Abstract
The clinical laboratory is moving from an activity focused on the analysis to an in vitro diagnostic service network. Laboratory analysis is a disassembly process, very different from manufacturing processes which are assembly processes. Specific quality goals must be discussed before adopting robots. As an example, in order to achieve the ultimate quality goals in our laboratory, it was necessary to use automation for sample handling. We developed automation in a new multidisciplinary model at the Institute of Biology of Nantes. Our multidisciplinary laboratory included clinical laboratories, research laboratories, and start-up companies in an open space of 10,000 m2 shared between two floors. Sample transportation is automated from clinical wards to laboratory workstations.
INTRODUCTION
Automation is often presented as the driving force of the evolution of the clinical laboratory and the full automated laboratory as the ultimate target to reach.
The activity of the clinical laboratory is not a manufacturing process and manufacturing tools should be used only if they fit with the specific requirements of laboratory professionnals. The first step in laboratory reengineering is to organize all the disciplins of the clinical laboratory in a technical platform to optimize sample handling, and equipment utilization ratio.
FROM SAMPLE ANALYSIS TO AN IN VITRO DIAGNOSTIC SERVICE NETWORK
Traditionally, clinical laboratory activity could be defined as a qualitative or quantitative determination of an analyte or a cell in a patient sample; analytical systems, data processing, quality control, and a good know-how are necessary to perform sample analysis.
Currently, all the steps from sample collection to tests results reporting to the practitioner and the patient are included in quality assurance. A new definition of our profession is necessary to establish the basis of quality requirements:
• From a patient sample, collected in the right container, according to doctor's prescription, transported without damage to the laboratory, produce relevant information, within minimal delay, for prescribing the doctor and patient.
It is crucial to keep in mind this enlargement of our responsibility as many laboratories are merging to face with cost containment, thus increasing sample handling, automation, and information processing. Exchanges of samples between specialized laboratories require a strict customer-supplier relationship similar to subcontractors in industry business.
QUALITY REQUIREMENTS FOR LABORATORY AUTOMATION
Considering various examples of full automated laboratories (1,2), often discussed as the future of the Clinical Laboratory, we would like to make some comments:
It is obvious that such laboratories answer only to a part of the first level of In Vitro Diagnostic (IVD), a kind of screening activity.
Robotic or conveyor belts technologies were transferred from manufacturing processes to IVD activities by copying sample handling done by the operator without looking at the eventual added value, lost after job elimination.
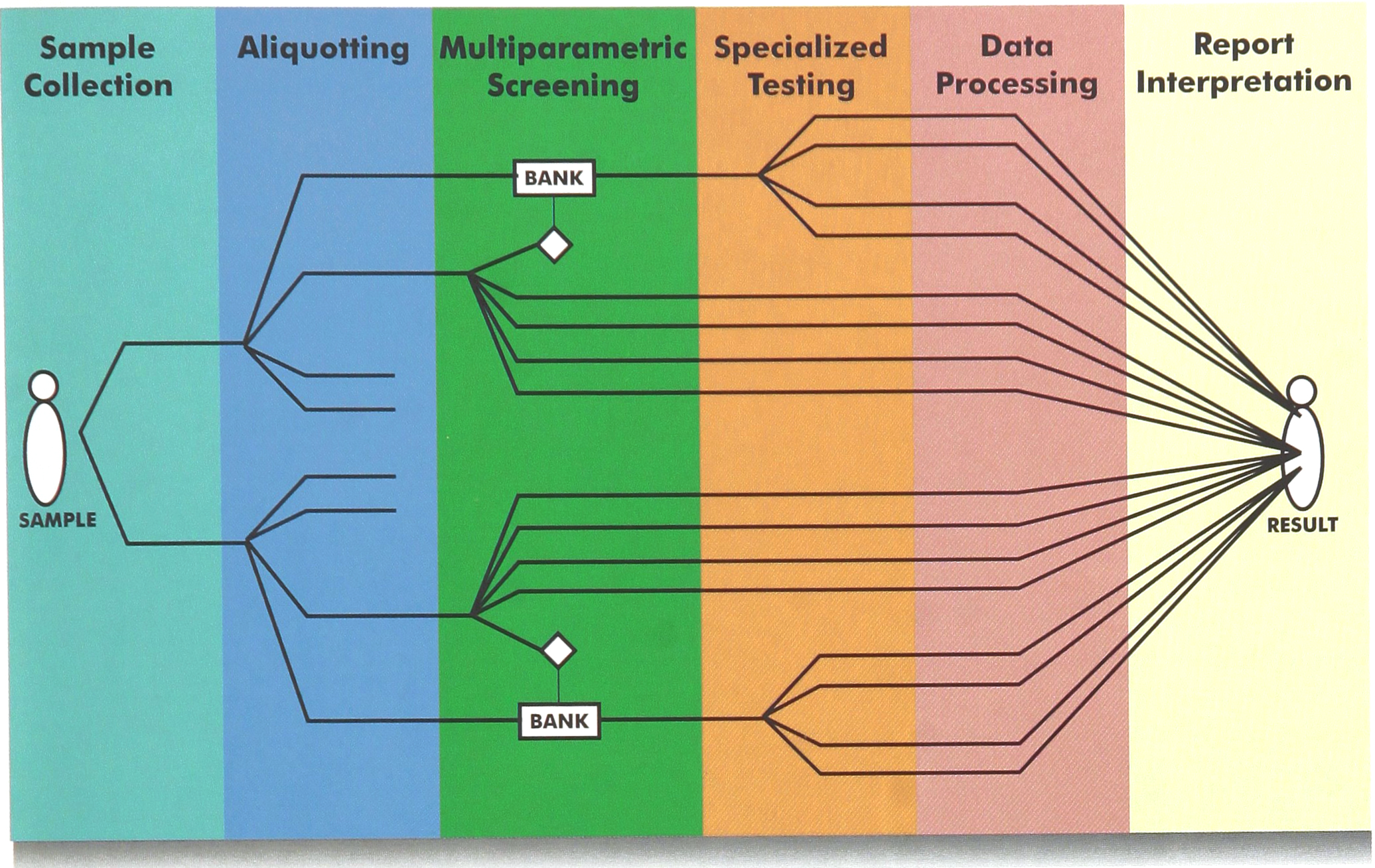
Full automated laboratories are based on a assembly line concept which is an assembly process (Figure 1); the complete Clinical Laboratory process includes different samples from one patient, different levels of investigations involving various automated or manual methods, used in a deductive reasoning: it is a disassembly process until final synthesis in the report of the patient (Figure 2).
Fully automated laboratories are based on a assembly line concept which is an assembly process A complete Clinical Laboratory process includes different samples from one patient, different levels of investigations involving various automated or manual methods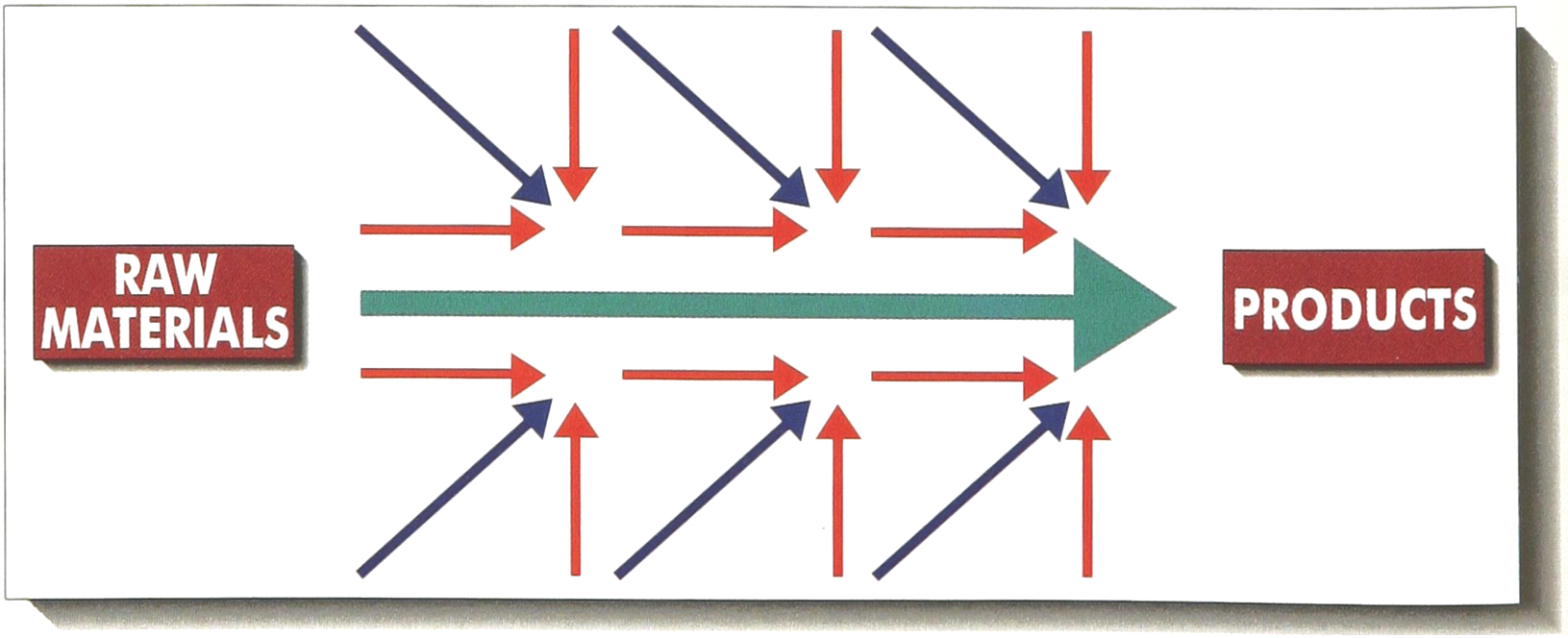
Specific quality goals should be established between laboratory professionals before choosing automated tools; we will discuss here the example of sample handling, and quality goals in this area may be divided in four items:
Patient and prescriptor: avoid mistakes requiring a new sample collection.
Sample: reduction of sample volume, identification and tracking all along the process, preservation, biosafety.
Operator: decrease sample handling, locate and contain biohazard (3).
Cost containment: decrease human labor, number of tubes, and paper work.
We can apply this quality goals to various steps of sample handling: sample transportation, centrifugation, detection, and aliquotting:
Sample transportation devices: controlled access to the samples, protection against breakage and leakage, separation between samples and documents, easy decontamination, thermal stability, tracking and traceability, time limit.
Automated centrifuges: traceability (rotor, speed, duration, and temperature), biosafety (isolated area, closed covers for centrifugation baskets to avoid aerosols), automatic detection of eventual tube breakage before further robotic handling, easy decontamination.
Sample detection: volume, clots, hyperlipemia, haemolysis, and hyperbilirubinemia.
Sample aliquotting: ability to use different forms of secondary tubes, checking of identification before aliquotting, no carryover by tips or fluidic device, compatibility with sample storage.
Sample loading: final control relating to the status of the sample to be analyzed in a specific workstation, eventual false identification, but also to the medical and biological history of the patient.
If we compare to analytical systems, first robots were only copying operator's manipulations, then analytical systems using sophisticated sensors were developed (3,4,5); the same evolution should occur in automation of sample handling but, until that day, it is preferable to keep automated sample handling under human control as it is the case in automated workstations, so-called islands of automation.
THE INTEGRATED LABORATORY PLATFORM
Approved technologies can be integrated in a multidisciplinary laboratory platform; this concept was implemented in the Technopole of the University Hospital of Nantes (France) with 10,000 m2 of laboratories, including clinical laboratories, but also research laboratories, and some start-up companies (Figure 3). Per day, 20,000 tests covering all the state of the art in Clinical Biology are performed by 100 technicians at the lowest cost in France for University Hospitals.

Technopole of the University Hospital of Nantes (France)
Samples are transported automatically from clinical wards (50 stations) to the laboratory reception area in closed containers mounted on 2.5 km of rails (Figure 4).

Closed container transport.
After sample sorting and eventual centrifugation in an isolated safe area, samples are aliquotted if necessary using a Tecan Genesis liquid dispenser which was adapted to our requirements (Figure 5).

Automated sample aliquotting.
Then, the racks of tubes are transported to the workstations by an internal carrier using the same system of rails.
The open-space architectural concept (Figure 6) allows easy ergonomic installation of any automated analytical system, but also of a specialized activity requiring a specific environment (air-pressure control, clean rooms, etc.). Since the opening in 1990, 40% of the surface were modified for different purposes.

Open-space laboratory architectural concept.
DISCUSSION: GUIDELINES FOR LABORATORY ENGINEERING
From our experience, we can discuss some guidelines about laboratory engineering:
Choose a flexible evolutive architecture with mobile walls.
Organize automated sample handling following quality requirements with a common reception area for all the laboratories under the responsibility of a clinical chemist.
Structure and manage different levels of technical investigation, the first one using technologies dedicated to the various disciplines (Biochemistry, Hematology, etc.), the second one using specialized activities shared by all the disciplines (DNA assays, HPLC, Image analysis, Flow cytometry, etc.).
Promote research and revelopment activities around clinical laboratories to anticipate new domains but also assist with the amortization of investments in expensive equipments.
Beside analytical activities, set up transversal logistic and engineering activities: purchasing office, quality assurance, evaluation, sample bank, information, etc.
Beside Quality of the results, integrate Quality Assurance as a management tool between internal and external partners.
CONCLUSION
In 1987, C. Burtis reviewed the evolution from analytical instruments to analytical systems (4) and concluded that our profession succeeded in adapting new technologies to the daily workload because a good basis of standardization in analytical methods already existed, directed towards the quality of the results.
Nearly ten years later, the same author described in a recent review (7), how the technological revolution has affected how clinical laboratories are organized, staffed, equipped, and operated.
Our problem is the lack of standardization in sample handling and information processing and that for such domains, we have to deal with many external partners. The only way to adapt ourselves to this new challenge is to be able to re-think our profession with a global Quality Assurance approach issued from our analytical know-how, but directed towards the patient.
The first step to improve laboratory services is to move from our classical analytical approach which is separated into different disciplines to a system approach modeled on the deductive reasoning of in vitro diagnostic.
Then it will be easy to integrate selected automation and technologies according to the fact that one sample, tracked all along the process, must symbolize one patient with a unique medical history, and that all the results collected on the final report representing a biological portrait of the patient, must benefit of the knowledge of a network of clinical laboratory scientists.
