Abstract
Background
Disease-modifying therapies for persons with spinal muscular atrophy (SMA) has led to greater need and demand for relevant outcomes assessments. Such tools help monitor disease progression, assess treatment response, and inform clinical management. Canadian SMA clinicians participated in a Delphi process to achieve a 2021 national consensus on recommended outcomes, recognizing future reassessment would be essential given the evolving field and gaps in patient-reported and bulbar measures.
Objective
Derive updated Canadian consensus of recommended outcome measures for adults with SMA.
Methods
A national consensus workshop was held with SMA clinicians, patient/family representatives, research leaders, national registry and advocacy organizations. Clinics and registry experience and data were presented and discussed, utility of current and additional outcomes reviewed. Long and short lists of measures were generated, with voting to derive consensus.
Results
Practical implementation, value of data, and relevance to persons with SMA were key considerations. Consensus was achieved to ‘think outside the box’, recognizing a spectrum of function and need to choose the right outcome measure for the right patient at the right time. Measures with greater acceptance for adults were selected, and bulbar measures introduced. Eight outcome measures are recommended; 4 motor, 2 respiratory and 2 patient reported domains, with use based on the individuals level of function. The Revised Upper Limb Module was deemed to have broadest applicability except in the strongest and weakest adults. Additional measures are included as optional and exploratory.
Conclusions
Employing measures meaningful for clinicians, researchers, and persons living with SMA is essential to ensuring quality data collection and an engaged patient-centred clinical team. Clinicians should select measures based on the person's functional ability and goals. Building a national community of practice to support clinical and research practice, including standardized outcome measure training, will be a key next step in dissemination and advocacy.
Background
Spinal muscular atrophy (SMA) is a recessive neuromuscular disease characterized by progressive muscle weakness and atrophy due to degeneration of anterior horn cells, resulting in lower motor neuron loss. Over 90% of SMA is caused by a deletion or loss-of-function mutation on chromosome 5q13.2 of the Survival Motor Neuron 1 gene (SMN1),1,2 which is crucial in producing Survival Motor Neuron protein (SMN protein).3,4 The loss of SMN protein results in aberrant splicing and transport of ribonucleoproteins leading to improper function and eventual loss of lower motor neuron cells.3,5 SMN2, a paralogous gene, can produce a small amount of stable SMN protein, with the number of SMN2 gene copies generally corresponding to disease severity.3,5
The incidence of SMA has been reported as one in every 11,000 live births, with routine newborn screening being implemented worldwide, leading to presymptomatic treatment and improved outcomes.6,7 Recent 2020 and 2021 Canadian data from genetics laboratories, surveillance programs and national registries estimated 1 in 21,472 live births with a prevalence of 0.85 per 100,000 population ages 0–79 years. 8
There is a wide range of SMA phenotypes, which are formally subtyped into 5 types, ranging from in-utero SMA (Type 0) to adult-onset SMA (Type 4). 1 SMA subtypes depend upon the age of onset, SMN2 copy number, previous life expectancy, and motor milestone achievement. With disease-modifying therapy (DMT), expected functional achievement and life expectancy are no longer reliable in determining the SMA subtype. A functional classification system also exists, which identifies persons with SMA (pwSMA) as being non-sitters, sitters or ambulators. This functional classification is used independent of the SMA subtype and better classifies functional ability among pwSMA.
Due to progressive weakness, pwSMA will experience impairments in various domains of function, including gross motor skills (sitting, standing, ambulation), fine motor skills, respiratory, speech, and bulbar function. Additional non-motor symptoms include fatigue, cognitive changes, and gastrointestinal issues.9–11 Medical treatment for pwSMA outside of clinical trials was supportive in nature up until 2017 when DMTs became available. Now, gene replacement therapy via AAV9 (omnasemnogene abeparvovec) delivering functional SMN protein means that infants are surviving and meeting motor milestones. 7 Others are treated with SMN2 splicing therapies, including the antisense oligonucleotide nusinersen or risdiplam, a small molecule therapy, to achieve motor skills and reduce expected disease progression. 12 The DMT era has shifted disease expectations and altered natural history, creating entirely new challenges and opportunities in the management of adults with SMA (awSMA).
In response to the need to improve the evaluation of treatment effects on disease progression and function for clinical purposes as well as overall management of adults living with SMA whether on DMT or not, the Canadian clinical SMA community undertook a national consensus exercise to select a ‘toolkit’ of recommended outcome measures that all clinics would strive to accommodate. Thirty-four measures were identified through the literature and clinician feedback, and information tables on each measure were provided with details on published use, reliability, validity, functional domain, and time required to administer. Participants recognized and acknowledged the particular challenges in making recommendations for the adult population; there is a paucity of both clinical trial and real-world evidence data specific to adults, and for some the disease is a slower progression, limiting outcome measure responsiveness over shorter terms. After multiple rounds of voting and meeting, the resulting consensus published in 2021 recommended 8 different outcome measures performed for one or more of the function levels of non-sitter, sitter, and ambulator (Figure 1). 13
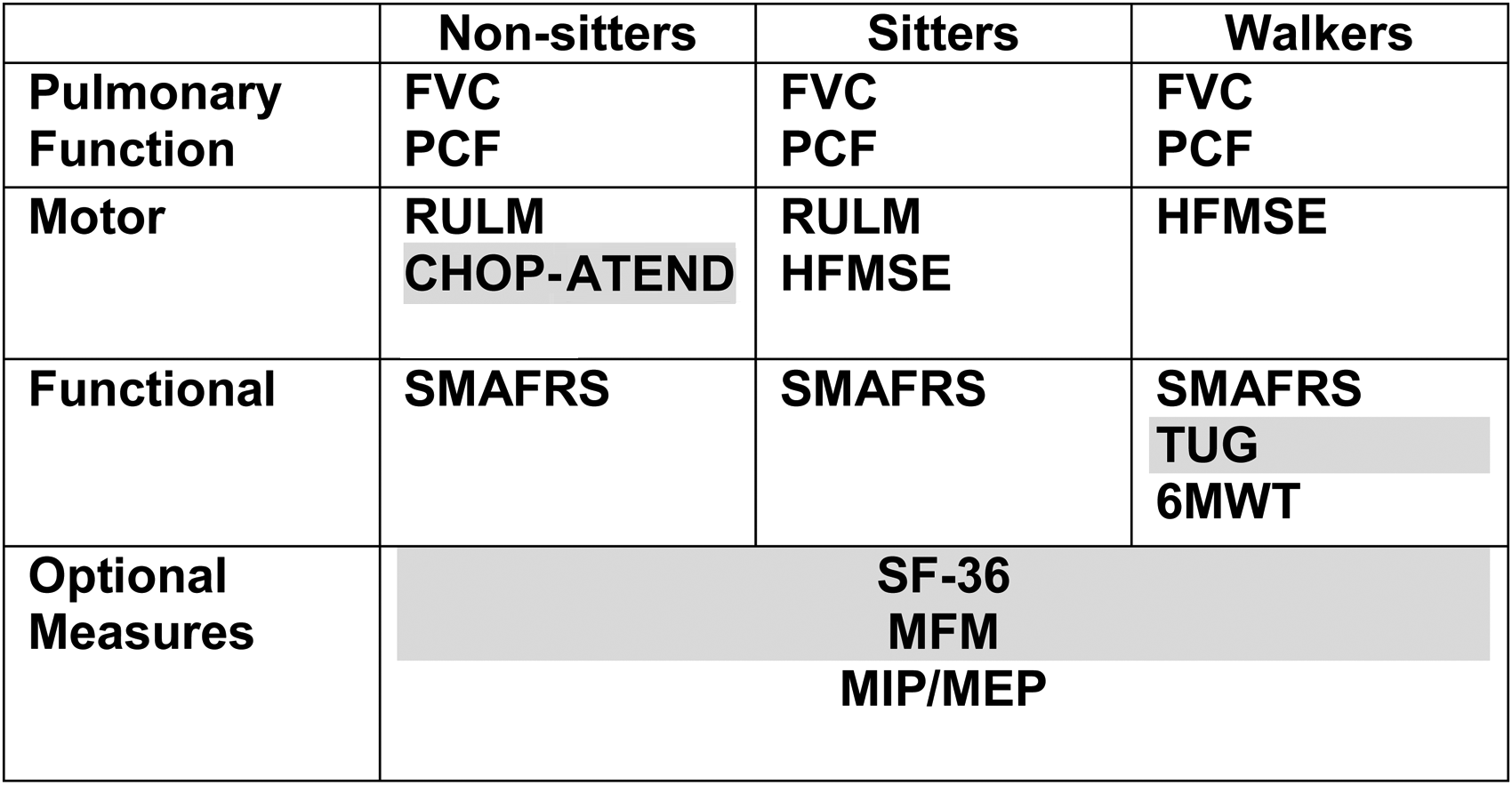
Outcome measures selected as Canadian consensus 2021 for adult SMA.
The national outcomes consensus aims to facilitate and encourage streamlined data collection and build a robust national database, providing a rich understanding of treatment effects, natural history, and real-world outcomes. Across Canada, 38 clinics participate in the Canadian Neuromuscular Disease Registry (CNDR), a national longitudinal registry study that collects epidemiological, clinical, genetics and outcomes data for several neurologic diseases, including pediatric and adult SMA. 14 Launched in 2012, the CNDR SMA registry aims to ensure accessibility and use of data by the broad neuromuscular community and has participated in 88 SMA-related studies (data releases, clinical trial inquiries, surveys, and others) since inception. The dataset items were expanded in 2019 to support an understanding of safety and effectiveness of novel disease-modifying therapies. Currently, the CNDR SMA registry hosts data for 416 participants (280 pediatric SMA and 136 adults with SMA). Data on response to therapy in awSMA has supported numerous posters as well as analyses for regulatory bodies within Canada and internationally. Such consensus also provides valuable solidarity and support in advocacy, as was demonstrated by the collective response to payer demands for frequent patient assessments that clinically were not indicated. 15 The development of robust outcome measure platforms to support clinical research is a priority of the Neuromuscular Disease Network for Canada (NMD4C), a pan-Canadian collaborative network of clinicians, researchers, patients, and advocacy organizations. 16
With the initial consensus, it was well recognized that a future review would be essential due to a rapidly evolving outcome measure landscape, apparent gaps in domains of function for communication/speech and swallowing, and a lack of patient-reported outcomes. 13 Newer measures such as the Motor Function Measure (MFM) were promising and would need reconsideration as data and clinical experience increased. Indeed, since the initial consensus, work on SMA-specific patient reported outcome measures have been published including topics of fatigue and bulbar function,9,17 further supporting the need to re-visit our recommendations.
In Canada, national regulatory approval by Health Canada exists for DMT for all age groups including adults. While coverage for adults is provided by some private payers, public reimbursement criteria is determined at a provincial level, and currently is only covered for adults in the province of Quebec. This variable reimbursement has led to gaps in outcomes data collection in some provinces given treatment-naïve adults do not have routine assessments performed. As such, Quebec clinicians have developed expertise in coordinating and administering assessments, and the first re-evaluation of the toolkit occurred in Quebec. Following an evaluation requested by the main university hospitals (medication management program, “Programme de gestion thérapeutique des médicaments”), which highlighted the heterogeneities and inconsistencies of the outcomes being performed and collected, a group of health professionals gathered to reach a provincial consensus on the clinical tools.
The Quebec group met in the fall of 2022 and comprised adult and pediatric therapists including medical specialists (physiatrists, neurologists), physiotherapists, occupational therapists, speech therapists, nurses, and a clinical coordinator. The outcome measures were selected based on if the test was internationally recognised, the duration of the test, cost, and accessibility. The goal was to find tests that mitigate a floor or ceiling effect when measuring patient ability.
This Quebec consensus to standardize assessment measures across the province was obtained and ratified at the beginning of 2023 after the formation of the Réseau québécois sur l'amyotrophie spinale (RQAS) (Figure 2). A first training session on these tools took place in Quebec on March 23, 2024 with 56 participants across 5 disciplines, focusing on 6 outcome measures.
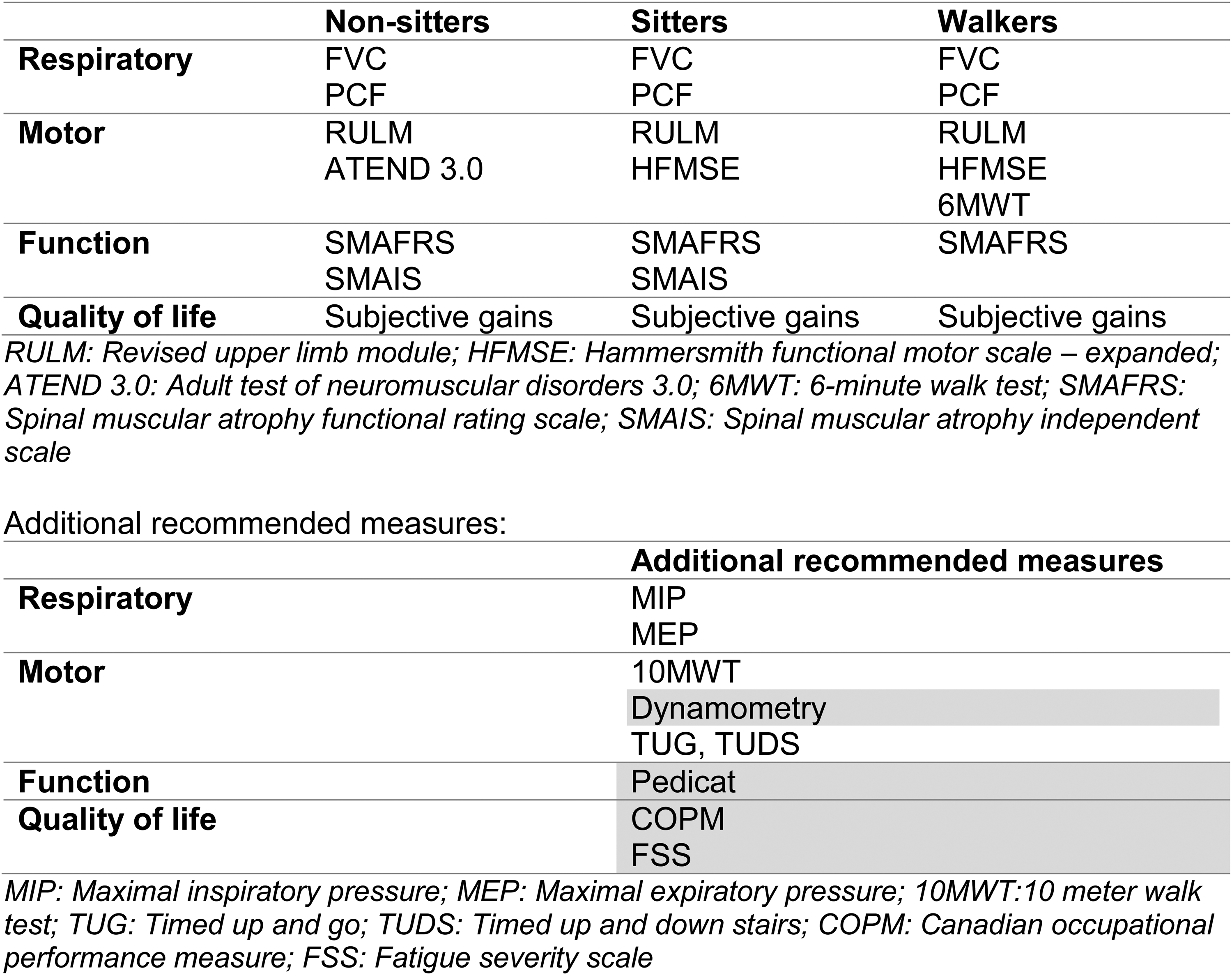
Quebec updated outcome measures 2023.
With the Quebec momentum and support of national stakeholders, the SMA community set a goal of establishing national consensus on updated recommended outcome measures to be used in adults with SMA and to align our national CNDR SMA data collection accordingly.
Methodology
In late 2023, a national 2-day SMA outcomes workshop was held in Ottawa, Canada. Representation included clinical and research network members from NMD4C and CNDR, advocacy organizations Cure SMA (family) and Muscular Dystrophy Canada, clinicians and researchers from CNDR SMA clinics across Canada. A facilitator and note-takers were engaged to support the process.
In the months leading up to the meeting, all participants were provided with pre-meeting materials including workshop objectives, relevant publications, and outcome measure metrics tables. All participants have clinical, research, advocacy or personal experience in SMA, as well as other neuromuscular diagnoses including muscular dystrophies and amyotrophic lateral sclerosis (ALS). The CNDR also manages the national disease registries for both Duchenne Muscular Dystrophy and ALS, which allowed leveraging of practical and research insights with outcomes assessments across other conditions.
Workshop activities to inform the consensus decisions included:
Overview of workshop objectives and importance of building national consensus Review of the Quebec SMA provincial consensus exercise (Figure 2) Review of initial Canadian SMA recommended outcomes toolkit
13
Overview of the SMA CNDR data collection experience and outputs Discussion on patient reported outcome measures and bulbar measures Functional level definitions Recommendations on frequency of patient assessments.
Collectively, the participants shared and reviewed their experience with current outcome measures and evaluated and discussed newer outcome measures and gaps in assessments currently being used. The CNDR provided data from the national SMA database, highlighting relevant needs and challenges. At the time of the workshop, CNDR had data on 271 pwSMA aged 13 and older. According to data reported within the CNDR, 61% of registered adults with SMA have a motor measure reported. The Hammersmith Functional Motor Scale Expanded (HFMSE), Revised Upper Limb Module (RULM), Six-minute walk test (6MWT), and variations of the Children's Hospital of Philadelphia Adult Test of Neuromuscular Disorders (CHOP-ATEND), are the most routinely collected with 88%, 85%, 23%, and 35% of adults with reported scores respectively. However, completion rates vary across the country.
Furthermore, over the past few years, there has been substantial variability between clinics in the version of CHOP-ATEND being used, and variable outcome measure use during the transitional phase from pediatric to adult care. The ATEND 3.0 was recognized as being preferred for adults, with clinics in Canada already implementing instead of CHOP-ATEND. Similar variability exists for ambulators, with certain clinics administering the 6MWT, the 10MWT, and/or the 2MWT, depending on space, time, and preference. The SMA Functional Rating Scale (SMAFRS) was routinely collected in only three clinics. The Short Form 36 (SF-36) has not been implemented. Other patient-reported outcome measures were being captured intermittently or through local pilot studies. Pulmonary function testing is generally routine in certain patient populations where clinically indicated.
When recommending or rejecting a particular measure, participants were instructed to consider factors including required resources, value of the information, ease of measure performance, burden for the person with SMA, and other factors as presented in Table 1.
Factors considered when selecting recommended outcome measures.

Each measure for discussion, both current and proposed measures, were written on individual cards and posted to a large board with sections for functional levels, and subheadings of ‘recommend’, ‘other suggested’, ‘exploratory to pilot’, and ‘reject’ following rounds of discussion. Voting for each measure took place following the close of discussion. The final discussion addressed the recommended frequency of assessments.
Results
Agreement was established to remain consistent with functional levels defined as: Non-sitter – unable to sit unassisted for > 3 s Sitter – able to sit independently (without assistance) for > 3 s Walker – able to walk 4 steps without aid (physical or technical)
However, participants stressed the importance of recognizing a more fluid transition across functional abilities and the need to choose measures that are appropriate to the aawSMA, considering their values and abilities and potential ceiling or floor effects of tools used. The importance of selecting the right measure for the right person at the right time was emphasized.
A unanimous consensus was achieved with a toolkit of 8 recommended measures from which a clinician may choose those most appropriate based on the functional abilities and personal values of the awSMA. As few as 1 or 2 measures may be sufficient for an awSMA based on the individuals’ level of function; for example the 6MWT may be sufficient for a strong ambulator . The RULM was deemed to have the broadest applicability, with good utility in all but the strongest and weakest awSMA. The main recommended changes from the 2021 consensus included use of ATEND 3.0 instead of CHOP-ATEND, change of TUG from ‘recommended’ to ‘other suggested’, and introduction of the SMAIS-ULM as a PROM. An additional 5 measures are ‘other suggested’, with their use depending upon clinic resources and patient and clinic needs. Four patient reported outcome measures to be further explored were selected, including the emerging SMAIS-Ambulation (SMAIS-AMB). 18 Experience and data from individual clinic use of the “other suggested” and “exploratory” measures will inform and guide future consensus and toolkit revisions. All 17 outcome measures comprising the SMA recommended toolkit -revised (SMART-R) are described in Table 2. A graphical visualization was designed to capture the concept of a fluid outcome measure recommendation without rigid classification limiting tool choice (Figure 3) and encourage use of the right tool for the right person at the right time.
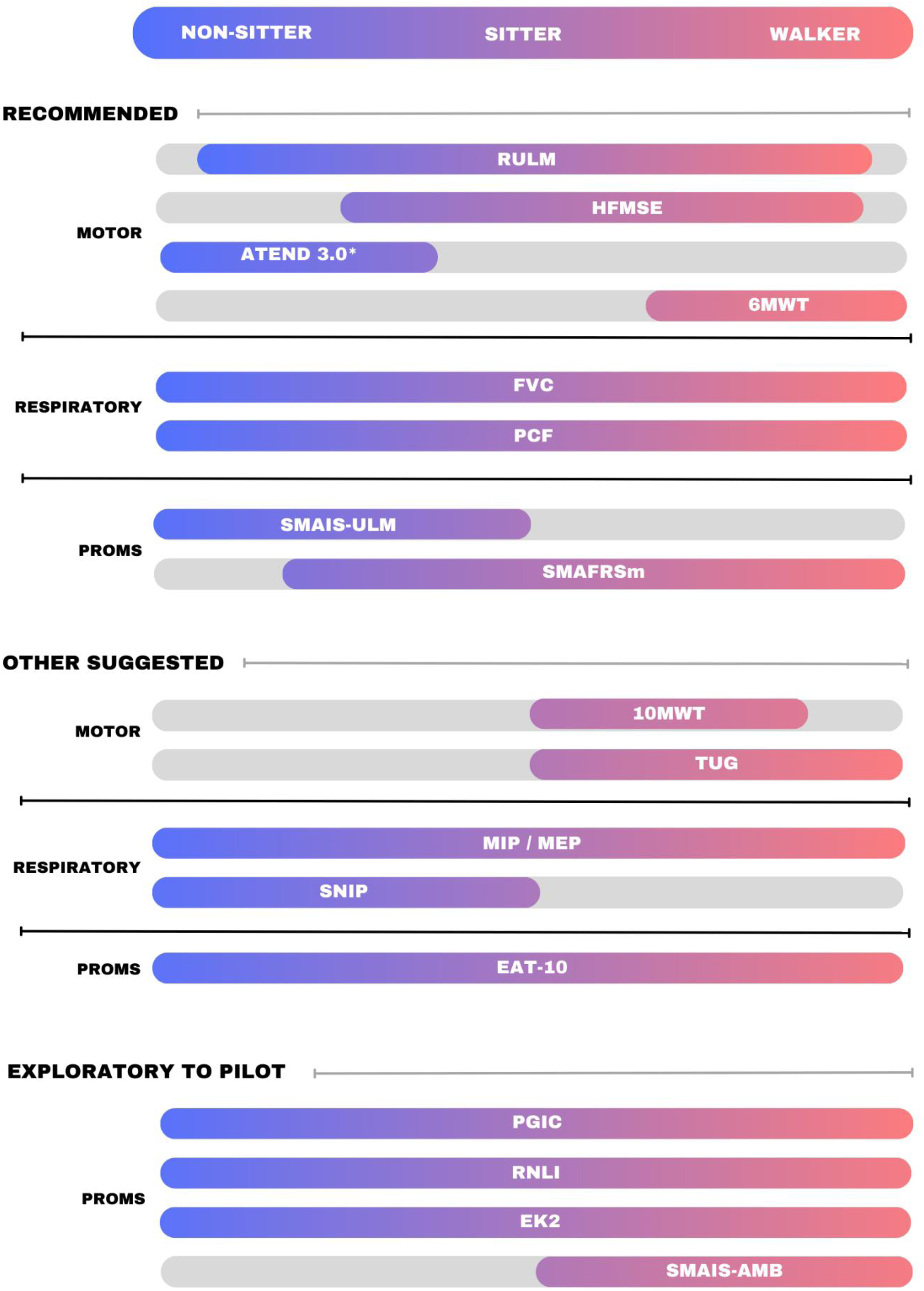
Canadian SMART-R outcomes (spinal muscle atrophy recommended toolkit – revised outcomes) for initiating a disease-modifying treatment, assesses at baseline, 6 months and 1 year. Assess annually as part of routine care when on stable dose of disease-modifying treatment or when treatment naïve. Recommended: FVC – Forced Vital Capacity; PCF – Peak Cough Flow; RULM – Revised Upper Limb Module; HFMSE – Hammersmith Functional Motor Scale Expanded; ATEND 3.0 – Adult Test of Neuromuscular Disorders 3.0; 6MWT – 6-min Walk Test; FVC – Forced Vital Capacity; PCF – Peak Cough Flow; SMAIS-ULM – Spinal Muscular Atrophy Independence Scale Upper Limb Module; SMAFRSm – Spinal Muscular Atrophy Functional Rating Scale-modified
Canadian SMART-R outcomes (spinal muscle atrophy recommended toolkit – revised outcomes) brief descriptions.

Discussion
There was overall consensus that the outcome measures used in clinical visits need to be meaningful to patients and families and need to reflect what functional activities are important in their daily lives. Reliable and feasible outcome measures that mitigate floor and/or ceiling effects and are responsive to change over time are essential to ensure an accurate representation of patient function and disease progression. Patient fatigue and of clinic visit length was considered when discussing the different outcome measure options, as many patients have long days when travelling and attending clinic appointments.
A major change from 2021 was the removal of the CHOP-ATEND; notably it was difficult to perform with adults who could not easily be examined on a plinth/exam table, it doesn’t take into consideration the different body types of patients, the contractures that adult SMA patients may have, and the fact that some patients can’t put their bodies into the positions required to complete the test. Participants shared the experience of patient and family frustration and discouragement with this assessment. The ATEND 3.0 was recently introduced in Quebec and New Brunswick, and although the test is not yet validated, patient feedback supports that ATEND 3.0 is a more appropriate test for adults, and that patients are more willing to participate in it.
From published literature and participants experience, the RULM was determined as the most appropriate for many patients but will have a floor effect for the weakest of non-sitters and ceiling effect for the stronger walkers.52,53 Participants reflected that for a pwSMA and their family to hear scores annually of “zero” is emotionally difficult and adds little value to clinicians. For those individuals who are non-sitters and have hit a floor on the RULM, it was suggested to add the ATEND 3.0. Overall agreement was to use clinician's best judgement for which test would best measure meaningful patient function when a patient doesn’t fall within a clear functional category or overlaps between two functional categories or has abilities within the extremes of function. For high functioning walkers, additional measures can be used to better assess their function, such as TUG or 10MWT. Using these additional measures for high functioning walkers will mitigate ceiling effects and provide meaningful results.
Overall agreement was that pulmonary function is necessary to measure in adult SMA patients. It was recommended to record the absolute value of FVC (forced vital capacity) in L/mL in addition to the currently captured % predicted. This is important as % predicted may not reflect true changes in respiratory function, rather than reflecting changes in age or anthropometric measures that impact the prediction algorithm.
A key point of the discussion was the lack of bulbar function measured in the currently used SMA outcomes. It was agreed upon by the group that bulbar function needs to be captured, and part of this consensus should include an outcome measure to pilot that will capture bulbar function. Ultimately it was decided that the EAT-10 was likely the most feasible outcome measure to pilot in routine patient clinic visits, given no specialized equipment required and its use as a patient-reported outcome. 17 The participants acknowledged that open-source, free to use outcome measures requiring little-to-no equipment are more likely to be integrated into routine practice, as opposed to measures that are resource or cost prohibitive.
The group agreed that patient-reported outcome measures (PROMs) were of utmost importance to capture as they have the potential to more accurately reflect an individual's lived experience, beyond traditional motor outcome measures used. While previous reviews have identified a large body of PROMs used in SMA and other neuromuscular disease populations, there is limited consensus on their use.54,55 The SMAFRS is the PROM most widely used across Canadian clinics although it does not take into account bulbar function, and it was noted that the scoring is not always an accurate representation of patient function; for example, an individual scores a 0 on SMAFRS but hold a full-time job. The SMAIS (most recent version updated to SMAIS – Upper Limb Measure) can mitigate the floor effect of the SMAFRS, is a validated measure, and is specific to SMA. SF-36 and PedsQL were both discussed, and it was decided that neither should be recommended as they are not specific to SMA and are less responsive to change. The Reintegration to Normal Living Index (RNLI) was introduced as a possible PROM to assess quality of life. RNLI is a generic scale, is validated in multiple languages, including English and French, and can be self-administered. The language used in the RNLI is focused on ones’ satisfaction with various aspects of life including social and personal roles. It was decided that the RNLI could be an exploratory measure.
Frequency of routine assessments was discussed, and consensus confirmed the recommended annual assessment for pwSMA on stable dose of disease-modifying treatment or that are treatment-naïve, as part of routine clinical care. For adults initiating a disease-modifying treatment, consensus recommends assessment at baseline, at 6 months and 1 year, followed by annual assessments thereafter.
Proper training for clinical staff is required to implement some outcome measures and ensure standardization of scoring. Such training can be foundational in building a community of practice that will ensure consistent training and implementation of outcome measures across Canada (ensuring all clinic staff receive the same training). Notably, national training for ATEND 3.0 is recommended as it is not widely used outside of Quebec at this time. Training opportunities and resources will need to be accessible to clinic and research teams across the country and will benefit from an online presence for continuing education and support.
Finally, recognizing the evolving field of outcome measure evaluation in SMA, participants iterated the need to establish a committee and meet annually to re-evaluate the consensus decisions, review the piloted outcome measures and literature, and ensure we are aligning with international recommendations. All neuromuscular organizations, including patient organizations, such as CureSMA Canada, Muscular Dystrophy Canada, and NMD4C, should continually be involved in ensuring outcome measure alignment and consensus.
Conclusion and next steps
Based on the results of this consensus meeting, the Canadian Neuromuscular Disease Registry (CNDR) will update their dataset accordingly, and dissemination strategies including webinars, investigator meetings, publications, conference presentations and training workshops will target the SMA clinic and research community.
Care of pwSMA is best provided through multidisciplinary clinics resourced with the appropriate health professional team members, tools, skills, and time. As a national priority, an outcome measures training program and associated community of practice is identified as essential to support clinics in developing the skills and confidence of their teams. Improving access to standardized assessments can also be facilitated by use of remote or virtual platforms; recent work by Arteaga-Bracho et al. demonstrated utility of smartphone-based use of RULM and HFMSE. 56 Informed by the community through close partnerships with Muscular Dystrophy Canada and Cure SMA Canada, and lead by the national clinical and research networks Neuromuscular Disease for Canada and Canadian Neuromuscular Disease Registry, the development of a Canadian guidelines for multidisciplinary care in SMA – inclusive of outcome assessments – is a future goal.
Footnotes
Acknowledgments
All authors other than JS and HO participated in the workshop; all authors contributed to pre-workshop preparation and post-workshop collaboration.
Funding for this meeting to support attendance and logistics was provided by Roche Canada. Roche was not involved in the agenda, in the meeting content, activities, or results, and have had no participation in preparation of this workshop report.
The CNDR SMA program receives support from Biogen, Novartis, and Roche. No funding party is involved in the data collection or analyses of the registry.
HL receives support from the Canadian Institutes of Health Research (CIHR) for Foundation Grant FDN-167281 (Precision Health for Neuromuscular Diseases), Transnational Team Grant ERT-174211 (ProDGNE) and Network Grant OR2-189333 (NMD4C), from the Canada Foundation for Innovation (CFI-JELF 38412), the Canada Research Chairs program (Canada Research Chair in Neuromuscular Genomics and Health, 950-232279), the European Commission (Grant # 101080249) and the Canada Research Coordinating Committee New Frontiers in Research Fund (NFRFG-2022-00033) for SIMPATHIC, and from the Government of Canada Canada First Research Excellence Fund (CFREF) for the Brain-Heart Interconnectome (CFREF-2022-00007). HL is an Editorial Board Member of this journal but was not involved in the peer-review process nor had access to any information regarding its peer-review. CO's institution has received research grant support from Roche Canada, Biogen. CO, KS have received honorarium for advisory board and/or speaker fees from Roche Canada and Biogen.
ORCID iDs
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
