Abstract

In the October on-line edition of
At baseline, trial participants showed prednisone-equivalent steroid dosing of 0.60 ± 0.12 mg/kg/day (mean ± SD), 1 where the recommended dose for prednisone is 0.75 mg/kg/day (American Academy of Neurology practice guidelines 3 ). These suboptimal doses of corticosteroids were then supplemented with supra-therapeutic and variable dosing for the initial 12-week treatment period (Figure 1; drawn from published Supplemental Data 1 ).
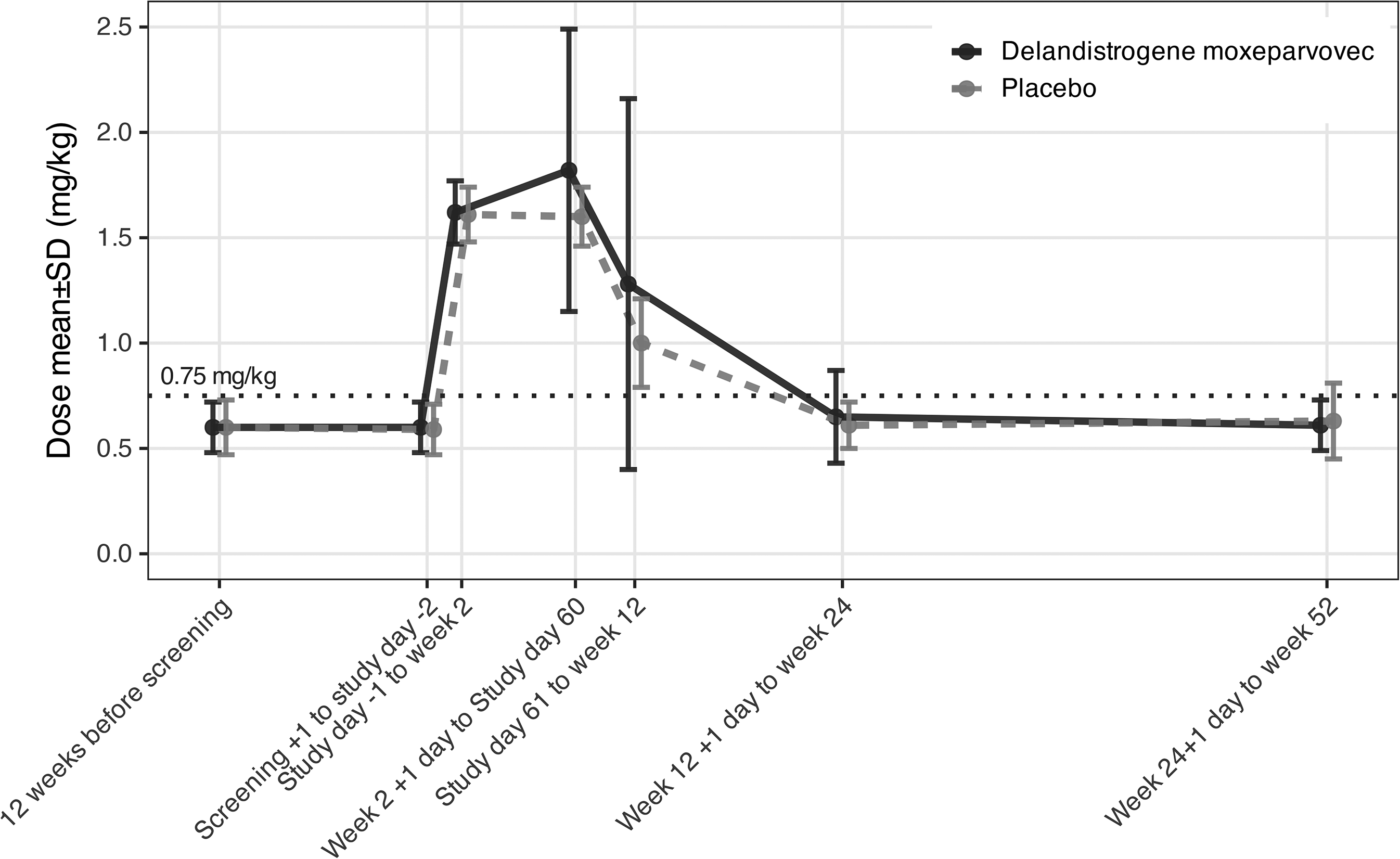
Corticosteroid dosing in prednisone equivalent dosage (mean ± 1 SD) in the EMBARK Phase 3 double blind clinical trial. Data as obtained from Supplemental Data. 1 The recommended dose of prednisone is 0.75 mg/kg/day in DMD (green dashed line). The EMBARK trial showed supra-therapeutic and highly variable dosing of corticosteroids that differed between placebo and drug treatment arms, and over time.
Corticosteroids in DMD are well documented to be effective, leading to marked improvements in motor outcomes, but the efficacy is also dose dependent.4–6 The markedly increased dosing of corticosteroids during the conduct of the EMBARK study would be expected to lead to improvement in motor outcomes, and thus reduce the statistical power of the trial to detect any ‘add on’ effect of delandistrogene moxeparvovec beyond the benefit of corticosteroids.
To illustrate the potential impact of increased corticosteroid dosing during the EMBARK trial, 1 we show the NorthStar Ambulatory Assessment (NSAA; primary outcome) of the EMBARK trial and the NSAA data from the VISION-DMD double-blind trial in DMD participants in a similar age range, including placebo and prednisone (0.75 mg/kg/day) arms (Figure 2). 4
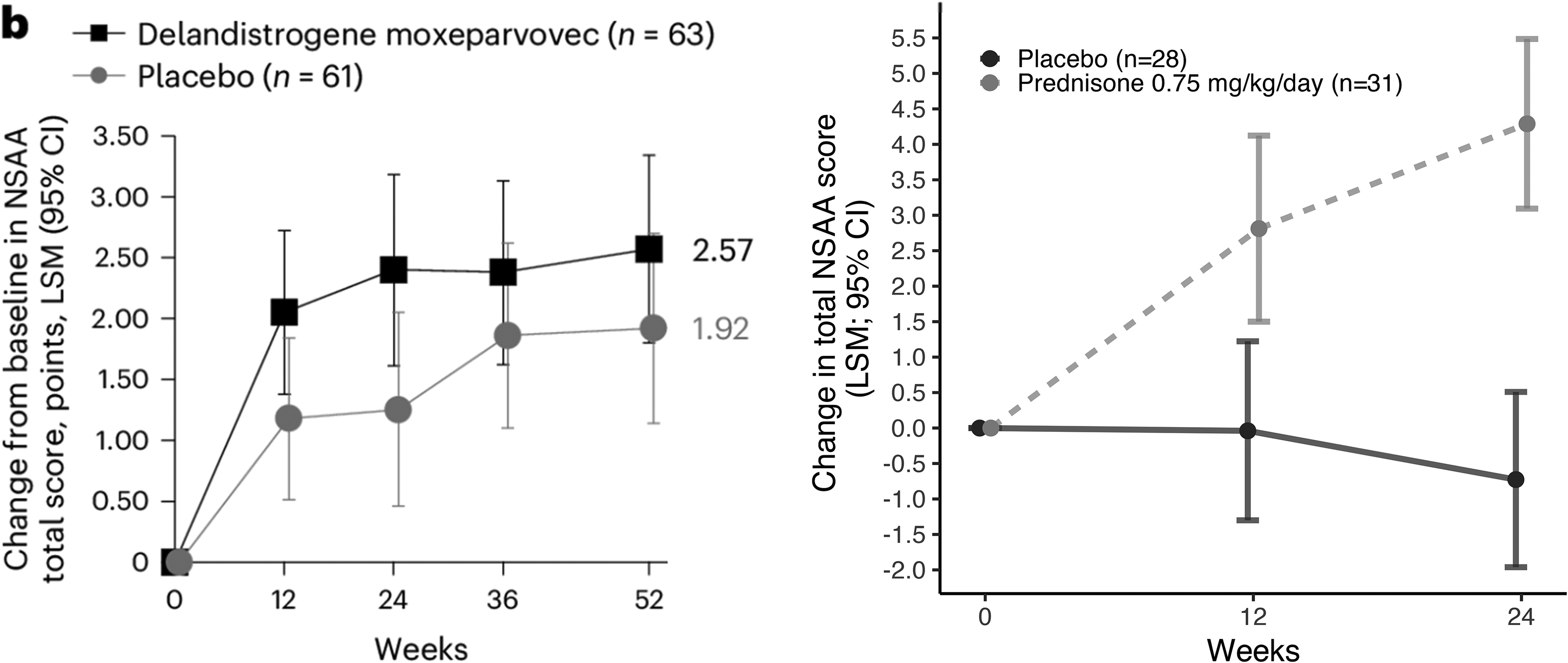
North Star Ambulatory Assessment (NSAA) in the EMBARK trial and VISION-DMD trial in Duchenne muscular dystrophy. Left panel: The EMBARK trial was a placebo-controlled trial of delandistrogene moxeparvovec as an add-on to corticosteroid therapy (LSM; 95% CI), published under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. 1 Right panel: The VISION DMD trial was a placebo- and prednisone-controlled trial of vamorolone in a similar age range following enrollment of steroid naïve participants, thus isolating steroid effect (LSM; 95% CI) 4 .
The EMBARK trial enrolled participants that were pre-treated with corticosteroids (0.60 ± 0.12 mg/kg/day (mean ± SD) at baseline; approximately 1 year since corticosteroid treatment started), whereas the VISION DMD trial enrolled steroid naïve participants. In the initial 12-week treatment period in the EMBARK trial, the steroid dosing was increased from 0.60 ± 0.12 mg/kg/day (mean ± SD) to 1.6 mg/kg/day (Figure 1). In the VISION DMD study, the magnitude of change in NSAA score was an improvement in over 4 points in the NSAA over a 24-week treatment period (from steroid dosing of 0 mg/kg/day at baseline to 0.75 mg/kg/day) (Figure 2; right panel). In the EMBARK study, the magnitude of change in NSAA score was an improvement of about 1 to 2 points, where steroid dosing went from 0.6 mg/kg/day at baseline to 1.6 mg/kg/day (an increase of 1 mg/kg/day). We feel it is likely that the improvement in placebo seen in the EMBARK is a consequence of increased steroid dosing.
In their Discussion, the EMBARK authors 1 interpret their lack of detection of gene therapy drug effect as follows: “The heterogeneity of disease progression is a challenge when designing DMD clinical trials, specifically trials of short duration (38). Particularly, during the ages of 4–7 years, motor function and coordination, including ambulation, may be still improving, maintaining or starting to decline from peak function as patients may be in the maturational or the plateau/early-decline phase (39).” Our recent Class I study of corticosteroid effect in DMD (VISION-DMD trial) demonstrated that the same motor outcomes used in the EMBARK trial can readily detect drug-related improvements in motor outcomes in this same age range and similar duration. Consistent with this study, a Class I study of edasalonexent vs. placebo in a similar age range and treatment period found that the placebo group showed either decline, or stability, but not improvement. 7 Thus, the suggestion of the EMBARK study authors that ‘heterogeneity of disease progression’ substantially contributes to the interpretation of their data is not supported by this literature. Rather, we believe that it is more likely that the heterogeneity of corticosteroid effect, introduced into the trial by variable dosing of participants during the trial, is a major contributor to the authors’ observed heterogeneity of disease progression. An analysis of total steroid exposure and association with functional improvements by the Sponsor would be edifying and helpful to the field.
A key limitation to our interpretation is that the effect of increasing corticosteroid dosage from 0.6 mg/kg/d to 1.6 mg/kg/d has not been systematically studied and cannot be directly compared to the effect size we observed in corticosteroid-naïve patients. A double-blind, placebo-controlled trial comparing two doses of prednisone (0.75 mg/kg/d, 1.5 mg/kg/d) to placebo carried out in 1989 in a wide age range of patients (5 to 15 years; mean ∼9 years) showed that both doses of prednisone showed similar efficacy vs. placebo over the 6-month treatment period. 8 However, no more contemporaneous study of higher doses of corticosteroids in DMD has been reported to our knowledge.
Footnotes
Acknowledgements
The figure image from the EMBARK trial was reproduced under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License 1
Authors contributions
All co-authors conceived of and wrote the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Stockholder and employee of ReveraGen BioPharma (EPH, LH). Collaborator, research grant co-investigators on vamorolone clinical trials (EPH, PRC, UJD). Consultant (ad hoc) ReveraGen BioPharma (UJD).
