Abstract
Background
The application of recombinant adeno associated virus (rAAV) in gene therapy is accepted as an effective strategy for the treatment of monogenic diseases. However, eligibility for such therapies is contingent upon the absence or minimal presence of antibodies against adeno-associated virus (AAV) capsid protein. While the passive transfer of maternal immunoglobulins in utero is well established, the potential impact of maternal antibodies transferred via breastfeeding remains less explored.
Objective
This study aims to quantify and compare the levels of anti-AAV9 and anti-AAVrh74 immunoglobulin G (IgG) and M (IgM) in both plasma and human milk from a group of healthy lactating mothers.
Methods
In this cross-sectional study, we analyzed plasma and human milk samples from healthy lactating mothers. We used an enzyme linked immunosorbent assay (ELISA) to determine the levels of circulating IgG and IgM antibodies against AAV9 and AAVrh74 capsids and compared their concentrations in the paired samples.
Results
Thirty-one paired plasma and human milk samples were analyzed. The median age at participation was 34 years (range: 25–43), median duration of breastfeeding at the time of sample collection was 7.5 months (range:0.7–33), and median body mass index was 23 Kg/m2 (range: 19.7–35.4). Median anti-AAV9 IgG in plasma and human milk, were 183 U/mL (range: 29–7214) and 1 U/mL (range: 1–33), respectively. Median anti-AAVrh74 IgG, in plasma and human milk were 138 U/mL (range: 23–8725) and 1 U/mL (range: 1–27), respectively. The differences in anti-AAV9 and anti-AAVrh74 IgG levels between plasma and human milk were statistically significant (p < 0.0001). Additionally, a strong correlation (r: 0.97, p: < 0.0001) was observed between anti-AAV9 and anti-AAVrh74 IgG levels in plasma.
Conclusions
The levels of anti-AAV9 and anti-AAVrh74 antibodies in human milk are remarkably lower than those in plasma. Consequently, breastfeeding should not be restricted for term infants who are potential candidates for AAV-related gene therapy products.
Introduction
Gene therapy has emerged as a proven approach for the treatment of a wide range of genetic diseases. Among the various gene delivery platforms, recombinant adeno-associated viruses (rAAV) have gained significant attention for their ability to deliver therapeutic genes to target cells. 1 In particular, rAAV-based gene therapy products address critical unmet clinical needs, especially in pediatric patients with monogenic genetic disorders, including fatal neurodegenerative diseases. 1 Adeno-associated virus (AAV), a member of the Parvoviridae family, is naturally found in multiple vertebrate species, including human and non-human primates. Importantly, AAV has not been associated with any human viral diseases. Structurally, it is composed of an icosahedral protein capsid of ∼26 nm in diameter and a single stranded DNA genome of ∼4.7 kb.1,2 Recombinant AAVs (rAAVs) share the same capsid sequence and structure as the wild-type; however, the encapsulated genomes within rAAV are modified to exclude all AAV protein-coding sequences, allowing them to carry therapeutic genes specifically designed to target tissues.1,2
The Food and Drug Administration (FDA) has approved several rAAV products for systemic administration. These products include onasemnogene-abeparvovec (Zolgensma®) for the treatment of spinal muscular atrophy (SMA), etranacogene-dezaparvovec (Hemgenix®) and fidanacogene elaparvovec (BeqvezTM) for the treatment of Hemophilia B, valoctocogene roxaparvovec (Roctavian®) for Hemophilia A, and delandistrogene-moxiparvovec (Elevidys®) for Duchenne muscular dystrophy (DMD).3–5 Further, it is anticipated that the FDA will approve several additional rAAV derived products in the upcoming years. Many of these upcoming treatments are poised to significantly impact infants and children, especially in the setting of newborn screening, as in the case for infants with SMA.
SMA is an autosomal recessive neurologic condition due to mutations in the survival motor neuron 1 (SMN1) gene resulting in progressive weakness and paralysis due to the lack of SMN protein. 6 Thanks to the approval of three disease-modifying therapies, including onasemnogene-abeparvovec, the outcomes of patients with SMA have dramatically changed in recent years.3,7–9
Eligibility for AAV related therapies is dependent on the absence or low levels of immunoglobulin G (IgG) antibodies against AAV. The seroprevalence of anti-AAV antibodies in the adult general population varies significantly, ranging from 14.8% and 72% depending on factors such as the AAV serotype, antibody type, the specific assay used, and the chosen titer cutoff.10,11 In a study performed in patients with DMD, the seroprevalence of total binding antibody levels against AAVrh74 was found to be 13.9% in patients aged 4 to 18 years, using a titer cutoff of <1:400. 12 Conversely, a different study in a cohort of infants with SMA revealed positive total antibody titers against AAV9 in 7.7% of the patients. This study used a different assay and a titer cutoff of 1:50. 10
Immunoglobulin A (IgA) is the dominant antibody isotope in human milk, and while IgG is also detectable, it is not present in significant amounts. 13 Further, these antibodies typically do not enter the infant's circulation in significant quantities.13,14 However, in several clinical trials in infants with SMA using onasemnogene-abeparvovec, such as START [NCT021222952], STRIVE-US [NCT03306277], STRIVE-EU [NCT03461289], and SPRINT [NCT03505099]), there was a precautionary measure for mothers; those who were seropositive for anti-AAV9 antibodies were advised not to breastfeed for 30 days post dosing. 10 This caution extended to infants consuming donor breast milk that had not been tested for antibodies; these infants were switched to formula prior to participation in the trials. 10
In this study, we quantified the levels of anti-AAV9 and anti-AAVrh74 IgG and IgM in a cohort of paired plasma and human milk samples from lactating mothers. AAV9 and AAVrh74 are the two AAV products that are currently used in the clinical setting. Additionally, we explored the relationship between these antibody levels in the respective sample.
Materials and methods
Study design
This study was performed through secondary analysis of plasma and human milk samples from lactating mothers recruited as part of a prospective observational study at the University of Florida for the evaluation of antibodies in plasma and human milk samples before and after SARS-CoV2 vaccination. Additional samples were collected cross-sectionally from a separate study to assess the seroprevalence of anti-AAV antibodies. Both studies were performed under institutional IRB approval (#IRB202003255 and #IRB201900677).
Recruitment of participants
Participants were recruited through the University of Florida's institutional email and advertising flyers posted at Shands Hospital for enrollment (IRB202003255) and via institutional email only as part of the cross-sectional study (IRB201900677). Inclusion criteria were: lactating women, ≥18 years old, and being able to provide consent. Upon agreement to participate, demographic information including age, race, ethnicity, weight, height and lactation duration was recorded.
Blood and human milk sample collection
Blood samples were collected by finger prick in ethylenediaminetetraacetic acid (EDTA) coated tubes (IRB202003255) and via peripheral venipuncture and collected in EDTA coated tubes (IRB201900677). Plasma was separated from cellular matter by centrifugation at 2000 g for 10 min at 4 °C. Plasma was stored undiluted at −20 °C and was not heat inactivated prior to analysis.
For consistency, mothers were instructed to collect up to 30 mL of the initial expressed human milk (foremilk) in the morning. Samples were stored at −20 °C within 4 h after collection. The samples were aliquoted into 2-mL tubes, then centrifuged at 500 g for 15 min at 4 °C. Using a 21 Gauge needle, the aqueous layer was separated from the fat layer and placed in a clean tube. This aqueous layer was then centrifuged at 3000 g for 15 min at 4 °C. The final aqueous layer was removed and stored undiluted at −20 °C and was not heat inactivated prior to analysis.
Anti-AAV9 and anti-AAVrh74 IgG and IgM antibody titers
Plasma and human milk samples were assayed for circulating antibodies to the AAV9 or the AAVrh74 capsid via enzyme linked immunosorbent assay (ELISA). Briefly, 96-well plates were coated with 1 µg/mL AAV9 or AAVrh74 overnight at 4 °C. A wash with phosphate-buffered saline (PBS)–Tween was followed by blocking for 2 h at 37 °C with blocking buffer (10% heat-inactivated fetal bovine serum (FBS) in 1× PBS). Paired plasma and human milk samples and a negative control (IgA/IgG/IgM depleted human serum (Sigma, St Louis, MO, USA)) were two-fold serially diluted in blocking buffer from 1:10 to 1:10,240 in a low binding plate. Each sample had a blank that contained all assay components except plasma or human milk samples. The standard used was pooled human serum from subjects exposed to AAV-based gene therapies and was initially diluted 1:75,000 (arbitrarily assigned a concentration of 10 units/mL) before being two-fold serially diluted in blocking buffer concurrently with the samples in a low binding plate (Thermo Scientific). At the end of the incubation, plates were washed once, and 50 µL of each standard and sample dilution were added to the ELISA plate and incubated overnight at 4 °C. Washing was followed by addition of the detection antibody at a dilution of 1:20,000 (goat anti-human immunoglobulin G or M, conjugated with horseradish peroxidase [HRP]; Sigma, St Louis, MO, USA) for 2 h at 37 °C. Finally, the plate was washed and exposed to tetramethylbenzidine (TMB) peroxidase detection substrate, stopped with 1 M phosphoric acid, and read at optical density (OD) 450 nm on a BioTek Plate Reader using the Gen 5 Microplate Reader and Image Software (BioTek, Winooski, VT, USA).
The linear portion of a four-parameter logistic standard curve was used to determine the concentration of each sample in units per milliliter (U/mL). Sample antibody titers (U/mL) were then read relative to a human standard curve generated from the same plate using the blank subtracted OD450 values for each standard (on the y axis) and the assigned concentration for each standard (on the x axis). Results from up to three sequential dilutions were averaged for each sample if the blank subtracted OD450 values were within the linear range of the standard curve. Samples with OD450 values below the linear range of the standard curve at all dilutions tested were reported as lower than the limit of quantification and assigned a value of 1 U/mL for descriptive statistics and tests.
A level of ≥50 U/mL was established as an exclusion criterion in previous clinical trials (Re-administration of Intramuscular AAV9 in Patients With Late-Onset Pompe Disease, AAV9-GAA_IM, NCT02240407; 15 Canavan-Single Patient IND, NCT05317780 16 ). A summary of the methods is presented in Figure 1.
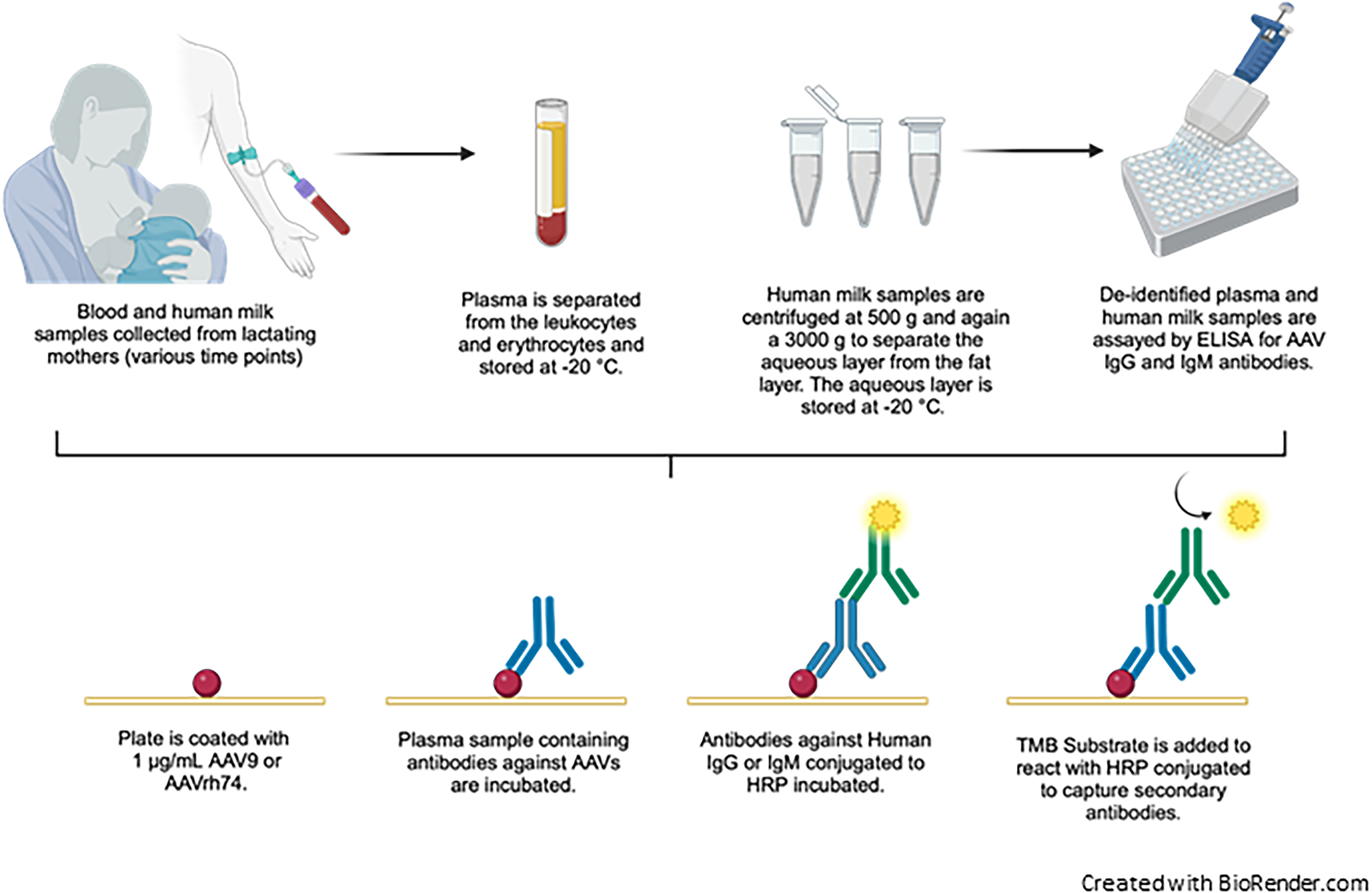
Analysis of anti-AAV9 and anti-AAVrh74 IgG and IgM titers via an enzyme linked immunosorbent assay (ELISA).
Statistical analyses
Descriptive statistics using median and ranges were included for continuous variables. Categorical data is presented as number and percentages. Wilcoxon matched-pairs signed rank test was used to compare matched plasma and human milk samples. Spearman's correlation was obtained between anti-AAV9 and anti-AAVrh74 levels. All statistical tests were two tailed. A p-value of <0.05 was considered statistically significant. Analyses and figures were performed using GraphPad Prism 9 (Graph Pad Software, San Diego, CA).
Results
Thirty-one lactating female participants met study criteria. Median age at recruitment was 34 years (range: 25–43). The majority of participants were non-Hispanic White. Median duration of breastfeeding was 7.5 months (range 0.7–33). Table 1 includes baseline characteristics of the participants.
Participant characteristics (N = 31).
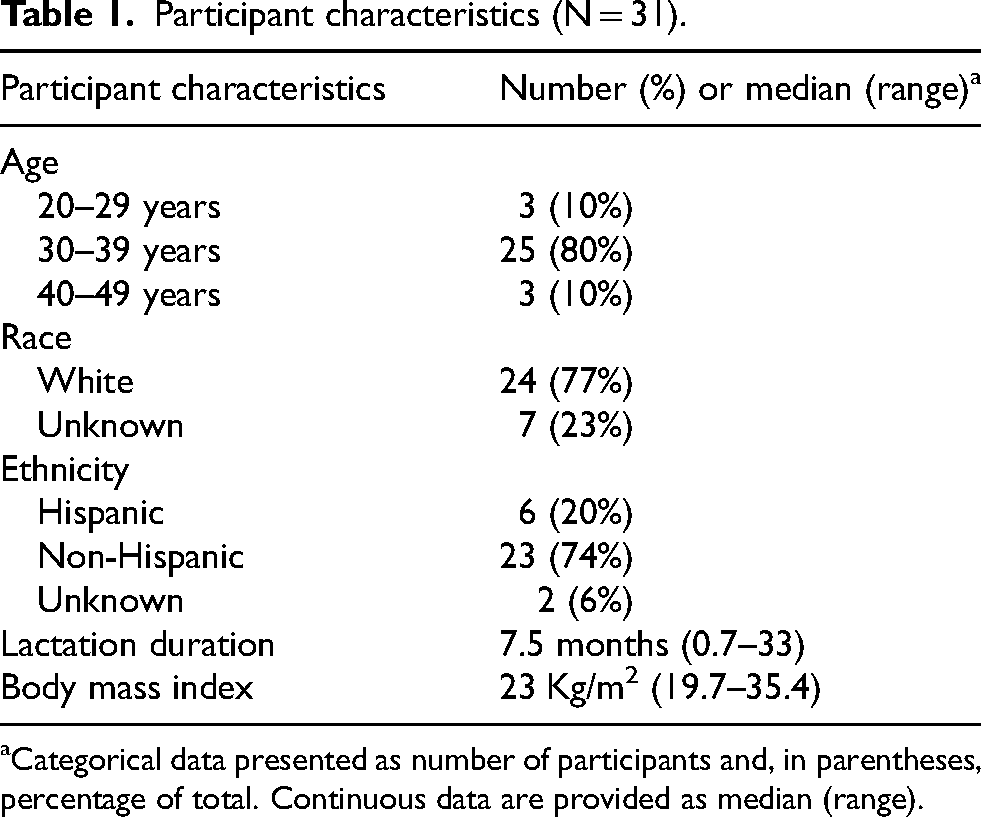
Categorical data presented as number of participants and, in parentheses, percentage of total. Continuous data are provided as median (range).
Anti-AAV9 IgG and IgM levels in plasma and human milk
Median (range) anti-AAV9 IgG in plasma and human milk were 183 U/mL (29–7214) and 1 U/mL (1–33), respectively. Anti-AAV9 IgG levels in plasma and human milk were compared using Wilcoxon signed rank test and were statistically different in all 31 pairs (p ≤ 0.0001) with an average decrease of 97.08% in human milk when compared to plasma levels
Median (range) anti-AAV9 IgM in plasma and human milk were 16 U/mL (1–396) and 1 U/mL in all subjects, respectively with an average decrease of 93.66% between plasma and human milk. Paired samples were compared using Wilcoxon signed rank test and demonstrated a statistically significant difference between the groups (p ≤ 0.0001). Figure 2 depicts the distribution of anti AAV9 IgG and IgM levels in plasma and human milk.
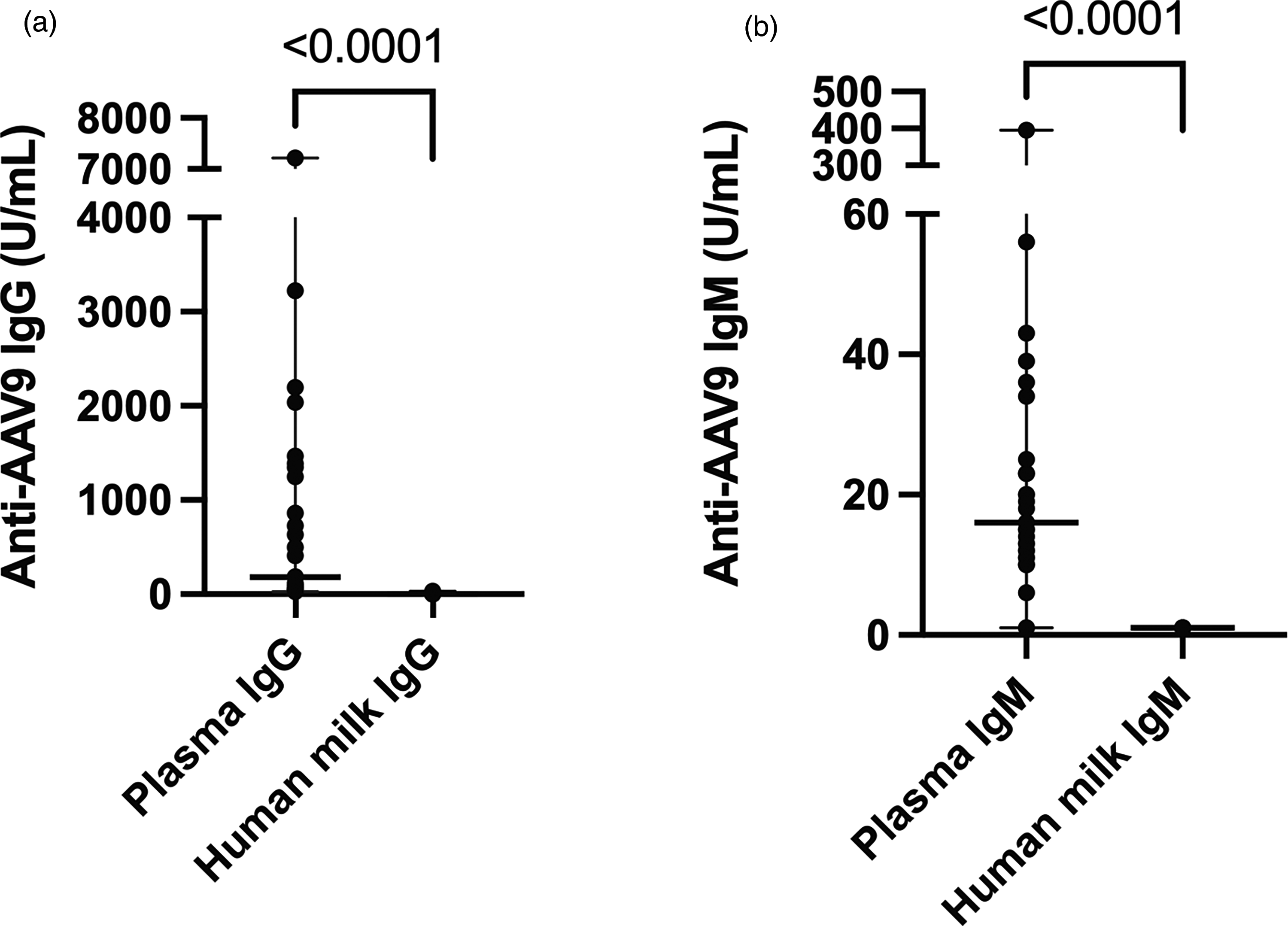
Anti-AAV9 antibody levels in paired plasma and human milk samples.
Anti AAVrh74 IgG and IgM levels in plasma and human milk
Median (range) anti-AAVrh74 IgG in plasma and human milk were 138 U/mL (23–8725) and 1 U/mL (1–27), respectively. Similarly, there was a statistically significant difference between plasma and human milk titers (p < 0.0001).
Median (range) anti-AAVrh74 IgM in plasma was 14 U/mL (1–65) and human milk Anti-AAVrh74 IgM was 1 U/mL for all subjects. There was a statistically significant difference between plasma and human milk titers (p < 0.0001).
Figure 3 includes Anti-AAVrh74 IgG and IgM levels in plasma and human milk of all subjects.
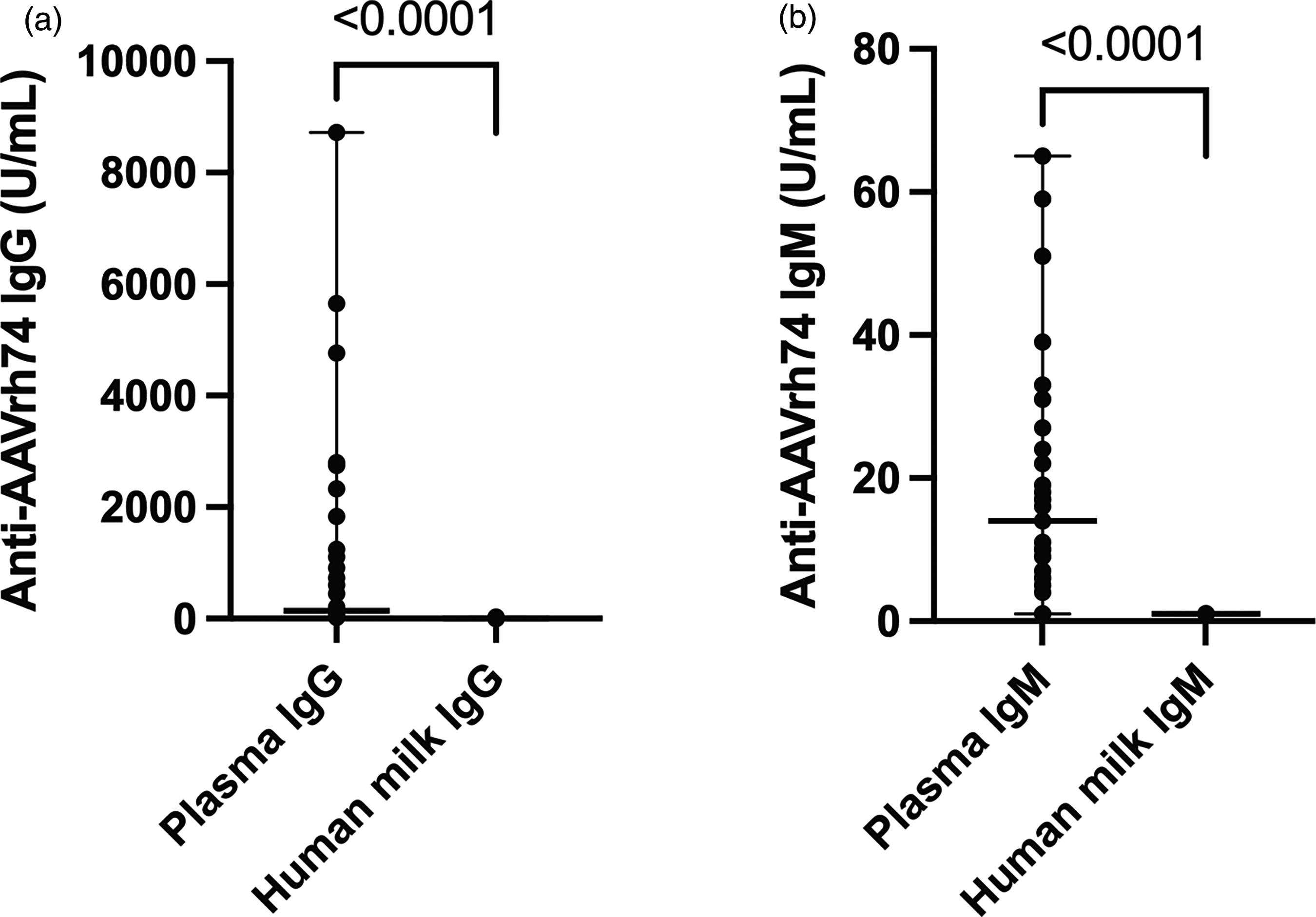
Anti-AAVrh74 antibody levels in paired plasma and human milk samples.
Correlation between Anti AAV9 and Anti AAVrh74 antibodies
Spearman correlation was performed between paired anti-AAV9 IgG and anti-AAVrh74 IgG levels in the study cohort and demonstrated strong correlation (p ≤ 0.0001, r = 0.9701). Similar results were found between anti-AAV9 IgM and anti-AAVrh74 IgM levels (p ≤ 0.0001, r = 0.6815).
Figure 4 shows the correlation between Anti-AAV9 and Anti-AAVrh74 antibody levels. There was no correlation between age and anti-AAV9 IgG levels, and between age and anti-AAVrh74 IgG levels in our cohort (p = 0.9053, r = 0.0223; p = 0.8367, r = 0.0386 respectively).
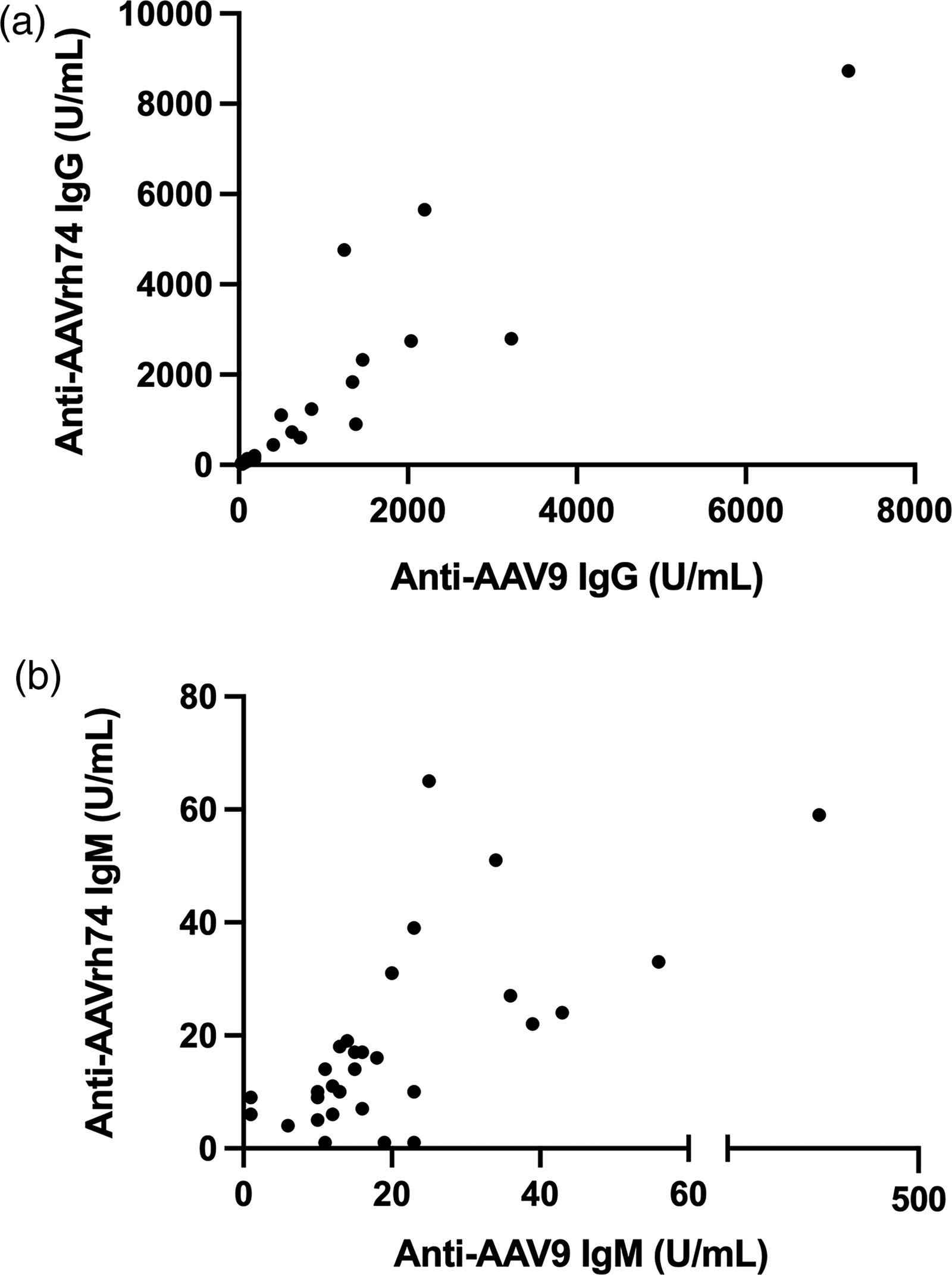
Correlation between anti-AAV9 IgG and anti-AAVrh74 antibody levels.
Discussion
Our study provides crucial insights into the mother-infant transmission of anti-AAV9 and anti-AAVrh74 antibodies through breastfeeding. The data suggest that such transmission is unlikely, as anti-AAV9 and anti-AAVrh74 IgG and IgM against AAV do not reach significant levels in human milk following environmental exposure. Consequently, the likelihood of seroconversion in breastfed infants due solely to breastfeeding is minimal. Further, the limited transfer of IgG from human milk into neonatal or infant circulation further supports this conclusion. 13 In primates, including humans, maternal IgG is primarily transferred during fetal life via the placenta. After birth, the infant's intestinal system undergoes developmental changes that restrict macromolecular transfer. Notably, secretory IgA becomes the predominant immunoglobulin in human milk.13,17–19
The absorption of IgG in the human intestine requires the expression of the neonatal Fc receptor (FcRn), which mediates the transepithelial transfer of IgG. 20 However, this process is largely lost during the process of development and maturation of the intestine until macromolecular transepithelial transfer undergoes “gut closure”, thereby significantly limiting antibody transfer from human milk. 18 Gut closure typically happens soon after birth, with a slight delay in premature infants. 21 Therefore, due to the limited ability of antibodies to reach circulation, their role is to primarily provide barrier immunity, rather than systemic immunity. 22
In our study, a cutoff of <50 U/mL was used, as it had been previously established as an inclusion criterion in clinical trials (NCT02240407, NCT05317780). In our cohort, 24 and 26 participants had Anti-AAV9 IgG and Anti-AAVrh74 IgG above this threshold, respectively; however, none of these participants exhibited antibody levels exceeding this threshold in human milk. The combined analysis of anti-AAV9 and anti-AAVrh74 IgM and IgG levels provided additional insights into past AAV infections within the cohort. While the IgM levels are not routinely assessed as part of the gene therapy candidacy requirements, their inclusion here helped identity recent AAV exposure in one participant, who exhibited elevated IgM and IgG levels in plasma. Despite this, antibody levels in the participant's human milk remained below the established cutoff of <50 U/mL (Figures 2 and 3).
These findings, coupled with the limited absorption of immunoglobulins in the intestinal wall, support the recommendation that breastfeeding should not be restricted for infants who are potential candidates for AAV mediated gene therapy. While at this time, the results of this study may only impact the care of infants with spinal muscular atrophy, we expect the future approval of AAV products for neonates with other conditions, particularly as newborn screening programs expand.23–25 Breastfeeding offers multiple benefits to infants, in addition to its nutritional value, including the provision of crucial anti-inflammatory cytokines that participate in the regulation and development of the immune system, as well as a protective effect against some chronic conditions, among others. 26
Importantly, the correlation between anti-AAV9 and anti-AAVrh74 antibodies demonstrates that a significant number of individuals may not be candidates for AAV mediated gene therapy even with the use of newer and better AAV products, unless novel immune modulation protocols are designed for safe AAV administration in seropositive patients.
This study is limited by the small sample size and the limited diversity in this cohort of lactating mothers. Variability in human milk composition has been attributed to factors such as genetic variation, maternal adiposity, maternal and infant infections and pathologies, prenatal smoking, and nutrition, among others.17,27–29 Further, the samples analyzed represent a single point assessment of a dynamic immune response as the total immunoglobulin concentration and profile changes during the course of lactation. The concentration of immunoglobulins in human milk changes significantly over time, with much higher levels in colostrum when compared to transition milk or human milk obtained after the first few weeks of breastfeeding, which is characterized by lower protein and immunoglobulin content. 17 Therefore, it is important to note that many of the samples in this study were obtained after the neonatal period given recruitment challenges, which could impact the anti-AAV antibody levels in human milk in this cohort. However, given the limited capacity of the intestinal mucosa to absorb IgG, and the significantly lower levels of IgG in human milk compared to plasma, we conclude that the risk for exclusively breastfed neonates to absorb important quantities of anti AAV antibodies to a degree that would have an impact in their gene therapy candidacy is negligible. As previously noted, gut closure can be delayed in premature patients, and therefore, the results of this study should be applied with caution to that population.
While thus far gene therapy candidacy relies on total or binding anti-AAV antibody levels, there is important variation in assay utilization to identify these and neutralizing antibodies. 30 The results obtained with each gene therapy product's companion diagnostic assay or at other institutions may vary.
Future studies should focus on the characterization of transplacental transfer of anti-AAV immunoglobulins, given the importance of passive immunization through this route in both human and non-human primates. 17 Such studies would contribute significantly to our understanding of maternal-fetal antibody transfer and its implications for AAV based gene therapy. Understanding the complexities of this transfer could provide crucial information for optimizing treatment strategies and ensuring the safety and efficacy of gene therapies in neonates and infants.
Footnotes
Acknowledgements
We would like to thank the study participants. We would also like to thank Megan Wichman for editing and reviewing the manuscript.
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Gerber Foundation and the Children's Miracle Network (VVL, LSS, JLIII, JN), U01-NS116752-01A1 (MC, BJB), University of Florida, Powell Gene Therapy Center (CLA, KC, SMS, MC, BJB). Funding used for data processing methods and supplies. There is no role of the funding source for writing the article or the decision to submit for publication.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study can be provided upon reasonable request.
