Abstract
Aurora Biosciences Corporation designs and develops proprietary drug discovery systems, services and technologies to accelerate and enhance the discovery of new pharmaceuticals. Aurora is developing an integrated technology platform centered around two technologies; 1) a portfolio of proprietary fluorescent assay technologies and, 2) an ultra-high throughput screening (“UHTS”) system designed to allow assay miniaturization and to overcome many of the limitations associated with the traditional drug discovery process. This approach takes advantage of the opportunities created by recent advances in genomics and combinatorial chemistry that have generated many new therapeutic targets and an abundance of new small molecule compounds. Aurora believes its integrated platform will accelerate the drug discovery process by shortening the time required to identify high quality lead compounds and to optimize those compounds into drug development candidates.
Background and Need
Recent developments in molecular biology and genomics as well as combinatorial chemistry have created significant opportunities to discover greater numbers of high quality lead compounds for development into new medicines. The advances in molecular biology and genomics have resulted in a greater understanding of the molecular and genetic basis of disease and have led to the identification of many new genes as potential therapeutic targets for drug discovery. Many companies have used combinatorial chemistry to create quickly libraries of hundreds of thousands or even millions of small molecules for screening against established and novel targets. However, the increasing numbers of targets and compounds have created severe bottlenecks in the drug discovery process. These bottlenecks result from the difficulty of analyzing quickly the function and disease relevance of newly discovered targets, the complexity of incorporating the many different types of targets into screening assays, and the inability to screen extensive compound libraries quickly and at a reasonable cost.
The UHTS system
Targets are specific biological molecules, often proteins such as receptors, enzymes or ion channels, which are believed to play a role in the onset or progression of disease. Most drugs work by binding to a target and modulating the target's biological function or activity. Thus, most drugs are discovered by identifying compounds that modulate an established target's biological function. Until recently, pharmacologists and molecular biologists had identified only a few hundred targets using conventional methods.
Assays are employed to determine the effect of a compound upon a particular target. When applied methodically, assays can be used as screens to identify active chemicals, referred to as “hits,” that may produce a desired effect upon a target's function.
Lead compounds can be identified by additional screening of hits and may then be optimized to generate candidate compounds for development as potential medicines. Screening is the process of methodically testing libraries of compounds for potential therapeutic value by using assays to determine if any compounds affect a selected target. Primary screening determines if any of the compounds tested are hits. Re-testing confirms initial hits and secondary screening refines the initial evaluation of hits. For example, secondary screening may measure a hit's potency (the amount of the hit compound required to exert its effect) and specificity (the degree to which the hit does not affect unintended targets). These secondary screens help in selecting lead compounds for further discovery efforts to identify candidate compounds for development.
The discovery and development of new medicines remains an expensive, time-consuming and often unsuccessful process. Although many pharmaceutical, biotechnology and clinical research organizations have improved the efficiency of the drug development phase significantly, only about five to ten percent of candidate compounds entering development will ultimately be approved for marketing. Candidate compounds that are identified in discovery frequently fail in the development phase due to insufficient therapeutic benefit or unexpected side effects. To date, efforts to improve the initial discovery process have been inadequate. If the discovery process were sufficiently improved, pharmaceutical and biotechnology companies could discover, quickly and efficiently, larger numbers of higher quality candidate compounds that have a greater chance of development into medicines that meet significant unmet needs.
Until the last five to ten years, screening was a labor intensive manual process in which it was generally possible to test only tens of compounds per day. Today, pharmaceutical and biotechnology companies with advanced drug discovery programs use semi-automated or robotic high throughput screening systems to perform 96-well assays. These systems, however, are unable to accommodate miniaturized assays that are critical for screening at increasing rates. Certain current best practice screening systems can operate at throughputs of up to approximately 10,000 discrete compounds per day per system, but typically such systems operate at throughputs of less than 3,000 discrete compounds per day.
Aurora's Solution
A principal component of Aurora's integrated technology platform is its UHTS system, which is being designed to screen over 100,000 discrete compounds per day in miniaturized assays. The two principal components of Aurora's integrated technology platform are its proprietary fluorescent assay technologies and its highly automated ultra-high throughput system that is being developed for screening miniaturized assays. This unique platform results from the innovative integration of many different disciplines, including fluorescence chemistry, biophysics, molecular biology, protein engineering, automation, process control, optics, microfluidics, informatics and software development.
Aurora's fluorescent assay technologies are being used today to facilitate drug discovery. Fluorescent assay technologies are highly sensitive, and are designed to permit more rapid screen development and the development of miniaturized assays important for cost-effective high throughput screening. They utilize the phenomenon of fluorescence resonance energy transfer which occurs when two fluorescent molecules interact as donor and acceptor over very short distances.
When the donor is illuminated with light at its wavelength of excitation, instead of giving its usual color of emitted light, it transfers energy to the acceptor that now emits at the acceptor's characteristic wavelength. This transfer decreases rapidly if the two molecules move apart, for example if a chemical linker is cleaved, and the fluorescence emission now changes to the wavelength of the donor. The ratiometric signal change generated can give a highly sensitive and reliable readout of molecular proximity.
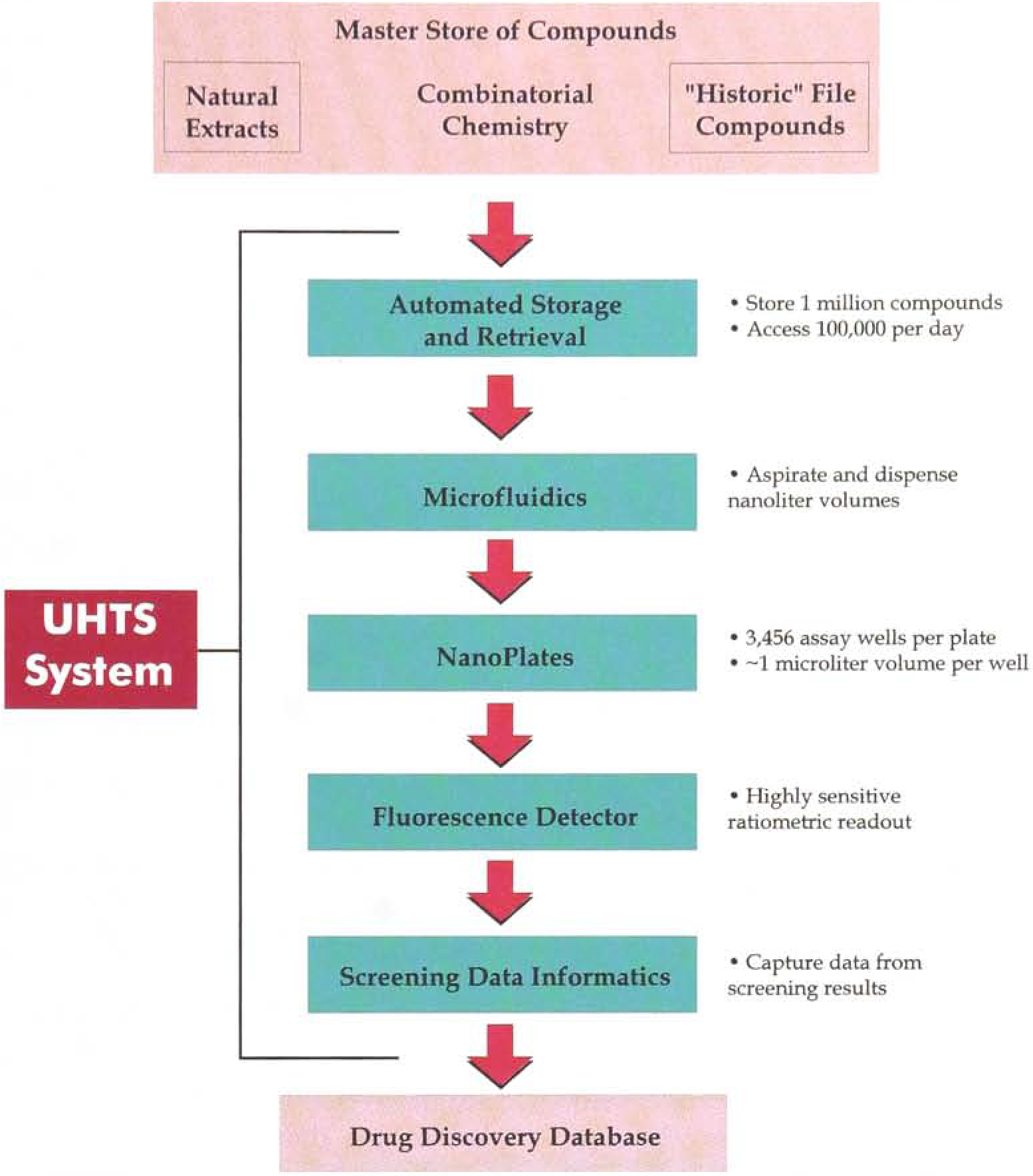
The UHTS system (Figure 1) will combine an automated storage and retrieval system, microfluidic dispensing devices, and NanoPlates in which miniaturized assays are performed.

Automated Storage and Retrieval System Automated Access to Compound Libraries
Specialized fluorescence detectors are designed to record and process the signals from the NanoPlates, with advanced software and informatics to capture the resulting data. In developing the UHTS system, Aurora is applying to the drug discovery process technological advances that have already been deployed successfully in other industrial processes, while adding its own proprietary innovations. In this regard, the company has entered into strategic technology alliances with several technology leaders, including Packard Instrument Company and Carl Creative Systems, Inc.
Ultra-high throughput screening requires automated rapid access both to organized collections of large compound libraries, as well as to individual compounds in the collection for re-testing. Aurora is unaware of any system currently available with such capabilities. The UHTS system's automated storage and retrieval system (Figure 2) is designed to house over 1,000,000 compounds in solution for immediate access. The system is designed to allow ready replenishment of the compound store from libraries in the master store. The robot that operates between the racks of compound storage plates is designed to deliver (and once sampled, return to the store for further use) over 100,000 selected compounds per day for primary screening and over 2,000 hits for re-test and potency determination. The ability to deliver selected compounds to the screen at ultra-high rates under computer control is a key advance offered by the Aurora plat form. The automated storage and retrieval system is also designed to facilitate high throughput screening in conventional 96 and 384-well plates. The robotic systems are being adapted from other industrial settings where automated, rapid access to very large stores of small items have been reliably deployed. Aurora is customizing the system for ultra-high throughput screening. This approach brings to the drug discovery process technological advances that have already been deployed successfully in other industrial processes.
Advanced Instrumentation for Small Volumes
Aurora has developed novel screening plates (NanoPlates) and innovative microfluidics to enable miniaturized assays in volumes approximately 100 times smaller than conventional screens. Smaller volumes reduce the amount of expensive or scarce reagents that may be required in a screen.
A key component of Aurora's UHTS system is the NanoPlate (Figure 3), which has 3,456 miniaturized wells in which fluorescent assay screens may be performed. A vital feature is the small assay volume (approximately 100 times smaller than conventional screening assays), which is critical for reducing cost per test and conserving compounds made by combinatorial methods that produce only very small amounts of each test compound.
Current technologies for dispensing small volumes of liquid cannot meet the requirements for screening in NanoPlates. Aurora, together with Packard, is developing microfluidic technologies to transfer, accurately and rapidly, microscopic volumes of the compounds into the miniature assay wells of the NanoPlates at rates of up to 10,000 wells per hour. While current screening systems can dispense volumes down to a microliter, Aurora is developing miniaturized screening dispensers capable of accurately dispensing sub-nanoliter volumes (less than one billionth of a liter) required for the UHTS system. These sample dispensing devices are designed to remove small amounts of the compounds from the storage plates and dispense at high speed precise sub-nanoliter volumes into the appropriate wells of the NanoPlates.

A key component of Aurora's UHTS system is the NanoPlate, which has 3,456 miniaturized wells in which fluorescent assay screens may be performed.
There are a number of fluorescence plate readers presently available which enable the use of Aurora's proprietary fluorescent assays for high throughput screening in 96-well plates. However, none of these provide the necessary sensitivity and precision to enable ultrahigh throughput miniaturized screens. Aurora is also collaborating with Packard to develop highly sensitive fluorescence detectors capable of measuring miniaturized fluorescent assays in NanoPlates. The detector is designed to record and process, in real time, data from more than 100,000 assays in 24 hours.
Aurora's detector is designed to measure the signals generated from the various fluorescent assays over the range of different wavelengths that the various reporters require and to acquire rapid ratiometric data.
Informatics and System Integration Successful overall integration of the UHTS system will require a strategy for user-friendly computer control.
Aurora will be required to link the operation of the automated storage and retrieval system to existing chemistry information databases and master compound store inventories in the discovery operations of its collaborators. The integration of the entire system will need to ensure that the large amount of screening data from the UHTS system is efficiently captured, processed and deposited in a centralized database.
Aurora is integrating advanced software tools and systems from leading providers. While the basic building blocks from leading suppliers, the supervisory control systems, the subsystem controllers for the instruments, the data analysis tools and overall system architecture and database structure for the UHTS system are being developed by the in-house informatics team.
Conclusion
In order to address these issues, the Company is integrating advanced technologies to develop superior assays, to enable analysis of gene function in mammalian cells and to miniaturize and accelerate compound screening.
Aurora's goal is to become the leader in the development and commercialization of technologies that will accelerate and enhance the discovery of new medicines.
Aurora seeks to diversify business risk by generating revenue from multiple collaborators seeking to exploit Aurora's fluorescent assay technologies and UHTS system in many different drug discovery programs.
Aurora expects to generate revenue by developing screens, providing screening services, developing and providing UHTS systems to syndicate members, licensing its proprietary technologies, and realizing royalty and milestone payments from the development and commercialization of drug candidates identified using Aurora's technologies.
To date, Aurora has entered into collaborative agreements with BMS and Lilly to license Aurora's fluorescent assay technologies for their internal discovery research, to collaborate on screen development and as initial members of a syndicate to co-develop Aurora's UHTS system.
In addition, Aurora has also entered into agreements to develop screens for or provide screening services to Sequana Therapeutics, Inc., Allelix Biopharmaceuticals, Inc. and Roche Bioscience. Aurora has also entered into agreements with Alanex Corporation and ArQule, Inc. providing Aurora with non-exclusive access to certain of their combinatorial chemistry libraries.
Footnotes
Acknowledgments
The authors would like to thank the researchers, engineers, and business development staff for their contributions to the design and development of the system presented here and in composing this manuscript.
