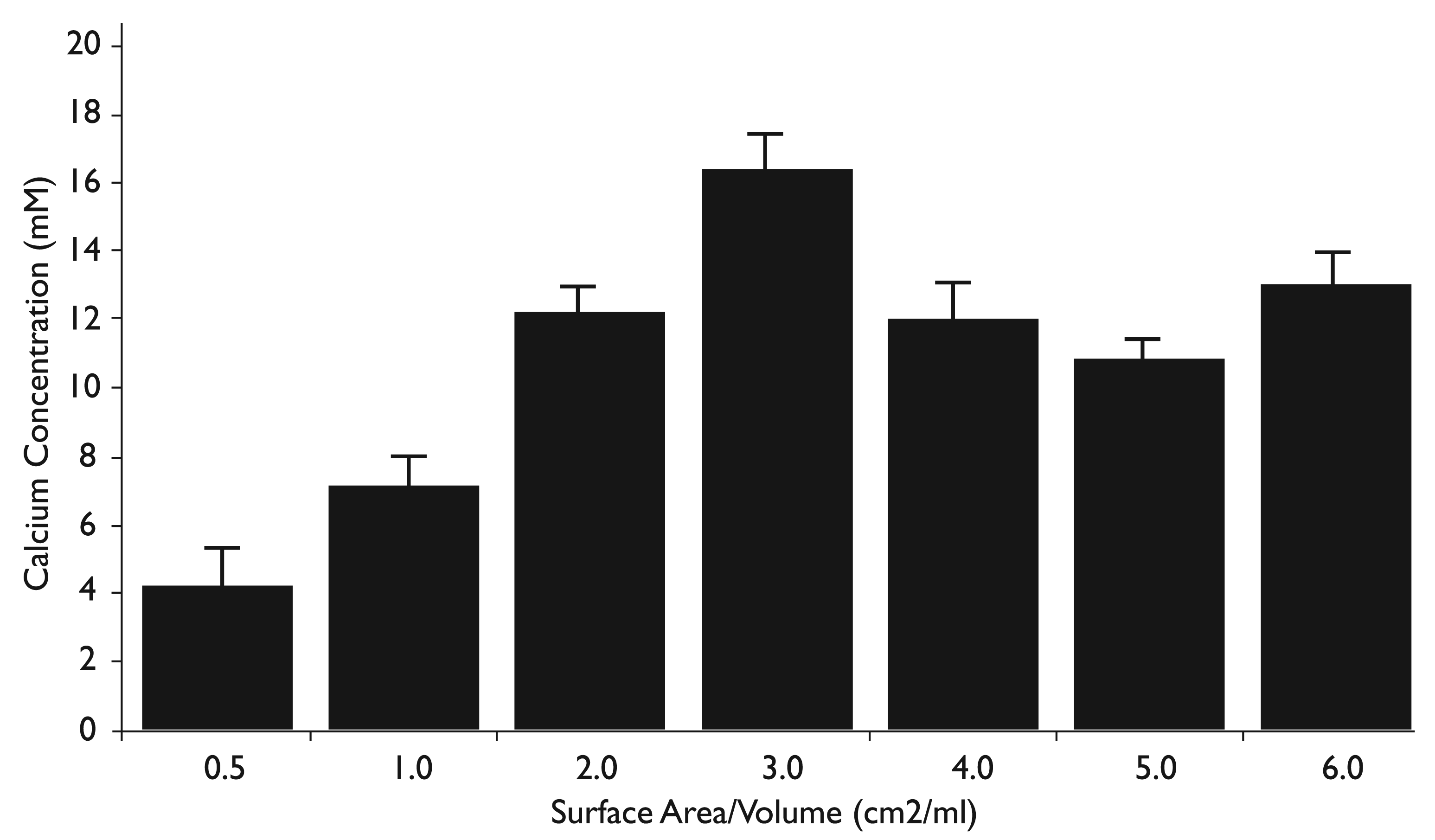
A reported characteristic of TheraCal is significant calcium release. 9 Calcium release by TheraCal LC pellets as a function of SA/Vol is shown in Figure 2. Using a colorimetric assay optimized for determining calcium in biological samples, at 0.5 cm2/mL, the lowest SA/Vol recommended by the ISO, calcium released by TheraCal LC measured 4.2 mM. This is approximately twice the calcium chloride concentration in the fibroblast medium used in the study, stated as 2 mM by the manufacturer. Calcium release varied with SA/Vol, with a concentration as high as 16.4 mM, eight times the normal calcium concentration in culture medium. It is interesting to note that the calcium released as a function of TheraCal SA/Vol generated a biphasic response, with the primary peak concentration at 3.0 cm2/mL and a secondary peak at 6.0 cm2/mL. All SA/Vol ratios generated a significantly higher concentration of calcium release compared to 0.5 cm2/mL (P < 0.01), related to acceptable cytotoxicity levels. All SA/Vol ratios additionally resulted in a significantly higher concentration of calcium release compared to 1.0 cm2/mL (P < 0.01), related to unacceptable cytotoxicity levels. Similar results were observed in an analysis of calcium release by TheraCal LC after 48 and 96 hours (results not shown).
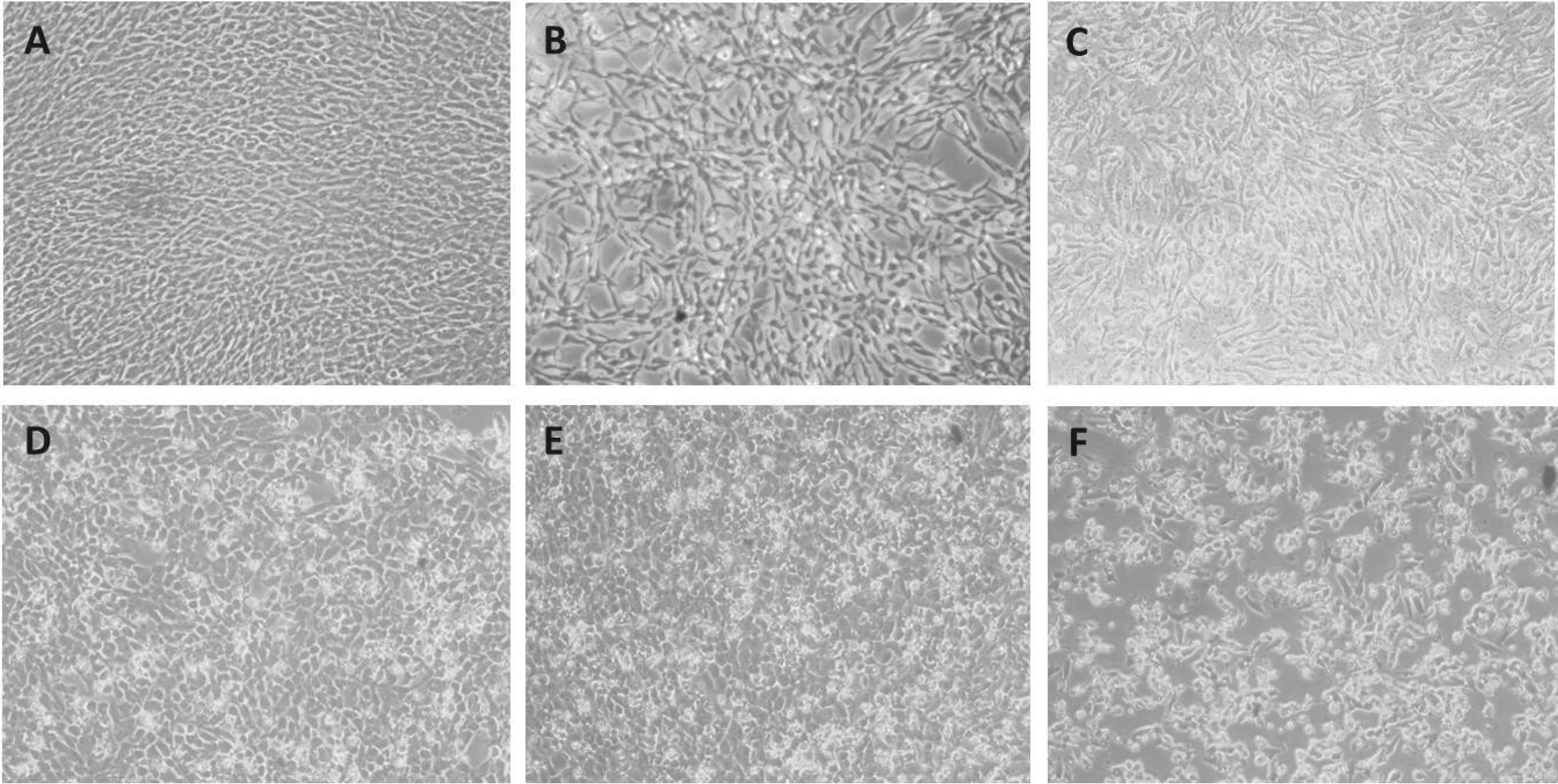
Relative Viability of Human Periodontal Ligament Fibroblasts Treated for 24 Hours with TheraCal LC Extractables
Discussion
MTT, WST, live-dead assays, and micronucleus assays are just a few of the mechanisms used to determine the in vitro cytotoxicity and biocompatibility of medical and dental devices and materials. 15 These assays are certainly appropriate for the end point in mind, have been used extensively by multiple researchers, and are available as commercial standardized kits. A standard cell line for cytotoxicity assays has been the mouse fibroblast L929; 16 however with the availability of oral-derived cells many other types are now used. Thus, it would be possible to standardize biocompatibility studies since cytotoxicity kits and many types of fibroblasts are commercially available.
A standardized protocol is also available through the ISO 10993-5 document, and a specified range of SA/Vol for testing is suggested (0.5–6.0 cm2/mL).7,8 A literature search of studies analyzing the in vitro cytotoxicity of endodontic materials shows that these guidelines are generally not followed. Specifically, for studies on TheraCal, often the protocol does not provide enough information to determine if the materials are being tested within the recommended SA/Vol ratios.11,13 Additionally, it does not appear that materials are tested using a range of ratios. In published studies on TheraCal which do provide sufficient information to determine SA/Vol ratios, often tests are being performed at ratios under the recommended range. 17 This makes direct comparisons between different studies difficult. For example, in a study comparing calcium release between TheraCal and Dycal, it appears from the article that materials were being tested at 0.05 cm2/mL. Results demonstrated that after 24 hours, 37.4 ppm (approximately 1 mM) calcium was “leaked into soaking water” by the TheraCal sample. 9 In the present study, TheraCal at 0.5 cm2/mL leaked 4.2 mM (approximately 160 ppm) into medium after 24 hours. More studies need to be done to determine if there is a linear relationship between surface area/volume and calcium release in pulp-capping materials.
In this study, significant TheraCal LC cytotoxicity was observed at 1.0 cm2/mL, and calcium release at this SA/Vol ratio was 7.1 mM. Normal extracellular calcium concentration is approximately 2 mM (similar to the culture medium used in this study). 18 Calcium above 10 mM showed cytotoxicity in mouse primary fibroblasts. 19 Thus, this current work suggests that if studies analyzing the biocompatibility of TheraCal LC employ test materials at SA/Vol ratios less than 0.5 cm2/mL cytotoxicity may not be significant because calcium release may not be deleterious to cells. Maeno et al. 19 showed that calcium in the range of 2 to 4 mM stimulated mouse primary osteoblasts; additional experimental work may show that TheraCal LC is beneficial to oral cells such as dental pulp cells at the appropriate SA/Vol. This, of course, would be difficult to translate to the clinical situation, and the influence of different cell types and different test materials used could also influence the outcome data.
In summary, this study shows that SA/Vol ratios should be considered when determining parameters of endodontic pulp-capping materials, such as cytotoxicity or biocompatibility, and calcium release. When assaying solid-form endodontic pulp-capping materials, the surface area/ volume ratio can have a significant effect on end-point assay results. Materials should be assayed for biocompatibility and other parameters using a range of surface area/volume ratios.
Footnotes
Acknowledgements
Supported by internal funding through the Student Research Program. The authors deny any conflicts of interest related to this study.
Declaration of Conflicting Interest
None.
Funding
None.
