Abstract
Background:
Myasthenia gravis (MG), a chronic and unpredictable autoimmune disease, is associated with multiple comorbidities and high disease burden.
Objective:
To assess the disease burden, healthcare resource utilisation (HCRU), and treatment patterns of patients with newly diagnosed MG in England.
Methods:
Data from Clinical Practice Research Datalink GP practices linked to the Hospital Episode Statistics database were used. Eligible patients had ≥1 diagnostic code for MG, with the first MG diagnostic code recorded between 01 January 2010 and 31 December 2019. Non-MG controls were selected if they had no recorded MG diagnosis and ≥12 months of data. Controls were matched for age, sex and GP practice in a maximum ratio of 5:1.
Results:
Mean follow-up duration was 2.8 and 3.1 years for the MG and non-MG cohorts, respectively. In the MG cohort, 56% of patients were male, with a mean age of 67 years at baseline. Incidence rates of all comorbidities assessed during follow-up were higher in the MG cohort than in controls. Almost two-thirds of MG patients experienced ≥1 myasthenic exacerbation during follow-up; incidence rates (95% confidence interval) of MG exacerbations and crises were 50.0 (44.7–55.9) and 1.3 (0.8–2.0) per 100 person-years, respectively. Visits to non-neurology specialists and outpatient clinics were the most common instances of HCRU overall, each being more frequent in the MG cohort than for controls. In the first year of follow-up, acetylcholinesterase inhibitors (AChEIs) and corticosteroids were used by 56.0% and 50.2% of MG patients, respectively; the use of AChEIs declined thereafter.
Conclusions:
Despite treatment, there is a high disease burden for patients with newly diagnosed MG in England, with high rates of MG exacerbation and HCRU use. Thus, there is a need for targeted treatments with sustained efficacy and improved safety to adequately manage MG symptoms and reduce MG-related disease burden.
Introduction
Myasthenia gravis (MG) is a rare, chronic, unpredictable autoimmune disease of the neuromuscular junction (NMJ). MG is characterised by fluctuating chronic muscle weakness and fatigue, caused by pathogenic immunoglobulin G (IgG) autoantibodies that inhibit signal transmission at the NMJ by binding to proteins at the postsynaptic membrane.1–3 The prevalence of MG is increasing globally, especially among people aged over 65 years. 4 In 2019, MG was estimated to be the second most common neuromuscular disease in the UK, with a prevalence of 33.7 per 100,000 people, after Guillain-Barré syndrome. 4 Similar estimates have been reported in Sweden (36.1 per 100,000 people in 2016) 5 and Germany (39.3 per 100,000 in 2019). 6 The years immediately following MG diagnosis are typically a period of high healthcare resource utilisation (HCRU), especially in the first year after diagnosis whereby emergency room visits, hospitalisations, and intensive care unit (ICU) admissions are all high. 7 This, in addition with quality of life and mental health, contributes to the overall burden of disease in MG.8,9 A greater awareness of the clinical burden experienced by patients with newly diagnosed MG, and the toll this takes on patients’ emotional wellbeing, may help to improve treatment strategies and management of this population.
For those who have been diagnosed with MG, guidance from the Association of British Neurologists recommends initial treatment with acetylcholinesterase inhibitors (AChEIs), with or without corticosteroids (CSs). 10 Non-steroidal immunosuppressive therapies (NSISTs) are recommended for patients who do not receive sufficient clinical benefit from conventional MG treatments. 10 Despite many therapeutic agents available to manage MG symptoms, side effects (such as increased risk of infection) are common, and patients’ disease is often not effectively controlled. 11 Some patients with MG experience myasthenic exacerbation or crisis, during which symptoms require rescue therapy and, in the case of crises, can be life-threatening.12,13 Rescue therapy includes intravenous immunoglobulin (IVIg) or plasma exchange (PLEX). 14
Overall, there is an evidence gap in the MG literature and the evolving disease landscape and burden is not well-understood. 15 The limited available data on the clinical burden and treatment patterns of adults with MG in England show that the rates of MG-related hospitalisations, exacerbations and myasthenic crises are highest in the first year after diagnosis. 16 However, to our knowledge, there are no published data specifically for patients newly diagnosed with MG. Here, we retrospectively analysed data from electronic health records to determine the disease burden, defined as comorbidities, MG-related HCRU, and MG-specific clinical events such as exacerbations and myasthenic crisis, and treatment patterns of patients with newly diagnosed MG in England. We also aimed to compare comorbidities and HCRU with a matched cohort of individuals without MG.
Materials and methods
Study design and data source
This was a retrospective cohort study of adult patients with newly diagnosed MG and matched non-MG controls identified from the Clinical Practice Research Datalink (CPRD) GOLD database, a primary care electronic health records database in the UK, and linked to the Hospital Episode Statistics (HES) database. The CPRD is a primary care electronic health records database containing longitudinal data from general practitioners (GPs) in England and other nations of the UK.17,18 CPRD can be split into two different databases, GOLD and Aurum, which differ by the system used to collect data.4,19 These datasets have been used recently to estimate the incidence and prevalence of MG in the UK, 4 and CPRD can be linked to UK mortality records from the Office for National Statistics and other datasets, including the HES database, which records hospital-based activity specifically in England. 20 HES is a secondary care data warehouse that contains pseudonymised records of all patients admitted to NHS hospitals in England, including patients who reside outside of England and private patients treated in NHS hospitals. HES stores data on hospital diagnoses, procedures, treatment and HCRU (including inpatient admissions, and attendance at outpatient and Accident and Emergency departments) for the past ten years. 21 Linkage of CPRD data to HES is available for approximately 75% of CPRD practices in England, representing 58% of all UK CPRD practices. 17 This linkage is only available for participating GP practices in England, and therefore this analysis was restricted to England. CRPD GOLD extracts data from clinical practices using the Vision® GP practice clinical software system. 19
Study population
Patients with MG were identified using clinical Read Codes for MG in the CPRD database (F380.00, F380z00, F380100, F383.00) or the International Classification of Diseases (ICD)-10 code for MG (G70.0) in the HES database, following the method of two prior CPRD-HES linked studies, by Harris et al., of patients with MG with data captured between 1997 and 2016.16,22 Eligible patients had linked data in the CPRD GOLD and HES databases and had ≥1 diagnostic code for MG, with the first MG diagnostic code (Read Code in CPRD or ICD-10 code in HES) recorded between 01 January 2010 and 31 December 2019 (the index date). Incident patients in a given year were defined as those newly diagnosed with MG in that year. A look-back period of 12 months prior to the index date (the ‘pre-index’ period) was applied to ensure that there was no prior mention of MG that would suggest an earlier diagnosis, thus capturing the first MG diagnosis.
A matched control cohort was created by randomly selecting individuals with no recorded MG diagnosis in the CPRD or HES databases, and with at least 12 months’ data available prior to the index date. These non-MG controls were matched for age, sex and GP practice in a maximum ratio of 5:1 with MG patients. Individuals in the non-MG cohort were each assigned the same index date as that of their matched MG case.
For each patient in the study, the pre-index period was defined as the 12-month period prior to the index date. The follow-up period was defined as the time between the index date and the end of follow-up, set as either the end date of the study (31 December 2019), the death of the patient, or the date of the last data collection by the GP practice, whichever came first.
Study outcomes
Comorbidities
The occurrence of comorbidities was assessed in patients with MG and non-MG controls pre-index and during follow-up. For simplicity, the term ‘comorbidity’ is also used to describe the conditions of individuals in the non-MG cohort. Clinical Read Codes (Medcodes), ICD-10 codes and OPCS codes were used to identify comorbidities, including: infections (including systemic infections and hospitalisation for infections), autoimmune diseases (ankylosing spondylitis, autoimmune thyroiditis, Crohn's disease, diabetes type 1, psoriasis, psoriatic arthritis, rheumatoid arthritis, systemic lupus erythematosus and ulcerative colitis), anxiety and/or depression, and other chronic conditions of interest (cardiac arrhythmias, diabetes type 2, dyslipidaemia, hypertension, obesity and osteoporosis) (Supplemental Table 1). A full list of the diagnostic codes used to identify comorbidities is in Supplemental Table 2. The numbers of patients with mild (1–2), moderate (3–4) and severe (5) Charlson Comorbidity Index (CCI) scores were also determined.
Clinical events, HCRU, and mortality
Myasthenic exacerbations and myasthenic crises were assessed in patients with MG during follow-up (Supplemental Table 1). Myasthenic exacerbation was defined as hospitalisation with MG (excluding hospitalisation for thymectomy) or treatment with IVIg or PLEX, and excluded cases of myasthenic crisis. Myasthenic crisis was defined as an ICU admission in combination with respiratory failure or mechanical ventilation or intubation.
HCRU was assessed in patients with MG and non-MG controls during follow-up. HCRU included overnight hospital stays, emergency room (ER) visits, GP visits, outpatient visits, ICU admissions, and neurology and non-neurology specialist visits. MG-related events among patients with MG were also assessed during follow-up.
Frequency of death, time to death (descriptive), and mortality rate were assessed only in MG patients. Mortality rate was calculated as the total number of deaths of patients with MG per patient-years at risk from start of follow-up until death. Further definitions of outcome variables are provided in Supplemental Table 1.
Treatment patterns
MG treatments among patients with MG during the pre-index period, patients’ transition to different treatment types during follow-up, and median time spent on each treatment were captured. The follow-up period was divided into treatment cohorts (“Segments”) based on the type of treatment received, if any (Supplemental Tables 3 and 4), to show the patient movement from one treatment type to another. This segment approach also enables easy categorisation and assessment of common treatment types for a large population, without needing to assess each treatment and combination of treatments. Segment 0 represented the time prior to a patient receiving any MG treatment, while Segment 10 represented permanent discontinuation of treatment or a treatment break. Segment 1 represented the first use of AChEI or CS on or after index date, and prior to use of any NSIST or biologics. Segments 2, 3, and 4 defined time of first, second and third NSIST use, respectively, while Segments 5 and 6 defined time of first and second biologic use, respectively. Patients could transition between segments or skip segments but could not be present in more than one segment at a time, and could only advance unidirectionally through Segments 0 to 6 (skipping of segments was allowed). Various treatment options were included in definitions of Segments 7 to 9 (e.g., time reinitiating NSISTs or biologics). AChEIs, with or without CSs, could be used as a background therapy in all segments, except Segments 0 and 10. Definitions of treatment types included are listed in Supplemental Table 3 and full definitions of treatment segments are described in Supplemental Table 4. Time on treatment was assessed by the time patients spent in each Segment, whereby the start of treatment is the prescription date; the end of treatment is the prescription date + days of supply. If days of supply are not available, 1 day of supply was assumed. Gaps were allowed for each treatment to consider a continuous treatment (e.g., 90 days for prescriptions). In the event that a patient resumed treatment after taking a treatment break while in any of Segments 1–6, without progressing to the next segment, time when patients re-initiated the treatment was added to the current segment (Supplemental Table 4). Treatments given to hospital inpatients are not captured by the CPRD database and are therefore not included in this analysis.
Acute and chronic (maintenance) use of IVIg during follow-up among patients with MG was also assessed, including and excluding use to treat myasthenic crisis (Supplemental Table 3).
Statistical analyses
Descriptive statistics were used throughout. Continuous outcome variables are presented as mean, standard deviation (SD), median, interquartile range (IQR; calculated as the difference between the third and first quartiles), and minimum and maximum values. Categorical variables are presented as n (%). Categorical data were analysed using a Chi-square test, and continuous variables were assessed using the t test.
Incidence rates for events of interest were calculated using the number of patients with a first-ever occurrence for the specific event during the period of interest. Patients at risk for events of interest were all those included in the MG cohort. Person-time at risk was the sum of person-years (PY) of patients at risk from the start to either the end of the period or the end of follow-up (whichever occurred first; for patients who did not have the event), or date of the first occurrence within that period (for patients who did have the event). The risk of each event was estimated using an incidence rate and its 95% confidence interval (CI) and expressed per 100 PY. Prevalence was calculated by the proportion of patients with an event divided by the entire population.
In alignment with CPRD data privacy policies, data cell sizes of five or fewer individuals were suppressed in our analyses.
Results
Patient demographics
Between January 2010 and December 2019, 5169 patients were identified from the CPRD GOLD database, from a total of 857 GP practices. Of patients who had linked data in the CPRD-HES database, 480 patients met the inclusion criteria, including an MG diagnosis in a given year between the period of 2010 to 2019, and were therefore defined as newly diagnosed and included in the MG cohort of this study. Data for 2237 matched non-MG controls were identified (Table 1).
Baseline demographics, comorbidities and follow-up duration.
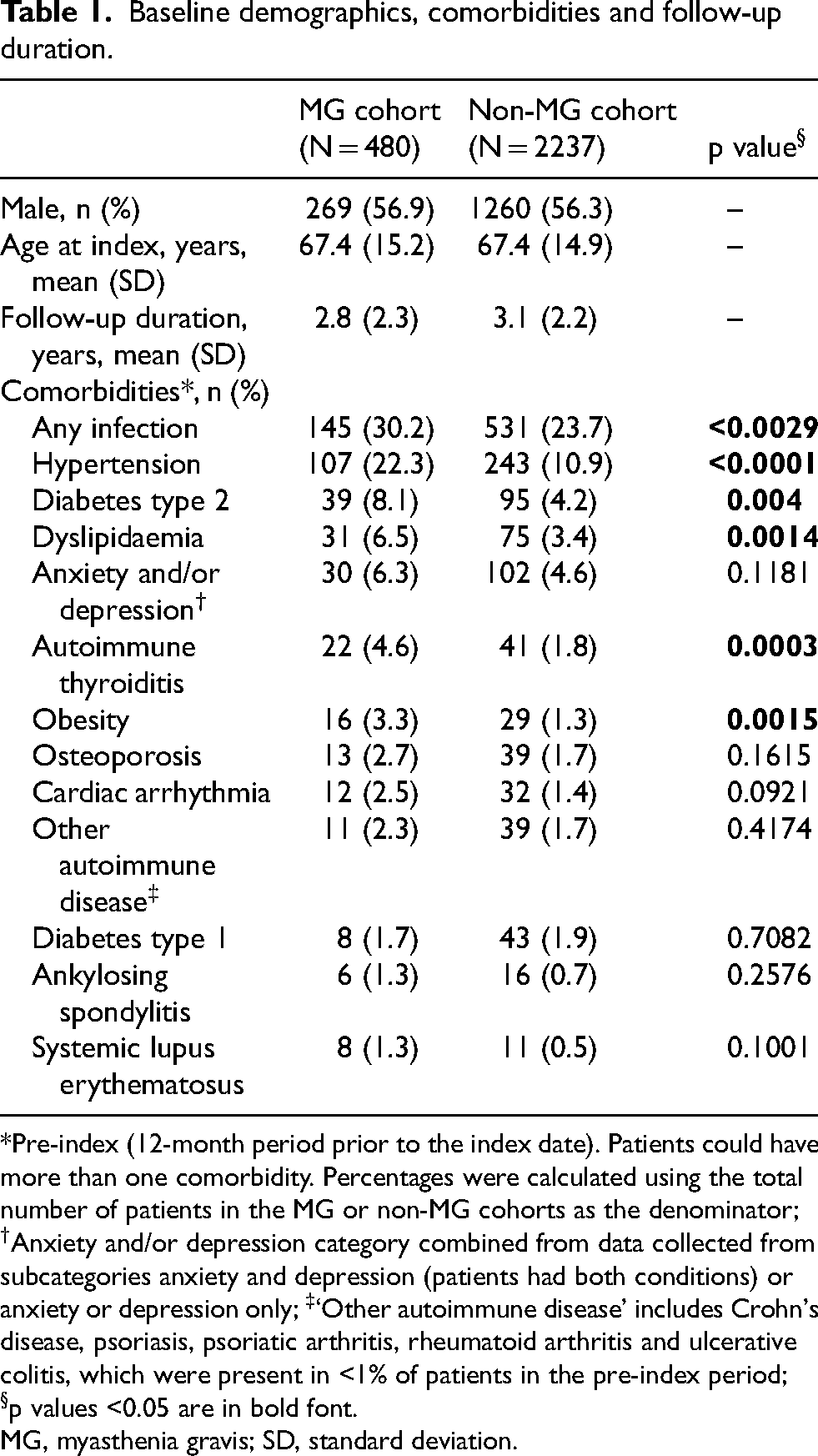
*Pre-index (12-month period prior to the index date). Patients could have more than one comorbidity. Percentages were calculated using the total number of patients in the MG or non-MG cohorts as the denominator; †Anxiety and/or depression category combined from data collected from subcategories anxiety and depression (patients had both conditions) or anxiety or depression only; ‡‘Other autoimmune disease’ includes Crohn's disease, psoriasis, psoriatic arthritis, rheumatoid arthritis and ulcerative colitis, which were present in <1% of patients in the pre-index period; §p values <0.05 are in bold font.
MG, myasthenia gravis; SD, standard deviation.
Comorbidities
Comorbidities in the pre-index period
The occurrence of several comorbidities in the pre-index period was statistically significantly higher in the MG cohort compared with the non-MG cohort, including infection (any type) and autoimmune disease (any type) (Table 1). The pre-index mean (SD) overall CCI score was statistically significantly higher in the MG cohort compared with the non-MG cohort: 0.3 (0.7) vs 0.2 (0.7); p = 0.0036. A higher proportion of patients with MG had mild grade CCI (1–2) than non-MG controls (23.8% vs 15.9%; p = 0.0011), while a similar proportion of patients had moderate grade CCI (3–4; 1.7% vs 1.3%). Eight (0.4%) non-MG controls had severe grade CCI (>4); no patients with MG had severe grade CCI.
Comorbidities during follow-up
Overall, the most reported comorbidity during follow-up was ‘any infection’, occurring in 35.8% and 28.0% of patients in the MG and non-MG cohorts, respectively (Figure 1). The incidence rates of all comorbidities during follow-up were proportionately higher in the MG cohort than in the non-MG cohort.
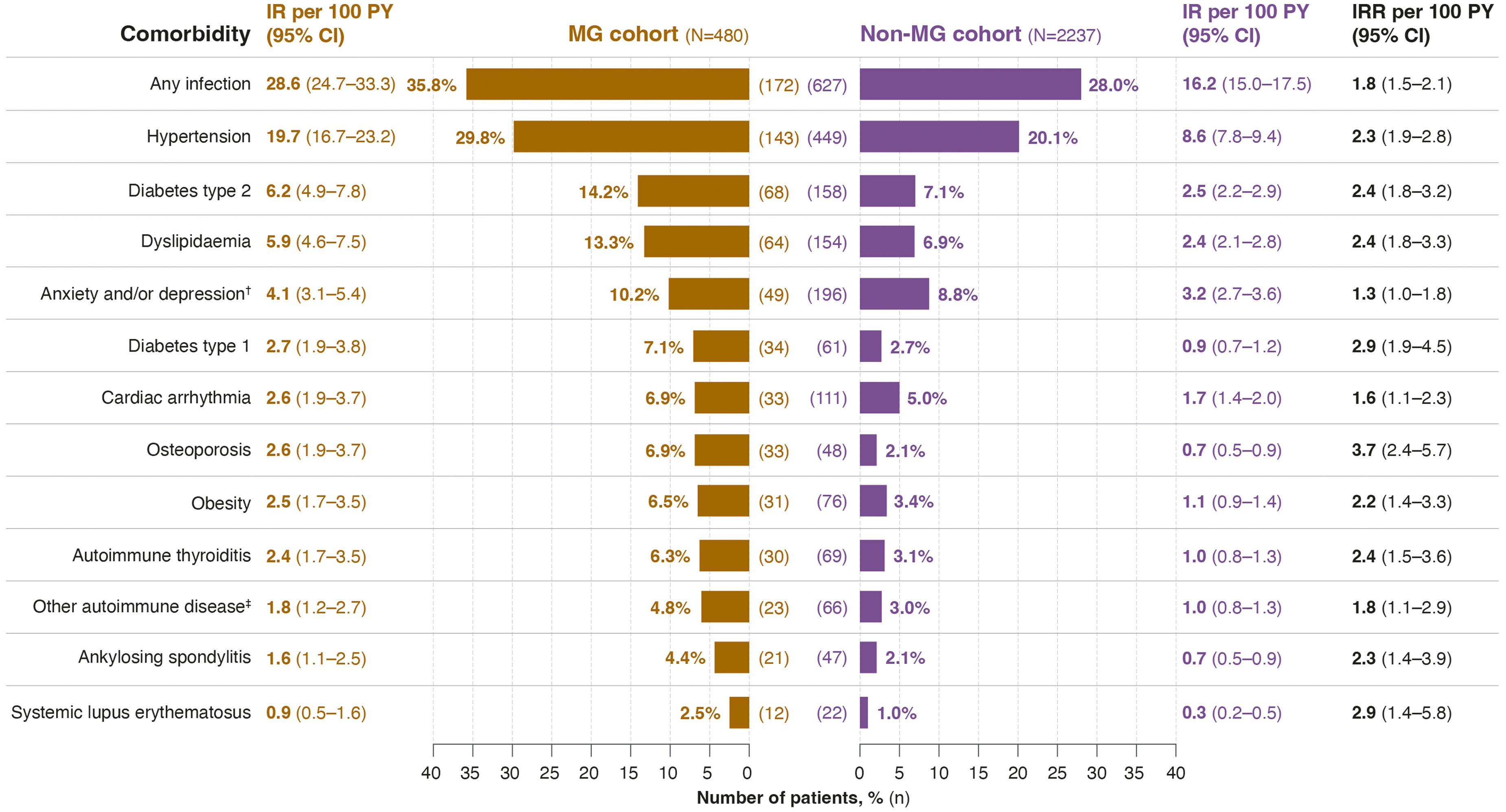
Prevalence and incidence of comorbidities during follow-up*. *Patients could have more than one comorbidity. Percentages were calculated using the total number of patients in the MG or non-MG cohorts as the denominator; †Anxiety and/or depression category combined from data collected from subcategories anxiety and depression (patients had both conditions) or anxiety or depression only; ‡‘Other autoimmune disease’ includes Crohn's disease, psoriasis, psoriatic arthritis, rheumatoid arthritis, and ulcerative colitis, which were present in <1% of patients in the pre-index period. CI, confidence interval; IR, incidence rate; IRR, incidence rate ratio; MG, myasthenia gravis; PY, patient-years.
Clinical events and mortality
Almost two-thirds (63.5%) of patients with MG experienced at least one MG exacerbation during follow-up, with a mean (SD) of 2.6 (3.0) exacerbations per patient. The incidence rate (95% CI) of exacerbations was 50.0 (44.7–55.9) per 100 PY. Myasthenic crisis was experienced by 17 (3.5%) patients with MG during follow-up. Of these 17 patients, the mean (SD) occurrence of crisis was 1.3 (0.8) per patient. The overall incidence rate (95% CI) for myasthenic crisis among patients with MG was estimated at 1.3 (0.8–2.0) per 100 PY.
During follow-up, 79 (16.5%) patients with MG died; the rate (95% CI) of all-cause mortality among patients with MG was 5.8 (4.6–7.2) per 100 PY. The mean (SD) time to death was 602.6 (621.7) days for patients with MG who died. Among the 308 patients with MG who experienced exacerbation during follow-up, 68 (22.1%) died with an all-cause mortality rate (95% CI) of 7.5 (5.9–9.5) per 100 PY. The median (IQR [min–max]) time to death for patients with MG who experienced exacerbation during follow-up was 294.0 (709.0 [3–2518]) days. Of 170 patients with MG who did not experience an exacerbation or crisis during follow-up, 10 (5.9%) patients died, with an all-cause mortality rate (95% CI) of 2.2 (1.2–4.1) per 100 PY.
HCRU
Visits to specialists other than neurologists were the most common HCRU event in both cohorts, with at least one visit occurring for almost all patients with MG and non-MG controls (Table 2). The proportion of patients with outpatient, GP, and neurology specialist visits was numerically higher for the MG cohort than the non-MG cohort, and almost twice as many patients with MG as non-MG controls had overnight hospital stays during follow-up. ICU admissions were the least frequent HCRU event for both cohorts.
All-cause and MG-related HCRU during follow-up.
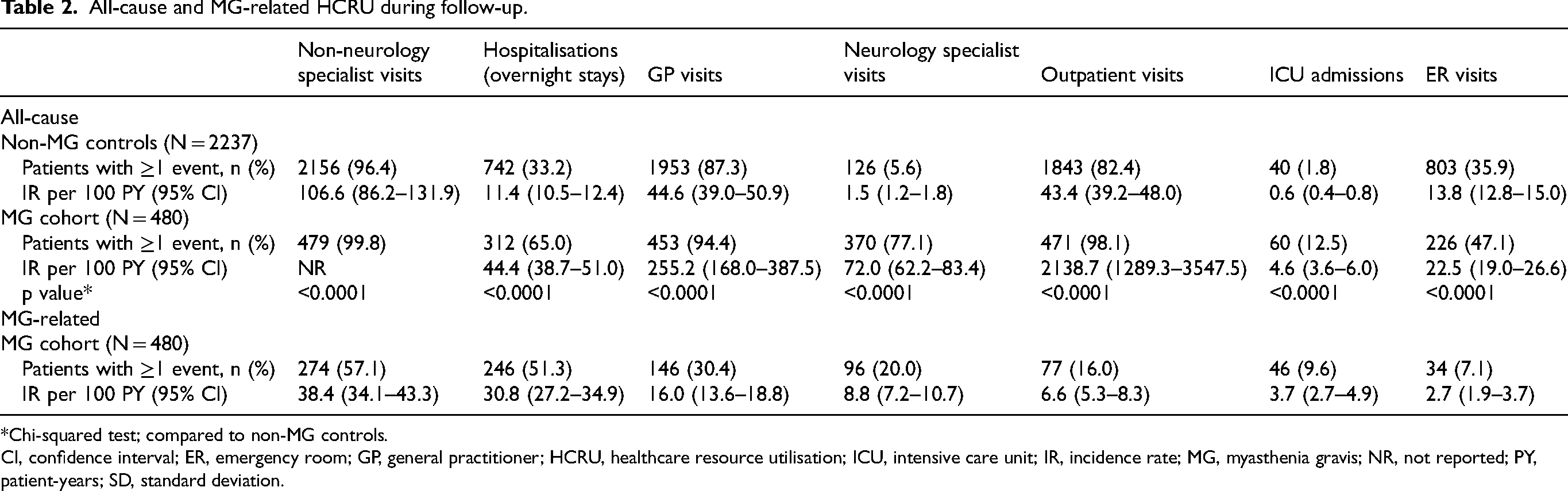
*Chi-squared test; compared to non-MG controls.
CI, confidence interval; ER, emergency room; GP, general practitioner; HCRU, healthcare resource utilisation; ICU, intensive care unit; IR, incidence rate; MG, myasthenia gravis; NR, not reported; PY, patient-years; SD, standard deviation.
More than one-third of patients with MG (37.3%) visited a neurology specialist after index, compared with 4.4% of non-MG controls. Proportions of patients with GP visits, ER visits, and outpatient visits that occurred for the first time after index were similar or greater for the non-MG cohort compared with the MG cohort (Table 2).
The mean duration of overnight hospital stays during follow-up was almost twice as long for patients in the MG cohort as for patients in the non-MG cohort. The mean (SD) duration of overnight hospitalisation was 28.0 (37.1) days and 21.2 (30.4) days for patients with MG and non-MG controls, respectively. The median (IQR [min–max]) length of overnight hospital stay was 14.5 (26.5 [1–314]) days and 10.0 (22.0 [1–289]) days for patients in MG and non-MG cohorts, respectively. In the MG cohort, the occurrence of hospitalisation for infection was more than double of that of the non-MG cohort (24.0% vs 10.0%).
MG-related HCRU
There were 640 MG-related visits in total by patients with MG to a non-neurology specialist (visit to a specialist other than a neurologist), equating to a mean (SD) of 2.3 (2.4) visits per patient and more than half of patients (57%) with at least one visit (Table 2). Approximately half of the MG cohort had at least one MG-related overnight hospital stay, and almost 10% of patients had at least one MG-related ICU admission (Table 2).
Treatment patterns
Of the MG cohort, 57 (11.9%) patients started treatment for MG on the index date with either AChEI or CS (n = 52) or their first NSIST (n = 5). In the first year of follow-up, AChEIs and CSs were used by 56.0% and 50.2% of patients in the MG cohort, respectively. From Years 1–9, the use of AChEIs progressively declined, and from Years 2–7, a higher proportion of patients with MG used CSs than AChEIs each year. NSISTs were used by 29.6% of patients with MG during follow-up, with the most frequently used NSIST being azathioprine (22.9%). Use of IVIg and PLEX was reported for 10.2% and 3.1% of patients with MG overall, respectively, but only in <5 patients per year from Year 3 of follow-up. Thymectomy was performed for 26 (5.4%) patients with MG during follow-up; no use of biologics was captured during this period (Supplemental Tables 5 and 6).
Patient movement between treatment Segments 1–3 is shown in Figure 2. On the index date, 423 patients (88.1%) were not receiving any MG treatment and therefore started in Segment 0. During follow-up, approximately one-quarter (n = 112; 23.3%) were not treated with any of the MG treatments included in the treatment segment definitions, and thus remained in Segment 0 until the end of the study. A total of 318 patients (66.3%) were in Segment 1 (AChEIs or CSs) at any point during follow-up. Of whom, 92 patients (19.2% of the total population) moved to Segment 2 (first NSIST), which comprised 142 patients in total during follow-up (Figure 2). Seventeen (3.5%) patients subsequently switched from Segment 2 to a second NSIST, entering Segment 3 (Figure 2). Patients spent a median (mean; SD; IQR; range) of 307.5 (515.9; 587.9; 615.0; 1–3145) person-days in Segment 1 (AChEIs or CSs), 618.5 (781.1; 732.9; 1092.0; 19–3201) person-days in Segment 2 (first NSIST), and 538.0 (652.5; 418.9; 635.0; 119–1491) in Segment 3 (second NSIST). Additional data on transition of patients between treatment Segments are reported in Supplemental Tables 6 and 7.
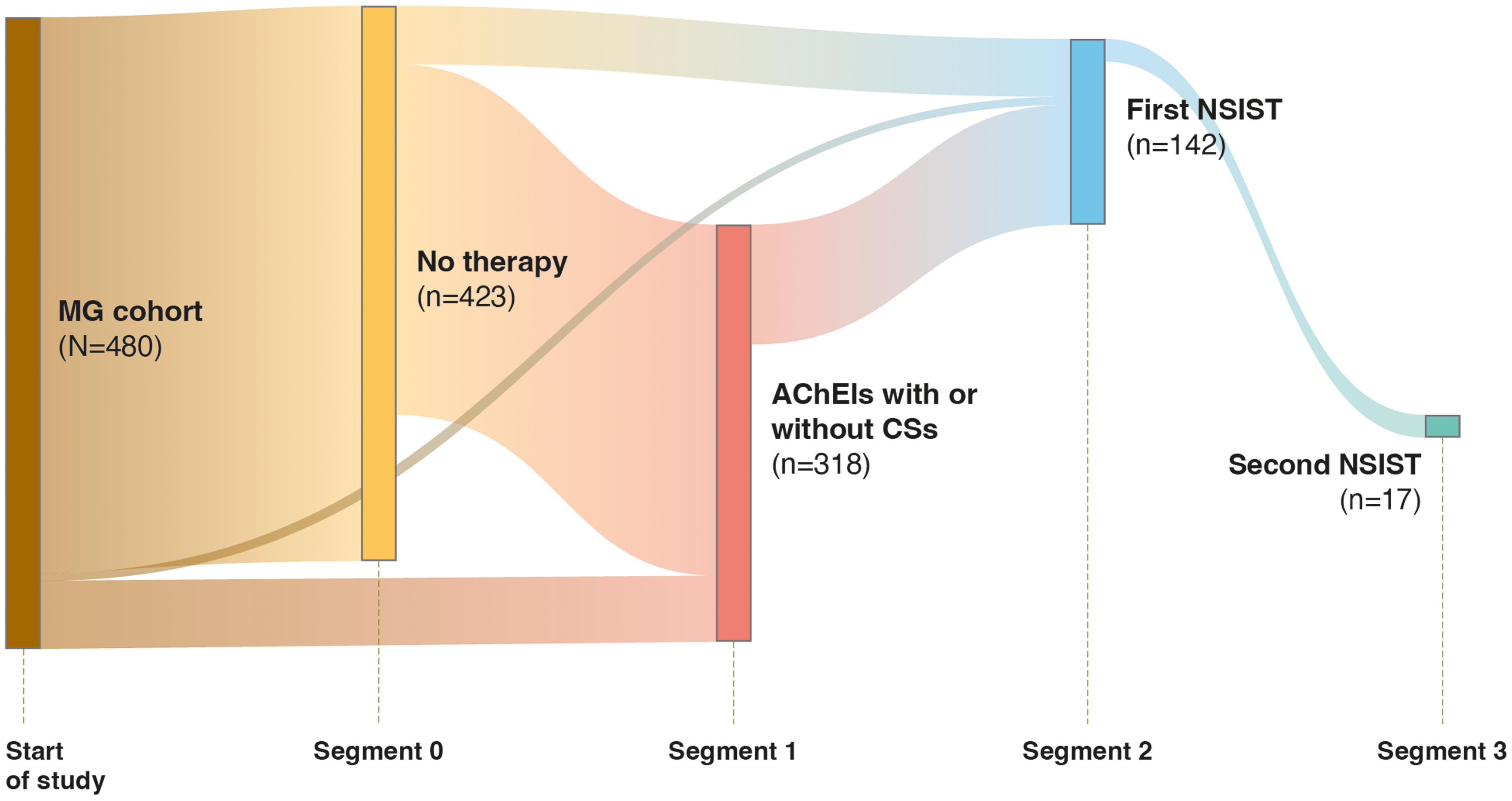
Movement of patients in the MG cohort from treatment Segment 0 to treatment Segment 3. On the index date, 423 patients (88.1%) were not receiving any MG treatment and therefore started in Segment 0. 112 of these patients were not treated with any MG treatments included in the treatment segment definitions during follow-up, and thus remained in Segment 0 until the end of the study. AChEI, acetylcholinesterase inhibitor; CS, corticosteroid; MG, myasthenia gravis; NSIST, non-steroidal immunosuppressive therapy.
IVIg use during follow-up
Over 10% of patients with MG were prescribed at least one first-ever IVIg cycle during follow-up. The highest rate of IVIg use was in the first year after index (8.1%; incidence rate = 10.1 per 100 PY, Supplemental Table 8).
Discussion
This retrospective analysis of disease burden, HCRU, and treatment patterns found that patients with newly diagnosed MG have a greater frequency of comorbidities and increased HCRU, compared with non-MG controls.
A greater proportion of patients in the MG cohort had comorbidities both in the pre-index period and during follow-up vs non-MG controls. These observations align with results from a US cohort study in the same timeframe that reported a greater prevalence of baseline comorbidities, including autoimmune diseases, dyslipidaemia, diabetes type 2, hypertension, obesity, osteoporosis, anxiety, and depression among patients with MG than matched controls. 7 Similarly, a longitudinal study of 327 adults with MG from the Oxford Myasthenia Centre UK registry, found 94.5% of patients with MG had at least one comorbidity, and that comorbidities accumulated with age. 23 The Oxford study also found that some comorbidities in older patients with MG, such as hypertension, may be associated with age rather than MG disease burden. 23 In our cohort, we found that hypertension was common among patients with MG, which aligns with the Oxford study, and previous studies from China, Taiwan, and the US.7,23–25
In our study, approximately one-third of patients with MG had an infection, both pre-index and during follow-up, whereas, in a 2008–2019 US cohort, infection at baseline was reported for over half of the patients with MG. 26 This higher rate of infection may be explained by the observation that a third of patients in the US cohort were being treated with NSISTs at index date 26 compared with just 5% of patients in the current study. The causes of these infections were not captured in our dataset, but possibilities include inadequate treatment for MG, whereby patients were not sufficiently medicated, or use of immunosuppressive MG treatments, which have a known increased infection risk from prolonged use. 13 A different US cohort study reported a 3.5-times higher risk of infection-related hospitalisation for patients with MG compared with healthy controls, also attributed to the use of NSISTs. 7 In the present study, almost one-quarter of patients in the MG cohort were hospitalised for infection. An analysis of hospital admissions of patients with MG in Finland between 2004 and 2014 found that the duration of overnight hospital stays was longer when infection was the primary diagnosis for hospitalisation, compared with hospitalisations for a non-infection primary diagnosis. 27 Further, in our study, the proportion of patients with MG who required ICU admission was three times higher than the proportion of patients who experienced myasthenic crises, which ruled out crises as the prominent cause of overnight hospitalisation. Therefore, it is important to consider the challenges that comorbid conditions and treatment with conventional therapies may pose for MG disease management, which may be better served by personalised treatment approaches. 28
In our study, almost two-thirds of incident MG patients experienced at least one myasthenic exacerbation during a mean of almost three years of follow-up, while myasthenic crisis was experienced by a lower proportion (3.5%). Compared with a previous CPRD-HES study by Harris et al., which included data recorded in England between 1997 and 2016, we found a 39% higher exacerbation rate and 15% lower crisis rate. 16 This disparity in the proportions of patients who experienced exacerbation or crisis between our CPRD-HES linked study and that of Harris et al. likely reflects the different follow-up durations, definitions of exacerbation and crisis, and variations in patient profiles between cohorts, including the time since MG diagnosis. 16 For example, in the 1997–2016 CPRD-HES study, MG exacerbation included events coded as myasthenic crisis and exacerbation or IVIg and PLEX administration, although neither IVIg nor PLEX was confirmed as MG-related, and incomplete coding for CPRD and HES data was acknowledged as a potential limitation. 16 Most exacerbations and crises occurred within three years of MG diagnosis for patients in the 1997–2016 CPRD-HES study. 16 Collectively, these studies support the need for early intervention to mitigate the risk of exacerbation and crisis during the early years following MG diagnosis. 7
Our analysis revealed that AChEIs and CSs were the most commonly used treatments in incident MG patients, followed by NSISTs, which is consistent with UK 10 and international treatment guidelines.29,30 The reduction we observed in IVIg use during follow-up likely reflects patient attrition, as more than 60% of patients with MG experienced exacerbation during follow-up, suggesting that MG was inadequately controlled with conventional treatments. IVIg supply can be intermittent globally, 31 which may also account for low recorded patient use during follow-up. Given the similar effectiveness and safety profile of IVIg and PLEX, a recent cost-minimisation study suggested that, owing to advancement in treatment administration, the National Health Service should prioritise PLEX over IVIg use. 31 Rates of thymectomy were also low in our cohort with just 5% of patients having undergone a thymectomy during follow-up. This is lower than expected considering the British and international guidance that recommends thymectomy within two years of symptom onset in patients positive for anti-acetylcholine receptor antibodies.10,30 It is possible that more thymectomies were done before the pre-index data were captured, or were not captured by CPRD or HES. Approximately one-quarter of our cohort were not treated with any treatment for MG per our segment definitions. We can speculate that some of these patients may have been prescribed their initial treatment in a hospital setting, and thus the CPRD (linked to primary care prescriptions) is unlikely to capture such prescriptions. In addition, our patients were identified from 857 participating GP practices, which only covers approximately 13% of the total practices in England.
Due to the secondary nature of the analysis and the use of a database as the data source, there are some data gaps, and the results should be interpreted with caution. Despite this, the CPRD still provides a large, real-world dataset of anonymised primary care electronic health records,17,18 and its use represents a key strength of this study. While the HES linkage restricted the analysis to patients in England only, the HES database comprises a complete and unbiased sample of hospital admissions and episodes, and can therefore be deemed representative of National Health Service hospital admissions in England. 20
Other limitations of the databases include the accuracy of the results being dependent on the complete and precise coding of clinical events and treatments by general practices and hospitals. A risk of misdiagnosis of MG is likely to be low as the study used the same CPRD Read Codes and ICD-10 codes for MG diagnosis as in the CPRD-HES linked studies of patients with MG by Harris et al.16,22 As discussed above, MG hospital-based treatments prescribed to patients during their hospital stay are not included in CPRD records. Therefore, it is likely that the use of inpatient MG treatments such as IVIg and PLEX has been underestimated. A further limitation inherent to database studies is the inability to link the diagnostic code and prescription in the database. The HES database does not capture use of specific biologics administered outside of a hospital setting or primary care visits. Data for biologic use in general were lacking, possibly owing to most of the follow-up years being before rituximab was recommended for use in England. The data for this study were from the GOLD version of the CPRD database; and, given the relatively short follow-up time in our study, it is possible that some English GP practices may have switched to the CPRD Aurum database. Therefore, further analyses using the larger CPRD Aurum database may be warranted to confirm the disease burden and MG treatment patterns observed in this study. In addition, a 1-year incident look-back window was used, which may not guarantee that all patients included are truly incident MG. However, this is unlikely, as most patients diagnosed with MG would be expected to receive treatment within a 12-month period.
Notwithstanding these limitations, our data highlight the compelling need for improved treatments for MG. Specifically, more effective, targeted treatments with sustained efficacy and improved safety to adequately manage MG symptoms and reduced MG-related disease burden will be welcomed by patients and those managing patients’ care.
Conclusions
These data confirm the high disease burden and HCRU of patients in England with newly diagnosed MG and highlight the limitations of conventional treatments, including increased infection risk and inadequate symptom control. Treatment should be used in a timely and consistent manner, with appropriate patients given access to new treatments that target the underlying causes of MG and offer sustained efficacy to mitigate the fluctuating, debilitating course of the disease. More broadly, based on the available data in the literature, increased awareness and a focus on the importance of rapid diagnosis and intervention with high-quality and cost-effective care is also needed to reduce the burden on patients, their carers, and an increasingly strained healthcare system.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602241308194 - Supplemental material for Disease burden, healthcare resource utilisation, and treatment patterns in patients with newly diagnosed myasthenia gravis in England: A retrospective cohort study
Supplemental material, sj-docx-1-jnd-10.1177_22143602241308194 for Disease burden, healthcare resource utilisation, and treatment patterns in patients with newly diagnosed myasthenia gravis in England: A retrospective cohort study by Kerina Bonar, Nada Boudiaf, Piotr Zaremba, Thais Tarancón, Jiachen Zhou and Saiju Jacob in Journal of Neuromuscular Diseases
Footnotes
Acknowledgements
Medical writing support was provided by Rosalind Carney, DPhil, and Rachel Price, PhD, of Ogilvy Health UK, and funded by UCB, in accordance with Good Publication Practice 2022 guidelines (![]() ). The authors thank David Harrison, DPhil, and Veronica Porkess, PhD, of UCB for publication and editorial support.
). The authors thank David Harrison, DPhil, and Veronica Porkess, PhD, of UCB for publication and editorial support.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by UCB.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kerina Bonar, Thais Tarancón and Jiachen Zhou are employees of UCB. Nada Boudiaf and Piotr Zaremba are contractors employed by UCB. Saiju Jacob has served as an international advisory board member for Alexion, Alnylam, argenx, Immunovant, Regeneron, and UCB; is currently an expert panel member of the Myasthenia Gravis consortium for argenx; and has received speaker fees from Terumo BCT and Eisai Pharmaceuticals.
Data availabilty
Data from non-interventional studies is outside of UCB's data sharing policy and is unavailable for sharing.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
