Abstract
Background
The muscle weakness in patients with nemaline myopathy (NM) and related disorders (NMR) often affects the muscles used for eating and dining, and some scientific evidence of poorer nutritional status of patients with myopathy exists. However, comprehensive research on the nutritional status of persons with NM or NMR has not been done.
Objectives
Our aim was to conduct a comprehensive cross-sectional pilot study among adult Finnish patients with NM or NMR to investigate their food consumption, nutrient intakes, selected nutrient-related laboratory parameters in blood, and self-assessed functioning of dining and eating and of the gastrointestinal tract. We also aimed to test the suitability of the methods selected.
Methods
The methods included a questionnaire to investigate eating-related functioning, a food frequency questionnaire, food records, and laboratory analyses from blood samples.
Results
Of 32 invited patients, 20 returned the survey and food frequency questionnaire. Food records were returned by 17, and blood samples were obtained from 16. Food consumption as well as nutrient and energy intake were highly variable between the individuals. Low energy intakes, low consumption of healthy foods, such as fruits, vegetables, and whole grains as well as low intakes of vitamin D, calcium, dietary fiber, vitamin C, folate, and iron were observed. Low nutrient intakes were seen especially in non-ambulatory participants, who also reported challenges in eating and dining related functioning. The laboratory parameters did not indicate severe undernourishment in any of the participants.
Conclusions
Evaluation of food consumption and nutrient intakes were needed to find patients with risk of undernourishment. The results underline the importance of monitoring adequate intake of calcium and vitamin D in this group of patients, especially because of the immobility-induced risk of osteoporosis. Non-ambulatory patients reported more challenges in eating and dining. This, however, did not determine the nutritional status of the patient.
Keywords
Introduction
Congenital myopathies are defined by characteristic structural abnormalities on muscle biopsy, in the case of nemaline myopathy (NM) by rod-like aggregates of sarcomeric proteins. 1 The 12 known genes causing NM are mostly genes encoding sarcomeric proteins. 2 Most often, causative variants are found in the genes encoding nebulin (NEB) and α-actin (ACTA1). These genes may also cause other congenital myopathies without nemaline rods or with unusual features.3–8 In this study, these have been named NM-related disorders (NMR).
NM and NMR constitute a spectrum of ultra-rare disorders. The severity of these disorders varies from neonatally lethal to milder forms, which do not cause any considerable decline in the patients’ physical functioning.9,10 The muscle weakness in NM and NMR often affects the muscles used for chewing, swallowing, and breathing.11–13 In addition, weakness of the facial and bulbar muscles early in life may lead to secondary structural abnormalities of the oral region, further compromising eating and swallowing.11,12 In childhood and adolescence, persons with NM are often lean, especially after the period of rapid growth. In a Finnish follow-up study of NM children, the mean decrease in relative weight was on average 2.1 SD over a period of 10 years. 14 Some scientific evidence of poorer nutritional status of patients with myopathy exists, 15 but comprehensive studies into the nutrition of persons with NM or NMR have not been done.
The aim of this pilot study was to evaluate the suitability of the methods used for comprehensively investigating food consumption, nutrient intakes, selected markers of nutrition from blood, and the self-assessed functioning related to eating and dining in NM and NMR patients in Finland. In this study, dining means the functioning of the upper extremities (cutting food, lifting a plate or glass etc.), finding a good eating posture, and dining outside the home (for example in a restaurant or canteen). Eating-related items cover structural abnormalities as well as facial muscle weakness and problems in swallowing and salivation (i.e., functioning related to the facial and oral region). In this study, the GI tract means the lower GI tract. Thus, a further aim of the study was to investigate whether poorer eating- and dining-related functioning is associated with insufficient intake of nutrients in this group of Finnish patients, with a view to a future, internationally extended study, to gain more data from a larger number of patients, and to identify any challenges related to nutrition of persons with NM or NMR.
Methods
Recruitment and workflow
The NM database at the Folkhälsan Research Center includes 38 living NM patients diagnosed in one of the five university hospitals in Finland. The invitation letter was sent to 32 patients aged over 16 years and residing in Finland. The invited persons were from 26 families. The recruitment criteria were that the diagnosis of the patient had been verified at clinical and histological examination, and the causative gene variant(s) had been identified. Patients residing abroad, who were under 16 years of age, or whose molecular genetic diagnosis had not been identified were not invited. The recruitment has been described in Lehtokari et al. 2023. 10
In the present study, a cross-sectional observational study design was used; self-reported questionnaires, food intake assessment and blood parameters.
The first part of the study was a survey including questions about background characteristics, and self-assessed functioning related to dining, eating and the gastrointestinal (GI) tract. The first part also included a food frequency questionnaire (FFQ) to investigate food consumption. When the questionnaires had been returned, the instructions for the second part, the two 4-day food records with a half-year interval, were sent to each participant. Those who returned the food records were invited to laboratory tests, and blood samples were obtained.
The participants were personally informed about their laboratory findings by a medical doctor and all participants received written feedback on their overall diet based on their nutrient intake and the FFQ-based healthy diet index (HDI) score.
The study has been approved by the Ethics Committee of the Children's Hospital, and the Helsinki University Hospital, University of Helsinki, Helsinki, Finland (9/2021). Informed consent was obtained from the participants.
Design of the survey
The survey included generic background- and health-related questions in addition to questions (items) on eating, and dining ability and as well as the functioning of the GI tract. It also included open text fields for optional additional comments. The areas of functioning were selected based on previously published data and by panelists, i.e., five persons with NM and six research group (RG) members.11,12,16
Most items addressing functioning were selected from the validated Patient-Reported Outcomes Measurement Information System (PROMIS®) item bank.17,18 The panelists selected the PROMIS® items, according to the validation procedure described in Polit et al. 2007. 19 In addition, items were selected from the National FinSote2019 Survey 20 and a study investigating nutrition in multiply disabled adults with intellectual and developmental disabilities. 21 Where suitable items were not found in existing surveys, the RG developed such items. Body mass index (BMI, kg/m2) was based on the height and weight reported by the subjects.
The survey included the following subject areas (Supplementary Table 1): dining / eating posture, 21 dining-related functioning of hands, swallowing, salivation and drooling, abdominal pain, constipation, diarrhea, gas and bloating, reflux, nausea and vomiting, and burping (PROMIS®). 17 Also, difficulties related to tongue, oral cavity, facial muscle strength, and dining outside the home (e.g., in a restaurant) were addressed (RG). In addition, any need of assistance in shopping groceries, preparing a meal, and time used for eating a meal were addressed (RG).
The responses were given on the five-point Likert scale, and they were converted as follows: point 1; indicated “no problems” and point 5 indicated “a lot of problems” or “unable to”. In other words, a higher number indicated a worse functioning or more challenges. If more than one item was used to address a subject area, sum variables were formed. The internal consistencies of the sum variables formed were tested using Cronbach's alpha. 22 If the alpha value was above 0.7, the sum variable was considered consistent. The number of items and their sources are presented in Supplementary Table 1.
The final questionnaire was created using Webropol, and the subjects could fill it out either electronically or on paper. In cases of unclear responses, specifying follow-up questions were asked by phone as described in our recent publication in which we studied the physical, psychological and social functioning of the same patient group. 10
Food frequency questionnaire and healthy diet Index
Food consumption and quality of the diet were studied using the D2D-food frequency questionnaire (FFQ), validated in the Finnish adult population.23,24 The D2D-FFQ has been designed in a Finnish type 2 diabetes prevention program, FIN-D2D. HDI, based on the D2D-FFQ, evaluates the quality of the individuals’ diet in relation to Finnish national nutrition recommendations, 25 but not the energy or nutrient intakes. D2D-FFQ includes seven weighed domains: meal pattern (max 10 points), grains (max 20 points), fruit and vegetables (max 20 points), fats (max 15 points), fish and meat (max 10 points), dairy products (max 10 points), and snacks and treats (max 15 points). The total HDI is the sum of the scores of the domains; the maximum total HDI being 100 points. The higher the HDI is, the closer it is to the Finnish nutrition recommendations.
The total HDI and the HDI scores, and the percentages of HDI maximum scores, for each domain were calculated for each subject.
Food records and calculations of nutrient intakes
To investigate energy and nutrient intakes, participants completed two 4-day food records (3 weekdays and 1 weekend day), the first one in November or December 2020 and the second one in May or June 2021. The instructions for filling in the food records and the electronic picture booklet with photographs of foods in different portion sizes were sent to participants to help in evaluating the portions when a kitchen scale was not available. The participants were instructed to report any nutrient supplements also. At the end of the recording period, the study nutritionist reviewed the record with the participant to clarify any ambiguities and flaws, making necessary amendments to the record.
The nutrient intakes were calculated using the nutrient intake calculation website Fineli, 26 based on Finnish food composition data. We report mean intakes of energy, energy-yielding nutrients, and selected micro- and macronutrients indicating the quality of the diet. Intakes of energy-yielding nutrients, i.e., carbohydrates, fat and protein, were calculated as a percentage of total energy intake (E%), including the intake of different fatty acids (FA), saturated (SFA), polyunsaturated (PUFA), and monounsaturated (MUFA). In addition, the protein intake was calculated as grams per body weight (g/kg). Intake of fiber was calculated as grams per day. The vitamin and mineral intakes were calculated separately from foods and nutrient supplements. Low energy reporters were not considered under-reporters, because low nutrient intakes might be a consequence of low food consumption due to muscle disease-associated eating problems.
The nutrient and energy intakes of participants of this study were compared with Finnish national nutrition recommendations 25 and with the intakes of the Finnish adult population. 27
Laboratory assays
Blood samples were taken at the nearest healthcare center in the morning after at least 10 h of fasting and sent to the Helsinki University Hospital laboratory (HUSLAB) where the laboratory assays were carried out. HUSLAB has been accredited by the Finnish Accreditation Service and fulfils the requirements of the standards SFS-EN ISO 15189:2013. Plasma creatinine, albumin, transferrin receptor (TfR), ferritin, parathyroid hormone (PTH), alkaline phosphatase (AFOS), phosphate (Pi), magnesium, calcium, alanine aminotransferase (ALAT), fasting total, LDL and HDL cholesterol, triglycerides, glucose, serum folate, vitamin B12 (bound to transcobalamin 2, B12-TC2), 25-hydroxy vitamin D (D-25), and blood hemoglobin (Hb) and glycated hemoglobin (HbA1c) were determined. The selected parameters are markers of nutrition and related health. Plasma creatinine mirrors muscle mass and is an indicator of kidney function.15,28 C-reactive protein was measured at the same time, as recommended, to enable considering a possible impact of inflammation on some blood parameters. 29 Albumin, on the other hand, is a carrier protein for many micronutrients.
The details of the assays are described in the HUS Diagnostic Center online guide. 30
For plasma calcium concentrations, the albumin-corrected value was used. For calculations, serum B12-TC2 results >146 were transformed to 147, TfR values <0.5 to 0.4 and ALAT <9 to 8.
Data analysis
The means, maedians, maximum and minimum values, as well as, for the variables assessing functioning, standard deviations, were calculated dividing the patients by their primary diagnosis of NM or NMR, and the patients with NM were reviewed as either non-ambulatory or ambulatory to take into account the severity of the phenotype, which usually correlates with the ability to walk. Gender is not known to affect functioning in NM or NMR and therefore the genders were not reviewed separately in terms of functioning. Nutrient intakes, laboratory parameters and HDI results were analyzed separately for females and males, because the Finnish national nutrition recommendations and reference values for blood parameters are different for some nutrients and parameters for females and males. The percentages of the participants reaching the recommended intakes and whose blood values were within the reference range were calculated. Due to the small number of participants, statistical analyses were not reliable and were not used in this study. Consequently, the results mirror the nutritional status of this group of patients, only, and should be considered as observations rather than extrapolatable data.
Results
Characteristics of the participants
Of the 32 invited, 20 participated (response rate 63%). The participants were 15 females (75% of invited) and 5 males (42% of invited) from 17 families (Table 1). The mean age of all participants was 49 years (range 19–75), of male participants 37 years and of females 51 years.
Characteristics of the participants by their gender, primary diagnosis, and walking ability.
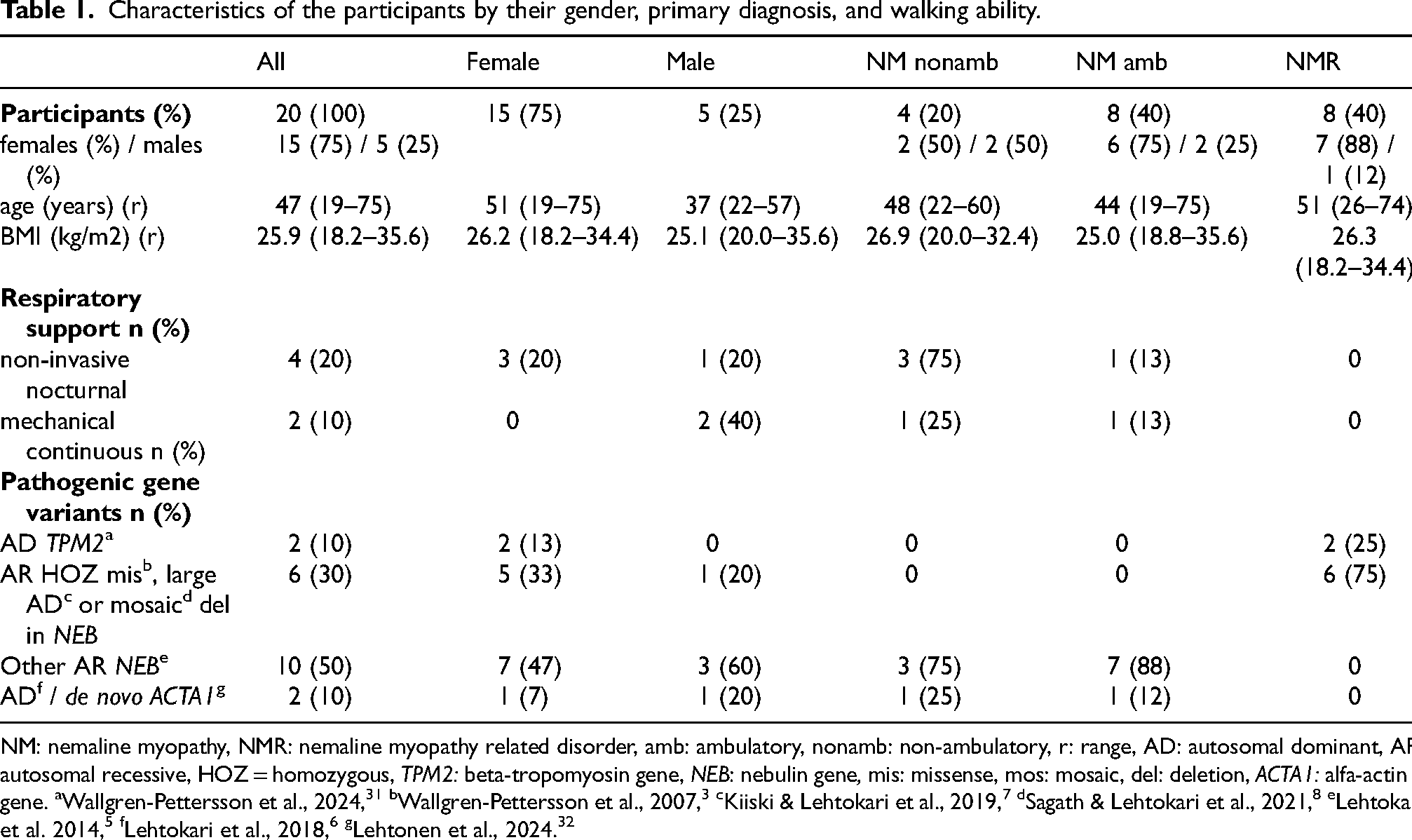
NM: nemaline myopathy, NMR: nemaline myopathy related disorder, amb: ambulatory, nonamb: non-ambulatory, r: range, AD: autosomal dominant, AR: autosomal recessive, HOZ = homozygous, TPM2: beta-tropomyosin gene, NEB: nebulin gene, mis: missense, mos: mosaic, del: deletion, ACTA1: alfa-actin 1 gene. aWallgren-Pettersson et al., 2024, 31 bWallgren-Pettersson et al., 2007, 3 cKiiski & Lehtokari et al., 2019, 7 dSagath & Lehtokari et al., 2021, 8 eLehtokari et al. 2014, 5 fLehtokari et al., 2018, 6 gLehtonen et al., 2024. 32
Out of 20 participants, 12 had NM and 8 had NMR. Of the NM patients 8 were ambulatory (6 females and 2 males), and 4 were non-ambulatory (2 females and 2 males). All 8 participants with NMR were ambulatory (7 females and 1 male). Six of the NMR patients had distal nebulinopathy and 2 an unspecified congenital myopathy with distal muscle stiffness as the predominant symptom. The clinical phenotypes and causative gene variants have been described previously.3–8,31–34
Of the 16 ambulatory patients, 11 used mobility aids or orthoses, either all the time or outdoors. Six participants with NM used respiratory support: two needed continuous invasive, and four nocturnal non-invasive support.
The body mass index (BMI) of the participants varied from 18.2 to 35.6 kg/m2. The mean reported BMI was 25.9 kg/m2. BMI was <18.5 in two participants (10%), > 25 in nine (45%) and 18.5–24.9 in nine (45%) participants.
The means, medians, max and min values of the age and BMI for all the participants, females, males, NMR, ambulatory and non-ambulatory participants are available in Supplementary Table 2.
Both participants tracheostomized reported having gained weight after the tracheostomy. They described that eating became easier because they did not have to concentrate on breathing while eating. Some of the non-ambulatory patients reported weight gain after transition to a wheelchair and some females who had children reported gaining weight postpartum.
Special diets were reported by 12 out of 20 responders; seven followed lactose-free or low-lactose, two gluten-free, and two pesco-vegetarian diets. In addition, two reported multiple food allergies and one followed a special diet due to reflux and constipation. The self-reported comorbidities diagnosed by a clinician were type 2 diabetes (one), hypercholesterolemia (one), hypothyroidism (three), osteoporosis (four; three non-ambulatory NM and one elderly NMR patient), and scoliosis (eight; three non-ambulatory NM, four ambulatory NM and one an NMR patient).
All 20 participants returned the survey and FFQ, 17 returned the food records (16 two food records and one participant one food record), and blood samples were obtained from 16 of the 17 participants who had returned the food records. One male with NMR and two ambulatory females with NM did not return the food records and one non-ambulatory male filled out the first, but not the second food record. The blood sample of one participant, a non-ambulatory male NM patient, was not obtained due to frail blood vessels.
Self-reported functioning related to eating, dining and gastrointestinal tract
In the non-ambulatory NM participants problems relating to functions requiring muscle strength, such as dining and finding a good eating posture, were more common than in ambulatory NM or NMR patients (Figure 1 and Supplementary Table 1). Especially problems relating to structural abnormalities of the oral cavity and facial muscle weakness were reported by the non-ambulatory participants. In swallowing, only minor difficulties were reported on the Likert scale, but in the open text field three non-ambulatory and one ambulatory participant with NM described problems relating to swallowing.
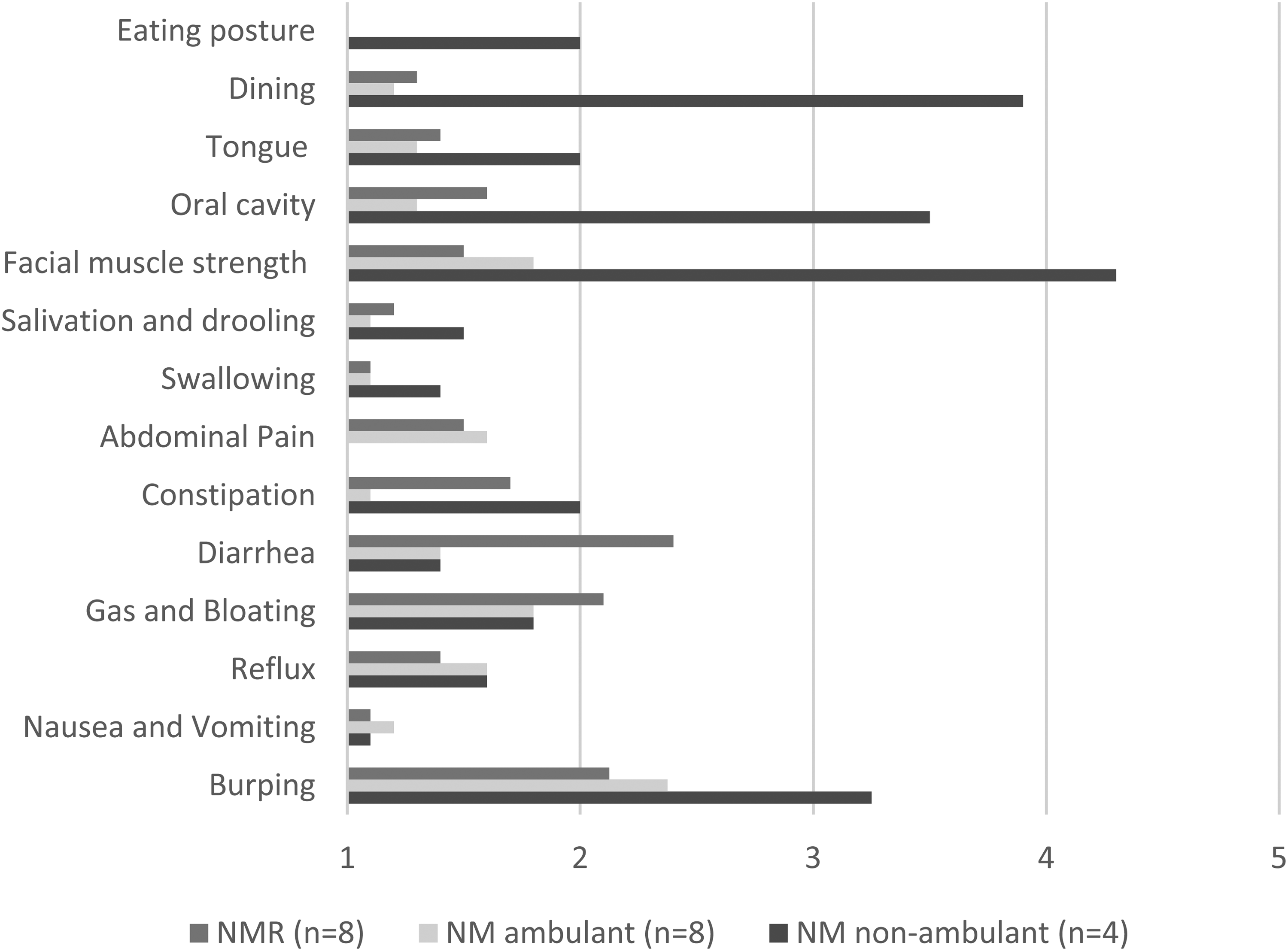
Mean scores of self-reported functioning of dining, eating, and gastrointestinal of non-ambulant and ambulant nemaline myopathy (nm) participants and participants with nm related (NMR) disorders on scale 1–5 (1 = never / no problems with / no need for assistance, 5 = lots of problems with / unable to / need for assistance in everyday life).
Problems with constipation and burping were reported more commonly by the non-ambulatory patients, whereas diarrhea, gas, and bloating where more often reported by the ambulatory patients. GI tract-related problems were usually not reported as being serious, but the inter-individual differences were large.
One non-ambulatory participant reported needing more than an hour for eating an average meal, one NMR patient and one ambulatory NM patient 30–60 min per meal and the remainder less than half an hour (Supplementary Table 1b).
All four non-ambulatory participants reported need for help on shopping groceries and preparing a meal, and three of them required assistance with dining. Of the eight ambulatory participants with NM, six reported needing assistance with shopping groceries. The vast majority of the ambulatory NM and NMR participants did not need assistance in preparing a meal or dining.
The non-ambulatory participants reported restricting dining outside the home because of difficulties in toileting, and some of them were afraid of attracting the attention of other people (Supplementary Table 1a).
The means, medians, maximum and minimum values for the self-assessed functioning for all the participants, females, males, NMR, ambulatory and non-ambulatory participants are available in Supplementary Table 2.
Consumption of foods: healthy diet Index
The mean total HDI of 20 participants was 55 (range 39–75) on the scale of 1–100 (Table 2). The mean total score was 58 (44–75) among 15 female and 48 (39–56) among the five male participants. Inter-individual differences were biggest in the consumption of fruits and vegetables (5–100) and grains (0–85), and smallest in the meal pattern (60–100) and the consumption of dairy products (27–73). On average, the participants with NM attained lower scores in the module for fruits and vegetables (31) as well as in the module of grains (36) than participants with NMR (54 and 56 respectively). The HDI score for fruits and vegetables was over 50 in only one out of 12 participants with NM (a non-ambulatory female), while in the same module HDI 50 was exceeded by five out of eight participants with NMR. The HDI for grains was over 50 in three out of 12 participants with NM and three out of eight participants with NMR. None of the four non-ambulatory participants reached HDI 50 in the module for grains. The inter-individual differences were large in all modules. The lowest and the highest individual total HDI (39 and 75) were found among the non-ambulatory NM participants.
Mean values (range) of D2D-FFQ-based Healthy diet index (HDI) (Max 100).
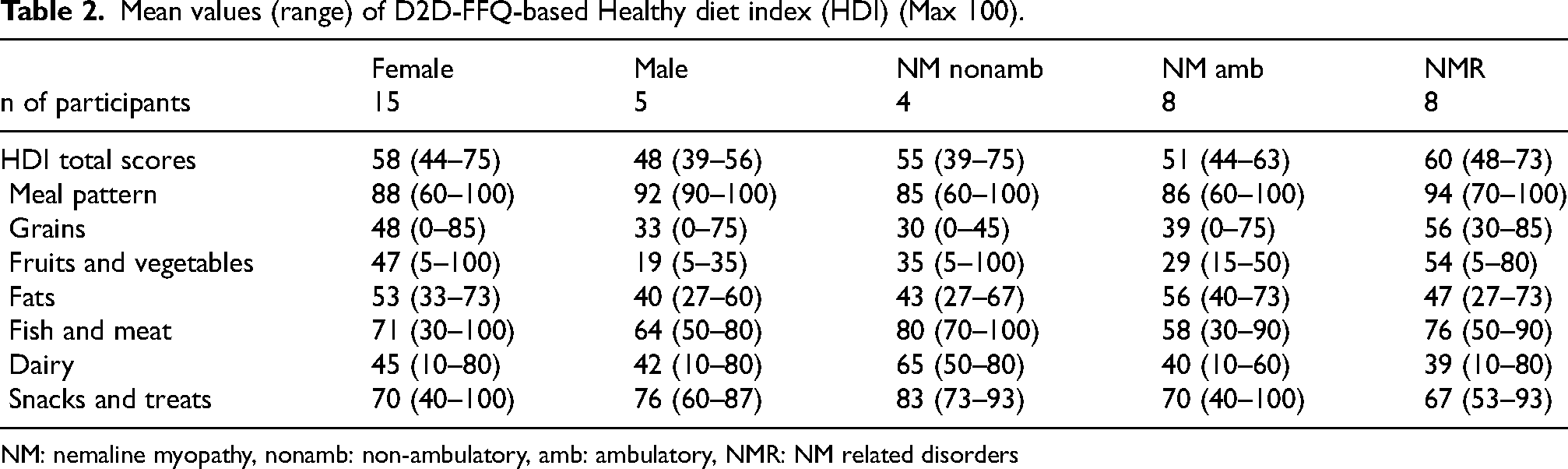
NM: nemaline myopathy, nonamb: non-ambulatory, amb: ambulatory, NMR: NM related disorders
Intake of nutrients
Food records were returned by 17 participants (13 females and 4 males). The energy intake of the participants varied between 845 and 2420 kcal; the mean energy intake of females was 1780 kcal and of men 1480 kcal (Table 3a). The mean energy intake of non-ambulatory participants (n = 4) was 1256 kcal and of ambulatory participants (n = 13) 1789 kcal respectively. The energy intakes of ambulatory NM and NMR participants were 1693 and 1870 kcal, respectively.
Mean intakes (range) of nutrients during 8 food record days (min-max) and % of participants whose intake meet Finnish Nutrition Recommendations (FNR).

Finnish nutrition recommendations 2014, b% of Finnish population whose total (food and supplement) intake meet FNR according to FinRavinto study 2017, cabove the lower intake level, UL = tolerable upper intake level of FNR (for vitamin C 1000 mg, folate 1000 µg), * intake per weight, Carboh. = carbohydrates, PUFA = polyunsaturated fatty acids (FA), MUFA = monounsaturated FA, SFA = saturated FA, suppl. = supplemented intake (total intake), **some participants exceed the upper tolerable limit.
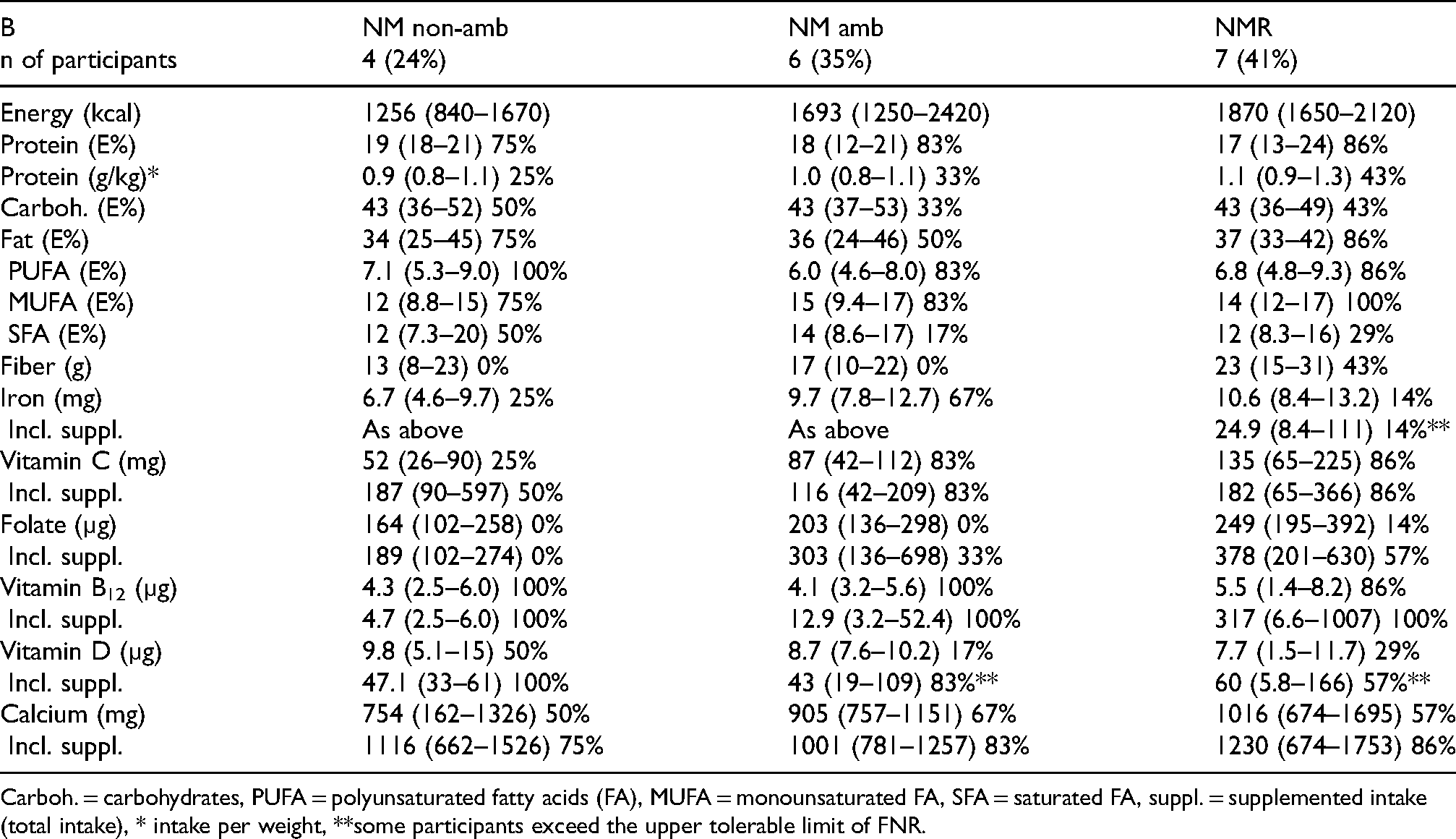
Carboh. = carbohydrates, PUFA = polyunsaturated fatty acids (FA), MUFA = monounsaturated FA, SFA = saturated FA, suppl. = supplemented intake (total intake), * intake per weight, **some participants exceed the upper tolerable limit of FNR.
The mean energy intake (E%) from carbohydrates was on average 43 E% in each subgroup (Table 3b). Seven participants reached the recommended intake of energy from carbohydrates. The mean daily fiber intake of all participants was 18 g (range 8–31 g): none of the males and only three females achieved the recommended fiber intake. None of the participants with NM, and three participants with NMR achieved the recommended fiber intake. The energy intakes of the total fat and unsaturated fatty acids were in line with the recommendation for the vast majority of participants, but 12 of the participants exceeded the recommended saturated fat intake. Protein intake as E% of all participants reached the minimum recommendation (three participants exceeded the upper recommendation). Intake of protein per body weight of most participants was slightly smaller than recommended, on average 1.0 g/kg.
The recommended iron intake was achieved by six participants (with and without supplements); one of four the non-ambulatory, and four of the six ambulatory NM participants and one of the seven participants with NMR. One patient reported use of iron supplementation (100 mg) for hemolytic anemia.
Vitamin C from foods reached the recommended level in 12, and when supplements were included, in 13 participants. The recommended level was reached by six of the seven NMR, five of the six ambulatory NM, and one (two with supplements) of the four non-ambulatory participants.
The mean intake of folate from foods was below the recommended intake in all but one participant. With supplements, four of the NMR and two of the ambulatory NM participants reached the recommended level.
The recommended intake of vitamin B12 from food was reached by 16, and when supplements were included, by all 17 participants. Two participants reported use of vitamin B12 supplementation 500 times the recommended intake (1000 µg/d). The recommended vitamin D intake was reached from food by only five participants. Vitamin D supplements were used by 16 participants and when accounting for the use of supplementation, 13 of the participants reached the recommended intake, although this led to the upper level of intake (100 µg/d) being exceeded by three participants.
The recommended calcium intake was reached by ten of the participants (14 with supplements). The non-ambulatory participants had the lowest mean calcium intake from food and even though all of them used supplementation, one of them did not reach the recommended intake.
The means, medians, maximum and minimum values for the nutrient intakes for all the participants, females, males, NMR, ambulatory and non-ambulatory participants are available in Supplementary Table 2.
Laboratory parameters and associated co-morbidities
Blood samples were gained from 16 participants. Suboptimal values outside reference values did not seem to be more common in any of the three subgroups (Table 4a and b). Albumin-corrected plasma calcium values above the reference value were, however, more commonly detected among non-ambulatory than ambulatory participants, while the serum vitamin D levels below 75 µmol/ml were detected in ambulatory patients only.
Mean values (range) and percentages of study participants whose blood values were within the reference range.
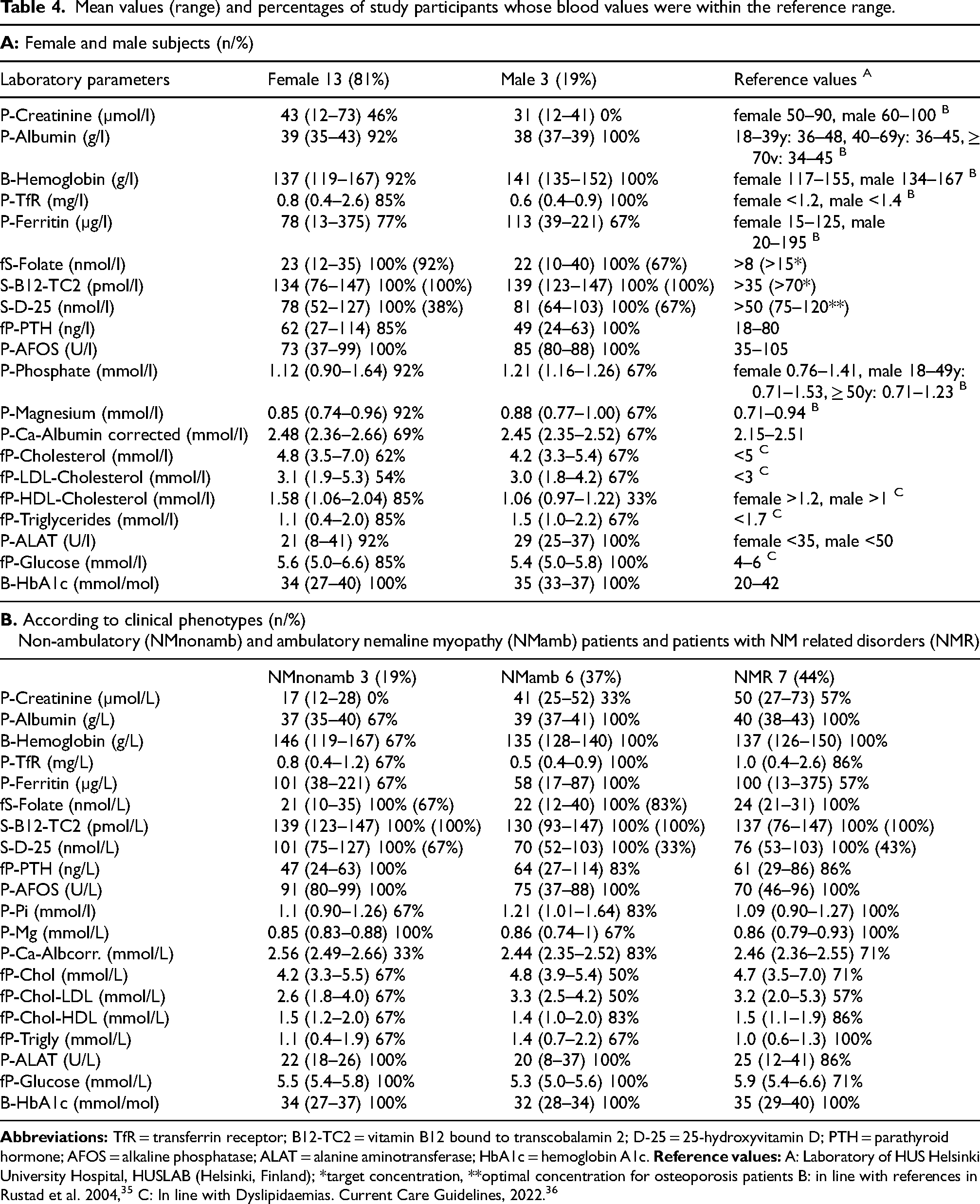
The plasma creatinine concentration was below the normal range in ten participants: in all three non-ambulatory, four of the six ambulatory NM and three of the seven NMR participants (Table 4b).
The plasma total cholesterol was above the reference value 5 mmol/l in six and LDL-cholesterol above 3 mmol/l in seven participants. In two of them, HDL-cholesterol was below and plasma triglyceride concentrations above the reference values. Both had been diagnosed with hypothyroidism, and one of them also had hypercholesterolemia. In addition, a person with high cholesterol levels had been diagnosed with hypercholesterolemia. Suboptimal cholesterol or triglyceride values were seen in all subgroups and did not appear more common in any of the three subgroups.
Of the glucose metabolism indicators, the blood HbA1c of all participants was normal. Fasting plasma glucose was above the normal range in two females, who did not report having been diagnosed with a glucose metabolism disorder. One male reported having type 2 diabetes. His glucose, cholesterol and triglyceride parameters were normal.
All three non-ambulatory participants and one elderly NMR patient sampled had been diagnosed with osteoporosis, while the bone density of the remaining 20 participants had not been investigated for osteoporosis, according to their responses. Of the bone health markers, elevated albumin-corrected plasma calcium (>2.51 mmol/l) was detected in five participants; in two of the three non-ambulatory and three of the 13 ambulatory participants sampled. The serum D-25 of all participants was above 50 nmol/l, but in eight ambulatory participants, it was below 75 nmol/l. Serum D-25 of one non-ambulatory female was above 120 nmol/l, while in other persons with osteoporosis the serum D-25 were between 74 and 103. Two ambulatory females with borderline low serum D-25 levels (55 and 52 nmol/l) had elevated PTH (86 and 114 ng/l) and one of them had elevated albumin corrected calcium level (2.55 mmol/l). Neither of them reported having osteoporosis. Elevated plasma phosphate or magnesium concentration was detected in four participants with NM.
Iron deficiency anemia was not observed in any of the participants, but blood hemoglobin of one non-ambulatory female was 167 g/l, i.e., above the normal range (plasma TfR was borderline high and plasma ferritin normal). In addition, TfR was above the normal range in one person with NMR. Plasma ferritin was low in one NMR participant and above the reference range in three participants (two NMR and one non-ambulatory participant).
The serum B12-TC2 of all participants was within the reference range (Table 4a). The serum folate of two participants was borderline low (10–12 µg/l) and of the remainder normal.
The means, medians, maximum and minimum values for the blood parameters for all the participants, females, males, NMR, ambulatory and non-ambulatory participants are available in Supplementary Table 2.
Discussion
This is, to our knowledge, the first comprehensive study using multiple methods to investigate nutrition and related self-reported functioning in persons with NM or NMR. The number of patients with the ultra-rare disorders such as NM or NMR in Finland is limited, and we do not attempt to extrapolate the results to concern all NM or NMR patients globally. Rather, our aim is to pave the way for extended studies in the future in order to gain more data on factors affecting or related to nutrition in persons with NM or NMR. The clinical symptoms and the degrees of severity, as well as the self-reported functioning of the participants varied noticeably.3–8,10,31–34 While the non-ambulatory participants reported major challenges in functioning related to eating and dining, the participants with NMR had only minor challenges, if any. The participants were categorized into three groups according to their primary diagnosis and walking ability as in our previous publication describing the self-reported functioning of the same patient group, 10 and because disease severity and walking ability likely affect the body composition (such as muscle mass) and energy expenditure. In this study, low plasma creatinine, reflecting a smaller muscle mass15,28 as well as lower energy intakes, were indeed in line with disease severity.
The BMIs of the participants varied widely (18.2–35.6). Many of them, however, had factors such as contractures and / or scoliosis affecting height, or diminished muscle mass, which complicate the interpretation of BMI and makes it an unreliable tool to evaluate a person's optimal weight. These factors underline the importance of utilizing other methods in future studies, such as bioimpedance measurement, as presented in the study by Jang and colleagues. 15
Even though obesity and dyslipidemias are globally significant health concerns, these have not been noted as common problems of persons with NM. However, 7 of the 20 participants in this study had significant or severe overweight (BMI > 30) and it was even more common in the study population than in the Finnish adult population in general. 37 On the other hand, two NMR participants (10%) were slightly under-weight. Both overweight and underweight may have significant clinical implications in NM patients. Excessive truncal fat mass increases the risk of sleep apnea, 38 and it is known that insufficient nocturnal respiration is a dangerous co-morbidity of NM, because NM often adversely affects the respiratory muscles more severely than other muscles. 13 Body mass affects bone health as well; excessive adipose tissue causes low-grade inflammation, possibly adversely affecting bone health,39,40 while low BMI is a known risk factor for osteoporosis. 41 People with physical disabilities, especially non-ambulatory persons, are at risk for osteoporosis due to immobility, which increases bone resorption and causes hypercalcemia.16,42–45 Therefore, the international recommendations for the standard of care for patients with congenital myopathies include supplementing calcium and vitamin D, and monitoring bone density. 16 Moreover, the risk for osteoporosis increases with age, especially among women, but also in ageing men.45,46
Four participants reported having a diagnosis of osteoporosis; three of them were non-ambulatory persons and one was a post-menopausal ambulatory female. Nutritional factors usually do not cause elevations in plasma calcium levels, thus the marginal elevations observed in five participants may possibly be explained by bone resorption due to immobility. 42 Two of them were non-ambulatory females diagnosed with osteoporosis while the bone density had not been examined in three others, who were ambulatory and below 50 years of age.
Serum-D-25 levels of <75 or >120 nmol/l have been suggested to predispose to osteoporosis.46–50 Although vitamin D deficiency (< 50 nmol/l) was not observed in this study (probably related to the use of vitamin D supplements), half (eight) of the patients had S-D-25 concentrations below 75 mmol/l. One person diagnosed with osteoporosis had serum D-25 concentration above 120 mmol/l, but her total vitamin D intake was below the tolerable upper intake level. 50 The intake of vitamin D of three other participants with osteoporosis was within the recommended, and their serum D-25 concentrations were 74–103 nmol/l.
The inter-individual differences in energy intakes were highly variable within the three subgroups. The mean energy intake of non-ambulatory persons was lower than that of the ambulatory participants. The energy intake was in line with the severity of the disease, and also with self-reported physical functioning. 10 The average daily energy intake of the females (1720kcal, range 1250–2110 kcal) in this study corresponded to that of the general Finnish adult female population (1756kcal), 27 while the mean energy intake of the male participants (1480 kcal, range 845–2420 kcal) was clearly lower than in the Finnish adult male population in general (2260 kcal), which may be due to the more severe phenotypes of the male participants. The non-ambulatory persons were not, however, underweight and some of them were even overweight. They reported gaining weight after starting to use a wheelchair and / or postpartum. Both participants with tracheostomy reported gaining weight after receiving a tracheostomy. They reported that when breathing had become easier, eating also became easier and they started to eat more. Both tracheostomy and transition to using a wheelchair may reduce energy expenditure and lead to weight gain.
If the energy intake is low, the intake of energy-yielding nutrient in grams may not meet the recommendations, even when the energy proportions (E%) of each energy-yielding nutrient (carbohydrate, fats and protein) were in line with the recommendations. This was seen in the protein intake of the participants in this study: the proportion of energy from protein of the total energy intake was normal or even high, although the intake in grams per body weight was rather low. The optimal protein intake of patients with congenital myopathies with relatively low muscle mass compared to the general population has not been determined. In addition, overweight complicates the interpretation of protein intake per body weight.
The number of participants exceeding the recommended maximum SFA intake and with elevated LDL-cholesterol (seven of the 16 participants sampled; one of the three males and six of the 13 females) was in line with that of the Finnish adult population. 37 The results of this study indicated dyslipidemias or risk for dyslipidemias in some of the participants, suggesting that attention to a balanced diet is important for avoiding an additional disease burden.
The low carbohydrate intake may predispose to low intake of some vitamins or minerals. Moreover, the same individuals who had low whole-grain consumption had low fiber and folate intake as well. In the Finnish population study, 27 21% of the females and 27% of the males meet the recommended fiber intake, and 5% of females and 21% of males reach the recommended folate intake. In the current study, the fiber recommendation was achieved by 23% (three) and folate from food by 10% (one) of the 13 females, while none of the male participants reached the recommended intake of fiber or folate from food. Despite this, the serum folate concentration of only two participants was borderline low, indicating that in this study, laboratory folate concentration was not an indicator of suboptimal folate intake. No blood sample of the person with the lowest folate intake was obtained due to frail vessels. Low energy intakes were seen in those individuals who had low intakes of fiber and several nutrients such as dietary fiber, vitamin C, folate, and iron. Intakes of these nutrients were clearly lower in male participants than in the Finnish adult male population. 27 According to the food records, non-ambulatory patients had on average lower nutrient intake, compared with ambulatory participants.
None of the 16 patients whose blood samples were analyzed showed severe undernourishment in the selected laboratory parameters, nor did the laboratory parameters reveal any congruent factors common to all participants or subgroups. The high ferritin level detected in some participants may be a consequence of a secondary cause rather than of nutrition. 29
Strengths and limitations
Study strengths include the use of several different methods to measure nutrient-related parameters, providing a more nuanced picture. The age range of the participants in the current study who filled the food records corresponds to the age range of the Finnish FinRavinto2017 population study (18–74 years), and the return rate of food records (53% of the invited) is similar to that of the Finnish population study, 27 enabling comparison of our results with the Finnish population data.
Selection bias cannot be excluded as the response rate was 63% for the food consumption questionnaire, 53% for food records and 50% for the laboratory measurements. In addition, only 25% of the participants were males, and 50% of males were non-ambulatory. All participants with NMR who returned the food record were females. Therefore, given the small study sample, the impact of random variation increases, which needs to be accounted for when interpreting the results. The small study sample did not allow for reliable statistical analyses. Thus, the generalizability of our results may be limited, which is often the case when studying ultra-rare disorders.
When the number of the participants is low, the use of mean values carries a risk for biased interpretations, as the mean values may be altered by one deviant value. To avoid this, we have specified the variation within each group and across the participants. We hope to expand this pilot work in the future internationally and nationally to be able to identify any significant differences between the groups.
Conclusions
In this clinically heterogeneous study population of NM and NMR patients, self-reported functioning related to dining and eating and consumption of foods as well as nutrient intakes were highly variable between the individuals. The variation was large also within the group of participants with the greatest disease severity. BMI, as well, varied from low to considerable overweight in this study population.
Taken together, the results indicate that especially patients with NM may be prone to problems with eating- and dining-related functioning, low consumption of healthy foods and low intake of energy and other nutrients and deteriorated bone health. At the same time, some of the patients are prone to overweight. Although, the laboratory parameters did not indicate severe undernourishment in the participants, evaluation of eating-related functioning, food consumption, nutrient intakes and selected laboratory parameters can help in identifying patients at risk.
The methods used are likely to be useful for similar studies in the future, if the study is expanded to other muscle disorders in Finland, or internationally.
Supplemental Material
sj-xlsx-1-jnd-10.1177_22143602241303374 - Supplemental material for Nutritional status of patients with nemaline myopathy and related congenital myopathies in Finland: A pilot study
Supplemental material, sj-xlsx-1-jnd-10.1177_22143602241303374 for Nutritional status of patients with nemaline myopathy and related congenital myopathies in Finland: A pilot study by Vilma-Lotta Lehtokari, Minna Similä, Marianne Tammepuu, Pirjo Isohanni, Mari Auranen, Sinikka Hiekkala, Carina Wallgren-Pettersson and Sonja Strang-Karlsson in Journal of Neuromuscular Diseases
Supplemental Material
sj-xlsx-2-jnd-10.1177_22143602241303374 - Supplemental material for Nutritional status of patients with nemaline myopathy and related congenital myopathies in Finland: A pilot study
Supplemental material, sj-xlsx-2-jnd-10.1177_22143602241303374 for Nutritional status of patients with nemaline myopathy and related congenital myopathies in Finland: A pilot study by Vilma-Lotta Lehtokari, Minna Similä, Marianne Tammepuu, Pirjo Isohanni, Mari Auranen, Sinikka Hiekkala, Carina Wallgren-Pettersson and Sonja Strang-Karlsson in Journal of Neuromuscular Diseases
Footnotes
Acknowledgements
We are grateful to Jaana Lindström, Katri Hemiö, and Kirsikka Aittola for all their advice in using the D2D-FFQ -tool, research assistant Marilotta Turunen for irreplaceable help with practical measures, and Lydia Sagath for technical assistance. In addition, this work would not have been possible without the patients who participated and enabled this study.
The authors PI, MA and CWP are members of the European Reference Network for Rare Neuromuscular Diseases (ERN EURO-NMD).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study has been supported by the Otto A. Malm Foundation, the Finnish Brain Foundation, the Sigrid Jusélius Foundation, the Finska Läkaresällskapet, the Medicinska understödsföreningen Liv och Hälsa, and the Helsinki University Hospital Special Funding for University Level Health Research. Open access funded by Helsinki University Library.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The raw data is in Finnish. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
