Abstract

Abstract

The South Bend Medical Foundation, Inc., recently installed the Boehringer Mannheim / Hitachi Clinical Laboratory Automation System. The planning process and investigation of Total Laboratory Automation (TLA) started in January 1995 with installation and operations commencing in December 1996. During this two year process, information was learned that could benefit others in the clinical laboratory field as they plan for TLA. Information gained by the author from discussions with automation vendors, experts and colleagues, from attendance at various seminars and from having actually gone through the process forms the basis of this article concerning the planning process. The article is intended to be an overview of the planning process used by one laboratory and not a detailed discussion related to all issues that may arise when planning for TLA.
As the field of laboratory medicine faces centralization, networking and downsizing, laboratorians continue to debate the merits of the various strategies to meet successfully these challenges. Any action plan selected and implemented must be in agreement with the organization's long term strategic plan. The plan must support customer “added value”, improve organizational performance, enhance the strengths of the organization and assist the organization in its long term survival. One strategy being explored by many is the concept of Total Laboratory Automation (TLA). TLA can be defined as the automation of the pre-analytical, analytical and post-analytical functions associated with laboratory operations. The automation of these functions is achieved through use of computer systems, conveyor systems and self operating devices. Pre-analytical self operating components can include:
Primary tube sorter
Centrifuge
Tube de-capper
Aliquotter
Aliquot tube re-capper
Barcode labeler
Aliquot tube sorter
With a TLA system, specimens are automatically transported between pre-analytical components and analyzers by a conveyor system. Analyzers will either sample a specimen directly from the conveyor system or the specimen will automatically be loaded on to the analyzer. Specimens for storage can be transported to automated refrigerated units for later retrieval. A laboratory exploring TLA will determine which components are the most effective for its operation.
Numerous conferences on TLA have been attended by many laboratorians throughout the country over the last two years. The conferences have addressed the differences between a turn key and modular design systems, the merits of pre-analytical equipment versus a total automation system, and the potential costs and benefits associated with the equipment. One aspect of TLA that has not received much coverage is the planning. Many have asked the question—“Where do I start?” As we all know, the success of any project is related to the planning that takes place prior to any acquisition or implementation. This article will focus on the planning process utilized by the South Bend Medical Foundation, Inc. (SBMF) located in South Bend, Indiana for its total laboratory automation project. SBMF began installing the first Boehringer Mannheim / Hitachi Clinical Laboratory Automation System (CLAS) in North America on October 22, 1996. But, long before installation, the planning process started.
The planning process began in January 1995 and continued to evolve as the project unfolded. While there are key components to any planning process, details may be different from one organization to another. Readers should bear in mind that this was one laboratory's planning approach and is intended to be an overview of the planning process and not a detailed discussion related to all the issues that may come up when planning for TLA.
Personnel Involvement and Education

People throughout an organization can be affected by any project of this scope. Since they have information that is important and useful, it is recommended that a multidisciplinary committee be established to investigate TLA. Our multidisciplinary committee had managerial representation from the hospital operations area, the patient services area responsible for nursing home and outpatient drawing sites, the centralized specimen processing area and the central laboratory hematology / chemistry area. This group educated themselves and investigated the concept of total laboratory automation while keeping other managers and departments within the organization informed of our progress. As the investigative steps progressed, additional representation from chemistry, hematology and immunology was incorporated to focus on the more detailed aspects of the project and the automation system under consideration.
The committee members reviewed available literature on automation systems, arranged for presentations by various automation vendors and discussed this concept with colleagues. As the committee became better educated, a one hour in-service was prepared and given to over 300 of our 500 employees. This communication was important to educate further all employees on the TLA concept and to alleviate staff anxiety about downsizing concerns. The in-service included videos of the various systems under review and an explanation of how staff reductions would be handled if a decision was made to move forward with implementation. In our organization, staff reductions are handled through attrition and reassignment to needed vacant positions rather than layoffs. In addition, personnel were assigned to evaluate and establish new test procedures for in-house applications instead of referring to a reference laboratory and to investigate new developing areas such as Molecular Diagnostics. Once committee members became familiar with the features of automation systems, desirable automation components were identified. Initially, our focus was on pre-analytical equipment, but we quickly discovered that automated lines would have to come from the vendor selected for pre-analytical equipment. We decided, given current circumstances, that “mixing and matching” automation equipment from different vendors would not work. We also determined that a system which included both pre-analytical and analytical processes should be considered.
As the workstations that were to be part of the automation project were identified, it became evident that—regardless of the automation vendor chosen —not all current instruments would be adaptable to the systems. As TLA is still in its infancy stages, equipment vendors and automation vendors have a long way to go to achieve compatibility with one another. Not all current equipment may be compatible with the selected automation system. New instruments may have to be considered. The last educational activity undertaken was a trip to Japan to visit five different laboratories. These laboratories were approximately the size of our laboratory based on number of vacutainer tubes handled per day.
Data Collection
Through the educational process, it became evident that the traditional data—revenue, number of tests performed based on inpatient and outpatient statistics — were not the most crucial elements for review. More important data included: the number of tubes collected by tube type, by time of day, by arrival rate based on where the automation system would be housed, by the number of aliquots prepared hourly from each tube and, lastly, how tests were ordered in combination with one another.
A number of different forms were created to gather and collate this data. All the data were collected manually with the exception of test ordering combinations. One form used to gather data is shown in Figure 1: “Total Number Of Tubes Collected By Type And Collection Site.” This form summarized the number of tubes collected by each site throughout our laboratory system. From the data, one could also approximate the number and type of tubes that would require pre-analytical processing. Supporting forms were used to identify the actual number of tubes centrifuged.
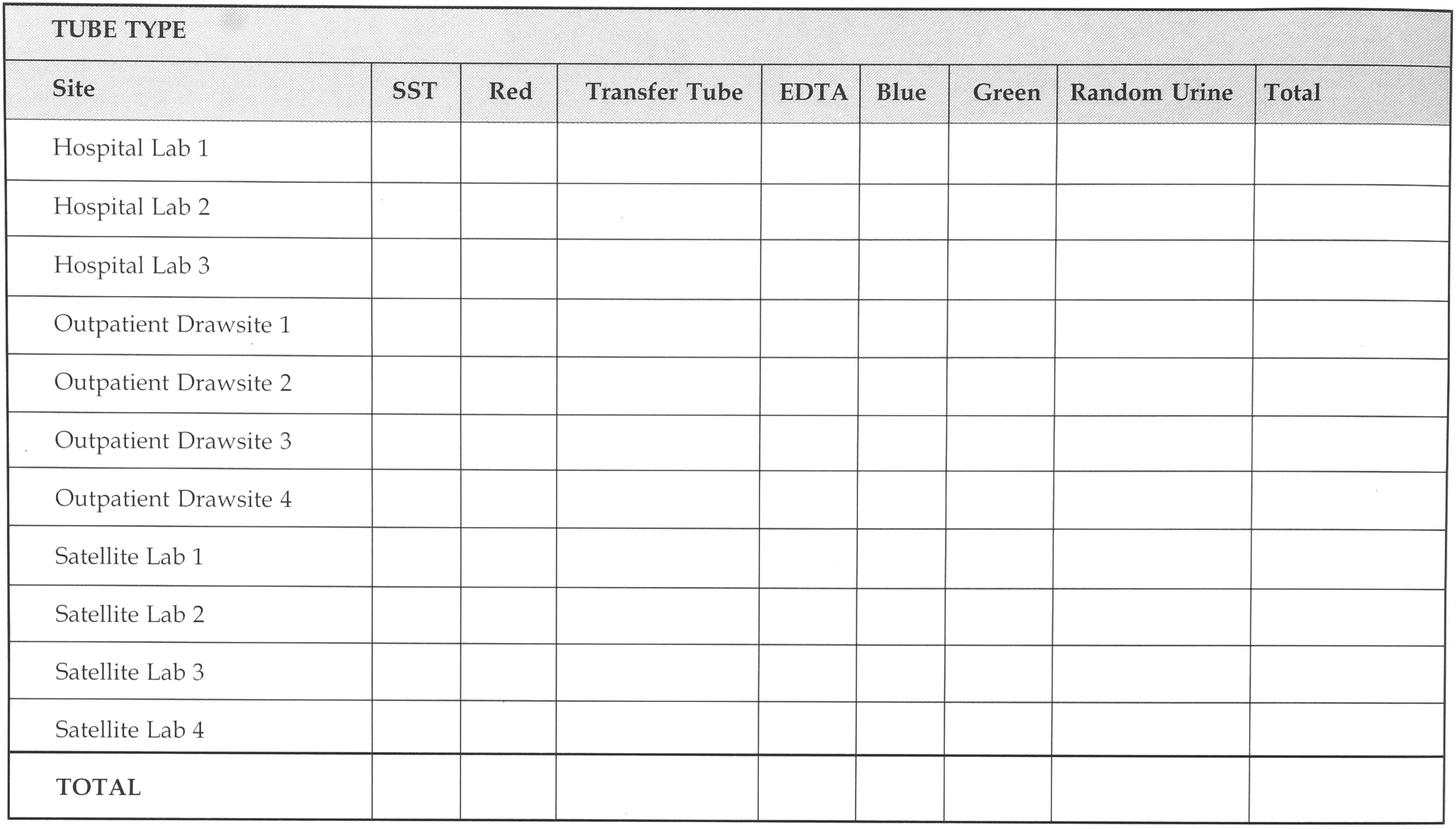
TOTAL NUMBER OF TUBES COLLECTED BY TYPE AND COLLECTION SITE
A similar illustration to Figure 1, can be used to identify the total number of tubes by hour that could be placed on the automation system as well as the number of tubes to remain on site for a particular laboratory. Not all specimens from all laboratory sites could be transferred to the Central Laboratory.

Data on the number of manual aliquots made by hour from serum separator tubes and red top clot tubes was captured on the form. This information was useful to evaluate the need and capacity requirements for an aliqoutter, aliquot tube re-capper, barcode labeler and aliquot tube sorter. Supporting forms identified the average number of aliquots made from each primary tube.
Other forms were used to tabulate the number of microtainer specimens, as well as data related to the type and size of collection and transfer tubes used within our laboratory system. We worked with our Laboratory Information System (LIS) partner, Cerner Corporation to extract the LIS ordering data and load it into a PC for further stratification and analysis by Boehringer Mannheim Corporation, our TLA partner. All of the information collected needs to be reviewed by your potential automation vendors. This information, coupled with forecasts of future volume increases, can help vendors size, configure and price the automation system. The data will identify workload “peaks and valleys.” It can also be used to calculate total volume capacity for the TLA system as well as identify potential hourly capacity problems for any particular module. If the automation vendor provides simulation modeling, this data can show the impact of TLA on laboratory operations.
Standardization
When considering automated pre-analytical equipment, tube standardization is required. All the various collection tubes and transfer tubes used within the organization and received from outside sources need to be identified. It may be necessary to convert to plastic collection tubes and standardize on certain sizes of vacutainer tubes (i.e., 13mm × 75mm, 13mm × 100mm).
Information supplied by automation vendors will contain the tube limitations—height and diameter—for the particular system being considered. Should tube standardization not be achievable or desirable by the organization, pre-analytical equipment most likely will not be a feasible part of your automation project. “Exception” specimens must be identified and possible changes considered. “Exemption” specimens may include:
Specimens received in transfer tubes instead of vacutainer tubes
Frozen specimens
Specimens or requests for special handling
Microtainer collection tubes
Urine containers
STAT specimens
Inappropriate sized tubes
Will these situations be handled manually or can solutions be implemented to reduce the number of “exemption” specimens?
Automation effectiveness is predicated on reducing the number and nature of “exception” specimens. Effectiveness is enhanced when all specimens are handled in a consistent manner. “Exception” specimens can be a major issue, especially in those laboratories that handle both hospital inpatient work and outpatient / outreach work. The amount of time necessary to sort, process and test a specimen through the TLA system may determine the system's effectiveness related to “exception” specimens.
Management Structure and Staffing
Converting to an automation system may necessitate review and changes to the management structure. The scope of an automation system can include the technical disciplines of hematology, chemistry, coagulation, immunoassay and urinalysis. In many laboratories, these areas may each have their own pathologists, managers or supervisors. Responsibility for the automation system will need to be assigned. The technical disciplines affected will also need to be determined.
The magnitude of the TLA project, centralization activities undertaken and an organization's management philosophy will determine what changes can be made to the management structure. In our organization, changes in management responsibilities and reductions in management numbers have resulted due to implementing TLA and the further centralization of laboratory testing.
The extent of cross-training of technologists / technicians in the disciplines that are part of the TLA system is a consideration. Staff members should be versatile in all the technical disciplines that comprise the automation system. Having a narrow focus in one discipline may impede system optimization. The automated laboratory is geared towards high volume work and a “generalist” in many disciplines may be preferred over a “specialist” in one discipline. This author believes the territorial boundaries related to the technical disciplines of hematology, chemistry, coagulation, immunoassay and urinalysis need to be modified to achieve operational success. Department alignments and responsibilities should be based on “like functions and focus,” not on traditional disciplines. Advances in automation technology requires one to think differently in order to optimize the impact of these new tools. As the TLA project progresses, staffing forecasts and functions to be performed should be considered and communicated. The major savings from TLA is in reduced labor needs. Staff members should be informed as early as possible on how staff reductions will be handled. Site visits, vendor discussions and critical review of operations can approximate the labor force needed to staff the TLA system.
Secondarily, decisions will be needed on how staff members for the new automated laboratory will be selected and what qualifications are needed—how many technologists, technicians, laboratory assistants or clerks. With a TLA system, some functions are best handled by technologists, some by laboratory assistants. Preventive maintenance, quality control and quality assurance functions still must be performed. Racks and tubes need to be placed on the system or taken off; aliquot tubes, caps, barcode labels and pipette tips need to be loaded onto the appropriate TLA modules; reagents and supplies need to be loaded on to analyzers.
After we determined the type and number of positions needed in the automated laboratory, these positions were posted throughout our organization. As this was viewed as a new department, all interested and qualified personnel could apply. Applicants were interviewed and selections made. Training was then planned and implemented for the new automated laboratory staff members.
Facilities

A critical review of one's facility is needed. Not only space, but the electrical, HVAC, plumbing and water supply requirements need to be assessed and possibly enhanced. Structural supports may need to be reviewed by the appropriate experts, based on where the system is to be located. Elevators and hallways need to be large enough to accommodate the size of the various automation components that need to be moved into the laboratory. If you are installing the TLA system in an existing laboratory, remodeling may be needed. Physical barriers separating departments may need to be removed. Rearrangement of departments that receive aliquots from the system may be desirable so they are in close proximity to the TLA system. Working with your laboratory architect and TLA design experts will assist in developing the best laboratory configuration. An automation system can be installed within an existing laboratory, but it requires more detailed planning about how operations will continue as reconstruction and installation occur. Patience and perseverance may be needed by all! Our automated laboratory is located on the second floor. Not all components could be moved up with existing elevators, so the automation components were lifted up and passed through an opening made in the exterior wall. Taking advantage of a construction lull allowed us to lift the components and slide them into the second floor space.
Laboratory Information System
An integral part of a laboratory automation system is the data supplied by the LIS. Typically, the LIS will download test orders and accession numbers. Prior to implementing TLA, our accession number was assigned by patient, regardless of how many different tubes were collected from that patient. Unless each tube has a unique identifier, the TLA system cannot process it. In order to generate a unique accession number for each tube, we implemented container tracking software. The unique tube identifier allows the TLA system to sort the collection tubes, apply the appropriate pre-analytical steps and route the specimens to the appropriate testing analyzers. Access to the system is dependent on its ability to read the barcode and process the sample accordingly. If labels are applied incorrectly, a clot tube can be passed down the hematology line.
Additional LIS features to consider include auto verification in order to maximize results reporting. Storage location of the primary tube or aliquot tube for future testing may also be desired. Should this storage information be stored in the LIS or the TLA system? Thought must also be given to the routing of instrument interfaces—directly back to the TLA system computer or to the LIS. The LIS capabilities must be assessed and early discussion with your LIS vendor is recommended.
Cost / Benefit Analysis
The cost of implementing a TLA system is not only the TLA system cost, but also includes the cost of any new analyzers, computer interfaces, remodeling of facilities and process reengineering. As a TLA system is custom designed for a particular laboratory's operation the cost of remodeling and process reengineering will be different for each laboratory. There are four primary benefits of TLA, but not all benefits can be easily quantified financially. As more work is consolidated in one location, and department boundaries modified, the greater impact TLA can have on operations. The benefits include:
Reduced labor costs due to workstation consolidation and the automation of the pre-analytical, analytical and post analytical functions. Increases in workload can be handled easier and need not always result in more staffing. The efficiencies gained by using TLA will allow us to handle additional workload and reduce staffing by approximately 15 FTE's.
Improved safety due to the less frequent handling of specimens.
Improved turnaround times for all specimens due to the consistent and timely application of pre-analytical functions and the automated transportation to analyzers.
Improved quality due to reduction in manual aliquotting and relabeling errors as well as reduced specimen collection from patients.
Conclusion and Summary
The planning process for a Total Laboratory Automation project is vast and complex. The success of the project will correlate to the amount and depth of planning that was done. The planning process involves not only the automation vendor, but also many departments within an organization as well as instrument vendors. The LIS vendor must be brought into the discussions early to assess its needs and issues related to an automation system. Keeping staff members informed and involved in the process is critical for acceptance. New information is needed to assess the need for a TLA system and to identify challenges which need to be overcome. As more laboratories install automation systems and discuss their planning process, data elements important to the planning process will become second nature to all of us.
Footnotes
Acknowledgements
The author acknowledges the help and assistance of many members of the South Bend Medical Foundation Management Team who reviewed this manuscript.
