Abstract

The ARCHITECT i2000 (Figure 1) heralds a new development in the production of automated analyzers for the clinical laboratory. As well as being one of a family of instruments, the i2000 is also part of a modular system which can provide automated immunoassay and clinical chemistry testing on a single platform.
FIGURE 1 OF ANALYZER
Table 1 gives a brief description of the ARCHITECT family which is designed to accommodate a wide range of laboratory workloads as well as to provide both immunoassay and clinical chemistry testing (c8000™). The i2000 and c8000 may be linked directly together to a maximum of four analyzers, in a variety of combinations, to meet the large workload needs of major hospitals. For example, four i2000s may be linked together to form an i8000™ or, two i2000s, may be linked to one or two c8000s to give a c8000/i4000™ and a c16000™/14000™ respectively. The modular system provides total automation of a wide range of analytes on a single tracking system operated by a single work station. Therefore, the need to aliquot specimens and move specimens between different analyzers is reduced. There is more economical use of specimens as only one dead space has to be considered. Finally, because the analyzers are directly linked together they occupy a smaller footprint than linking individual instruments via an independent specimen tracking system.
Features of the Abbott ARCHITECT family of instruments

The ARCHITECT family provides flexibility as the system can be adapted to the customer's needs, and scalability as the system may be upgraded as the laboratory's workload increases. This may be done without the need to change reference ranges as the same immunoassay technology is used on all the immunoassay instruments. The system also uses a common processing unit so that the basic operating and management procedures are the same across instruments. Therefore, introduction of a different analyzer from the ARCHITECT family requires little training and no loss in operating time.
The ARCHITECT i2000 is the first of the family to be launched onto the market. Chemiluminescent immunoassay technology is used incorporating a very stable patented acridinium derivative tracer. This has enhanced light emission giving greater analytical sensitivity and extended dynamic ranges. Calibration of assays is only required every thirty days and the reagents have a long shelf-life of several months. One-step, two-step and fully automated pre-treatment assays can be accommodated.
The instrument sample carrier accepts primary tubes from 10mm (0.4inch) to 16mm (0.6inch) up to a height of 105mm (4.15inch) as well as aliquot tubes and sample cups. A 25 or 50 sample queue is available providing either 125 or 250 total sample loading capacity. Samples are identified by barcode and a range of barcodes can be read. The sample probe has clot, gel, and bubble detection giving a secure sampling system.
The analyzer is operated through a single processing centre operating under Windows NT The software is designed to make it user friendly and the whole instrument may be easily run by a single operator. Throughout its operation, the processing centre keeps an up-to-date log of reagent usage and alerts the operator to low reagent volumes. A maintenance history log is also recorded. A modem link may be fitted providing remote diagnostics from the company. It is the company's intention that customers will also keep a stock of easily replaceable parts in order to reduce down time and engineer costs.
An evaluation of the reproductive peptide assays is presented in this paper.
EVALUATION DETAILS
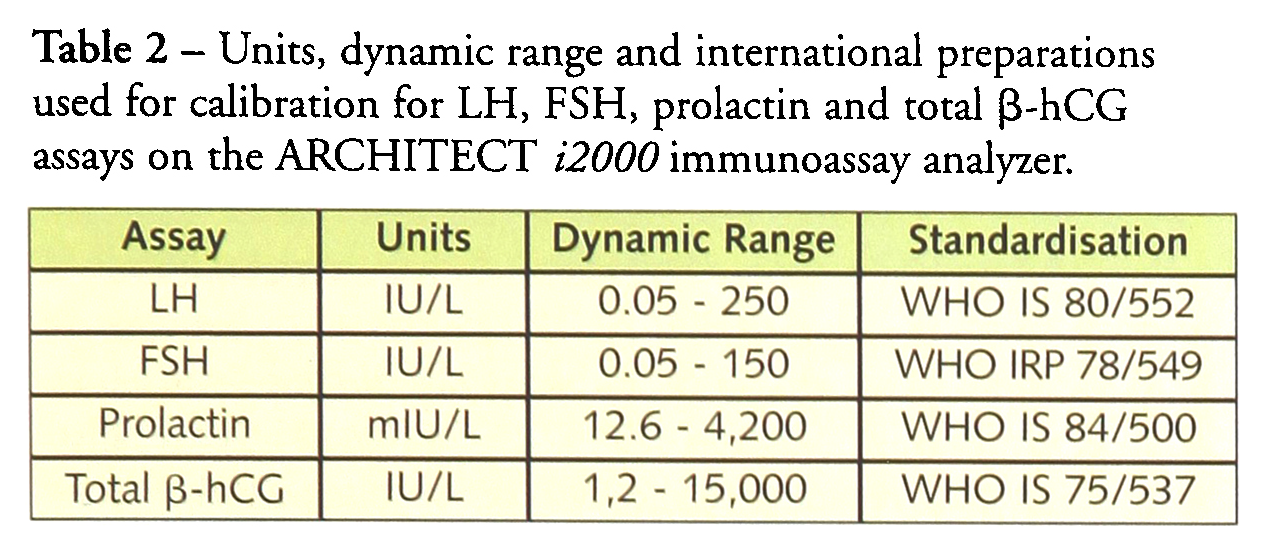
LH, FSH, prolactin, and total β-hCG have been evaluated and Table 2 gives the dynamic ranges of these assays and the reference preparations used for their standardisation. Analytical precision, within-, between-and total precision and linearity have been assessed. The results obtained with the ARCHITECT i2000 have been compared with the results obtained with the Abbott AxSYM™ and Bayer ACS:180™.
Units, dynamic range and international preparations used for calibration for LH, FSH, prolactin and total β-hCG assays on the ARCHITECT i2000 immunoassay analyzer.
Evaluations were carried out using two reagent lots for each assay with two runs (on different days) for each reagent lot. Analytical sensitivity was determined from the analysis of ten replicates of the zero standard and four replicates of the first level calibrator. The signal mean of the zero standard was calculated and two standard deviations of the mean was read from the calibration curve to give the analytical sensitivity. Linearity was determined by diluting ten patient samples with concentrations between the top two calibrators. Duplicate measurements were carried out on each dilution. Precision was also determined using two reagent lots per assay. Five replicates, of three commercial BioRad controls and three Abbott controls were analyzed in five separate assays, run over five days, for each reagent lot. Precision was calculated using NCCLS guidelines. Routine samples measured by the laboratory's routine method (ACS: 180) were reanalyzed on the ARCHITECT i2000 and the AxSYM within 24 hours. Passing-Bablock regression analysis was used to examine the correlation of the results from the different methods.
RESULTS
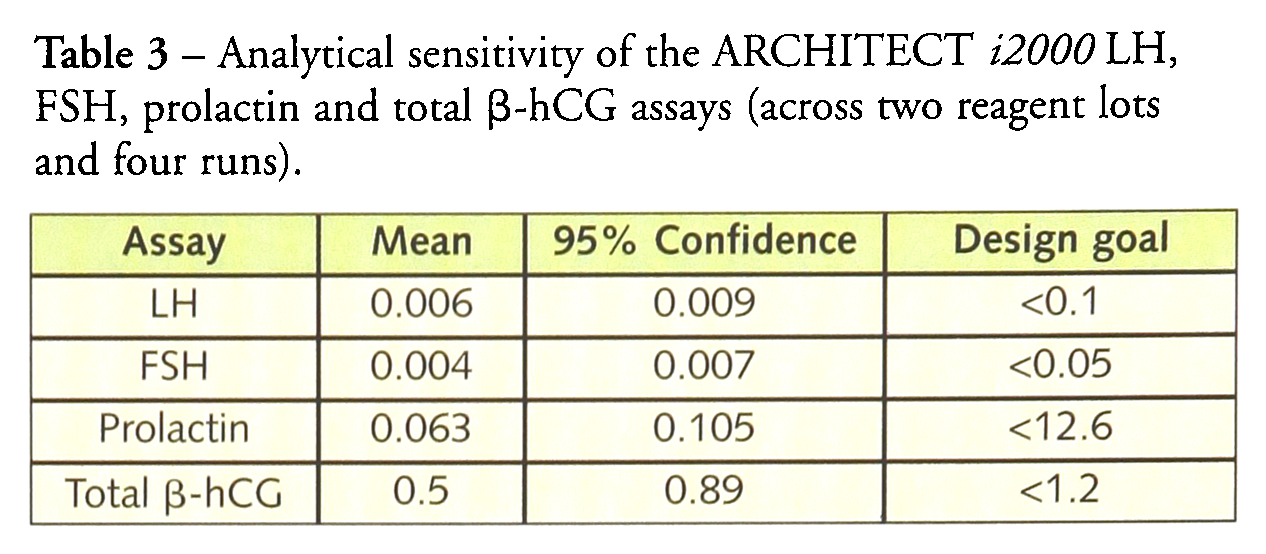
The analytical sensitivity of the four analyses is given in Table 3. The mean analytical sensitivity calculated for the two reagent lots with the 95% confidence limits is compared with the design goals for analytical sensitivity set by the company at the beginning of the development period. In all cases the sensitivity obtained exceeded the design goals that had been set.
Analytical sensitivity of the ARCHITECT i2000 LH, FSH, prolactin and total β-hCG assays (across two reagent lots and four runs).
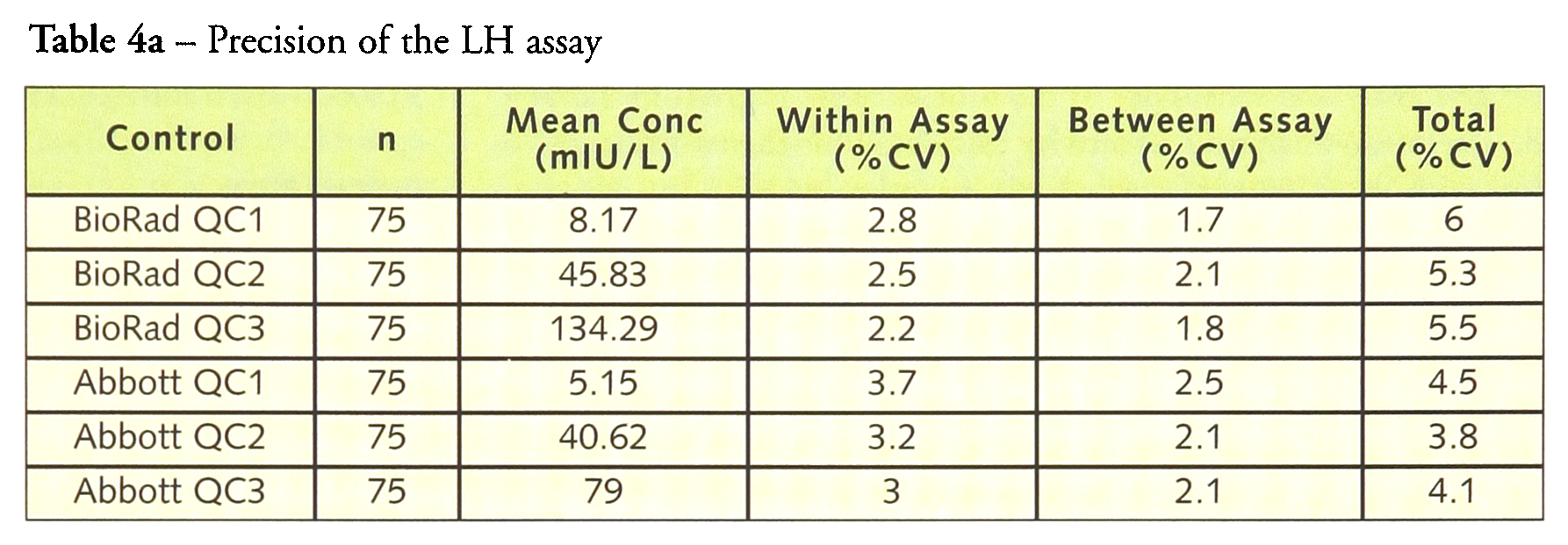
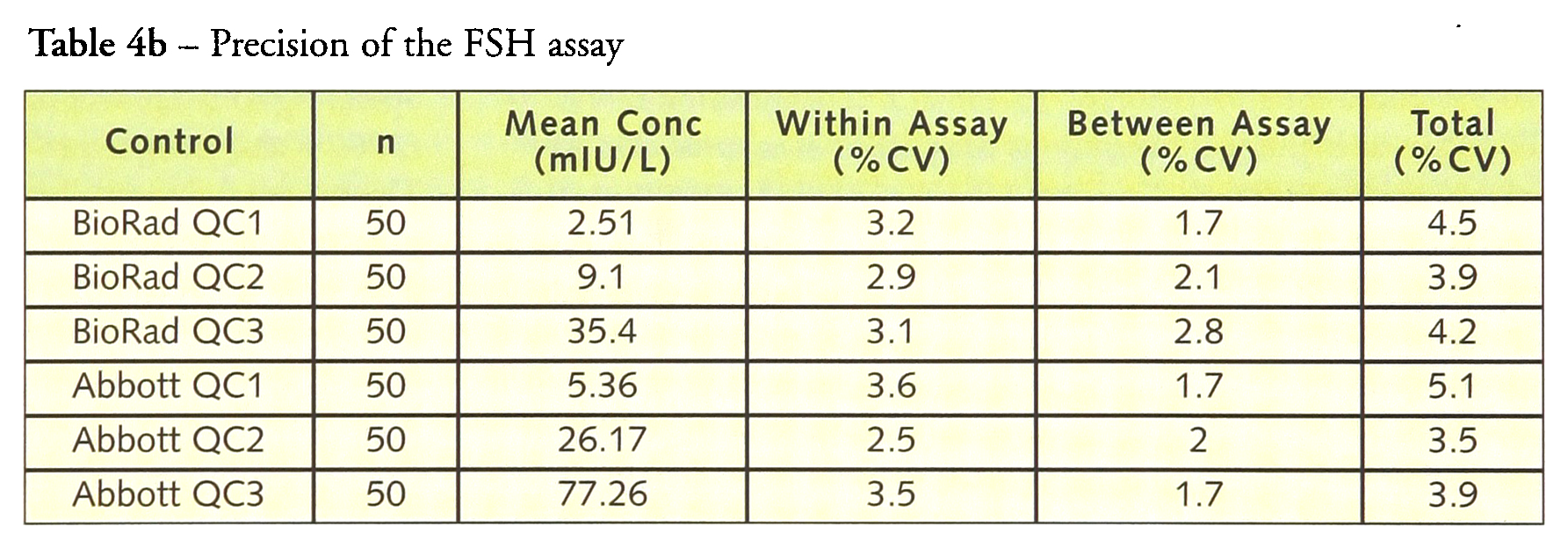
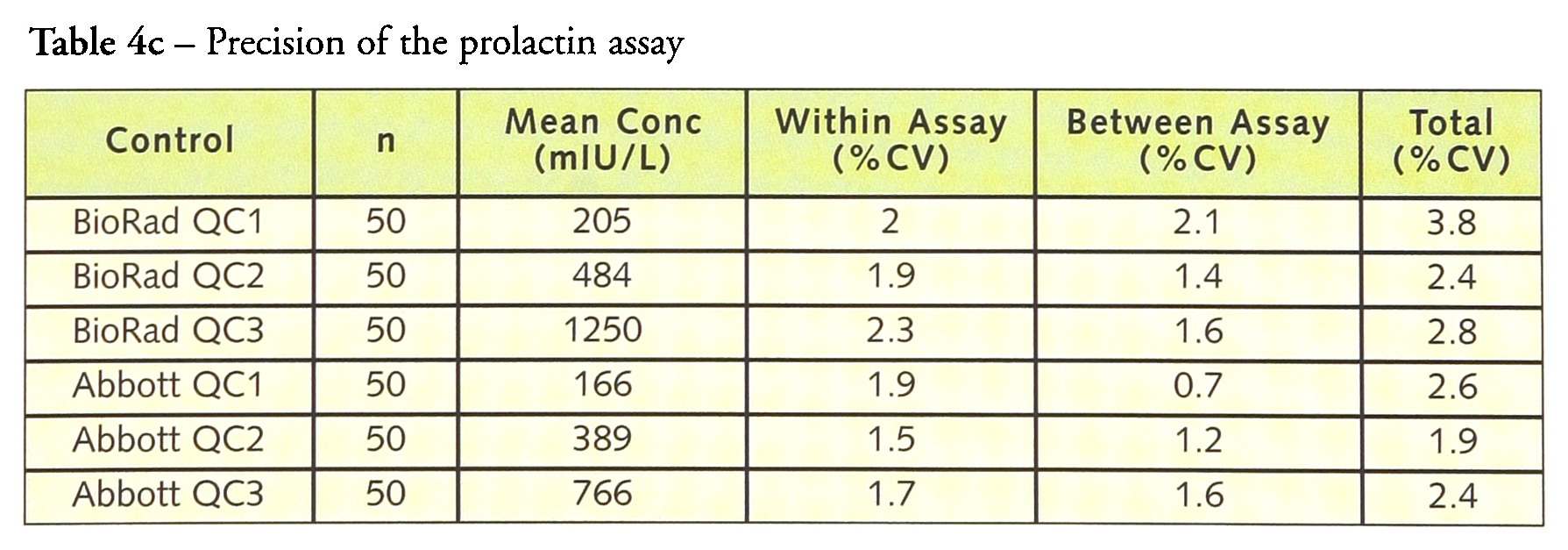
Tables 4a–4d give the within-, between- and total precision for each of the assays. For LH, within-assay precision was <4% and total imprecision was <6.0%. Similar results were obtained for FSH with the within-assay precision <4% and the total imprecision <5.5% while prolactin had a within-assay precision <2.5% with a total precision of <4%. hCG has a within-assay precision of 16.9% and a total precision of 19.1% at a concentration of 1.5 IU/L but at higher concentrations the within-assay precision was <4.5% and the total precision <5.0%.
Within-, between- and total imprecision of the ARCHITECT i2000 LH, FSH, prolactin and total β-hCG assays calculated from the analysis of six quality control samples (between lot CVs are not shown, but are included as a component of the total CVs)
Precision of the FSH assay
Precision of the prolactin assay
Precision of the β-hCG assay
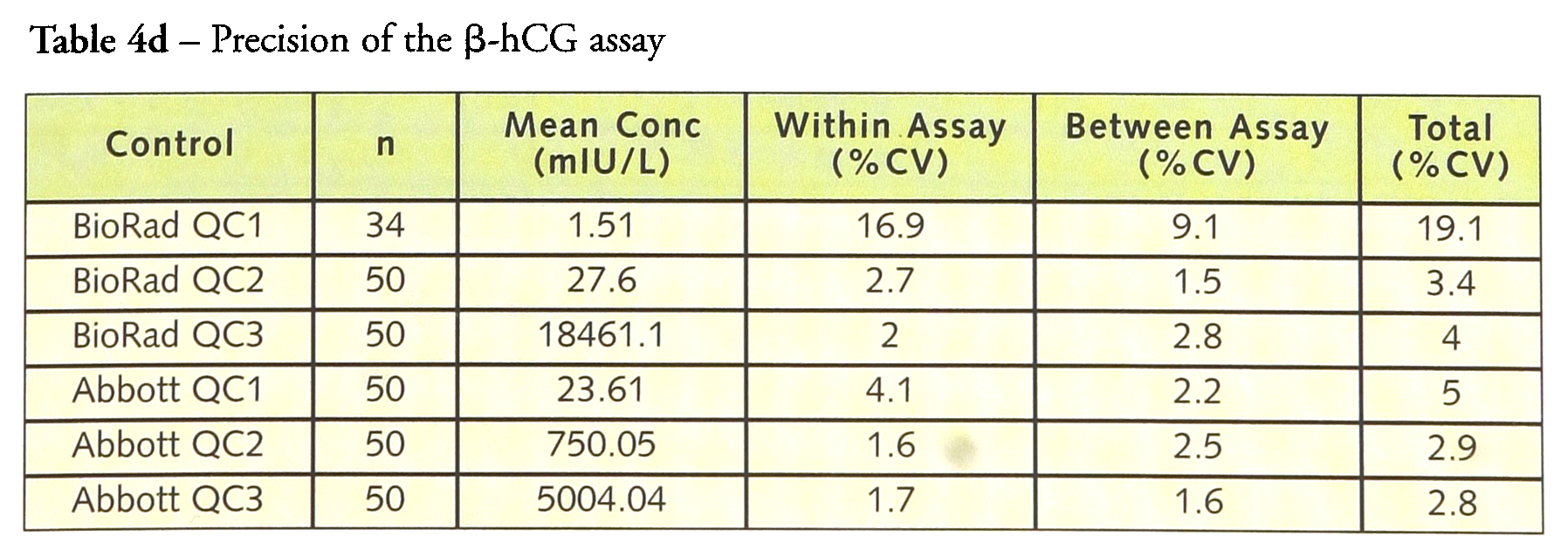
Table 5 gives the regression analysis of the data for the linearity experiments which was excellent for all assays. Table 6 gives the correlation data for the comparison between the ARCHITECT i2000 assays and the results for the same sample analysed on the AxSYM and the ACS:180 analysers. For FSH, there was excellent agreement between the results from all three instruments. There was also very good agreement between the ARCHITECT i2000 and AxSYM prolactin assays. There was more scatter about the regression line when the ARCHITECT i2000 results were compared with those from the ACS:180 assay, but the slope was very close to unity. Surprisingly the ARCHITECT i2000 hCG results showed better agreement with the ACS:180 results than the AxSYM results. There were some marked outlines in both comparisons which would require further investigation. The slope of 0.77 between the ARCHITECT i2000 and the AxSYM results also needs further investigation. However, it should be noted that this slope includes results from autodilution of samples above the dynamic range for each assay (>1,000 IU/L for AxSYM and >15,000 IU/L for ARCHITECT).
Regression analysis data for the linearity on dilution of samples in the ARCHITECT LH, FSH, prolactin and total β-hCG assays

Passing-Bablock correlation data for the comparisons between the ARCHITECT i2000 and the Abbott AxSYM and the Bayer ACS: 180 respectively
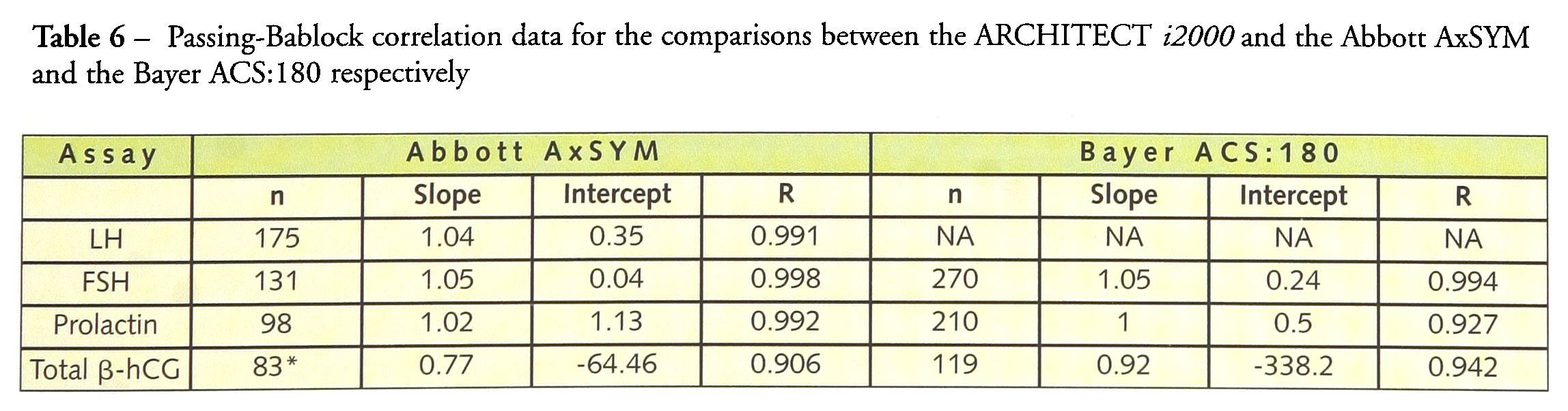
samples above the dynamic range were run using the autodilute function of each assay
DISCUSSION
The ARCHITECT i2000 heralds a new trend in the automation of immunoassays in the clinical laboratory. The software and immunoassay technology used on the i2000 is also used on two other smaller instruments of the ARCHITECT family, the i500™ and the i1000™ analyzers currently in development. In this way Abbott is able to meet the needs of laboratories with quite different workloads. In addition, if a laboratory takes another ARCHITECT analyzer of a different size, the assays will be the same, obviating the need to re-establish reference ranges. In addition, the operating software will have only a few differences. Further, the i2000 is also designed to bolt to a chemistry analyzer, the c8000. The modular design allows the laboratory to automate the majority of their assays on one machine which itself can be expanded to up to four analysers in any combination. This approach has a number of advantages. The footprint of the instrument is smaller than linking individual instruments with a sample tracking system, thus saving what is usually limited space. The combined analytical processing provides economy of use of sample and reduction in the need for aliquoting and movement of specimens around the laboratory from one instrument to another.
Our evaluation of the reproductive peptide assays has shown that these assays are highly sensitive and have excellent precision. It should be noted that the use of commercial QC material has not allowed the precision to be examined at low concentrations in the LH and FSH assays. Concentrations down to about 0.5 IU/L are found in normal subjects and precision should be examined at these low levels. The prolactin concentrations of the QC samples do cover the concentration range found in most clinical samples and excellent precision was demonstrated. At the very low concentration of 1.5 IU/L, the hCG assay had a total precision of 19% suggesting that the functional sensitivity of this assay is possibly about 2.5 IU/L. This is adequate clinically although some technologists and clinicians may prefer a lower functional sensitivity.
Dilution of serum specimens with high concentrations of analyte showed that all assays demonstrated excellent linearity. This demonstrates that the diluent is appropriate and that as the proportion of serum decreases, there are no matrix effects due to the diluent. Because of the wide working ranges of these assays dilution of sample should not be required with the LH and FSH assays and only very occasionally in the prolactin assay. Dilution of samples will be more common in the hCG assay where concentrations > 100,000 U/L can be encountered.
The ARCHITECT i2000 LH, FSH and prolactin results showed excellent correlation with the Abbott AxSYM results, which will enable a laboratory to transfer assays to the new instrument with little or no change in the reference ranges. These assays showed good agreement with the ACS: 180 with a little more scatter about the correlation line in the prolactin results. A personal communication with a representative of the company indicated that the company had endeavoured to develop assays on the i2000 which gave results in close agreement with the results from the AxSYM assays. It is surprising therefore to find that the hCG results from the i2000 are about 25% lower than those from the AxSYM. A better agreement was found between the i2000 and the ACS:180 results. It is not known whether this was an intentional development by Abbott.
One feature which is missing from this instrument is full automatic reflex testing of specimens. Samples track from the left side of the instrument, across the front where the specimen is pipetted, and on to the right side where they are held ready for manual removal. However, the software identifies the samples that need dilution or further analysis and indicates to the operator which rack, and in which position in the rack, the specimen is located. The operator may then transfer the specimen or the whole rack back to the left side of the instrument for further analysis.
The instrument evaluated in this laboratory was a pre-launch analyzer which did not have the most up-to-date software. Nevertheless, the machine was found to be very reliable during the four months it was used in this laboratory. Problems encountered were all in the first two weeks after which it was used every day for most of the day without trouble. The software was very user friendly and took only about a day to learn. Maintenance is straightforward and easy to carry out. Daily maintenance takes about ten minutes with a weekly maintenance of about thirty minutes. The instrument has been designed so that boards and many parts can be readily changed by the operator. It is the intention of the company that a stock of replaceable parts should be kept at the user laboratory. The range of parts has yet to be decided but there may be a basic range of parts, identified as needing more frequent replacement, held by all laboratories. A much wider range of parts may be held by one laboratory which would be available to other users in the area. Together with a direct modem link to technical services downtime should be kept to a minimum.
In summary, Abbott have developed a fully automated random access analyzer which is both part of a family of instruments and part of a modular system. This approach provides flexibility, scalability and commonality to meet the ever changing needs of today's laboratories. The modular system allows the automation of both immunoassay and chemistry assays on one instrument within a minimum footprint. It offers consolidation of pathologies, better deployment of staff and economic use of patient specimens.
