Abstract
Background:
To date, assistive gait devices (AGDs) for people with neuromuscular diseases (NMDs) have not been systematically evaluated.
Objective:
This systematic review evaluated AGDs for people with NMDs.
Methods
Suitable tools were used to assess the study quality and the certainty of evidence. If feasible, a descriptive and quantitative synthesis was conducted.
Results:
Forty studies were included on: gait-assisting exoskeletons (GAEs), orthopaedic footwear (OF), knee and/or ankle-foot-orthoses (AFOs) and non-invasive neuroprostheses that provide functional electrical stimulation (FES). Pairwise meta-analysis could not be performed due to a lack of homogenous data. The within-group pre-post meta-analysis in GAE studies showed an improvement in 2-min walk test (2MWT) (m) (standardized mean difference [SMD] = 0.36, 95%-CI: 0.17 to 0.54), but not in 10-meter walk test (10MWT) (sec) (SMD = 0.33, 95%-CI: −0.10 to 0.76) and in functional independence measure (FIM) (SMD = −0.002, 95%-CI: −0.21 to 0.21). In AFO studies, there was no improvement in 10MWT(m) (SMD = −0.11, 95%-CI: −0.76 to 0.53). A single session with AFO or OF vs no-AFO or OF post-intervention meta-analysis did not reveal an improvement in walking speed (m/s) (SMD = 0.39, 95%-CI: −0.03 to 0.83).
Discussion:
GAEs seem to help patients to walk longer. The meta-analyses showed no significant results to support the effect of AFOs or OF. Neuroprostheses and knee orthoses showed no evidence.
Conclusion:
No clear conclusions can be drawn on how AGDs affect NMDs. The positive AGD effects are based on very low certainty of evidence.
Introduction
The term neuromuscular diseases (NMDs) encompasses a large number of different disorders that are localized either in the motor anterior horn cells of the brainstem, the spinal cord, the peripheral nerves, in the structures of the motor end plates of striated muscles, or the muscle fibers. 1 Common features and symptoms of NMDs include muscle weakness, foot lesions, balance problems, pain, fatigue, and limitations in activities of daily living. 2 The survey by Jerath et al. 3 summarized the functional priorities of patients with disabilities due to NMDs in the upper and lower extremities. According to their results, if given a choice between upper and lower extremity functional correction, 45% of patients would prefer lower extremity correction. The most common activities for which patients wanted to improve their independence were mobility and transfers (46%), followed by toilet use and hygiene (32%). 3 In the last decade, new assistive gait devices (AGDs), mostly adapted from other technical and engineering areas, have been applied to replace a loss of motor function or improve patients’ quality of life. We define the term AGD as assistive devices worn on the lower limb(s) that directly assist the gait of the patient's lower limb(s) in performing a functional movement, including gait-assisting exoskeletons (GAEs), knee and/or ankle-foot orthoses (AFOs), orthopaedic footwear (OF), and non-invasive neuroprostheses that provide functional electrical stimulation (FES). It is important to distinguish between assistive devices used in daily living and substitution and rehabilitation devices used in rehabilitation programs. Our definition of AGDs includes devices used to assist patients with walking activities of daily living. It does not include, in the case of GAEs, rehabilitation devices such as a robotic treadmill training system (lokomat) and an end-effector-based gait trainer or, in the case of AFOs, automatic mechanized knee or foot flexion devices used for passive extension of the joint. For neuroprostheses, it does not include devices and systems that apply somatosensory stimulation using transcutaneous electrical nerve stimulation (TENS). The neuroprosthesis refers to the use of non-invasive functional electrical stimulation, applied to the peripheral leg nerves, to activate muscles during the swing or stand phase of ambulation. It provides active dorsiflexion and may reduce foot drop by facilitating voluntary muscle activity and can be used as an alternative to the AFOs.4,5
Especially in slow-advancing NMDs, patients and medical staff are interested in maintaining mobility and independence in activities of daily living.6,7 A recent systematic review 8 summarized current evidence on the effectiveness of wearable assistive technologies for upper limb support during activities of daily living for patients with NMDs. However, our preliminary PubMed search did not identify any systematic reviews that summarized the effects of different commonly used AGDs by individuals with NMDs. We found systematic reviews summarized only one of the possible commonly used AGDs separately, such as knee and/or ankle-foot orthoses (AFOs),9,10 gait-assisting exoskeletons (GAEs)11–13 or orthotic devices for knee or foot instability 14 in neurological and orthopaedic diseases generally. However, we did not find any review that summarized and evaluated the clinical effects of the most commonly used AGDs specified for NMDs only.
In the field of NMDs, clinicians are challenged to help patients by clarifying options and making the right choice between different AGDs. Knowledge of appropriate evidence-based AGDs in the heterogeneous field of NMD gait disorders could help clinicians determine which appropriate clinical outcome can be used when testing and prescribing AGDs. Due to the rare disease phenotypes and their heterogeneity, knowledge about intervention and pharmacological treatment of NMDs is mainly based on observations and case studies. 15 It is believed that these data can provide sufficient evidence if the study structure and risk of bias are reduced to a minimum. 16 If the majority of identified studies are pre-post studies, case reports, and case series, it will be challenging to identify sufficiently homogenous outcome data and to assess the overall body of evidence. Randomized control trials (RCTs) are considered the most robust research design for investigating the cause-and-effect mechanisms of interventions. 17 However, when RCTs are unavailable, other study designs and alternative limited methods, such as pre-post meta-analysis, should be considered. Such alternative methods have already been proposed 18 or employed 19 to synthesize and grade the evidence from study designs that are not-RCTs.
Considering the evidence gap described above, the purpose of the present systematic review was to evaluate the kinematics and kinetics gait measures (velocity, step time, step length) and functional assessments, such as scores, scales and functional tests, used to assess the gait pattern effectiveness of different commonly used AGDs that can be integrated into the daily lives of people with NMDs.
Objectives: First, to assess the efficacy of AGD interventions on NMD patientś gait patterns and activities of daily living. Second, two types of meta-analyses were planned. A conventional pairwise meta-analysis in which the mean difference between AGD and their comparison group was calculated, and a pre-post meta-analysis of AGD intervention groups were performed.
Materials and methods
Study selection
The study protocol was prospectively registered at the open science framework (OSF) with the registration: https://doi.org/10.17605/OSF.IO/28M65 on the 18th of November 2022, and a final updated protocol was registered on the 02th of January 2024. The PICO 20 model was implemented to answer the primary clinical question (Table 1). This systematic review used “The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement 2020”. 21 All search strategies are available on the OSF project registry (https://doi.org/10.17605/OSF.IO/TDHK4). The research team defined inclusion and exclusion criteria in advance. Reference sections of relevant reviews and research articles were used to identify additional pertinent articles. Screening of all records and reports published in English and German was performed independently by two authors (MM, CWP) using the Rayyan QCRI software. 22 No automated tools were used in the process. Disagreements between reviewers were resolved by consulting a third and fourth reviewer (PY, BS). If required, additional information was requested from the article authors.
PICOS Consort criteria for primary question.

The final search strategy via Pubmed was: (neuromuscular disease*[Title/Abstract]) OR (neuromuscular disorder*[Title/Abstract]) AND (foot orthose*[Title/Abstract]) OR (AFO[Title/Abstract]) OR (ankle foot orthose*[Title/Abstract]) OR (orthotic device*[Title/Abstract]) OR (brace*[Title/Abstract]) OR (knee orthose*[Title/Abstract]) OR (exoskeleton*[Title/Abstract]) OR (neural prosthese*[Title/Abstract]) OR (man machine system*[Title/Abstract]) OR (assistive device*[Title/Abstract]) OR (hybrid assistive limb[Title/Abstract]) OR (cyborg*[Title/Abstract]) OR (functional electrical stimulation[Title/Abstract]) OR (end-effector device*[Title/Abstract]) using following filters: Humans, English, German, Studies from (2010–2024).
A team of nine healthcare professionals, including three physiotherapists (MM, CWP, AJ) and six physicians (KG, MA, NG, SW, PY, BS), designed the study, including the aim with primary outcome measurements, the search strategy, and its eligibility criteria. The following databases were searched: Pubmed/MEDLINE®, Web of Science, Scopus, Embase, and MEDLINE (via EBSCO). The search was limited to studies published between 01/Jan/2010 and 01/Jan/2024, as the preliminary PubMed search revealed the first studies written on the topic of GAEs.
As different types of trials were included in this review, it was necessary to use various tools to assess the risk of bias. For the management and assessment of internal validity of case reports/studies and case series, the steps and instructions described in the study protocol by Nambiema et al. 18 were adopted. We followed recommendations from the Cochrane Handbook 23 to assess the risk of bias in observational studies that compare the health effects of two or more interventions, using the Cochrane Risk of Bias in Non-randomised controlled Trials (NRCT) tool (ROBINS-I) developed by Sterne et al. 24 Finally, we used the Cochrane risk-of-bias tool for randomized trials (RoB 2). 25
Inclusion criteria
The AGDs and types of studies included are in PICO Table 1. Studies were included if participants were assigned to one or more devices, the effects of device use were compared with no device use or alternative interventions, or there was no control group. The following NMDs were eligible for inclusion: amyotrophic lateral sclerosis, chronic inflammatory demyelinating polyradiculoneuropathy, inclusion body myositis, dystrophinopathies, limb-girdle and facioscapulohumeral muscular dystrophies, myotonic dystrophies, myasthenia gravis, Charcot-Marie-Tooth disease (synonym for group of hereditary motor and sensory neuropathies), Pompe disease, and spinal muscular atrophy. Moreover, we included degenerative diseases such as spinocerebellar degeneration, spinocerebellar ataxia, or hereditary and spontaneous spastic paraparesis.
To evaluate the patients’ improvement using AGD, clinical outcomes assessing gait ability based on time, distance or metabolic energy consumption were included when the device was used in a clinical setting or domestic environment, e.g., Stair climbing, Chair rise, 6-min walk test or 10-meter walking test (10MWT). Other assessment tools to measure patient independence, i.e., gait stability and dependence during walking in a domestic environment or fatigue, were included e.g., Fugl-Meyer Assessment of Lower Extremity or Berg Balance Scale, etc.
Exclusion criteria
The following diagnoses were not included: stroke, spinal cord injury, diabetic neuropathy, and mechanical nerve compression conditions. In addition, the following assistive devices and equipment were excluded: lokomat, treadmills, end-effector-based gait trainers, walkers, crutches or canes, manual and electric wheelchairs (with and without powered standing function), implanted devices, e.g., joint replacements, implanted brain or muscle stimulation and somatosensory stimulation using transcutaneous electrical nerve stimulation (TENS).
Data extraction
Data regarding the number of subjects, study design, type of assistive device, duration of treatment and training protocol was extracted from each study. An important basis for evaluating an AGD is whether it satisfies the needs of the disabled consumer. However, the factors consumers consider when deciding whether a device meets their needs are unknown. 26 Therefore, we additionally searched and extracted data from primary included studies that examined the benefits, limitations, acceptance, and implementation of AGDs in activities of daily living from the perspectives of patients, caregivers, and medical staff. All included surveys and questionnaires were only subjected to a qualitative synthesis by describing their significance. No other instrument was used to assess psychometric properties.
Methodological quality
Using an adapted protocol from Nambiema et al., 18 case reports/studies and case series were first assessed to determine whether they were well documented, scientifically rigorous and followed ethical practices according to the CARE (Case Reports) guidelines,27,28 and the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Case reports/studies and Case Series. 29 The CARE guidelines for case reports help authors reduce the risk of bias, increase transparency, and provide early signals of what works, for which patients, and under which circumstances. Case reports following the CARE guidelines support the measurement of clinician- and patient-assessed outcomes, the effectiveness of clinical practice guidelines and the return on investment. 28 Subsequently, the risk of bias within included case studies was assessed using the Navigation Guide tool,30,31 which covers nine domains of bias for human studies: (a) source population representation; (b) blinding; (c) exposure or intervention assessment; (d) outcome assessment; (e) confounding; (f) incomplete outcome data; (g) selective outcome reporting; (h) conflict of interest; and (i) other sources of bias. For each section of the tool, the risk of bias was rated as “low risk”; “probably low risk”; “probably risk”; “high risk”; or “not applicable.” Finally, the overall risk of bias in individual studies was assessed.
The “Quality Assessment Tool for Before-After (Pre-Post)”, developed by the National Institutes of Health (NIH), was used to rate the methodological quality of pre-post studies without a control group. 32 The questions in the NIH quality assessment tool were designed to help reviewers focus on the key concepts for evaluating the internal validity of a study. Critical appraisal of a study involves considering the potential for selection bias, information bias, measurement bias, or confounding. A high risk of bias translates to a rating of poor quality; a low risk of bias translates to a rating of fair and good quality. 32
The Cochrane risk of bias in NRCTs tool (ROBINS-I) 24 was used to assess the risk of bias in observational studies that compare the health effects of two or more interventions. ROBINS-I is a tool for evaluating the risk of bias in estimating the comparative effectiveness (harm or benefit) of interventions from studies that did not use randomization to allocate units (individuals or clusters of individuals) to compare groups. ROBINS-I fundamental underlying principle is to compare the risk of bias associated with the current evaluated NRCT with a target RCT hypothesized to be conducted with the same group of participants, even though this RCT may not be feasible or ethical. 33 The ROBINS-I tool includes seven domains to assess the risk of bias that may arise in a NRCT: (I) bias due to confounding; (II) bias in the selection of participants into the study; (III) bias in classification of interventions; (IV) bias due to deviations from intended interventions; (V) bias due to missing data; (VI) bias in measurement of outcomes (or detection bias); (VII) bias in selections of the reported results. The categories for risk of bias judgments are: “low risk”, “moderate risk”, “serious risk” and “critical risk”. The risk of bias is assessed for each domain, and then the overall judgment of the study's risk of bias is made. 24
The Cochrane Risk of Bias Tool (RoB 2) 25 for RCTs was used to assess the risk of bias in a single estimate of an intervention effect reported from a randomized trial. RoB 2 is structured into a fixed set of domains of bias, focusing on different aspects of trial design, conduct, and reporting. Within each domain, a series of questions (‘signaling questions’) aim to elicit information about features of the trial that are relevant to risk of bias. The domains included in RoB 2 cover all types of bias currently understood to affect the results of randomized trials. These are: (I) bias arising from the randomization process; (II) bias due to deviations from intended interventions; (III) bias due to missing outcome data; (IV) bias in measurement of the outcome; and (V) bias in selection of the reported result.
Data synthesis and meta-analysis
If a quantitative synthesis was not possible, a qualitative synthesis was carried out by presenting the significance of the outcome between the AGD and specific NMD. As we included RCTs and NRCTs, pre-post studies without a control group and case series, not all results found could be combined systematically. We are aware of the Cochrane recommendation 23 that different study designs should be expected to differ systematically, resulting in increased heterogeneity, and that observational studies which used different study designs (or which have different design features), or RCTs and NRCTs, should not be combined in a meta-analysis. If this was impossible due to clinical heterogeneity, only a pre-post meta-analysis was performed. A similar quantitative synthesis methodology has already been used in the study by Aspinall et al. 19 Meta-analyses were performed with the Comprehensive Meta-Analysis V4 (Biostat, Inc., USA) software, using standardized mean difference (SMD) and standard deviation with a random effects model to calculate the change from baseline for each outcome separately. Numerous studies did not report the standard deviation of the mean change but provided other data that allowed the calculation of standard deviations as follows. If mean change from baseline with either 95% confidence intervals or standard error were provided, standard deviations were calculated based on formulae from section 7.7.3.2. according to the Cochrane Handbook. 23 The Comprehensive Meta-Analysis V4 software can calculate this data automatically according to Cochrane Handbook guidelines. To calculate the pre-post intervention effect besides mean change and standard deviation, a correlation between the pre- and post-results is needed, as recommended by Borenstein et al. 34 Since we could not identify any published estimates of the correlation between pre- and post-outcomes, we ran a sensitivity analysis by calculating the variances twice, based on high and low assumed correlations (0.75 and 0.25, respectively). We found minimal variation in the meta-analysis results whether we assumed a correlation of 0.75 or 0.25 for calculations of combined variance. Therefore, the decision was made to use a 0.75 correlation. The meta-analysis did not include case studies that included less than three participants. Moreover, a meta-analysis was performed if a minimum of three studies with at least 20 participants were found. A subgroup analysis divided by each disease separately was planned if sufficient data were established.
The overall certainty of evidence
The overall certainty of evidence and strength of recommendation was assessed using the Grades of Recommendation Assessment, Development, and Evaluation (GRADE) handbook methodology. 35 According to the GRADE approach, the certainty of evidence is graded as high, moderate, low, or very low. Furthermore, a body of evidence from observational studies begins with a low certainty of evidence rating, which can be downgraded for five reasons: risk of bias, indirectness, inconsistency, imprecision and publication bias. Three factors permit rating up the certainty of evidence: large magnitude of an effect, dose-response gradient, and effect of plausible residual confounding. 35
Results
The systematic search yielded 13430 references after excluding 2915 duplicate studies using Rayyan QCRI software. 22 The search from PubMed database resulted in 9351 records, Embase database in 892 records, MEDLINE database (via EBSCO) in 706 records, and the Web of Science/Clarivate resulted in 5396 records. After screening of records, 304 reports were included for full-text reading. Additionally, 7 articles were found after the screening of reference lists. Finally, forty articles were included in the review after applying the inclusion and exclusion criteria. The PRISMA flow diagram presents the search process (Figure 1).
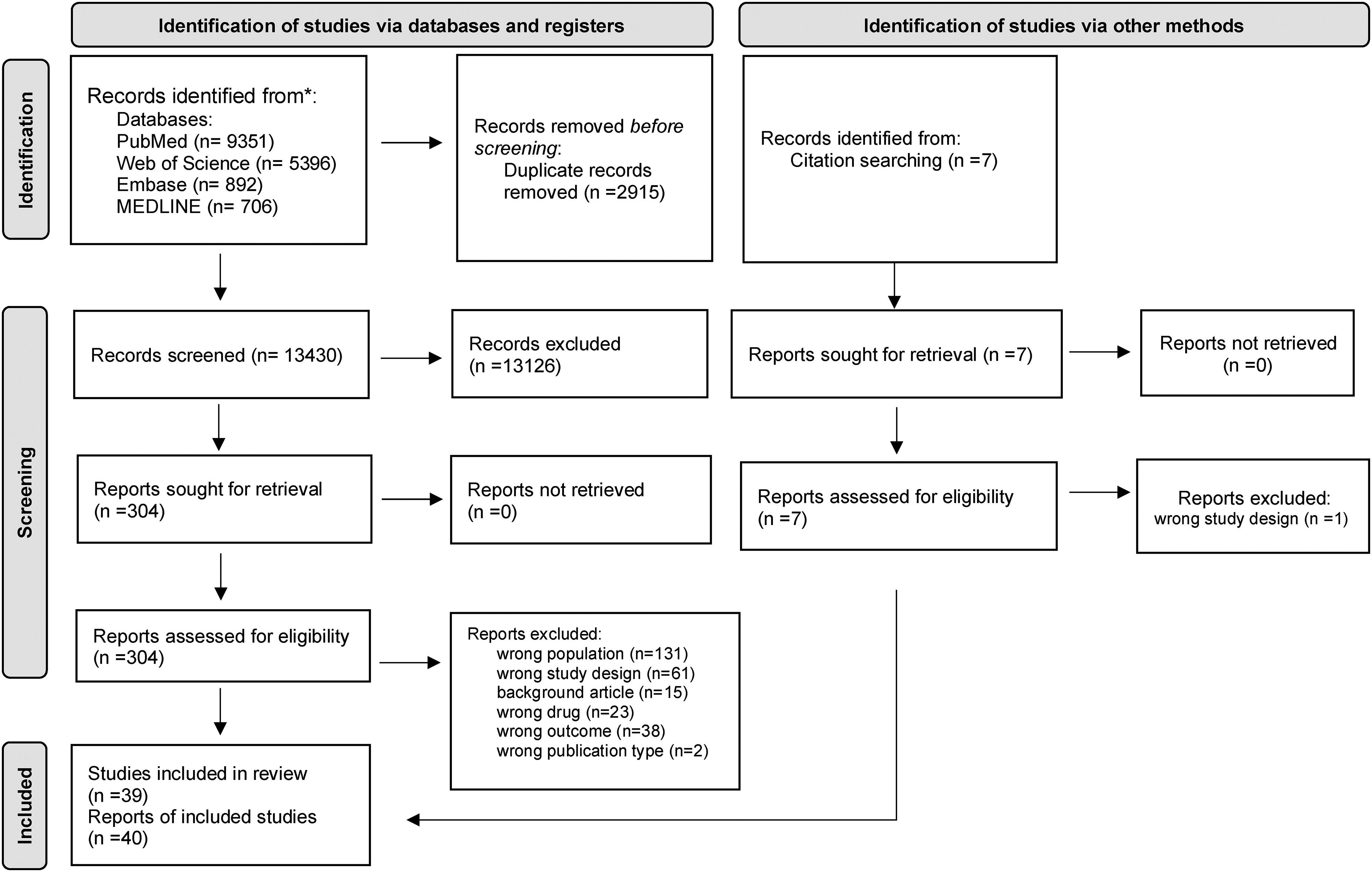
PRISMA flow-chart.
Assistive gait devices, outcome measurements, and neuromuscular disorder
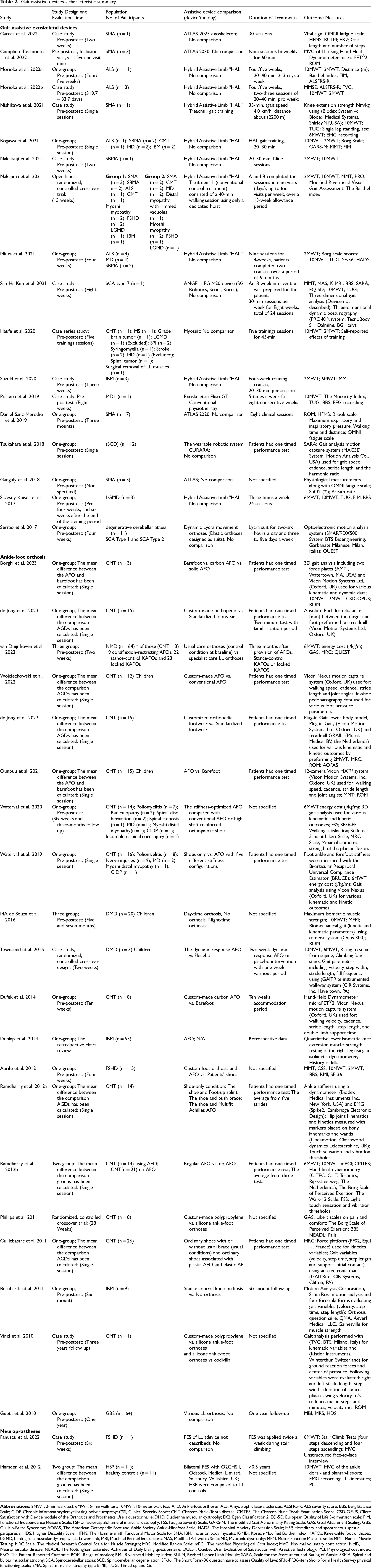
From forty included studies, eighteen studies tested various GAEs36–53 and nineteen studies tested various AFOs, braces or orthopaedic footwear (OF).54–72 One study tested a stance-control 63 and one a locked-knee-ankle orthosis and an AFO. 66 Two studies tested neuroprostheses73,74 (Table 2). The total amount of participants in the reviewed studies was 596. The following diseases were investigated: spinal muscular atrophy (n = 20), amyotrophic lateral sclerosis (n = 30), spinal and bulbar muscular atrophy (n = 7), Charcot-Marie-Tooth disease (n = 190), myotonic dystrophy (n = 13), facioscapulohumeral muscular dystrophy (n = 19), limb-girdle muscular dystrophy (n = 6), Miyoshi myopathy (n = 6), Duchenne muscular dystrophy (n = 38), inclusion body myositis (n = 68), other NMDs (n = 183), and other non-NMD pathologies (n = 16). The most commonly assessed outcomes in studies evaluating GAEs were 10MWT, 2MWT, Functional Independence Measure (FIM), Time up and go test and Borg Scale score. In studies evaluating AFOs, the most commonly assessed outcomes were a 6-min walk test, 10MWT, as well as gait speed, cadence, stride length and joint angles using the Vicon Nexus (Oxford, UK) motion capture system. In studies using neuroprosthesis, maximal voluntary contraction and a 6-min walk test were rated. All data are summarized in Table 2.
Gait assistive devices - characteristic summary.
Methodological quality
Two RCTs (Figure 2), three NRCTs (Figure 3), twenty-two pre-post studies without control group (Table 3) and thirteen case studies (Table 4) were included. Although the studies51,61 used randomisation, no low overall risk of bias judgment using RoB 2 tool was found (Figure 2). Moreover, low overall risk of bias in NRCTs,59,72,74 which can be compared to a well-performed randomized trial, was not found in any of the included studies. Furthermore, two studies were classified as having serious risk of bias59,74 using the ROBINS-I tool. One study was evaluated with moderate risk of bias due to confounding bias, selection of participants, and bias in measurement of outcomes 72 (Figure 3).

Risk-of-bias of included studies according to the RoB2 tool.

Risk-of-bias of included studies according to the ROBINS-I tool.
Methodological quality of included studies according to the “Quality Assessment Tool for Before-After (Pre-Post)”.

Abbreviation: CD, Cannot Determine.
The risk of bias for case reports/studies using the Navigation Guide tool.
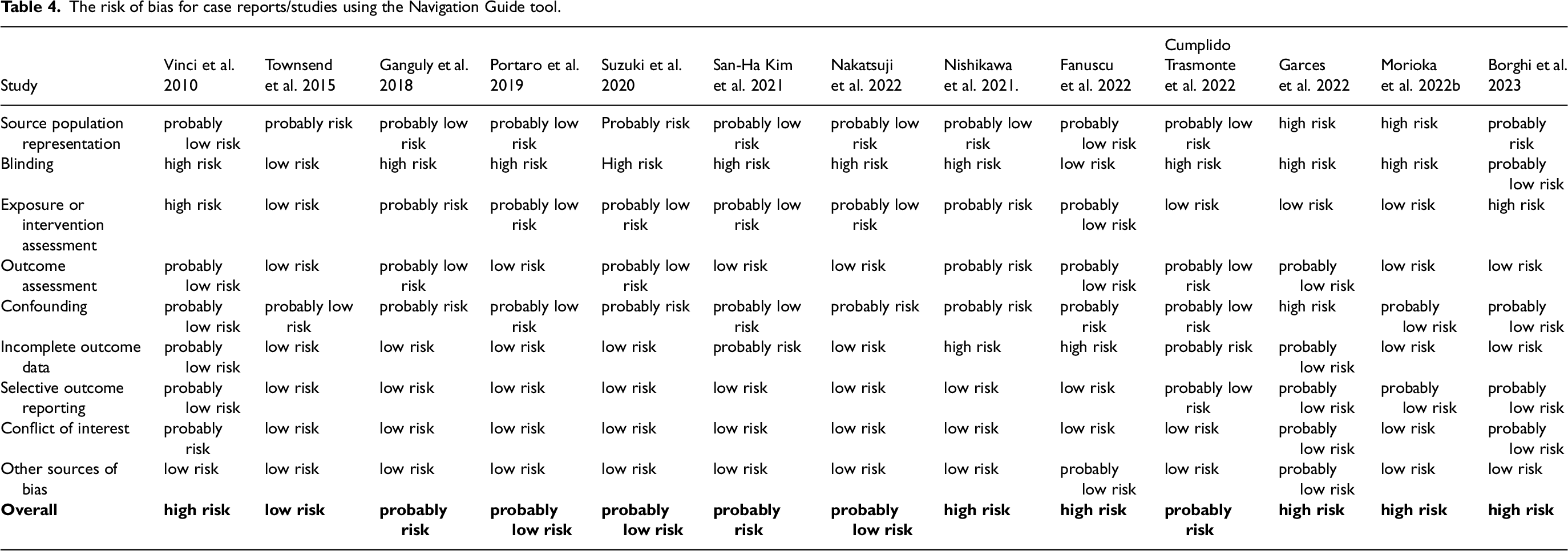
By the NIH tool, six pre-post studies without a control group had good,42,52,55,67,70,71 twelve studies had fair,45,47,49,57,58,60,63,65,66,68,69,75 and four had poor38,41,44,62 methodological quality (Table 3). The eligibility criteria and the outcome measures of pre-post studies were prespecified, clearly defined, valid, reliable, and assessed consistently across all study participants. According to the NIH tool, the sample size should be large enough to provide confidence in the findings. Only three studies66,70,71 met this critical criterion. Moreover, outcome assessors should be blinded to the participantś exposures and interventions. None of the studies used a blinded design. Thirteen pilot studies using the CARE Checklist indicated sufficient methodological transparency to be included in further evaluation (Appendix A). Using the Navigation Guide methodology tool to evaluate risk of bias determinations, we found one case study with low risk of bias. 56 Three studies had probably low risk,39,40,50 three probably risk,37,43,53 and six high risk of bias.36,46,48,54,64,73 The risk of bias for case reports/studies using the Navigation Guide tool is presented in Table 4.
Synthesis of results
A conventional pairwise meta-analysis was impossible due to differences in interventions and outcome assessments in included RCTs and NRCTs. Pre-post meta-analysis was performed for the outcomes 2MWT, 10MWT, FIM with GAE (Figure 4) and 10MWT with AFO (Figure 5). In addition, a matched group post-intervention meta-analysis was performed between AFO and no AFO use for the outcome walking speed (m/s) (Figure 5). Neuroprostheses were restricted to eligible studies. Although sufficient GAE and AFO data were found for the outcomes range of motion, manual muscle testing and 3D motion analysis, the researchers examined different sets of muscles and joints, which did not allow us to perform a meta-analysis. No subgroup analysis was conducted.
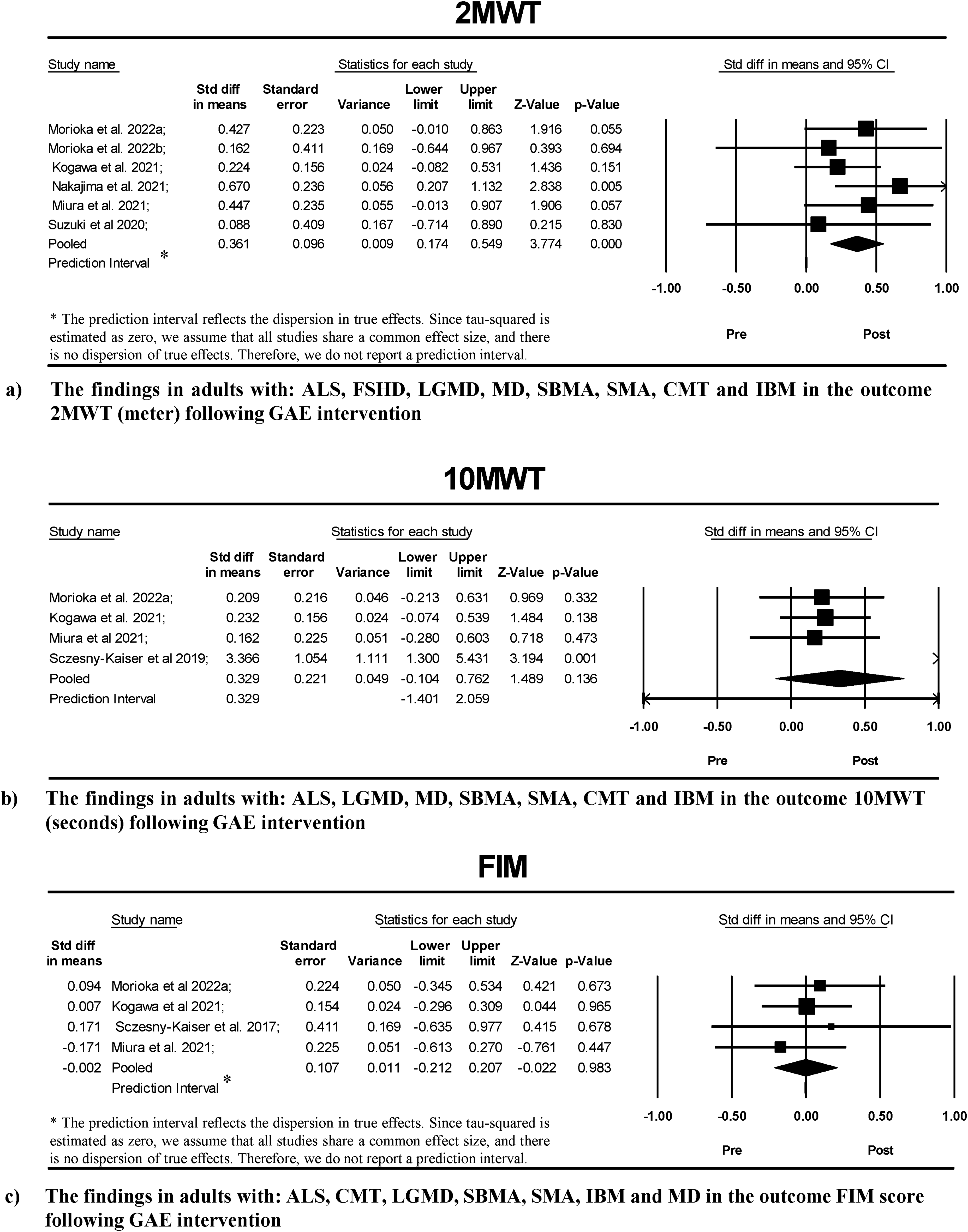
Meta-analyses of GAE intervention in pre-post outcomes: (a) 2MWT, (b) 10MWT and (c) FIM.
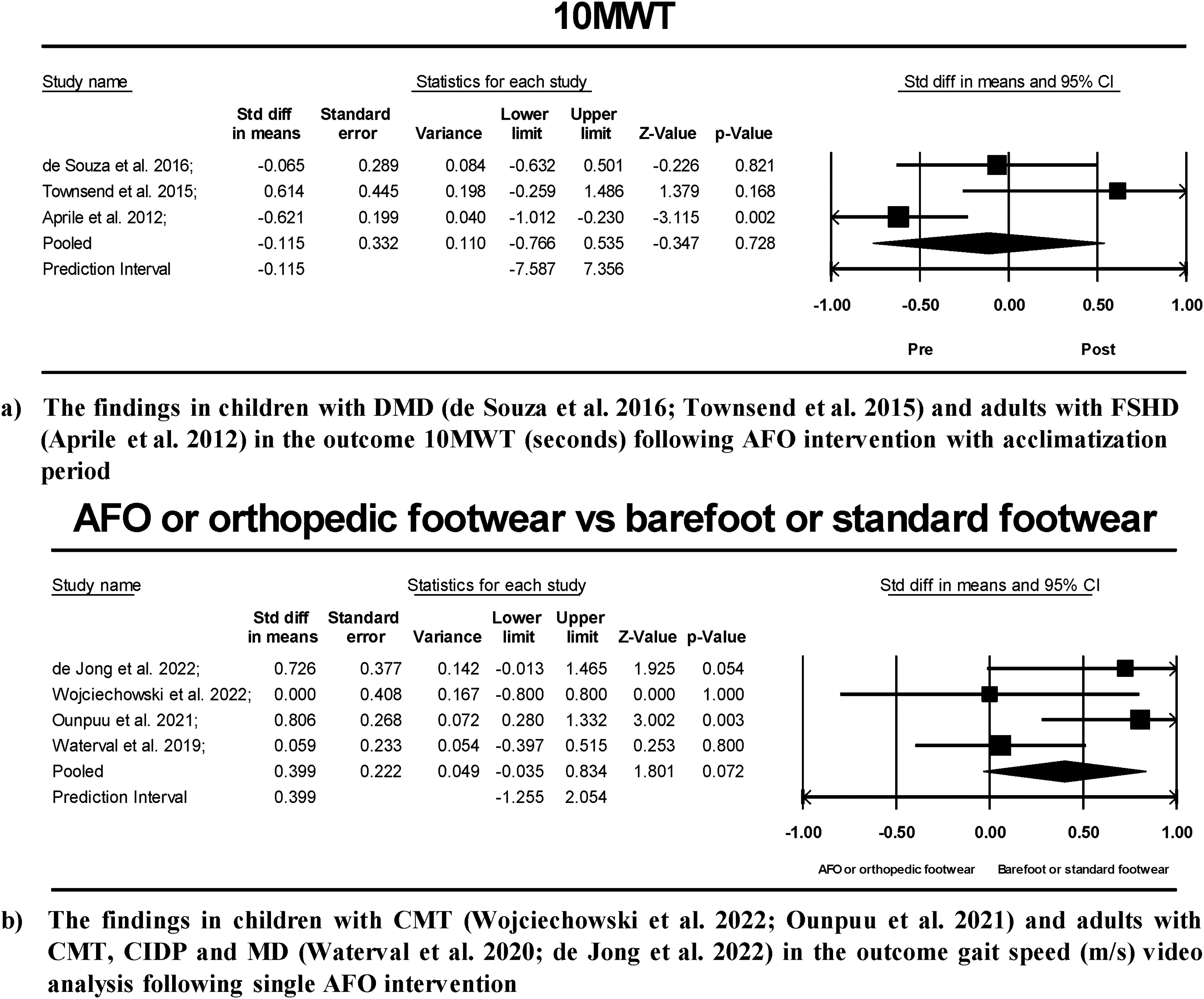
Meta-analysis of AFO intervention in pre-post 10MWT outcome (a), and matched group post-intervention meta-analysis between AFO/OF and barefoot or standard footwear in outcome (b) gait speed video analysis (m/s).
Gait assistive exoskeleton outcome results and effects of training as reported by patient and rater
Patient- and evaluator-reported outcome measures such as the Berg Balance scale, the Timed up and go test and the Short-Form Health Survey were included, among other tools, questionnaires, and rater-based scores. Timed up and go test decreased in case studies with spinocerebellar ataxia, 53 myotonic dystrophy 40 and limb-girdle muscular dystrophy. 44 However, a study conducted in patients with amyotrophic lateral sclerosis, spinal and bulbar muscular atrophy patients and myotonic dystrophy found no significant differences. 52 The Berg Balance scale showed changes in case studies of myotonic dystrophy, 40 spinocerebellar ataxia, 53 and limb-girdle muscular dystrophy. 44 Kogawa et al. 49 revealed significant improvements of gait posture in patients with amyotrophic lateral sclerosis, myotonic dystrophy, inclusion body myositis, Charcot-Marie-tooth disease and spinal and bulbar muscular atrophy. Even though the majority of the patientś and evaluatorś scored outcomes didn’t reveal significant differences in the use of GAE, the study by Serrao et al. 45 indicated that over 80% of participants using the GAE were satisfied with the device used.
Gait assistive exoskeletons meta-analysis
The GAE meta-analysis for 2MWT (m) (N = 59) was based on combined variances calculated with a pre-post correlation 0.75. The analysis was carried out based on six studies. The pre-post standardized mean difference was SMD = 0.36 (95%-CI: 0.17 to 0.54) (Figure 4), revealing a statistically significant result. There was no evidence for statistical heterogeneity based on the I2 test (I2 = 0%), which was confirmed by the Q test (3.37 with 5 degrees of freedom; p = 0.64) and a tau2 = 0.
The GAE meta-analysis for 10MWT (m/s) (N = 45) was based on combined variances calculated with a pre-post correlation of 0.75. The analysis was carried out based on four studies. The pre-post standardized mean difference was SMD = 0.33 (95%-CI: −0.10 to 0.76) (Figure 4), which was not statistically significant. Evidence for substantial statistical heterogeneity (I2 = 66%) was confirmed by the Q test (Q = 8.94 with 3 degrees of freedom; p = 0.03) and a tau2 = 0.11. Assuming that the true effects are normally distributed (in d units), we can estimate that the prediction interval is −1.40 to 2.05. The true effect size in 95% of all comparable populations falls in this interval.
The GAE meta-analysis for FIM score (N = 44) was based on combined variances calculated with a pre-post correlation 0.75. The analysis was carried out based on four studies. The pre-post mean difference was SMD = −0.002 (95%-CI: −0.21 to 0.21), which was not statistically significant. There was no evidence of statistical heterogeneity based on the I2 test (I2 = 0%), which was confirmed by the Q test (Q = 0.93 with 3 degrees of freedom; p = 0.81), and a tau2 = 0.
Orthopaedic footwear, knee and ankle-foot orthosis outcome results and effects of training as reported by patient and rater
In people with Charcot-Marie-Tooth, OF improved gait adaptability compared to standard footwear, as indicated by a precision stepping task on an instrumented treadmill projecting visual targets 68 and gait speed. 55 No loss of dynamic gait balance was observed.55,68 Furthermore, people with facioscapulohumeral muscular dystrophy showed significant improvement in 10MWT tests with custom-made OF. 58 The study by Ramdharry et al. 60 on the Charcot-Marie-Tooth individuals found a significant reduction in hip flexion amplitude during the swing phase with the foot-up splint device, push brace and AFO. It should be noted that the AFO showed the best results. Compared with the other subgroups, knee-ankle-foot orthoses showed no significant differences in walking speed and energy cost. 66 However, there was a significant improvement in weight-bearing stability when ascending or descending stairs. 66 In the case of inclusion body myositis, 63 subjects walked slower and with a lower cadence after six months of intervention. When evaluating the range of motion with the AFO, ankle dorsiflexion and plantar flexion were significantly reduced or increased, depending on the gait phase.54,69,72 Patients highlighted that the stiffness-optimized AFOs have a lower weight, which made walking feel easier. However, the stiffness-optimized AFOs made it difficult for patients to climb stairs and drive a car, mentioning reduced stability. 70 In addition, increase in falling frequency were identified using dynamic response AFOs in patient with Duchenne muscular dystrophy. 56 At one site, three mount AFO interventions reduced walking energy cost66,70 in the 6-min walk test; at the other site, more energy effort was reported during a single test with no AFO adaptation period. 59
Ankle-foot orthosis and/or orthopaedic footwear meta-analysis
The AFO meta-analysis for 10MWT (s/m) (N = 24) was based on combined variances calculated with a pre-post correlation of 0.75. The analysis was carried out based on three studies. The mean difference was SMD = −0.11 (95%-CI: −0.76 to 0.53), which was not statistically significant (Figure 5). Evidence for substantial statistical heterogeneity (I2 = 73%) was confirmed by the Q test (Q = 7.45 with 2 degrees of freedom; p = 0.002) and a tau2 = 0.23. Assuming that the true effects are normally distributed (in d units), we can estimate that the prediction interval is −7.58 to 7.35. The true effect size in 95% of all comparable populations falls in this interval.
A matched group post-intervention meta-analysis was conducted between AFO and no AFO use (N = 94) in outcome walking speed (m/s) (Figure 5). The analysis was based on four studies. The mean difference was SMD 0.39 with a (95% of −0.03 to 0.83), which was not statistically significant. Evidence for substantial statistical heterogeneity (I2 = 51%) was confirmed by the Q test (Q = 6.13 with 3 degrees of freedom; p = 0.10) and a tau2 = 0.09. Assuming that the true effects are normally distributed (in d units), we can estimate that the prediction interval is −1.25 to 2.05. The true effect size in 95% of all comparable populations falls in this interval.
Neuroprosthetic outcome results and effects of training as reported by patient and rater
Two case reports showed signs of enhancement in walking speed and muscle strength by hereditary and spontaneous spastic paraparesis 74 and facioscapulohumeral muscular dystrophy 73 using neuroprostheses. The case study by Fansucu et al. 73 found a substantial improvement in the patient's ability to participate more comfortably in his/her daily school life due to the increased walking distance and ability to climb stairs.
Overall quality of evidence assessment
Due to the primary aim to analyse pre-post AGD intervention outcomes, included studies were first graded as having low certainty of evidence. However, the certainty of evidence of each outcome was downgraded due to limitations in study design (risk of bias) or imprecision. The overall certainty of evidence was very low for all outcomes assessed (Appendix B). There were no legitimate reasons to rate up the certainty of evidence of any outcomes. Therefore, no conclusive recommendation for or against using of GAEs and AFOs for NMDs can be made.
Discussion
Gait assistive exoskeletons
The meta-analysis results in outcome 2MWT suggest that GAEs may help NMD patients to walk longer. There was insufficient evidence to confirm that patients improved in gait speed, coordination and balance tasks. Furthermore, fewer studies reported and assessed patients’ outcomes in home-based settings. Unlike robotic gait therapy used during inpatient rehabilitation, GAEs must be used in the patient's home for extended periods to assist with daily activities or to replace lost walking function. The FIM score, used to assess a patient's level of disability and a change in patient status, showed no significant change in pre-post meta-analysis (Figure 4). We recognize that GAEs are still in the early stages of development and that new technologies are needed to achieve the goal of home use. However, GAE developers should consider involving clinical professionals working in the field of NMDs by asking which impairments are common and how they can be overcome. A good example of this is the fact that none of the GAE devices included have been tested for ascending or descending stairs, despite the fact that the majority of patients are experiencing this limitation. 76 In the field of robotic, such solutions are already available. 77 Another important aspect that was not disclosed is the cost and disposability of devices. Most devices are expensive for the majority of patients and are not covered by health insurance. Due to the heterogeneity of NMDs, the use of GAEs may need to be considered on a case-by-case basis and may not be accessible to many patients. The fear of falling is one of the parameters that should not be underestimated, and there is a strong need for further studies to analyze this phenomenon. As previously shown, 75 anxiety of falling contributes to reduced quality of life. To provide a complete picture of GAEs, upcoming studies, articles and research protocols need to include information from the patient's perspective using patient-reported outcome measures such as the SF-36 Medical Outcome Study 36-item used in,52,58 the Nottingham Extended Activities of Daily Living questionnaire used in, 61 or the Quebec User Evaluation of Satisfaction with Assistive Technology used in studies.45,66
Orthopaedic footwear, knee and ankle-foot orthosis
The OF, AFO and knee orthosis have a long history of use in most orthopaedic and neurological conditions. The OF may improve gait quality in comparison to standard footwear, as demonstrated by an improvement in a precision step task on the treadmill, 68 gait speed, 55 10MWT, 58 and by reduction in hip flexion amplitude during the swing phase. 60 No barriers to the prescription of an appropriate OF have been identified. However, only two studies looked at long-term walking outcomes.63,65 Therefore, there is insufficient evidence on potential barriers. In the matched group post-intervention meta-analysis between single AFO and no AFO use, gait speed (m/s) did not change significantly. This may indicate that practice and familiarisation time are required to ensure a stable gait after AFO prescription. However, a more rigorous pairwise meta-analysis is needed to confirm this suggestion. When testing ankle dorsiflexion and plantar flexion with an AFO, there was a significant reduction or increase in ankle range of motion, depending on the phase of gait, compared to no AFO use. This can indicate improved gait, foot-ankle stability, and weight distribution.54,68,69,72 In addition, three mount AFO interventions reduced walking energy cost66,70 in the 6-min walk test, compared to improved energy effort during a single test with no AFO adaptation period. 59 AFOs can help with tasks that involve isometric muscle contraction, and in some specific cases, the optimized AFO can reduce the risk of falling, which may result in less harm. 75 The pre-post meta-analysis found no benefit for 10MWT. Due to the small sample size and only three included trials, the analysis was likely underpowered. It is important to note two following limitations: first, the diversity of NMDs was not considered separately, which may lead to uncertain results and misinterpretation due to the differences in the NMD clinical profiles; second, AFO meta-analyses were conducted in both adults and children together, as we intended to determine the overall positive or negative effect of AFOs on mobility and independence in activities of daily living in NMD patients. This review did not find sufficient evidence to support knee orthoses; a small indication is provided in the study 66 where the patient maintained their functional status between assessment dates.
Neuroprostheses
No evidence was found on the benefit and tolerability of neuroprostheses to support lower limb function during gait and activities of daily living for people with NMDs. Some cases show signs of enhancement in walking speed and muscle strength by hereditary and spontaneous spastic paraparesis 74 and facioscapulohumeral muscular dystrophy. 73 From a neurobiological point of view, in some NMD cases without inflammation, metabolic components could hypothetically show positive results. However, neuroprostheses using FES, which are effective in treating some neurological diseases, 78 are fundamentally limited because they must employ charge-balanced stimuli to avoid the evolution of irreversible electrochemical reactions and their byproducts at the interface between metal electrodes and body fluids. 79 Further research is needed on each NMD to assess the advantages and disadvantages of using neuroprosthetic systems.
Implications for future research
The most relevant activities for which patients wanted to improve their independence (mobility & transfers, toilet use and hygiene)3,6,7 were not sufficiently addressed in the included studies. Moreover, patients’ ability to dress and apply the AGD independently was not considered. There has been no evaluation of patient tolerance to prolonged or excessive use of the devices. This information may be necessary because the persons will use the AGD at home for a extended period to support their daily activities. Future research should focus on implementing clinical protocols by including patient-reported outcomes that assess patients’ social role, emotional and mental health, and moving away from the concept of using only functional outcomes in a purely clinical setting, as only a satisfied patient will be likely to use the AGD at home.
Considering the diversity of NMDs and the challenges of conducting RCTs, the concept of the symptom approach, including several NMDs sharing the same symptoms in one trial, may be a helpful way to provide evidence for different interventions in non-pharmacological trials with less confounding bias and high overall certainty of evidence. Our final look at the clinicaltrial.com website found few protocols that followed the symptom approach, at least for assistive device trials. This can open new perspectives, and this systematic review showed that such trials are possible if protocols are strictly followed to reduce methodological difficulties.
Limitation
As different studies were included in this review, using different tools to assess risk of bias was necessary. The use of several tools did not cover all areas of quality of evidence and strength of results. Therefore, the results should be treated with caution from the outset. We could not perform more rigorous meta-analyses (such as pairwise meta-analyses) due to a lack of RCTs and homogenous NRCTs. With small sample sizes and only three to six studies included in each meta-analysis, the analyses probably needed to be more balanced. Sometimes, a small analysis finds a benefit, as in the case of the pre-post 2MWT outcome meta-analysis with GAE intervention. However, the result may not be statistically significant because the study is underpowered. In that case, the apparent benefit should be viewed cautiously and considered when designing future studies to replicate the finding. 80 The fact that no comparison with a control intervention could be performed in our synthesis should be considered a design-related limitation. Therefore, and because of the very low certainty of evidence, no robust conclusions can be drawn.
Conclusion
The results suggest that GAEs may help patients with NMDs to walk longer. However, GAEs are not yet suitable for home usage. It may be recommended that AFOs and OF need be fitted individually to each patient; a period of familiarization might be required to help the patient become comfortable. There is no evidence to support the use of knee orthosis and neuroprostheses. The results are based on very low certainty of evidence. Furthermore, more consideration should be given to patient preferences in future research.
Supplemental Material
sj-xlsx-1-jnd-10.1177_22143602241289216 - Supplemental material for The assistive gait devices and their implementation in activities of daily living for patients with neuromuscular disease: A systematic review and meta-analysis
Supplemental material, sj-xlsx-1-jnd-10.1177_22143602241289216 for The assistive gait devices and their implementation in activities of daily living for patients with neuromuscular disease: A systematic review and meta-analysis by Marko Mijic, Corinna Wirner-Piotrowski, Andres Jung, Kristina Gutschmidt, Marcela Arndt, Natalia García-Angarita, Stephan Wenninger, Peter Young and Benedikt Schoser in Journal of Neuromuscular Diseases
Supplemental Material
sj-docx-2-jnd-10.1177_22143602241289216 - Supplemental material for The assistive gait devices and their implementation in activities of daily living for patients with neuromuscular disease: A systematic review and meta-analysis
Supplemental material, sj-docx-2-jnd-10.1177_22143602241289216 for The assistive gait devices and their implementation in activities of daily living for patients with neuromuscular disease: A systematic review and meta-analysis by Marko Mijic, Corinna Wirner-Piotrowski, Andres Jung, Kristina Gutschmidt, Marcela Arndt, Natalia García-Angarita, Stephan Wenninger, Peter Young and Benedikt Schoser in Journal of Neuromuscular Diseases
Footnotes
Abbreviations
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
