Abstract
Background:
Spinal muscular atrophy (SMA) is a rare, progressive neuromuscular disease associated with a significant burden of illness to both patients and caregivers; however, there is little evidence available regarding how patients and caregivers evaluate potential treatment benefit–risk profiles. As access to SMA disease-modifying therapies increases, it is imperative to understand which treatment attributes drive treatment choices.
Objective:
To identify which treatment attributes drive treatment choices in adults with SMA and caregivers of children with SMA across nine countries in Latin America.
Methods:
A discrete choice experiment (DCE) survey was developed for market research using data collected via qualitative interviews and consultations with medical experts. Adults with Type 2/3 SMA and caregivers of children with Types 1–3 SMA were recruited by patient advisory groups and physician referrals. Respondents completed a 30-min, online survey that collected patient demographics, disease-specific information, and quality of life data (via the EQ-5D-5L), and included the DCE, in which respondents were asked to choose between 14 sets of hypothetical treatment profiles. Data were pooled for analysis, as the country-level sample sizes were small. Raw data were aggregated in Microsoft Excel. Statistical testing was performed using data tables and SPSS (as appropriate). Demographic data were summarized descriptively.
Results:
A total of 143 respondents (45 adults with SMA and 98 caregivers) completed the online survey. Most respondents were from Argentina (35.0%) or Brazil (19.6%). Breathing function outcome was the most important treatment attribute for caregivers, while adults with SMA placed greater importance on motor function outcome. Both adults with SMA and caregivers placed the greatest importance on improvements in function compared with worse or stable outcomes.
Conclusions:
Understanding treatment attribute preferences at a regional level will improve shared medical decision-making for individuals with SMA.
Introduction
Spinal muscular atrophy (SMA) is a rare, progressive neuromuscular disease caused by reduced levels of the survival of motor neuron protein.1,2 SMA is associated with a significant burden of illness to both patients and caregivers, including higher healthcare utilization, decreased quality of life (QoL), and increased financial costs.3–6 Despite this, there is little understanding of how both patients and caregivers evaluate the benefit–risk profiles of potential treatments, 7 particularly at regional levels. Three disease-modifying therapies (DMTs) have been developed to treat the cause of SMA, and, as new therapies are being developed,8,9 it will be vital to further understand patient and caregiver perceptions of treatment attribute preferences.
Nusinersen, an intrathecally administered antisense oligonucleotide, was approved by the Food and Drug Administration (FDA) and the European Medicines Agency (EMA) in 2016 and 2017, respectively. 10 Onasemnogene abeparvovec is an intravenously administered gene therapy, approved by the FDA in 2019 and the EMA in 2020. 10 Risdiplam, an orally administered, small-molecule splicing modifier, was approved by the FDA in 2020 and the EMA in 2021. 10 As access to SMA DMTs broadens globally, approvals and regulations will vary across countries/regions.
Patient preference studies are increasingly being used to elicit benefit–risk assessments of treatments and the risk tolerance of patients, particularly in rare diseases. Knowledge of patient preferences informs product development and evidence-generation planning, supports regulatory and reimbursement submissions, and aims to increase patient centricity in healthcare and improve shared decision-making.11,12 Multiple regional-based patient preference studies have been conducted in patients with SMA and their caregivers, including in the UK, 13 the USA,7,14 Australia, 15 and Europe; 16 however, SMA treatment attribute preference studies in other regions of the world are lacking, including in Latin America.
It is important to conduct patient preference studies at a regional level to compare how treatment attributes are viewed and to determine how treatment choices are made, as results from one country/region may not be directly translatable to another. Patient preferences may vary between countries/regions due to population demographics, population size, cultural and social factors, and healthcare access. Indeed, access to healthcare is generally more challenging in Latin America than in the USA or Europe, with many individuals deferring medical care due to difficulties accessing and affording treatment. 17 Many individuals in Latin America with neuromuscular disorders remain undiagnosed due to a lack of access to specialized medical centers, 18 and families living in rural areas frequently need to relocate to large urban centers to access care for their relatives once they are diagnosed. 19 Additionally, out-of-pocket healthcare costs in Latin America and the Caribbean are much greater than in the European Union and USA (24% of total healthcare costs vs. 14% and 10%, respectively), 17 which can influence treatment uptake and preferred routes of administration. Cultural differences may also play a role in determining preferred routes of treatment administration. 20
To address the gap in knowledge about patient treatment preferences in Latin America, we conducted a market research study to identify key attributes that influence treatment choice in SMA and to determine their importance in driving the choices made by patients and caregivers across Argentina, Brazil, Mexico, Chile, Costa Rica, the Dominican Republic, Panama, Paraguay, and Peru.
Materials and methods
Study design
To collect information about patient preferences, an online survey was developed for market research, which included a discrete choice experiment (DCE) to understand preferences for SMA treatment attributes. 21
DCEs represent a widely accepted methodology to identify and evaluate patient preferences.22,23 A DCE uses hypothetical scenarios and asks respondents to choose between two options. In the current study, the DCE was used to validate the importance of attributes in SMA treatment choices/preferences. A DCE mimics a respondent's real-world trade-offs when making decisions and allows the comparison of relative importance of various treatment attributes.
Survey development
Part 1: qualitative survey
To develop the market research survey, it was first important to identify the key attributes that drive SMA treatment choices in patients and caregivers in Latin America. To do this, twelve 45-min, web-assisted, recorded, qualitative telephone interviews were conducted with six patients and six caregivers across Argentina, Brazil, and Chile (n = 4 respondents per country). Interviews were conducted by a moderator in the local language using a discussion guide. Interviews collected data on SMA diagnosis, key symptoms, perception of SMA (using word and image association), QoL, treatment experience, unmet needs, and treatment attribute preferences.
Participants also submitted a self-completed form designed to capture attribute importance. This was achieved using a 100-point allocation exercise, in which participants were asked to distribute 100 points between attributes to prioritize them: attributes allocated more points had greater importance. This method produces interval-level data that can be averaged and quantified.
A copy of the discussion guide used in the qualitative interviews can be found in the supplemental materials (online; Supplementary Table 1).
Part 2: development of the DCE experiment
Results from the qualitative survey (see supplemental materials [online]) were used by the research team to develop a draft list of attributes for use in the DCE in the main market research survey.
Three expert healthcare providers (HCPs) from reference centers in Chile, Argentina, and Brazil, as well as one international expert HCP, were consulted to validate the potential treatment attributes identified in the qualitative survey. These HCPs were selected for their extensive experience in treating patients with SMA and in clinical research and publications. Feedback was obtained from a virtual advisory board conducted in December 2020, in which the HCPs were asked to provide feedback on the clinical relevance of the draft attributes and the clinical accuracy of the chosen range of attribute levels.
DCE survey questions were described using five attributes: motor function, breathing function, mode of treatment administration, treatment reactions, activities of daily living, and fatigue (adults only). Each attribute was categorized by three different levels (Table 1).
Motor function: The primary outcome tested in clinical trials to determine treatment efficacy. Attribute levels of worsening (worse by one level), stable, and improvement (better by one level) were determined using a motor function scale based on motor milestones relevant to functional abilities in SMA (sitting, standing, and walking; see Section 2 of the questionnaire in the supplemental materials [online] for further details). These levels were chosen as stabilization of, or improvement, in motor function is a relevant treatment outcome for people with SMA.27,28 Breathing: Respiratory function is a key treatment target for people with SMA.27,28 Breathing was categorized as either worse, stable, or better. Mode of administration: Administration methods were aligned with the DMTs approved/soon to be approved for the treatment of SMA in Latin America. Activities of daily living: Maintenance of independence is an important treatment outcome for patients with SMA.27,28 Attribute levels for activities of daily living were selected based on the ACTIVLIM
24
and Assessment of Caregiver Experience in Neuromuscular Disease (ACEND)
25
scales. Levels were categorized as a change in the ability to carry out daily activities: becoming more difficult to do, staying the same, and becoming easier to do. Fatigue: Reported as a clinical feature of SMA by patients and via functional and electrophysiological tests. Fatigue attribute levels were selected based on the Fatigue Severity Scale
26
and categorized as getting worse, staying the same, or getting better. Fatigue has a significant impact upon the everyday life of patients.
29
Discrete choice experiment treatment attributes and levels.
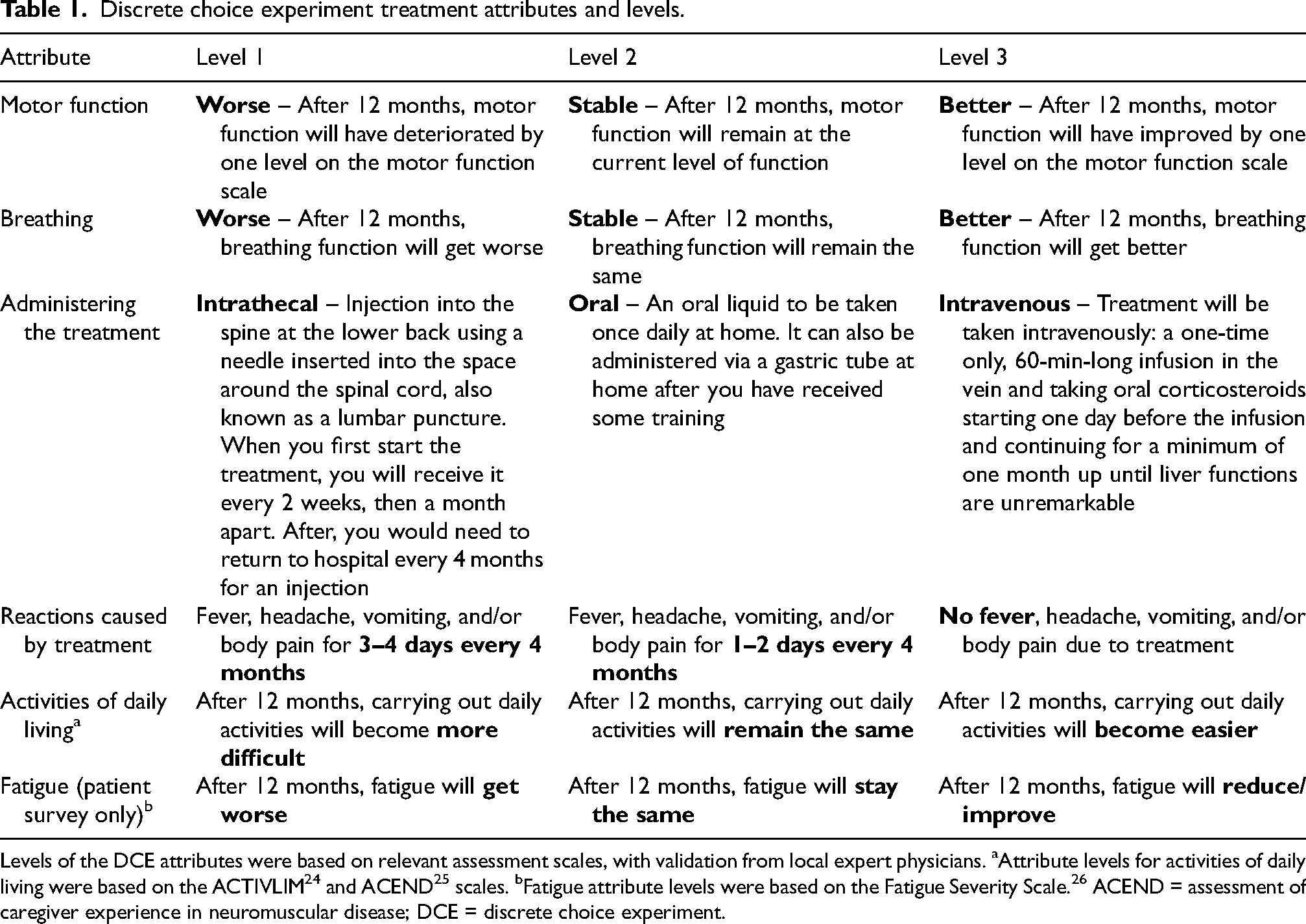
Levels of the DCE attributes were based on relevant assessment scales, with validation from local expert physicians. aAttribute levels for activities of daily living were based on the ACTIVLIM 24 and ACEND 25 scales. bFatigue attribute levels were based on the Fatigue Severity Scale. 26 ACEND = assessment of caregiver experience in neuromuscular disease; DCE = discrete choice experiment.
The choices in the hypothetical set were based on an experimental design that was derived through Sawtooth Software (Provo, Utah, USA), based on the number of attributes/levels in the DCE, factoring in the sample size. The experimental design aimed for orthogonality, so that each level for each attribute appeared an equal number of times. In the random design of a DCE, each level should appear roughly an even number of times in the left option. After each task, respondents were asked to indicate whether they would use the selected treatment to manage their/their child's SMA. Complete enumeration was used to produce the design for the DCE. Standard error was checked when constructing the design for each attribute level (this was <0.05, as recommended by Sawtooth). 30
Online survey and DCE
The main survey aimed to validate the importance of treatment attribute preferences that were initially identified in the qualitative interviews. Respondents completed a 30-min, online survey composed of three parts: a questionnaire to collect patient demographics and clinical data, the DCE to collect and understand preferences for SMA treatment attributes, and the EuroQol 5-dimension, 5-level (EQ-5D-5L) questionnaire 21 to collect data about the impact of SMA on QoL. Two versions of the online survey were developed: one for patients and one for caregivers (see supplemental materials [online] for the surveys).
Survey questions were designed to understand preferences for SMA treatment attributes relating to the currently available or soon-to-be-approved SMA treatment options (nusinersen, onasemnogene abeparvovec, and risdiplam). Surveys were conducted in the local language across Argentina, Brazil, Chile, Costa Rica, Peru, Panama, Paraguay, Mexico, and the Dominican Republic. Questionnaires were developed in English and professionally translated into Spanish and Portuguese, to ensure consistency of the content across countries.
The first part of the online survey was a questionnaire that collected demographic data, including country of residence, age, and disease-specific information such as SMA type, symptoms, diagnosis timeframe, and treatments received (see the surveys in the supplemental materials [online] for the full list of questions).
In the DCE component of the survey, respondents were presented with a series of hypothetical treatment choices, each with two options (‘Treatment A’ or ‘Treatment B’). Respondents were asked to decide which hypothetical treatment they would prefer for themselves (patients with SMA) or for their child (caregivers). An example DCE question is shown in Figure 1. There was a total of 14 set choices, which were randomly distributed to respondents.
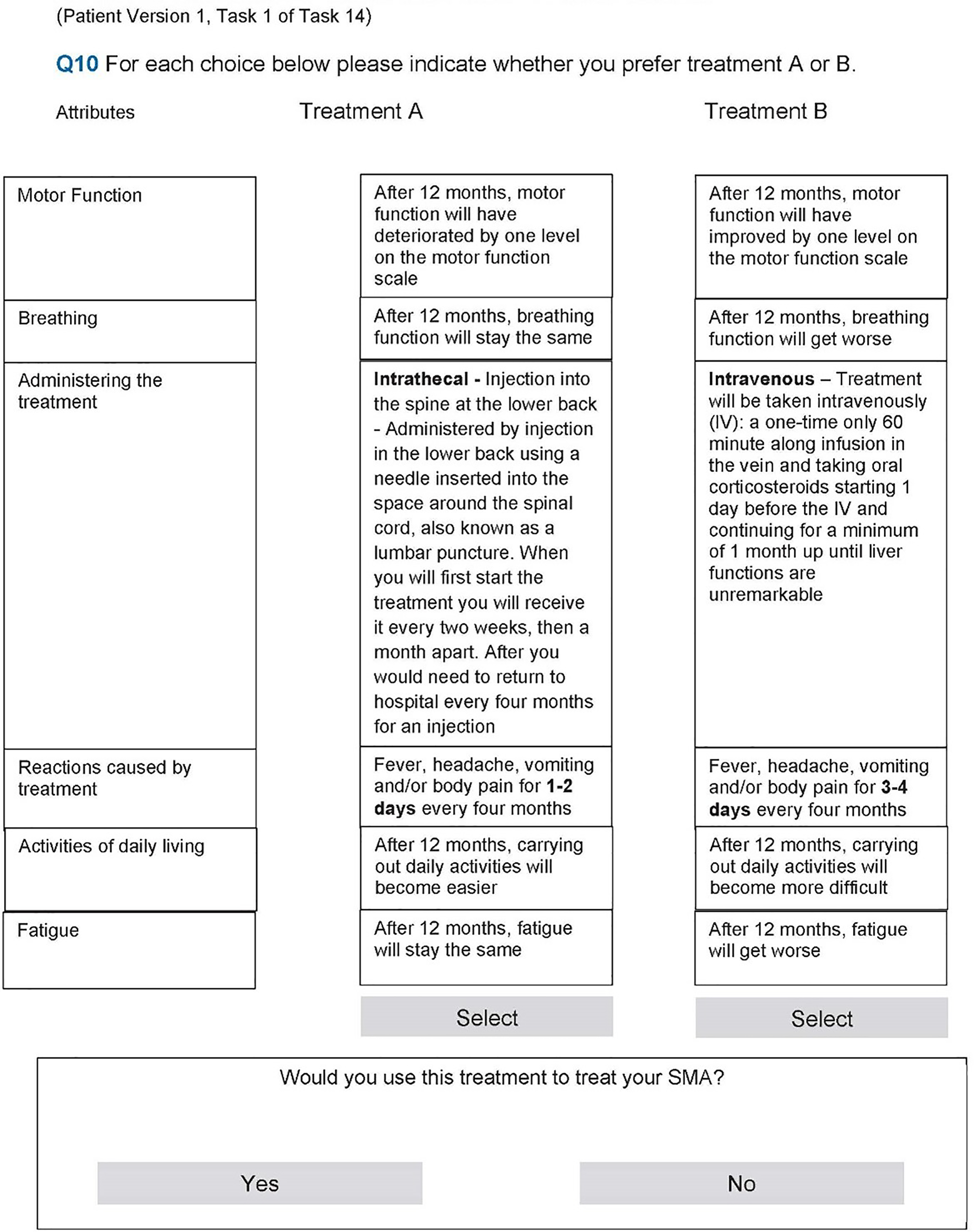
Example discrete choice experiment question. SMA = spinal muscular atrophy.
Patient QoL was also assessed in both patients with SMA and caregivers via the EQ-5D-5L questionnaire. Country-specific, validated versions of the EQ-5D-5L were used for each country (see the surveys in the supplemental materials [online] for further details). The EQ-5D-5L is a standardized measure of health to facilitate the calculation of quality-adjusted life years, consisting of five dimensions (mobility, self-care, usual activities, pain and discomfort, and anxiety and depression), each of which has five severity levels (no problems, slight problems, moderate problems, severe problems, and extreme problems) that are described by statements appropriate to each dimension. 21 A patient is asked to indicate their health state by ticking the box next to the most appropriate statement for each of the five dimensions. The results for the five dimensions can be combined into a five-digit number that describes the patient's overall health state. 31
Study population and recruitment
For both the qualitative interviews and the online survey, eligible participants included adults with SMA ≥18 years of age with Types 2 and 3 SMA and caregivers (≥18 years of age) of children <18 years of age with Types 1–3 SMA. People with Type 4 SMA were excluded, due to the significant difference between the natural disease history of Type 4 SMA compared with Types 1–3 SMA 2 and because, at the time of this market research, the DMTs investigated were typically not indicated for the treatment of Type 4 SMA. People who had been treated with nusinersen or onasemnogene abeparvovec were eligible to participate in the study; people who had received risdiplam were excluded, due to the regulatory heterogeneity concerning risdiplam across different Latin American regions. The approval status of nusinersen, onasemnogene abeparvovec, and risdiplam in participating countries can be found in the supplemental material (online; Supplementary Table 2).
Recruitment was facilitated by local patient advisory groups (PAGs) and physician referrals from specialists involved in treating SMA (neurologists, respiratory physicians, and orthopedic surgeons). A link was shared with the PAG/physician who distributed it directly to the people with SMA and their caregivers – participation was voluntary. No renumeration was given to the PAGs or physicians for facilitation of recruitment.
Recruitment and fieldwork for the qualitative survey were conducted from November 2020 to December 2020 in Argentina, Brazil, and Chile. A sample size of four respondents per country was chosen to cover both patients and caregivers and was based on the feasibility of recruiting respondents with a rare genetic condition who met the inclusion criteria.
The main online survey was conducted from August 2021 to March 2023, in nine countries (Argentina, Brazil, Mexico, Chile, Costa Rica, the Dominican Republic, Peru, Panama, and Paraguay). A total sample of 230–250 respondents across all countries was intended. This sample size was estimated to give a margin of error of <10%, based on a 95% confidence interval. Due to the rare nature and low incidence rate of SMA, final sample sizes were flexible.
All recruitment and research were conducted in accordance with the European Pharmaceutical and Market Research Association (EphMRA) code of conduct. Formal, written consent was obtained from all respondents using an informed consent form before they participated in the study. No remuneration was provided for completing the online surveys. Individuals who participated in the qualitative interviews were compensated for their time. The value of the incentive received was determined based on the market value and specifications of the project.
Data analysis
Qualitative interviews
Recordings of the qualitative interviews were analyzed by an analyst who spoke the local language. Relevant information was extracted and summarized descriptively, and key insights identified the key attributes driving SMA treatment choice. Analysis focused on answering the key market research objectives of the qualitative phase:
Understanding SMA disease burden and impact on QoL Treatment unmet needs How is treatment choice made? Spontaneous and probed identification of key attributes (e.g., motor function vs. breathing, oral vs. intrathecal)? What trade-offs are patients and caregivers willing to make? Relative importance of attributes.
These results were translated into English for use in developing a report. The report was then used to develop the DCE and for the validation of the attributes and levels by the expert HCPs.
Online quantitative survey and DCE
Pseudonymized data were pooled for analysis, as the country-level sample sizes were small. Raw data were aggregated in Microsoft Excel, and statistical testing was performed using data tables and SPSS (as appropriate). Demographic data were analyzed and summarized descriptively. While the sample size was not sufficient to run specific subgroup analyses or to test differences between groups, the demographic data were used descriptively to report results by SMA type.
For all questions (as appropriate), the following principles were applied:
numerical and percentage values were calculated to one decimal place (e.g., 25.1%); ‘don’t know’ or ‘not applicable’ responses were excluded, and the base revised to include only those who answered the question; all percentage/numerical questions were calculated in bands of 10 (e.g., 0%, 1–10%, 11–20%, etc.), unless otherwise specified.
In the DCE, specific attributes related to treatment preferences other than mode of administration (periodic intrathecal [IT], daily oral, and one-time intravenous [IV]) were not included. Hierarchical bayes was used to estimate the utilities for the outputs of the DCE. Preferences for treatment were assessed based on the utility scores for each level and were simulated together with the other attributes to see how preference share shifts with changes in an attribute. All other variables were kept constant, and sensitivity to changes in the levels was assessed. The relative importance of each attribute along with other attributes was also provided in the simulator. It was not possible to calculate statistical differences between adult patients with SMA and caregivers due to differences in the DCE grids shown to both groups.
Ethical review
Ethics committees reviewed and approved the study protocol in each participating country (Argentina, Brazil, Mexico, Chile, Costa Rica, the Dominican Republic, Peru, Panama, and Paraguay). The names of each committee and their respective numbers are included in the supplemental materials (online). All research was conducted in accordance with the European Union General Data Protection Regulation and EphMRA code of conduct.
Individuals provided informed consent before they participated in a qualitative interview.
Participation in the online survey was by choice. A link to access the survey was shared with participants via PAGs and HCPs.
In the online survey, participants were first asked to read a participant information sheet that described the objective, who is invited to participate, what happens if they take part, what types of information the study will be collecting, and how their information will be used and shared. Participants were then asked to indicate whether they consented to take part in the study before proceeding. Participants then completed a study screener and, if eligible, were directed to the main survey. Participants could stop the survey at any time. Copies of the online surveys, including the consent forms, can be found in the supplemental materials (online).
Data management
The Research Partnership (London, UK) was responsible for the data management of this research, on behalf of F. Hoffmann-La Roche Ltd, including quality checking of the data. The Research Partnership, in collaboration with a third-party fieldwork agency, Estudio Silvia Roca (Barcelona, Spain), produced the electronic questionnaire specifications for the research. Data were collected via an electronic data collection platform owned by Estudio Silvia Roca.
Data were pseudonymized before analysis. Individual respondent data were labeled with a unique respondent identification number to remove all personal identifying information. F. Hoffmann-La Roche Ltd and The Research Partnership were only granted access to the pseudonymized research data.
Data will be stored at The Research Partnership for at least 5 years after first publication, or as according to local regulations, if longer. Records and documents pertaining to the conduct of this research will be retained by F. Hoffmann-La Roche Ltd for at least 10 years after completion of the research, or as required by relevant national or local health authorities (whichever is longer).
Results
Demographics
In total, 572 respondents began the online survey. After removing ineligible entries, 143 respondents (45 adults with SMA and 98 caregivers) were included in the analyses (Figure 2). The greatest proportion of respondents was from Argentina (35.0%), Brazil (19.6%), Mexico (14.0%), Chile (13.3%), and Peru (10.5%; Table 2). Most caregivers reported caring for a child aged 2–6 years (42.9%). Adults with SMA were aged 18–53 years, with the majority aged 18–30 years (51.1%). Most adults with SMA and caregivers lived in urban or semi-urban areas (92.3%). Almost half of all patients in the study had Type 2 SMA (45.5%; Table 3). Most adults with SMA were treatment naïve (68.9%), with most of these adults reporting that they had not been assessed for treatment (58.1%). Over half of the children with SMA were treatment experienced (59.2%).
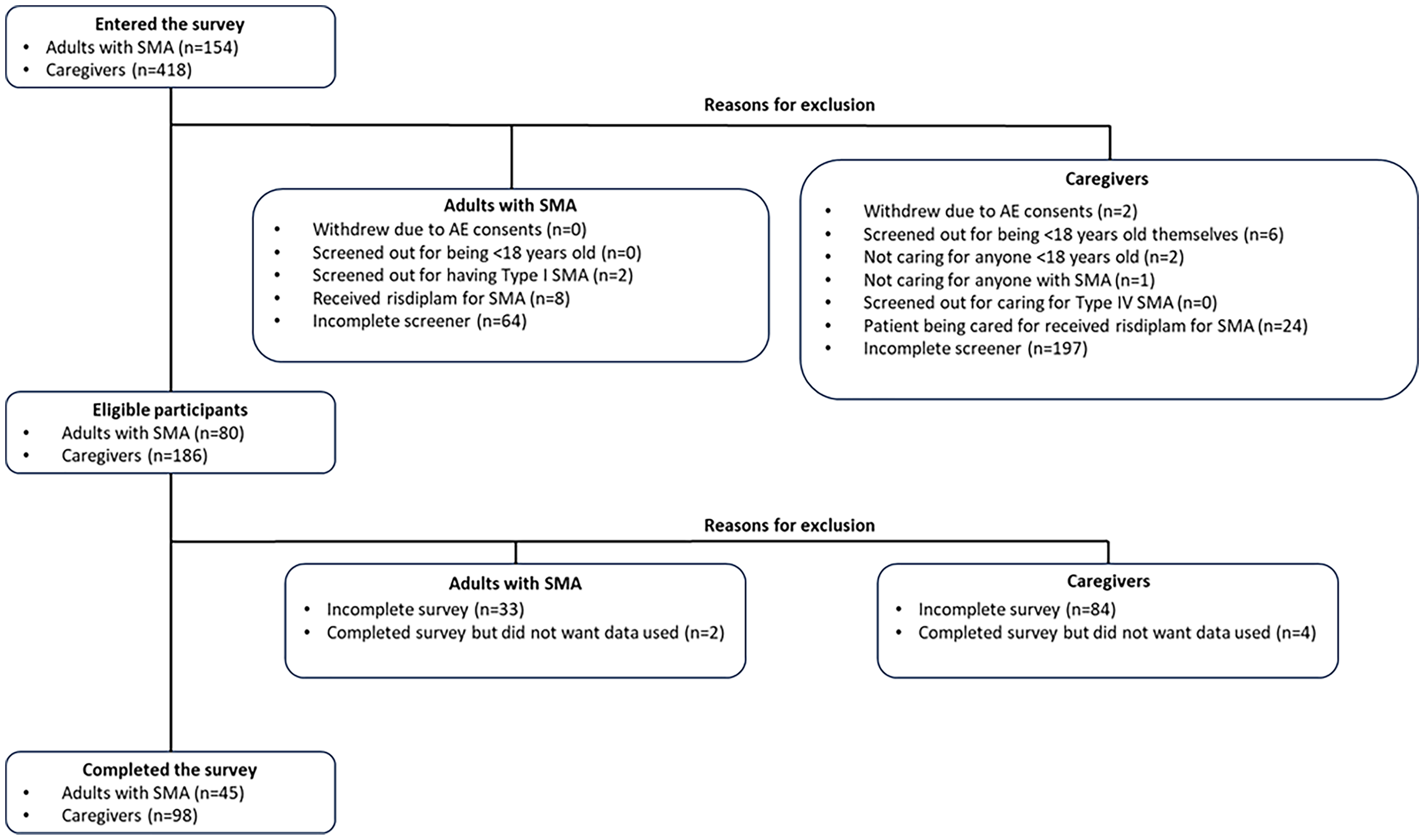
Patient attrition in the online survey.
Demographics of individuals with SMA as reported by survey respondents.
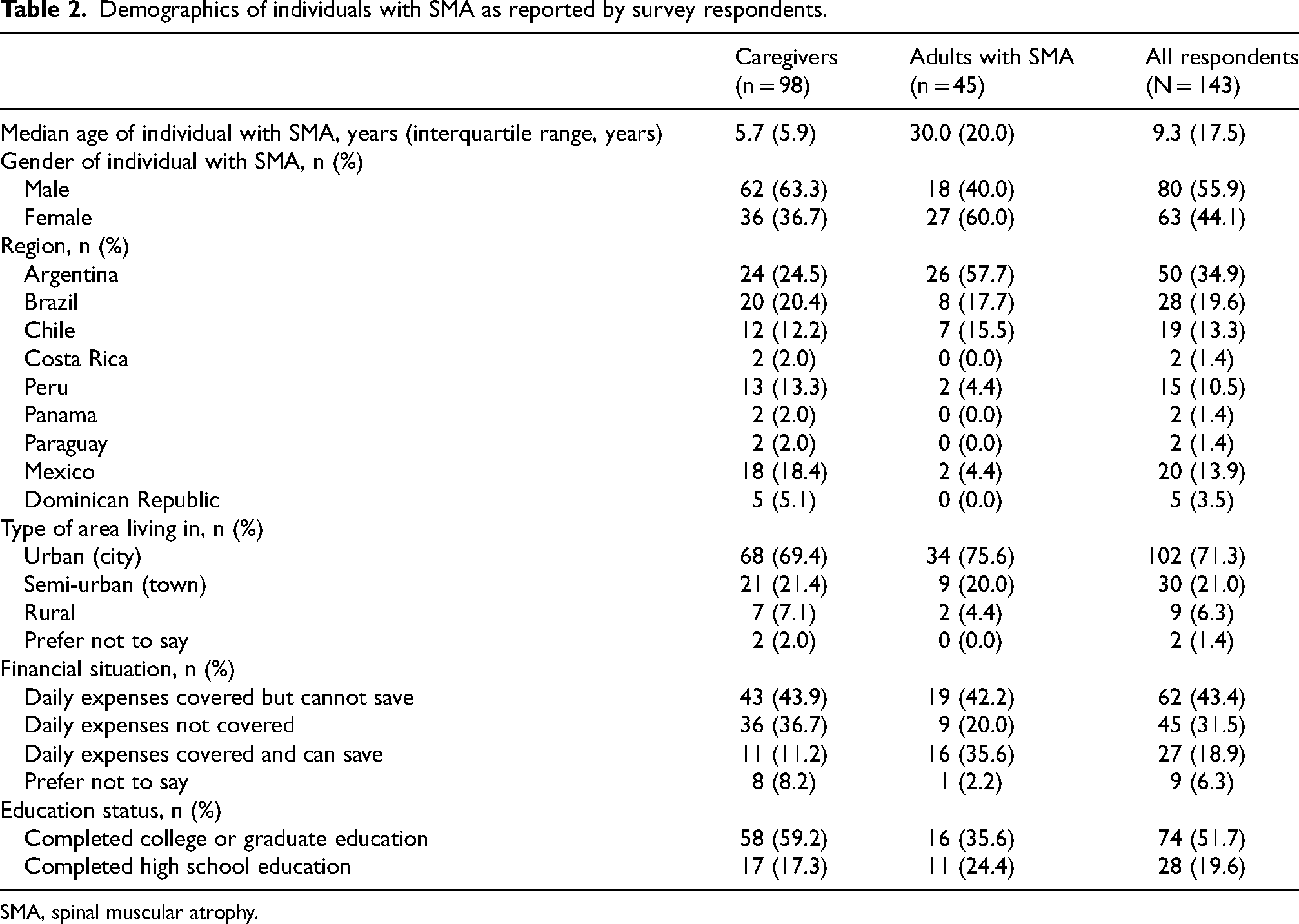
SMA, spinal muscular atrophy.
Clinical characteristics of individuals with SMA as reported by survey respondents.
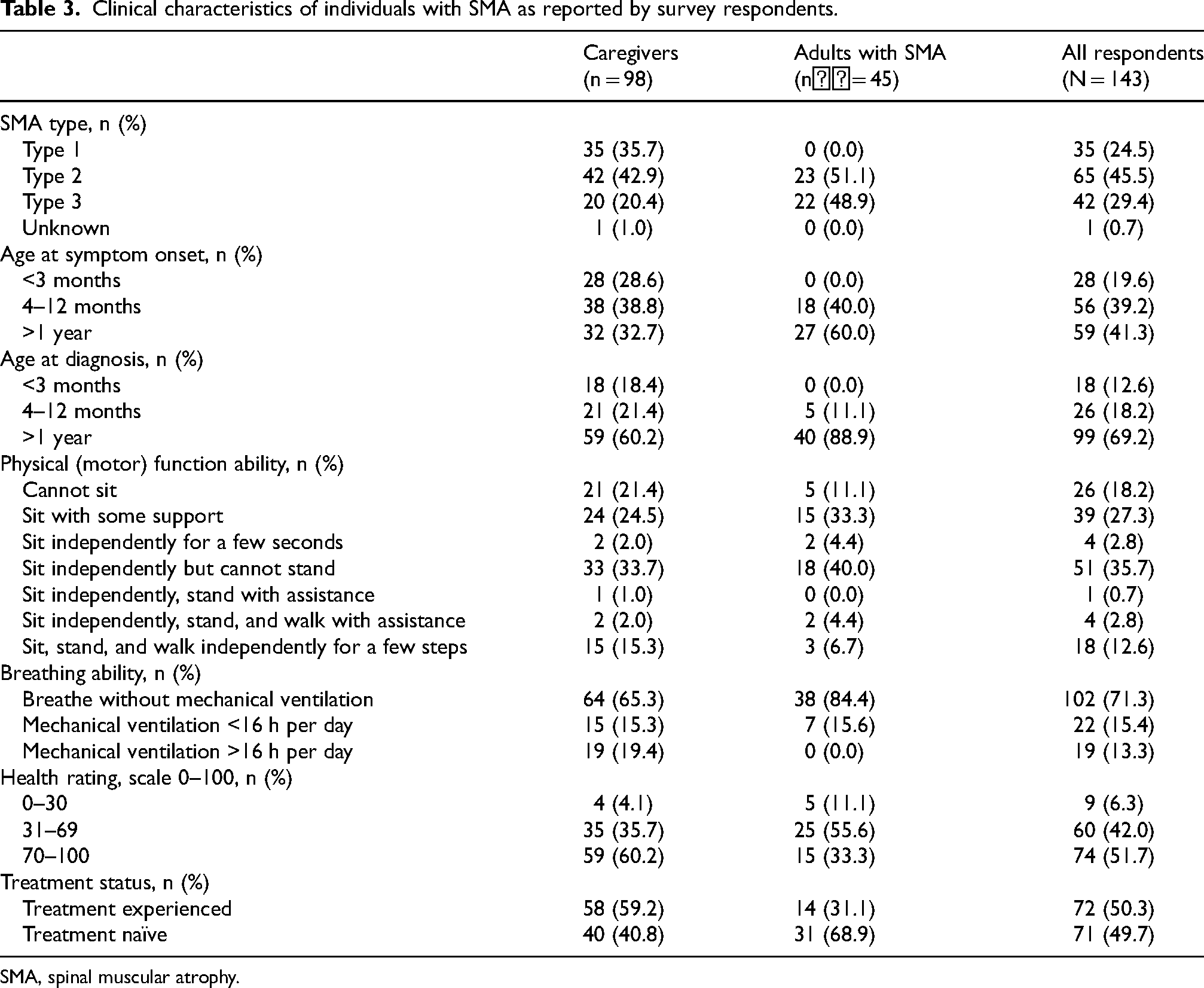
SMA, spinal muscular atrophy.
Treatment attribute preferences of adults with SMA and caregivers of children with SMA
Caregivers of children with Types 1–3 SMA reported that breathing outcome was the most important treatment attribute when considering treatment options (31% relative importance), while adults with Type 2/3 SMA considered motor function outcome the most important treatment attribute (24% relative importance) (Figure 3).
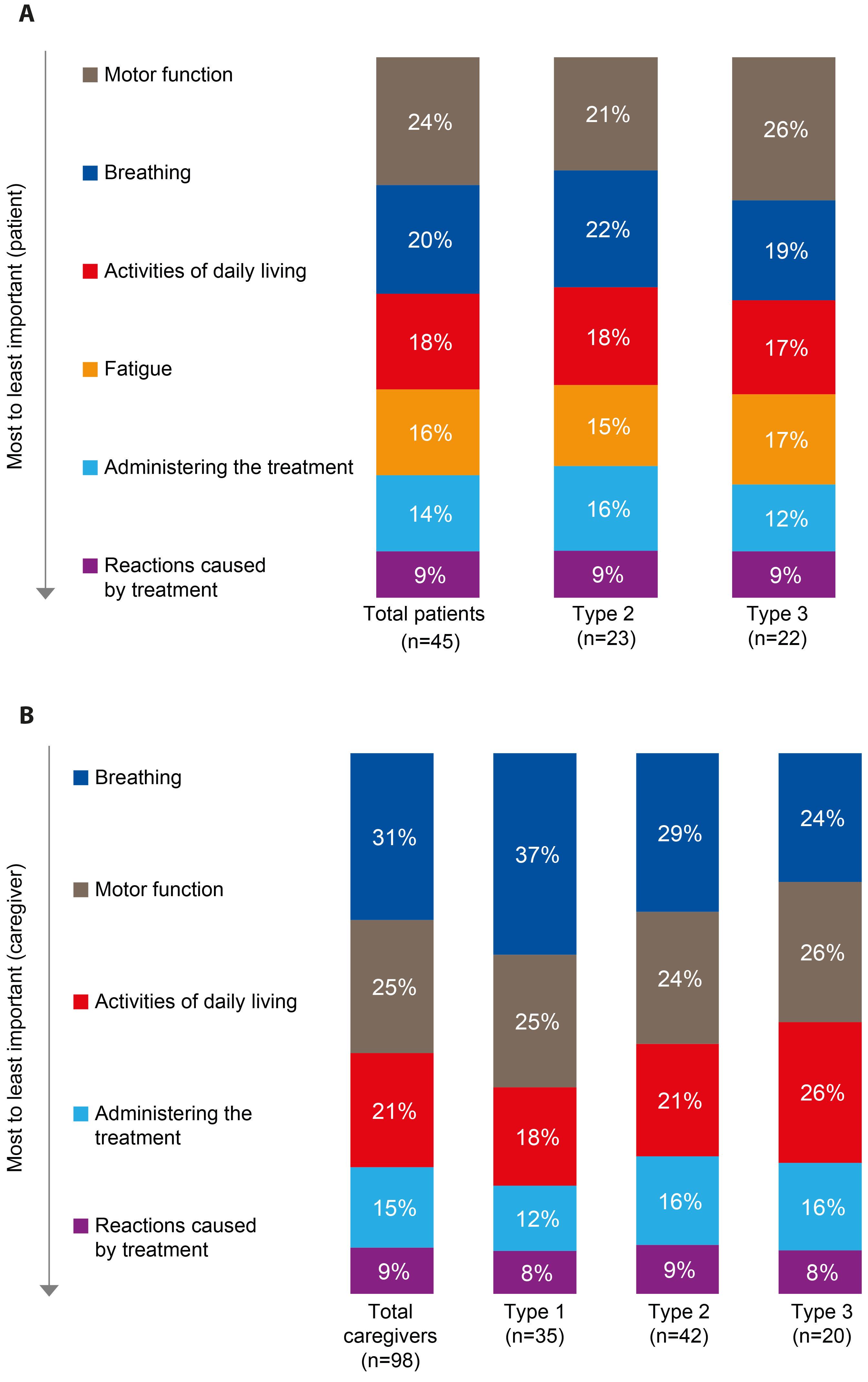
Relative importance of treatment attributes from the discrete choice exercise for adults with SMA (A) and caregivers of children with SMA (B).
Caregivers of children with Type 3 SMA reported motor function and activities of daily living as the most important treatment attributes (26% relative importance for each), whereas caregivers of children with Type 1 or 2 SMA reported that breathing outcome had the greatest relative importance (37% and 29% relative importance, respectively). Adults with Type 2 SMA reported breathing to be as important as motor function (22% and 21% relative importance, respectively), whereas adults with Type 3 SMA reported that motor function had the greatest relative importance (26% relative importance).
Caregivers of children with Types 1–3 SMA and adults with Type 2/3 SMA both placed the least importance on mode of treatment administration and reactions caused by the treatment (Figures 3A and B).
Across all treatment attributes related to functional outcomes, adults with SMA and caregivers of children with SMA gave the highest importance to improvements in function compared with worse or stable outcomes (Figures 4A and B). Although administration of the treatment was less important for both caregivers of children with SMA and adults with SMA, both groups declared that a daily oral treatment had the highest importance relative to one-time IV or periodic IT administration (Figures 4A and B). For adults with SMA, daily oral treatments had a much higher level of importance relative to one-time IV or periodic IT administration (58% for oral vs. 20% and 22% for IV and IT, respectively). Although daily oral treatment was still preferred by caregivers, it had only a slightly higher relative importance than one-time IV or periodic IT administration (43% for oral vs. 26% and 31% for IV and IT, respectively).

Relative importance of each of the three levels within the individual attributes for adults with SMA (A) and caregivers of children with SMA (B).
The EQ-5D-5L index value was highest for patients with Type 3 SMA; however, the EQ-5D-5L visual analog scale score was similar across all SMA types. Patients with Type 2/3 SMA who had received treatment had higher scores versus patients who were treatment naïve (Figure 5).
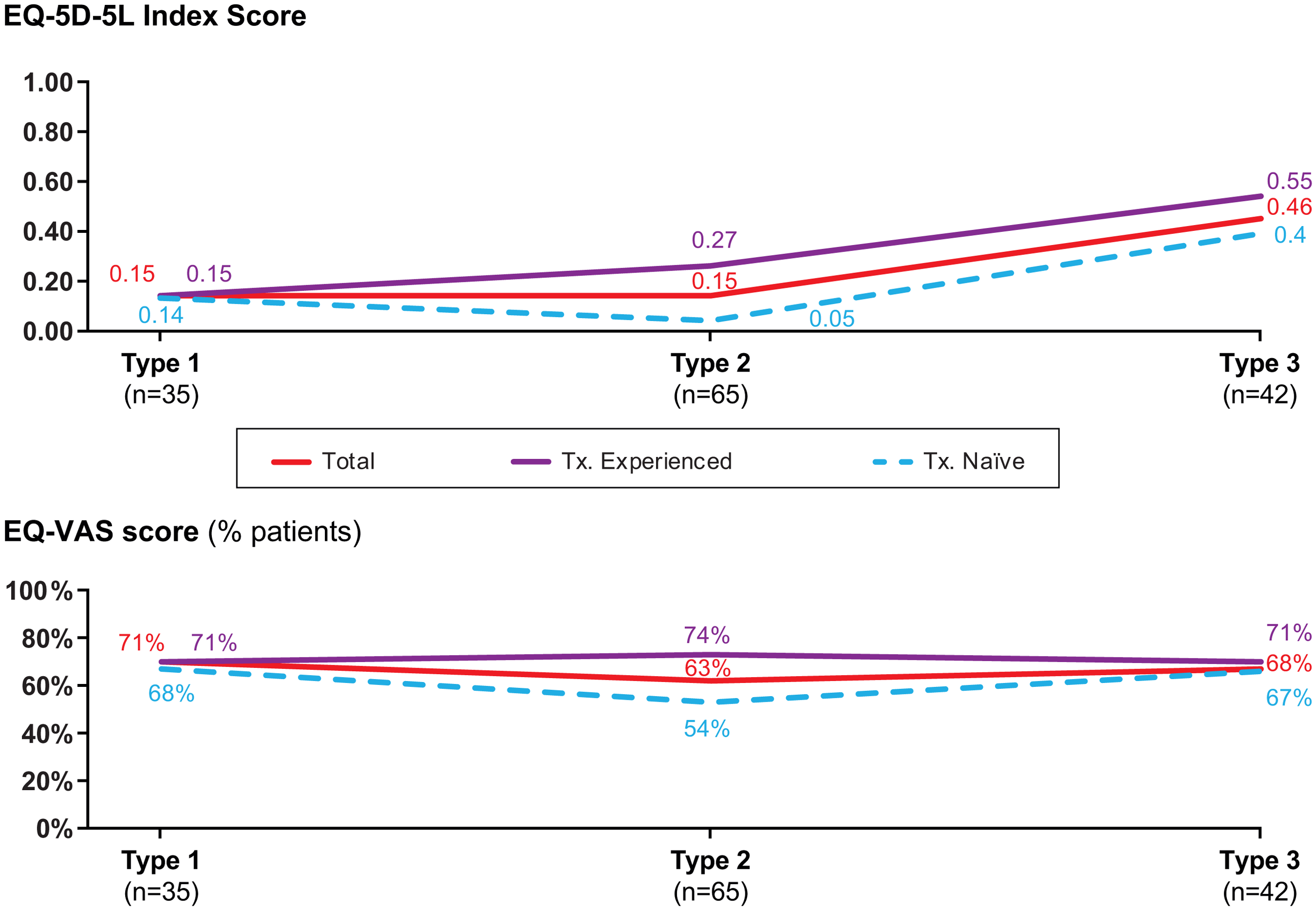
EQ-5D-5L index and EQ-VAS scores by SMA type and pharmacologic treatment history.
No statistically significant differences were reported within the patient or caregiver groups (e.g., by SMA type or treatment experienced vs naïve), due to small sample sizes.
Discussion
Our study illustrates SMA patient and caregiver treatment attribute preferences in Latin America. Caregivers of children with SMA and adults with SMA across Latin America placed different emphases on the most important treatment attribute when considering a DMT. While caregivers of children with Types 1–3 SMA placed greater importance on breathing function, adults with Type 2/3 SMA placed greater emphasis on motor function. These differences correlate with the varying symptoms between SMA types and with differences between caregivers and patients, and they also reflect the ages of these groups. For patients with less severe forms of SMA, breathing function may be less burdensome on QoL compared with patients with Type 1 SMA; therefore, greater emphasis is placed on motor function, which enables them to gain or maintain autonomy and could thus be perceived as a marker of treatment success. 32 This was most evident in the caregiver subgroup analysis, in which caregivers of children with Type 3 SMA emphasized the importance of motor function the most, compared with caregivers of children with Types 1–3 SMA, for whom breathing function was most important.
In the current study, adults with SMA and caregivers of children with SMA gave the highest importance to improvements in function compared with worse or stable outcomes. Studies have previously reported that meaningful change was relative to the individual involved and this can depend on SMA disease severity. 33 Stabilization of the disease can also be viewed as therapeutic progress. 34
The current study reported several similarities in results to studies in other regions. For example, in the USA (N = 101) and European (N = 65) studies, improvement in motor and breathing function was highly valued, both by adult patients and caregivers.7,16 Similarly, in the current study, both caregivers and adults with SMA reported breathing and motor function as the most important treatment attributes. In a UK study (N = 167), adults with SMA (n = 84) and caregivers (n = 83) alike valued improvements in motor function and breathing function, 13 a trend also observed in the current study; in this UK study, adults placed relatively greater emphasis on avoiding deterioration in motor function and breathing function, whereas caregivers of children with SMA prioritized gains in motor and respiratory function. This could be another indication of the subjective nature of how meaningful change is defined – adults with SMA are more likely to have gained motor functions that they now do not want to lose. Caregivers prefer treatment attributes that will improve the likelihood of gaining functions that will help their child lead a more independent life. In an Australian study including 1113 caregivers of people with SMA, participants also indicated a preference towards improved functioning (breathing, swallowing, and muscle strength), mobility (head control, sitting, standing, and walking), and daily oral treatments. 15
In this study, daily oral versus periodic IT or one-time IV administration was the preferred mode of SMA treatment administration by both caregivers and adults with SMA, and this preference appeared stronger among adults with SMA. Compared with other regions of the world, the proportion of respondents who were treatment experienced is within the range of what has been reported in other patient preference studies. Overall, 50% of respondents in the current study were treatment experienced (excluding risdiplam), compared with 68% treatment-experienced respondents (nusinersen or onasemnogene abeparvovec) in a US study, 7 40% treatment-experienced respondents in a European study (nusinersen only), 16 and 33% treatment-experienced respondents (nusinersen only) in a UK study. 13 Preference towards treatment type may depend on experience with one treatment versus another. For instance, patients aged >18 years might not have IV experience. This phenomenon has been underscored in prior research that has reported on treatment experience among people with SMA, in which authors reported that some patients may want to switch their treatment regimen, with reasons cited including convenience, desire to try something new, and safety. 35 Patients may also be influenced by treatment schedule. The perception of patients may be that onasemnogene abeparvovec is like a potential cure because of its requirement for only a single infusion, whereas risdiplam and nusinersen both require continuous treatment.
Considering that SMA is a rare disease and, therefore, recruitment of a target sample can be difficult, the final sample size was relatively large and is similar to prior patient preference studies in SMA.7,13,15,16 Of note, COVID-19 and local ethics committee approvals impacted the length of recruitment. To mitigate this, no strict quota for the ratio of caregivers and patients was set; however, this limitation may have affected subpopulation analyses. Patients were also recruited during different time periods, and country-specific regulatory changes could have happened during the recruitment period.
In addition, the dropout rate in this study was high. Only 25% of people who entered the survey completed the study, with 66% of participants not completing screening or not completing the main survey. Comparatively, there was a 25% dropout in a UK study, 13 a 15% dropout in an Australian study, 15 and a 32% dropout in a European study. 16 One explanation for this could be due to the survey being too long. The number of set choices in the DCE that each respondent had to complete was likely to directly impact respondent engagement. It is important to note that, to keep the survey as short as possible, the number of choices was limited to 14, due to the estimated sample size and the numbers of attributes and levels selected.
The exclusion of risdiplam was a limitation to the analysis of this data set. When patients using oral treatment are excluded, the opportunity to compare the experience they have had with oral treatment is lost. Additionally, although the DCE used in this study was tested internally, it was not piloted before launch to patients and caregivers. The attributes included in the DCE were informed by the results of Part 1 of the study, though further testing of the DCE could have been conducted to test its suitability for this study.
Furthermore, typical limitations of the specific methodology used should be considered. Specifically, given the method of data collection, results might not be representative of the overall population, but rather a representation of a subgroup, likely the most educated. Of note, over half of respondents overall (51.7%; 59.2% of caregivers and 35.6% of people with SMA) had completed a college degree or graduate education. There may also have been unintended bias introduced by the methodology. Access to a computer and internet were mandatory to complete the survey, and, therefore, patients and caregivers without this access were not able to participate.
Additionally, the questionnaires were developed in English and professionally translated into the local language. The EQ-5D-5L and EQ-VAS elements of the survey were performed and approved according to EuroQol, but the translation of other survey components was not conducted according to Functional Assessment of Chronic Illness Therapy guidelines.
In conclusion, this study elucidates that adults with Types 2 and 3 SMA and caregivers of children with Types 1–3 SMA across Latin America place different emphases on treatment attribute priorities when considering a DMT, highlighting differences in preferences among subgroups. Both subgroups expressed a pronounced preference for enhanced functional outcomes over the mere stabilization of existing motor functions. The preference for daily oral administration versus periodic IT or one-time IV routes was particularly strong, notably among the adults with SMA population. Understanding patient and caregiver SMA treatment preferences, particularly at a regional level, is important for shared medical decision-making. It is also important to replicate this study on a larger scale and in other regions of the world, including in patients treated with all available DMTs.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602251320267 - Supplemental material for Patient and caregiver spinal muscular atrophy treatment attribute preferences in Latin America
Supplemental material, sj-docx-1-jnd-10.1177_22143602251320267 for Patient and caregiver spinal muscular atrophy treatment attribute preferences in Latin America by Victoria Saenz, Marijana Chlistalla, Nayara Carlos, Claudia Castiglioni, Maria Soledad Monges, Laurent Servais and Edmar Zanoteli in Journal of Neuromuscular Diseases
Supplemental Material
sj-pdf-2-jnd-10.1177_22143602251320267 - Supplemental material for Patient and caregiver spinal muscular atrophy treatment attribute preferences in Latin America
Supplemental material, sj-pdf-2-jnd-10.1177_22143602251320267 for Patient and caregiver spinal muscular atrophy treatment attribute preferences in Latin America by Victoria Saenz, Marijana Chlistalla, Nayara Carlos, Claudia Castiglioni, Maria Soledad Monges, Laurent Servais and Edmar Zanoteli in Journal of Neuromuscular Diseases
Supplemental Material
sj-pdf-3-jnd-10.1177_22143602251320267 - Supplemental material for Patient and caregiver spinal muscular atrophy treatment attribute preferences in Latin America
Supplemental material, sj-pdf-3-jnd-10.1177_22143602251320267 for Patient and caregiver spinal muscular atrophy treatment attribute preferences in Latin America by Victoria Saenz, Marijana Chlistalla, Nayara Carlos, Claudia Castiglioni, Maria Soledad Monges, Laurent Servais and Edmar Zanoteli in Journal of Neuromuscular Diseases
Supplemental Material
sj-pdf-4-jnd-10.1177_22143602251320267 - Supplemental material for Patient and caregiver spinal muscular atrophy treatment attribute preferences in Latin America
Supplemental material, sj-pdf-4-jnd-10.1177_22143602251320267 for Patient and caregiver spinal muscular atrophy treatment attribute preferences in Latin America by Victoria Saenz, Marijana Chlistalla, Nayara Carlos, Claudia Castiglioni, Maria Soledad Monges, Laurent Servais and Edmar Zanoteli in Journal of Neuromuscular Diseases
Footnotes
Acknowledgments
The authors thank all the individuals who participated in this study. The Research Partnership (London, UK) was responsible for the data management of this research, on behalf of F. Hoffmann-La Roche Ltd. Data were collected by a third-party fieldwork agency Estudio Silvia Roca (Barcelona, Spain). Medical writing support was provided by Kiran Verma of Nucleus Global, UK, an Inizio Company, in accordance with Good Publication Practice (GPP) 2022 guidelines (![]() ).
).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by F. Hoffmann-La Roche Ltd, Basel, Switzerland. Medical writing support was funded by F. Hoffmann-La Roche Ltd.
Declaration of conflicts interest
VS, MC, and NC are all employees of Roche and hold stock/share options in Roche.
CC conducts research/educative events partially funded by Biogen, Novartis, Roche, and PTC Therapeutics. CC collects speaking fees from Roche and PTC Therapeutics.
MSM has received honoraria for advisory board participation and speaker's fees from Biogen, Novartis, Roche, and PTC Therapeutics. MSM is a principal investigator of a Novartis study.
LS has conducted consultancy/been part of the board for Biogen, Novartis, Roche, Biohaven, and Scholar Rock. LS conducts research/educative events partially funded by Biogen, Novartis, Roche, Biohaven, and Scholar Rock.
EZ is principal investigator of clinical studies from Roche, Biogen, and Novartis. EZ conducts educative events funded by Biogen, Novartis, and Roche.
Data availability
For up-to-date details on Roche's Global Policy on the Sharing of Clinical Information and how to request access to related clinical study documents, see here: ![]() . Anonymized records for individual patients across more than one data source external to Roche cannot, and should not, be linked due to a potential increase in risk of patient re-identification.
. Anonymized records for individual patients across more than one data source external to Roche cannot, and should not, be linked due to a potential increase in risk of patient re-identification.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
