Abstract
FORTITUDE-ALS was a study evaluating reldesemtiv in people living with ALS. Post-hoc analysis identified larger treatment effects in those with symptom onset ≤24 months and baseline ALSFRS-R ≤ 44 (24/44 criteria). Using the ENCALS risk score (RS), we analyzed how the 24/44 criteria changed the eligible population. Of the 272 participants meeting the 24/44 criteria, 73% had very short to intermediate RS compared to 18% not meeting the criteria. Though the 24/44 criteria enriched the FORTITUDE-ALS population with rapidly progressing patients, they did not completely exclude all patients with a very long predicted survival.
Introduction
Amyotrophic lateral sclerosis (ALS) is a progressive disease affecting motor neurons in the brain and spinal cord for which there is no cure, and approved treatments provide limited benefit. With the hope that better treatments can be developed, rethinking clinical trial design to improve the efficiency of therapeutic development is warranted.1,2
Clinical trial design in ALS faces many challenges including clinical disease heterogeneity. Because of this, clinical trials use inclusion criteria to select homogeneous populations of participants most likely to benefit from a particular treatment. One way to quantify the heterogeneity of ALS participants is using predictive models, such as the European Network to Cure ALS (ENCALS) predictive survival risk score (RS) which uses eight clinical variables to generate predictions on prognosis and/or survival. 3 Besides serving as eligibility criteria themselves, these predictive tools can also be used to quantify the expected heterogeneity of participants eligible for a future ALS clinical trial and be used to assess how heterogeneity is impacted when modifying conventional criteria, such as restricting the duration of symptoms or the extent of functional symptoms.
In this study, we illustrate in a real-world example how the ENCALS RS can be used to evaluate a change in conventional eligibility criteria in order to enrich the eligible population for a future study with more rapidly progressing patients.
Methods
FORTITUDE-ALS
The data used for this study originated from the FORTITUDE-ALS (NCT03160898) study. This was a 12-week, randomized, double-blind, placebo-controlled, phase 2 study of reldesemtiv in people living with ALS (pALS) with results previously described. 4 Reldesemtiv acts by increasing the sensitivity of the sarcomere to calcium, thereby increasing contractility of the muscle to produce greater force in response to neural stimulation. 5 In total, 458 pALS were randomized to either active or placebo treatment. The primary outcome was the change from baseline in % predicted slow vital capacity (SVC) at 12 weeks with the key secondary endpoint being the change from baseline in ALSFRS-R total score at 12 weeks. Key inclusion criteria included diagnosis of ALS ≤ 24 months, %predicted SVC ≥ 60%, and a stable dose of riluzole and/or edaravone.
The primary and secondary outcome measures in FORTITUDE-ALS were not met. 4 Post-hoc analysis of FORTITUDE-ALS found that pALS with a shorter time from symptom onset (≤24 months) and a more rapid progression prior to study initiation (ALSFRS-R ≤ 44) were more likely to respond to reldesemtiv. Of pALS in FORTITUDE-ALS with symptom onset >24 months and an entry ALSFRS-R total score of >44, 89% and 95% were in the slowest progressing tertile of subjects, respectively. 6 This formed the basis for the “24/44” criteria.
ENCALS survival prediction model
The development of the ENCALS RS model has been previously published. 3 Briefly, clinical data for pALS from 14 European ALS centers were collected. From these data, 16 participant characteristics were identified as potential predictors of time to death or respiratory insufficiency. Backward elimination with bootstrapping was applied for predictor selection; 8 predictors were selected and used to develop the final multivariable model, including age of onset, diagnostic delay, site of symptom onset, C9orf72 repeat expansion, ΔFRS, vital capacity, presence of frontotemporal dementia, El Escorial classification. RS were then grouped into five survival categories: very short, short, intermediate, long, and very long.
Statistical analysis
The ENCALS predicted survival RS 3 was calculated for all participants enrolled in FORTITUDE-ALS for the full analysis set (FAS). Participants were assigned into the five groups based on their RS, very short (G1) through very long (G5) predicted survival. The numbers of participants in each of the five RS groups were then compared between placebo and reldesemtiv treatments. Participants in FORTITUDE-ALS who met the 24/44 inclusion criteria were identified. The numbers of participants meeting the 24/44 criteria in each RS group were compared to the numbers of participants in each risk group from the FAS.
Results
When applied to the pALS in FORTITUDE-ALS, the distribution of ENCALS RS categories were well balanced between the two treatment arms. The mean (±SD) RS for pALS randomized to placebo was −4.65 ± 1.46 and to reldesemtiv −4.80 ± 1.85 (p = 0.4136) for the FORTITUDE-ALS FAS. The mean RS for all participants was −4.77 (Table 1). There were no statistically significant differences in RS distribution when comparing groups assigned to placebo and reldesemtiv (Table 1). Notably, participants in the G1 group were underrepresented if one expects a roughly equal distribution for each RS group in the general pALS population. This underrepresentation may have occurred for several reasons, including the applied eligibility criteria, the reduced willingness of very rapidly progressive participants to participate in clinical trials because of the perceived burden of trial visits, and the lower survival among patients with a poor prognosis between the time of diagnosis and trial enrollment. 7
FORTITUDE-ALS participants by category of ENCALS risk score.
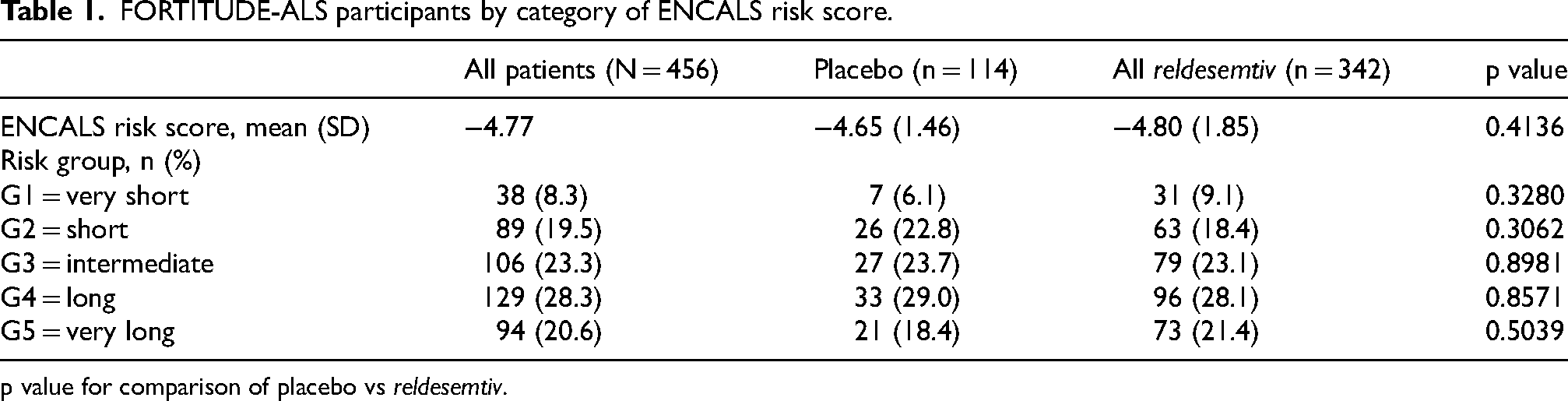
p value for comparison of placebo vs reldesemtiv.
To understand how the 24/44 criteria may affect pALS clinical heterogeneity, the ENCALS RS calculated for FORTITUDE-ALS participants meeting the 24/44 criteria compared with those who did not (Table 2). Of the participants who met the 24/44 criteria (N = 272), 73% were in the G1-3 groups whereas of those not meeting the 24/44 criteria, 18% (34/184) were groups G1-3. All the pALS in FORTITUDE-ALS assigned to the G1 RS group met the 24/44 criteria as well as 4.4% of pALS in G5.
FORTITUDE-ALS participants according to 24/44 criteria.

p value for comparison of groups who met vs did not meet the 24/44 criteria.
Discussion
In this study, we evaluated how revision of eligibility criteria impacted the prognostic heterogeneity in a trial population. The ENCALS predictive model is a well validated tool utilizing multiple clinical characteristics to make predictions for the survival time of pALS, and can be used to quantify the prognostic heterogeneity of a population. 3 A post-hoc analysis of FORTITUDE-ALS identified a subset of pALS that had a smaller decline in ALSFRS-R and SVC with reldesemtiv compared to placebo.4,6 These participants were found to be predominantly faster progressing pALS and had a baseline ALSFRS-R score of ≤44 and time since symptom onset ≤24 months – the basis of the 24/44 criteria. We show how these criteria, according to the ENCALS predictive model, selected a population of pALS who were faster progressing without completely excluding slower progressors. The 24/44 criteria did not select, however, all subjects with a faster disease progression or fully excluded those with a very slow progression rate. This is likely because the 24/44 criteria only considers two variables whereas additional variables are associated with ALS progression and survival, as evidenced by the independent prognostic value of eight variables in the ENCALS RS. 3 Moreover, the prognostic value of a total ALSFRS-R score of 44 may be different for each participant given the multidimensional build-up of the ALSFRS-R itself. 8
How modification of eligibility criteria – in this case, applying the 24/44 criteria – impact a trial population is insightful for the design and interpretation of future trials. Here, the 24/44 criteria enriched the FORTITUDE-ALS population with rapidly progressing patients, but did not completely exclude all patients with a very long predicted survival. From an efficiency perspective, this may be unwarranted as this would require a larger sample size. On the other hand, from a regulatory perspective, keeping some very fast or very slow progressing patients in the trial population may help to generalize findings to a broader population of patients, which could facilitate subsequent drug approval or insurance coverage. It should be noted, however, that in this case, the 24/44 criteria also led to adverse exclusion of patients in the “favorable” patient groups (e.g., adverse exclusion among G2 patients in Table 2). Thus, though there is a wider prognostic range in the enriched trial population using conventional criteria like the 24/44, the actual number of patients eligible may still be less compared to using the ENCALS RS directly as inclusion criterion. Hence, it is of interest to optimize patient eligibility criteria while weighing prognostic diversity, exclusion rates and trial efficiency, and evaluate how this may differ depending on the phase of drug development.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Cytokinetics, Inc., South San Francisco, CA, USA.
Conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TS, SK, FIM, LM, SAR, JW: Employees of and own stock in Cytokinetics. JMS reports compensation received as a consultant from Amylyx, Apic Biosciences, NeuroSense Therapeutics, Cytokinetics, Denali Therapeutics, GSK, Mitsubishi Tanabe Pharma America, Orphazyme, Orthogonal, Pinteon Therapeutics, RRD, SwanBio, Helixmith, Novartis, Sanofi, PTC, and EMD Serono; and research support from Amylyx, Biogen, Biotie Therapies (now Acorda Therapeutics), Cytokinetics, Mitsubishi Tanabe Pharma America, Alexion, MediciNova Inc, Ionis, Alector, and Orphazyme. RPAVE has no conflicts of interest to report.
Data availability
Qualified researchers may submit a request containing the research objectives, endpoints/outcomes of interest, a statistical analysis plan, data requirements, a publication plan, and qualifications of the researcher(s). Requests are reviewed by a committee of internal and external advisors. If approved, information necessary to address the research question will be provided under the terms of a data sharing agreement. There is no end date for eligibility to submit a data sharing request for this study. Requests may be submitted to medicalaffairs@cytokinetics.com.
