Abstract
The use of a sprocket driven carrier tape provides a unique solution for Ultra High Throughput Screening. 10-microliter wells in the 384 well pattern are embossed in a polypropylene or polycarbonate carrier tape. For compound handling, 100,000 compounds in 5 microliter aliquots may be stored, sealed and frozen in a roll 16 inches in diameter and 4 inches wide. The sprocket drive provides recall to any given compound. At time of use, the peelable seal is removed for access to the compound aliquots. Two levels of assay automation are provided. In Phase I the compound MicroTape is used to support assays in the standard 384 well plate format. In Phase II the compounds are transferred directly to the assay MicroTape for Ultra High Throughput Screening at the rate of 100,000 per day.
GENERAL DESCRIPTION
There are three basic areas of High Throughput Screening: 1) handling the compound library 2) lead discovery screening and 3) lead optimization. Handling the compound library is an essential element of the other two. Lead discovery and lead optimization tend to overlap. The objective of the lead discovery is to develop “hits” or what appear to be active compounds in specific assays. Lead optimization is a refinement of these “hits” so as to pass on qualified leads to medicinal chemistry for further development. Without this refinement medicinal chemistry is swamped and negates the discovery of more “hits”.
The primary objective of Ultra High Throughput Screening (UHTS) is to achieve more qualified lead compounds. In general terms UHTS has been described as the ability to screen a library of 500,000 compounds against 50 therapeutic targets a year. This equates to 100,000 compounds screened per day. The economics of this number dictates some form of miniaturization to conserve the precious reagents consumed.
Since the compound handling is the front end of both lead discovery and optimization, let's look at it first. The long term library storage is going to be in solid or semi-solid form, for stability reasons. However, for use in screening it must be converted to a liquid phase. The most commonly accepted method is to weigh out a small aliquot of the compound and solvate it with dimethyl sulfoxide (DMSO). Speed and convenience dictate weighing 10 milligram quantities as the minimum amount. These are then brought into solution form, yielding 5 to 10mL of solution. This is then subdivided into smaller aliquots of 0.5mL and stored frozen in sets of 96-well deepwell tubes (at −20°C or −80°C) as an archive library. This one labor intensive weighing of the compounds is expected to support the screening operation for as long as possible — hopefully several years.
Several areas of concern arise in going from this archive library to the soluble form for the assay. First is the concentration, many assays are tested at 10−5M or 10−6M concentrations. The majority of assays cannot tolerate much more than 1% DMSO. Thus a dilution from the archive library is required. However some compounds, while soluble in 100% DMSO are not soluble in lesser percentages. It is desirable to make the compound dilution in the final assay volume and not on a previous dilution step. Another concern is protecting the stability or validity of the archive compound. Freezing lengthens its shelf life. But to access the compound it must be thawed to remove an aliquot. Each time a freeze-thaw cycle occurs there is the potential for moisture to degrade the compound. Thus it is desirable to minimize these cycles. The real problem is how to transfer 100,000 discrete samples, per day, from the archive library to the assay, keeping the above constraints in mind. The MicroTape system is specifically designed to accomplish this function.
MICROTAPE SYSTEM
The MicroTape system consists of a carrier tape that is tractor driven with precision sprocket holes down each side (FIG. 1). A 384-well pattern is embossed into the carrier tape every 5 inches. Each well has a brim volume of 10 microliters. For compound storage, the carrier tape is inert polypropylene to withstand DMSO. The assay carrier tape is polycarbonate to be tissue culture compatible. This carrier tape format has been adapted from the electronic industry, where it is used for high speed automation in picking and placing small surface mounted devices.
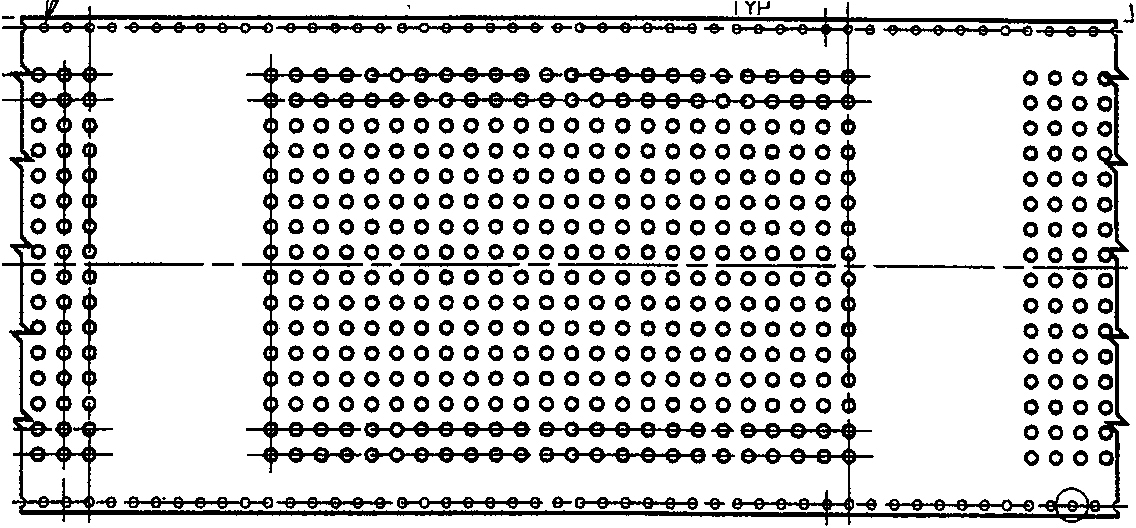
There are a number of reasons for choosing the 384-well format. First, it is a multiple of the existing 96-well format (i.e. 4 × 96 = 384). Secondly, there is a variety of liquid handling instrumentation on the market now for the 384 well format. Plate readers using photomultiplier tubes (PMT) are currently available. To go to a higher density system, such as 1536, complicates liquid handling. Plate reading at the higher densities implies imaging with CCD camera technology to obtain the desired throughput. It is questionable whether CCD systems and their associated software can match the sensitivity currently available with PMT's, although improvements are occurring in that field.
Probably a more compelling reason to use 384-well formats is the biology. Assay technology, currently running in 96-well formats at 100μL to 200μL can be more readily scaled down to the 10μL to 50μL volumes of 384-well. This is the objective of Phase I, whereby the compound library may be replicated in 384-well MicroTapes, but the actual assay is run in 384-well microplates. As assay technology is developed for 1μL to 5μL volumes then Phase II uses the MicroTape format for the assay.
MICROTAPE COMPOUND HANDLING
The MicroTape system uses a tape replicating system for the compound library (Figure. 2). Ten replicate MicroTapes are made at one time. Each finished MicroTape roll holds 100,000 discreet compounds. It is approximately 16 inches in diameter by 4 inches wide. The replicating system consists of four small volume 96 well pipettors, each fed by dual stackers. When one is depleted the switchover to the full stacker is automatic. The Stackers hold the 96-well archive plates in either microplates or deepwell plates. The pipettors are 96-well positive displacement systems, capable of delivering 0.5μL with a precision of 2% — 3% Cv.
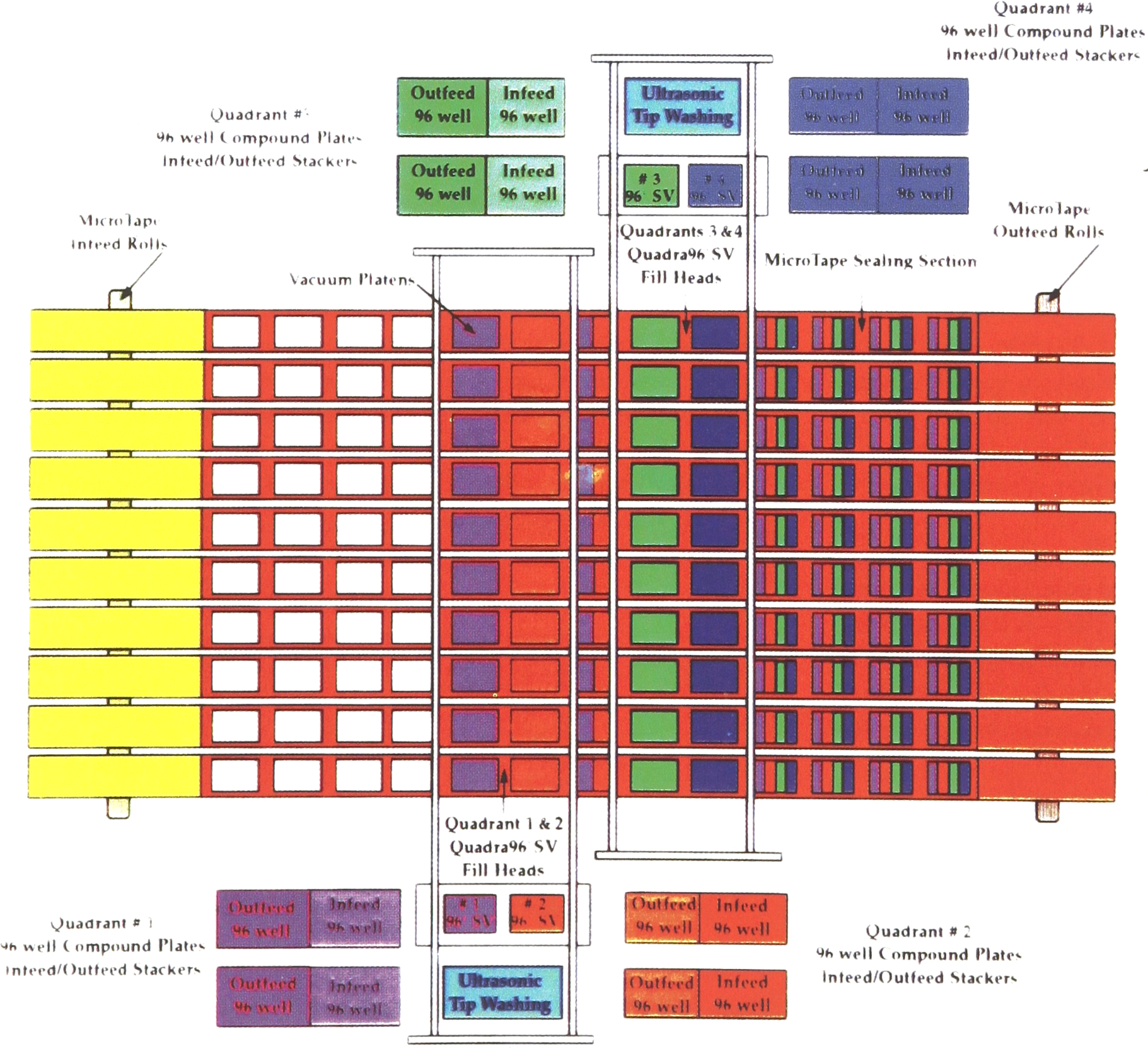
Each 96 well pipettor aspirates 50μL per well from its 96 well archive plate. The four 96-well pipettor heads then traverse across the 10 MicroTapes, dispensing 5μL into one quadrant of each 384-well pattern. The heads return and the pipetting tips are rinsed in an ultrasonic tip washing station, to eliminate carry over between samples. This cycle requires 5 minutes. Thus the four pipetting heads can create 10 replicate copies of forty-eight 96-well archive plates per hour. The MicroTapes are held flat in position by a vacuum platen. After all four quadrants are filled, both a man readable and machine readable number is printed on the bottom of each MicroTape pattern. This number is unique and relates to the four 96-well archive plates that went into the 384-well pattern. This maintains the audit trail of the compound library.
After the MicroTapes are filled, a peelable heat seal is applied to each pattern. The finished rolls are wound on 6 inch cores and transferred to the freezer. Thus from one freeze-thaw cycle of the archive plates, 10 replicate copies have been made in less than 22 hours. Each copy holds 5μL of 100,000 compounds, bar code identified and individually addressable. The compact storage of the rolls allows 2 million compounds to be stored in one −80°C freezer.
The MicroTape compound storage system is extremely flexible. Since each 384-well pattern is sealed and identified it may be cut from the roll and transported individually. It enables a central compound library to ship 5μL aliquots of 384-well compounds in 100% DMSO, to outlying investigators worldwide. The end user places the pattern in a frame, peels off the seal and may now accesses the compounds with existing pipettors — individual or multiple well. On the other extreme, the entire roll may be supplied to the high volume user to support HTS or UHTS.
MICROTAPE PHASE I ASSAY
In Phase I of the MicroTape assay system the compounds, in roll form, are used to support 384-well assay in the microplate format. In Phase II, for UHTS systems, the compounds are transferred from the compound MicroTape directly to the assay MicroTape. This assumes assays at 5μL have been developed.
The Phase I system uses a 384-well microplate system (Figure 3).
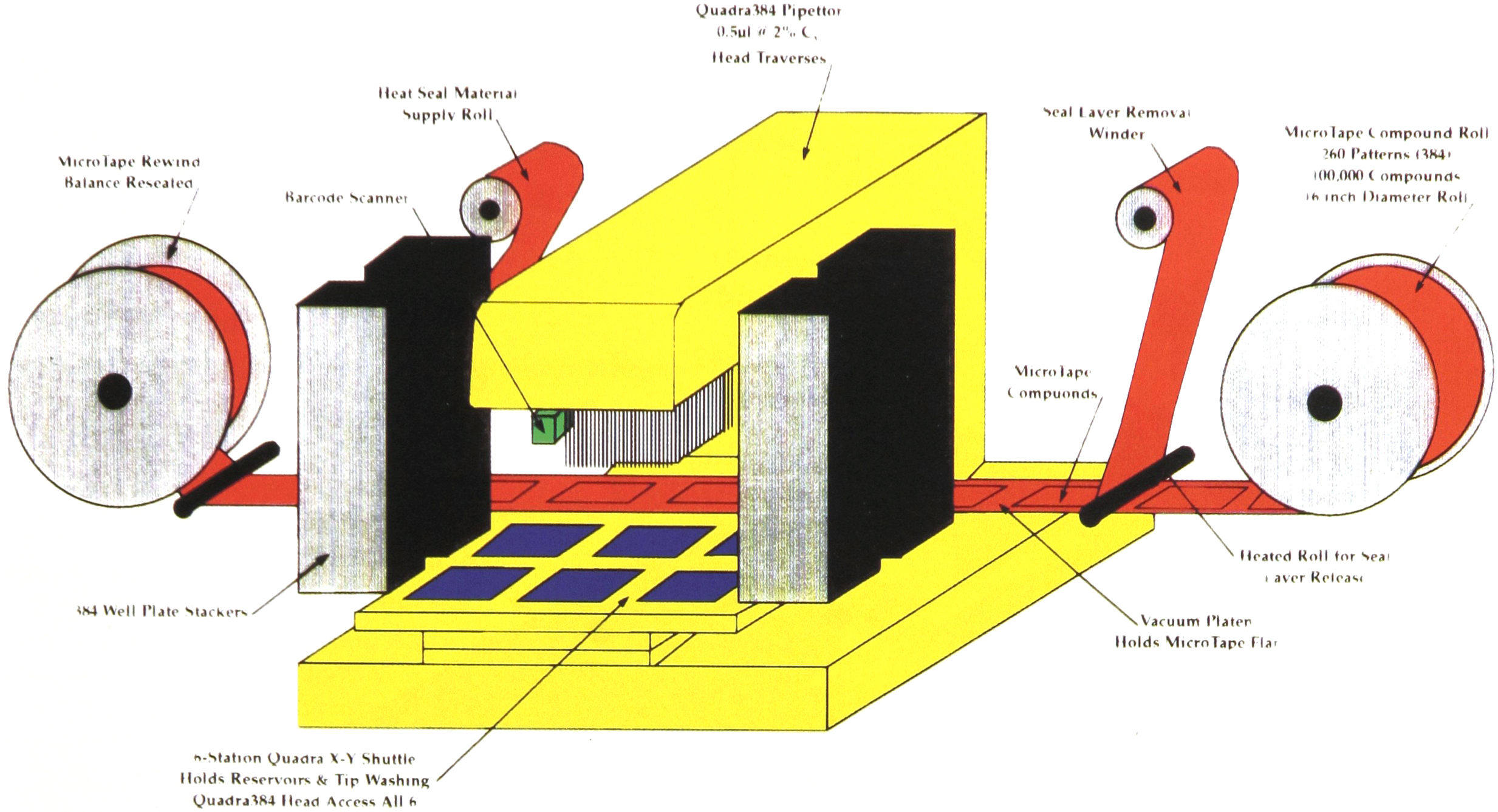
It consists of a 384-weIl pipettor with dual reversible stackers. That is each stacker can serve as an infeed or outfeed according to the program control. The stackers bring standard 384-well microplates to the 6-station shuttle. The 6-station shuttle holds reservoirs for reagents and the standards and controls. A separate station, accessible by the 384-well pipettor holds the MicroTape compounds. They are fed in from the infeed roll. The sealed MicroTape passes under a heated roller. The heat from the roll softens the heat seal bond. The peelable seal is automatically removed and rewound on a take up reel. The MicroTape is held flat by a vacuum platen. The vacuum platen is made of aluminum, through which cold ethylene glycol is circulated. This prevents any thermal damage to the compounds during seal removal. The open compounds can now be accessed by the 384-well pipettor tips. Following this, the balance of the compounds in the MicroTape may be resealed and rewound or, the MicroTape may be cut into pieces and trashed.
Using pipeline pipetting, the 384-well pipettor aspirates the various reagents in the assay using an air gap, for separating the reagents. The standards and controls are aspirated from special reservoirs to match the users format. The ability of the 384-well pipettor to aspirate 0.5μL permits aspirating compounds from the MicroTape in 100% DMSO while maintaining a 1% DMSO in the 50μL assay volume. In addition, to speed in processing, pipeline pipetting uses the high volume reagents (i.e. buffer) to wash out the small volume of compound from the tips, thereby maintaining the precision in the assay. After dispensing the tip volume in the 384-well assay plate, the plate is restacked, the 384-well pipettor tips are washed, in an ultrasonic tip wash station, the next plate obtained from the stacker and the cycle repeats.
MICROTAPE PHASE II ASSAY
In Phase II of the MicroTape assay system, the compounds are transferred from the compound tape directly to the assay MicroTape running in parallel alongside (Figure 4). The assay MicroTape uses the same 384-well format as the compound MicroTape. The difference is in the tape material. This is a function of the assay. The common material is polycarbonate. It may be clear or opaque in either white or black. The wells may also be supplied coated with biological reagents.
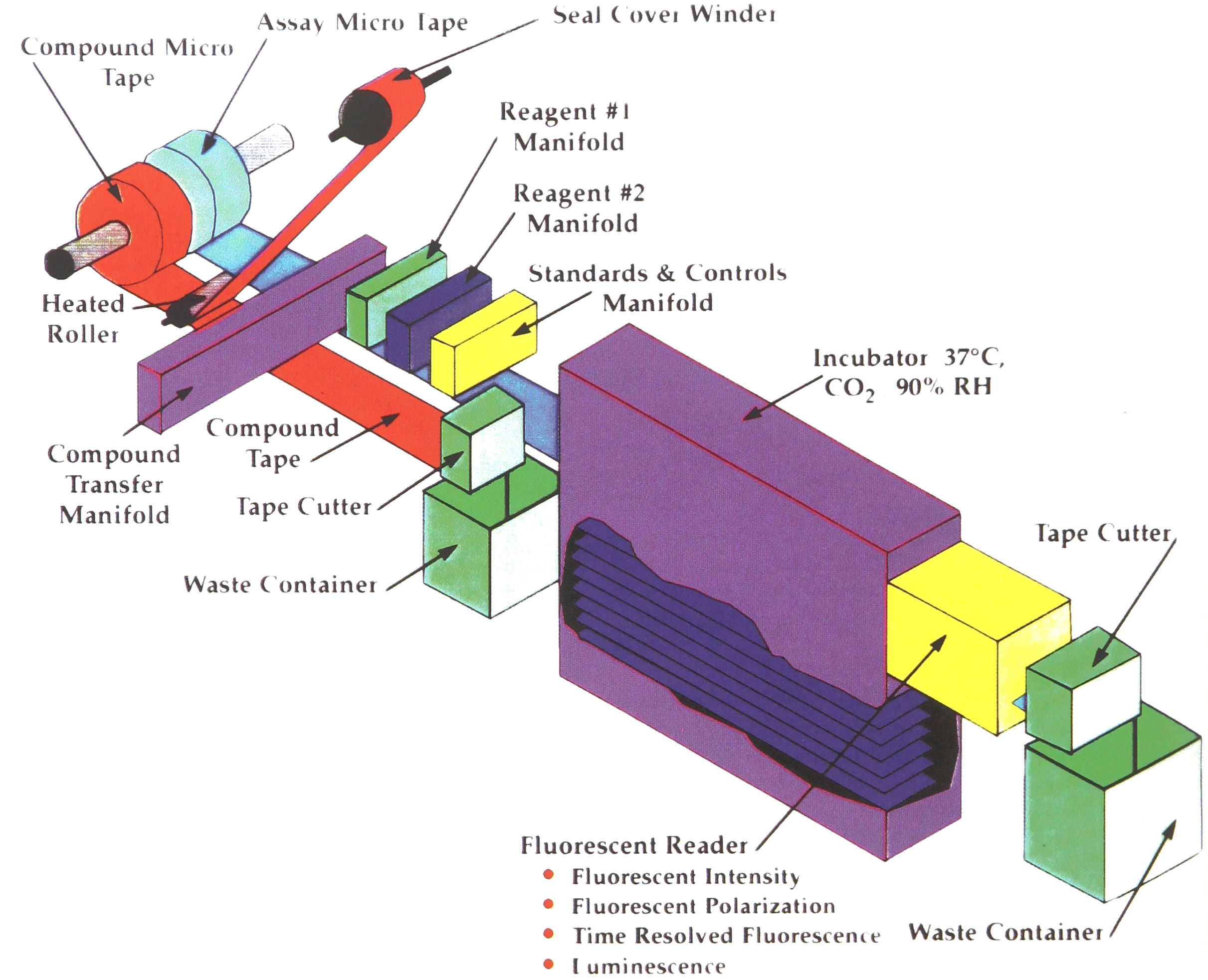
The primary difference between Phase I and Phase II is that the Phase II assays are designed for 5μL volume or less. Additional assay development will be required, for Phase II.
The small volume puts other stringent requirements on the system. Very small volumes of compound, in the range of 50 nanoliters, must be transferred. Systems using piezo electric and ink jet methods are being developed. Evaporation at 5μL is another problem to be addressed. As with the compounds, the assay MicroTape can be sealed. Work is in process testing both a peelable heat seal material and a peelable pressure sensitive material. Another alternative is a high humidity incubator. The incubator required for MicroTapes is very compact. It need be only 6 inches wide. Using shelves, 100,000 assay wells could be maintained in a unit 4 feet long and 2 feet deep. Since there is little thermal mass in the MicroTape, compared to polystyrene microplates, incubation times and temperature control to the assay can be greatly improved.
Additional development is under way regarding reading MicroTape assays. It is assumed the assay readout will be fluorescent in nature. It may be fluorescent intensity, fluorescent polarization, time resolved fluorescence, fluorescent energy transfer, or luminescent. At only 5μL working volume a sensitive system will be required. Development work is under way utilizing multiple PMTs. Work is also going forward with image based systems.
The above high throughput systems, particularly Phase II, are directed at lead discovery where high numbers and reagent conservation are high priority. For lead optimization the numbers will be much lower. It is envisioned that many of these will remain in the 96 well format with some going to 384-well. The higher assay volumes provide more signal. Due to the smaller numbers to be run, it may not be cost effective to convert existing 96 well formats to higher density systems.
SUMMARY
In summary the logic of HTS and UHTS demand a higher density system in addition to the existing 96-well protocols. To go directly from 96-well to 1536-well or higher means many different problems must be solved simultaneously. Not only the assay protocols themselves but also the mechanical problems of liquid handling, readout and evaporation. The MicroTape system allows an immediate stepwise approach, in 384-well formats. Currently instrumentation, both in liquid handling and readout, exists. Additional developments are under way. Assay development is less taxing going from 96-well to 384-well protocols than from 96-well to higher density, such as 1536-well. When 5μL assays are developed very little modification will be required in equipment and instrumentation.
