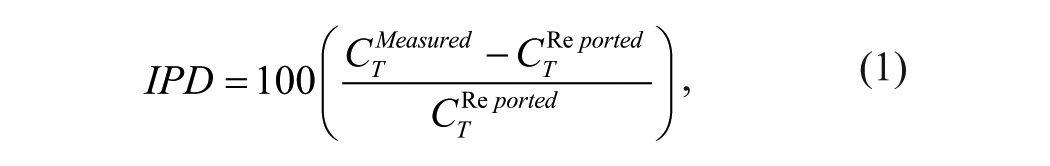Abstract
The aqueous solubility of a drug candidate is a vital physicochemical property that stops a drug candidate from proceeding further in the drug development processes. Classical solubility determination methods, which are commonly used in pharmaceutical laboratories, are expensive and time-consuming. In this work, an automated determination method is proposed that is based on a laser monitoring technique, and the validity of the measured solubilities is checked by comparing the measured solubilities of acetaminophen at various temperatures as proposed in various literatures. An additional set of acetaminophen solubilities in various concentrations of a surface active agent is measured at various temperatures, which has been reported for the first time, and it could be applied in the pharmaceutical industry, where solubilization of acetaminophen in aqueous solutions is required.
Introduction
Information about the aqueous solubility of a drug candidate is essential in drug discovery because poorly soluble compounds have low absorption rates and fail to proceed in drug discovery and development processes. In addition, low-soluble drugs create issues during the earlier stages of bioactivity tests, where it is necessary to make a solution for evaluating pharmacological or toxicological activities. Aqueous solubility investigations are important for oral or parenteral drug liquid formulations; are required for the determination of absorption, distribution, metabolism, and excretion; and are also an important topic in drug extraction and analysis.1,2
The most common method for the determination of thermodynamic solubility of a drug is the shake-flask method. 3 In this method, an excess amount of drug is added to the solubility medium. The added amount should be enough to make a saturated solution in equilibrium with the solid phase. On the basis of the dissolution rate and type of agitation used, the equilibration time between the dissolved drug and the excess solid could be varied. After equilibration, solid and solution phases are separated using either filtration or centrifugation, and the quantification of drug concentration in the saturated solutions is measured by ultraviolet (UV) spectrophotometric analysis. A fast, feasible, and automated setup should be used for the high-throughput screening studies. The shake-flask method is not recommended. The synthetic method,4,5 which is also called laser monitoring technique, 6 last crystal disappearance method, 7 or dynamic method, 8 is the next method for determination of thermodynamic solubility of a drug. The synthetic method is based on disappearance of the solid drug (from the mixture of solvent and drug) monitored by a laser beam. The disappearance of drugs could be achieved either by changing the temperature or by adding a known amount of the solvent.
The solubility apparatus consisted of a jacketed glass vessel (varying volume from 60 to 250 mL) maintained at the desired temperature by circulating water that was provided by a constant-temperature bath. The water temperature was controlled by a workstation with a temperature accuracy of 0.1 K, which is achieved by continuous stirring and a condenser (or a perforated rubber cover) that was fitted to reduce the solvent’s evaporation. A thermometer with an uncertainty of 0.01 K was used to determine the temperature of the system. A laser beam was used as a tool to observe dissolving the solid in liquid. The signal transmitted through the vessel was collected by a detector that decided the rate of temperature rise and estimated the equilibrium point of the given system on the basis of the signal change. The solute and the solvent were prepared using an electronic balance with the estimated uncertainty in the mole fraction of less than 0.001. A predetermined quantity of drug and solvent was placed into the jacketed vessel. The system was slowly heated (the rate of heat increase is 0.5 to 2 K/h) with continuous stirring. When the solute particles disappeared thoroughly, the signal approached a maximum value. The workstation judged the signal difference at 10-min intervals; if the interval was less than 10 min, the workstation gave an order to stop heating and record the temperature. The temperature recorded was equivalent to the temperature of a given solution upon the complete dissolution of the drug. 5 In another version of this setup, predetermined masses of drug and solvent were placed in the vessel, and the contents were stirred continuously at a constant temperature. As the particles of the drug are dissolved, the intensity of the laser beam increases gradually and reaches the maximum value when the drug is dissolved completely. Then an additional known mass of the drug is dispensed to the vessel, and the procedure is repeated until the laser beam cannot return to the maximum value, which means the last addition cannot be dissolved. The total amount of the added drug was recorded and used to calculate the solubility value. 9 The synthetic method is preferred over the shake-flask method for solubility determination of drugs in viscous solvents in which the separation of the excess solid from saturated solutions is not achievable. 10
Yuan and coworkers 11 proposed a rapid solubility determination setup and tested the solubility of 15 pharmaceuticals in which almost higher solubilities were obtained when compared with the shake-flask data. Wenlock et al. 12 reported an automated assay setup for the determination of aqueous solubility for high-throughput screening purposes. The system is based on shaking for 66 h, double centrifugations, and UV quantification analysis. It is capable of determining the solubility of 96 compounds and is validated using measured solubilities of 11 marketed drugs in which the average of percentage deviations is ~60% (four solubility data points were excluded from this analysis because their values were reported as less than a given solubility value). Repeatability of the setup was checked by 30 repeated experiments on glyburide, and the obtained solubility values varied from ~5 µM to 16 µM. After excluding four highly deviated data points, the range was reduced to 8 to 12 µM. 12 Dinter et al. 13 reported an automated solubility determination by combining automated solid dispensing, weighing, and solvent addition setups for the qualitative determination of the solubility of reactants, reagents, and products in aqueous and nonaqueous solvents. A given amount of the chemicals was added, and the observations were categorized as soluble and/or turbid. 13
This work aims to present an accurate, precise, low-cost, and feasible setup for the determination of drug solubilities on the basis of the laser monitoring technique. The setup was validated using the measured solubility of acetaminophen at various temperatures and the corresponding data points collected from the literature. A new data set of acetaminophen solubility in water in the presence of a surface active agent at various temperatures was also reported as an additional set.
Materials and Methods
The experimental aqueous solubility of acetaminophen as a model drug was determined using the developed instrument for validating the setup, and the obtained data were compared with the corresponding data available from the literature. In addition, the solubility of acetaminophen in ethanol and propylene glycol at 298.2 K was determined to check the validity of the setup.
After validation of the solubility data, the solubility of acetaminophen in aqueous solutions containing various concentrations of sodium dodecyl sulfate (SDS; an anionic surfactant) was measured at temperatures varying from 298.2 to 318.2 K.
Instrumentation
In the proposed setup (see Fig. 1 for a schematic representation and Fig. 2 for an actual photograph of the setup), glass tubes (syringes) with different diameters and constant length were used to dispense the drug powder to the dissolution vessel. Low-diameter syringes are suitable for very-low-soluble drugs because they dispense a very small mass of the powder in each dispensing, and larger diameters are suitable for relatively soluble drugs, in which more masses of drugs are required for saturation of the solution. The filled tube with drug powder was weighed using an electronic balance before and after saturation of the solution, and the mass difference determines the mass of drug added to saturate the solution. The saturated concentration of the solution is calculated concerning the mass and/or volume of the solvent added to the dissolution vessel. After introducing the experimental conditions, the setup adds a small amount of drug powder to the solution, and the contents were stirred continuously at a constant temperature with the given uncertainty defined by the user. As the particles of the drug are dissolved, the intensity of the laser beam is increased gradually and reaches the maximum value when the drug is completely dissolved. Then, another mass of the drug is dispensed to the vessel, and the procedure is repeated until the laser beam cannot return to the maximum value, which means the last added powder could not be dissolved. This was checked several times, and then the system was stopped, and the total amount of the added drug was recorded and used to calculate the solubility value. The signals for neat solvents were considered as the maximum intensity of the signals detected by the photoconvertor. Figure 3 illustrates a simple flowchart of the system, which is controlled by an in-house computer program. This setup allows the user to adjust (1) the temperature of the solution, (2) the signal intensity for neat solvent (in the absence of the solute as the background signal), (3) the rate of powder dispensing, (4) the length of time for dissolution of the added powder, (5) the number of equilibration check cycles, and (7) the stirrer rate. Further details can be provided by the authors on request.
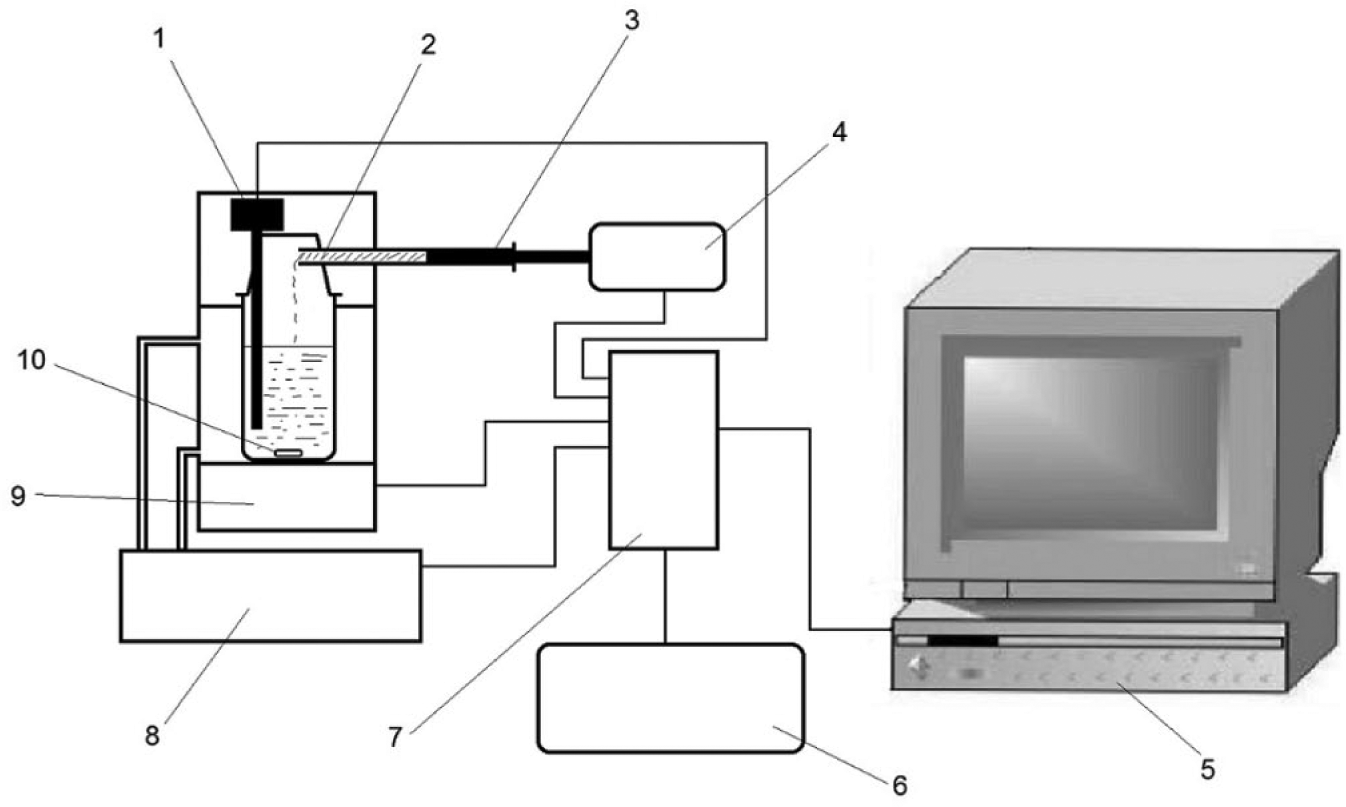
Schematic representation of the setup: (1) laser generator and photoelectronic convertor, (2) drug powder, (3) dispensing syringe, (4) syringe actuator, (5) display system, (6) power supply, (7) data processor and controller, (8) thermostate system, (9) magnetic stirrer, and (10) stir bar.

Actual photograph of the setup and syringes with different sizes.
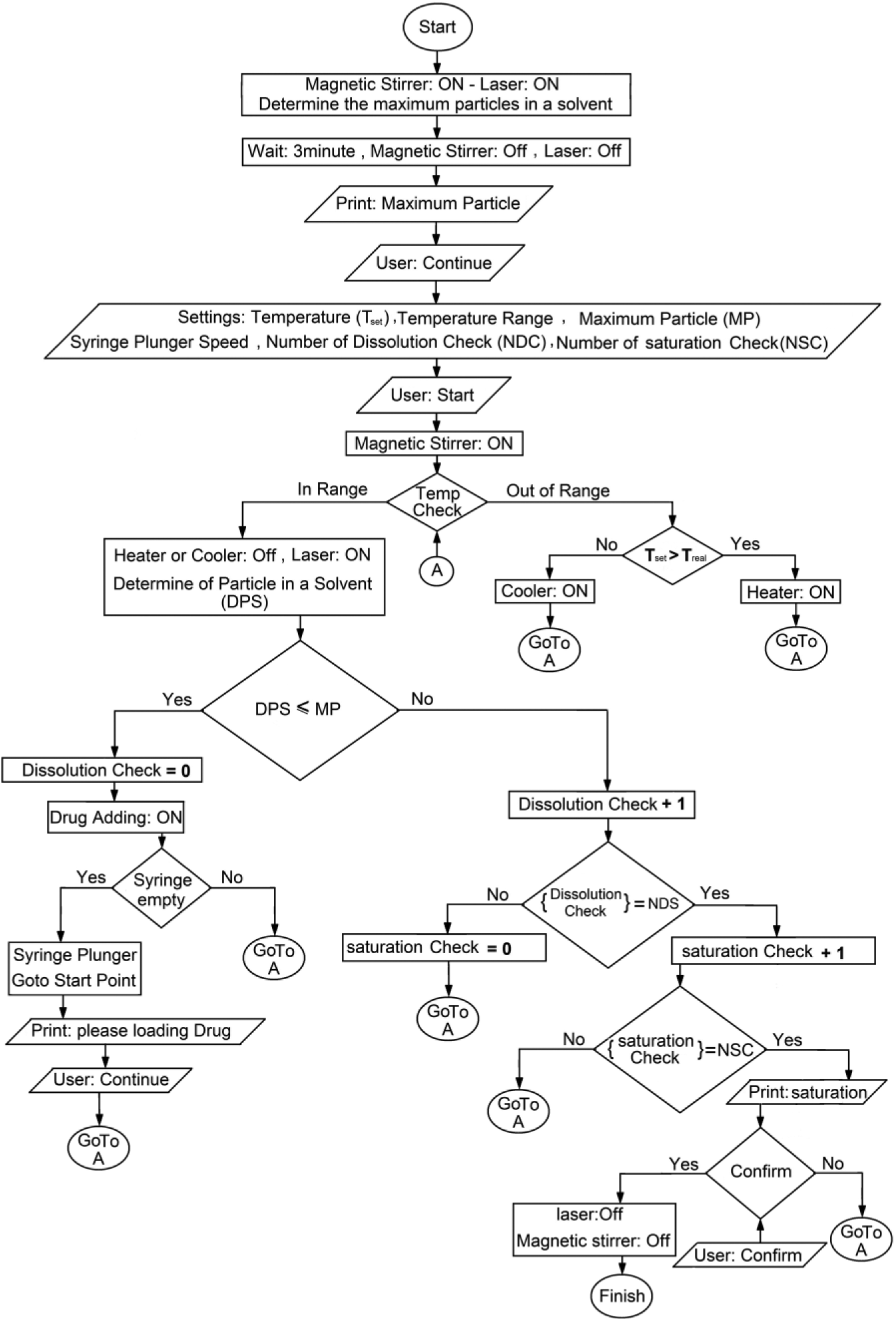
Flowchart of the developed setup.
Materials
Acetaminophen powder (purity of 99.0%) was gifted by Daana Pharmaceutical Company (Tabriz, Iran). SDS with purity of 99.0% was purchased from Acros (Geel, Belgium). Ethanol (mass purity of 99.5%) was purchased from Scharlau Chemie (Barcelona, Spain) and propylene glycol (mass purity of 99.5%) from Merck (Darmstadt, Germany). Double-distilled water was used throughout this work.
Validation of the Instrument
Measured acetaminophen solubilities in aqueous solutions at various temperatures were used and compared with the corresponding values obtained from the literature. The differences were computed using the individual percentage deviation (IPD) using
which is a similar value to the relative standard deviation values that were routinely used to check the repeatability of the experimental measurements in the solubility studies.
Results and Discussion
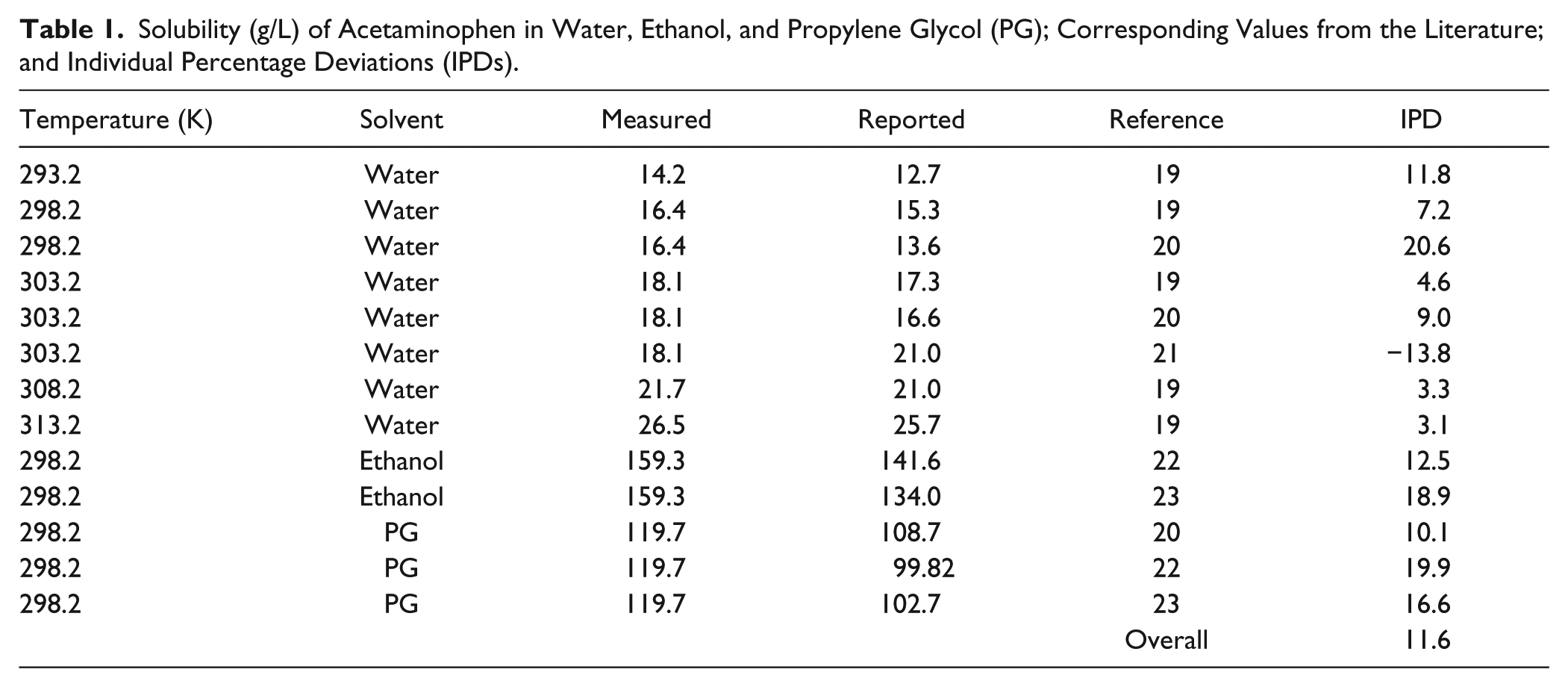
The measured solubility of acetaminophen in water at 293.2 to 313.2 K is in line with the van’t Hoff equation, with a correlation coefficient of 0.99 revealing that the experimental data are valid with regard to the temperature effects on drug solubility. When the measured data at a given temperature were compared with previously measured data using methods other than the laser monitoring technique (see Table 1 for details), the IPDs varied from −13.8% to 20.6%, with an overall IPD of 11.6%, which is less than the 25% reported by Wang et al. 14 Figure 4 shows a plot of the measured solubility data of acetaminophen in water, ethanol, and propylene glycol at different temperatures against their reported values taken from the literature. The correlation coefficient of 0.9985 reveals that there is very good agreement between the measured and reported data points and confirms the validity of the setup for solubility measurements. These variations could be considered as acceptable variations when the IPD values of 988% (for reported solubility of sulfapyridine by two different groups15,16) or 804% (for reported solubility of sulfadimidine by two different groups15,17) are considered. Solubility data of anthracene and fluoranthene were reported by 17 different laboratories using a standard method. The results showed that even when all variables were kept constant, the interlaboratory difference can still be very significant, with an average IPD of 51%. 18 A number of reasons could be proposed to justify such differences, including the purity of drug powders or solvents.
Solubility (g/L) of Acetaminophen in Water, Ethanol, and Propylene Glycol (PG); Corresponding Values from the Literature; and Individual Percentage Deviations (IPDs).
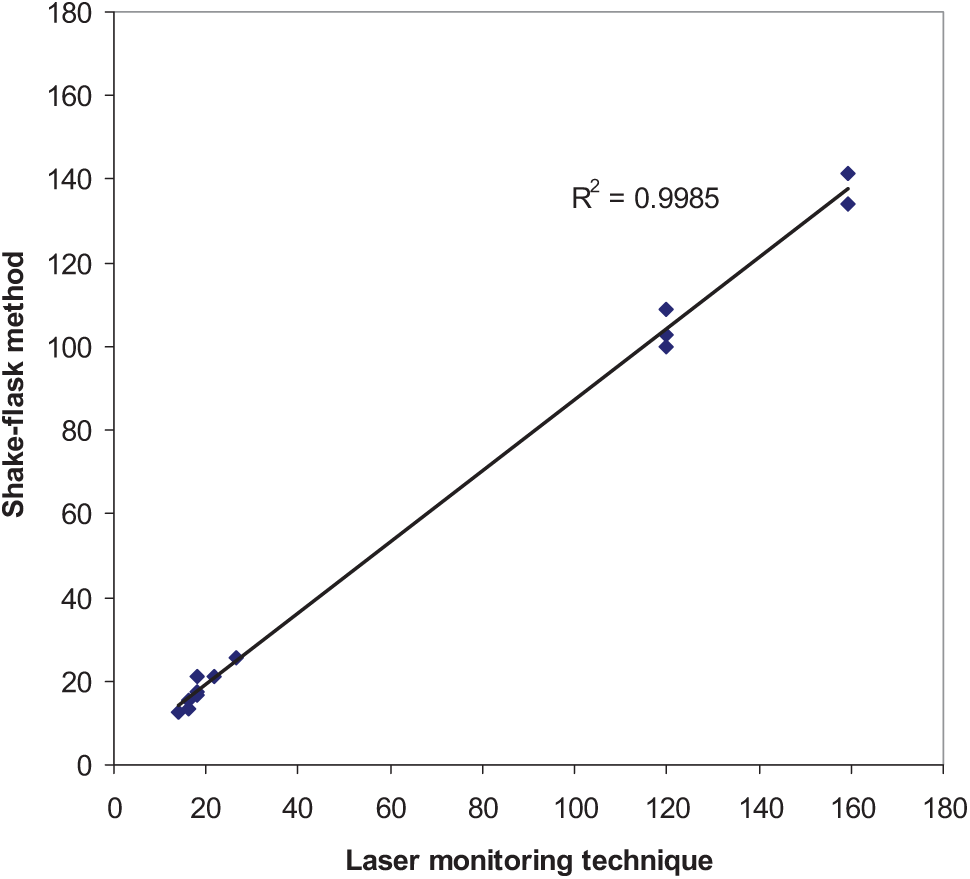
Plot of measured (g/L) solubility of acetaminophen by the proposed setup against the corresponding data measured using the shake-flask method taken from the literature.
In addition to the variation in the results for different laboratories, the relative standard deviation (RSD) values for the repeated experiments from the same laboratory are significantly high. As examples, the reported RSD values are up to 9.2%, 24 10%, 25 and 28%. 26 Higher solubilities in organic solvents (i.e., ethanol and propylene glycol) were obtained using the proposed setup when compared with those of the shake-flask method data taken from the literature, which is in agreement with the findings of Grant and Abougela. 10
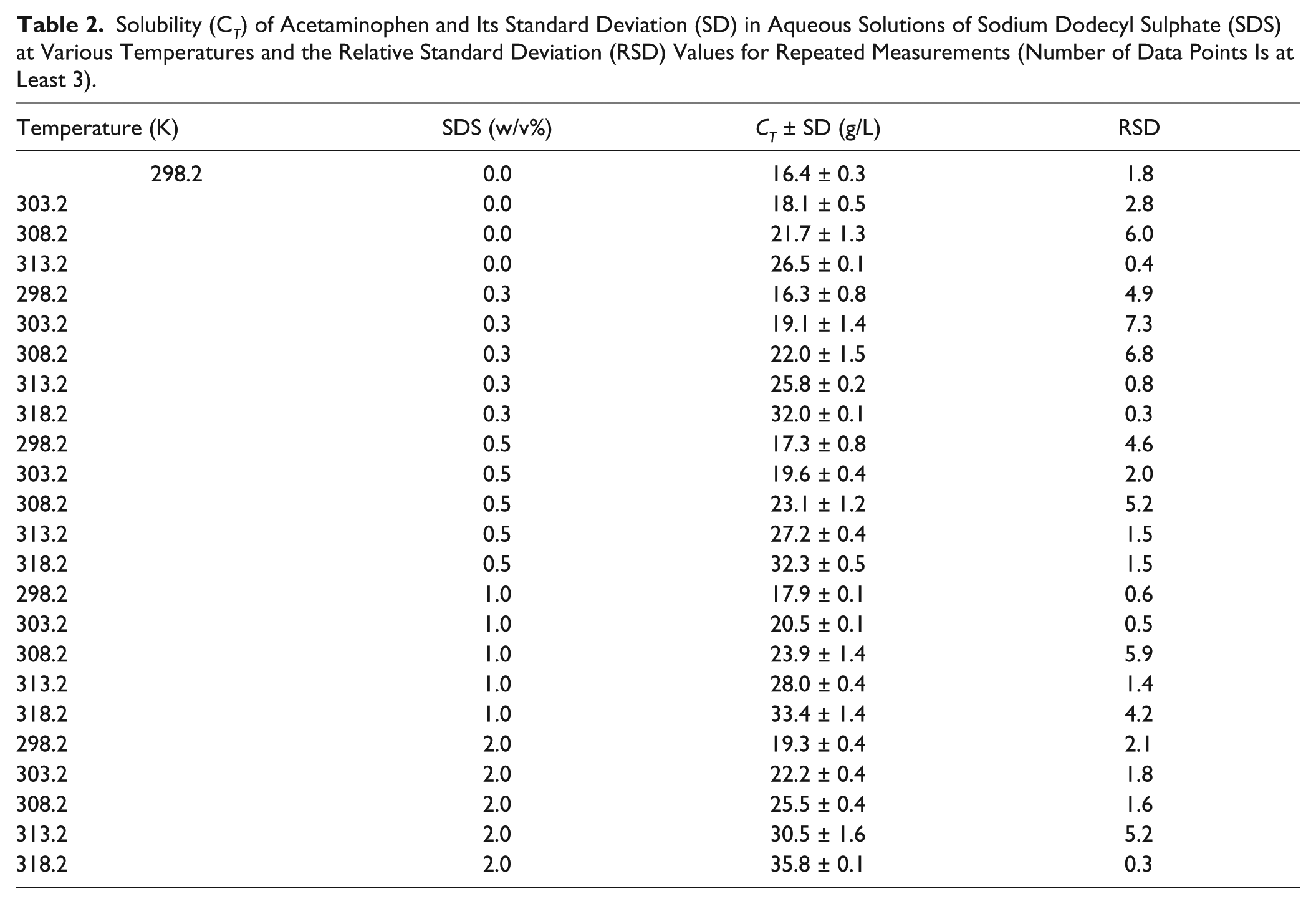
The aqueous solubility of acetaminophen in the presence of different concentrations of SDS at various temperatures was also measured using the developed setup, and the results are reported in Table 2 . The solubility was increased with the addition of SDS concentration at a given temperature as expected. On the other hand, the solubility was also increased with an increase in the solution’s temperature in a given concentration of SDS, and there are good linear relationships among the logarithm of the solubility, SDS concentration, and 1/T values, which are in agreement with previous findings.27,28 Concerning these observations, one might combine these linear relationships and propose the following multiple linear regression equation to represent the solubility data in the presence of various concentrations of SDS at different temperatures. The model is

where CT is the gram per L solubility of acetaminophen, T is the solution’s temperature expressed in Kelvin, and SDS is the mass fraction of SDS in the solution. Standard errors of the constants are given in the parentheses, and the constants are statistically significant (p < 0.0005). The correlation coefficient of eq 2 is 0.996, and the F value is 1189, which reveals a significant correlation with the probability of <0.0005. When C T values were back-calculated using eq 2, the mean (±SD) of the absolute values of deviations of experimental and calculated solubilities is 1.8% ± 1.2%.
Solubility (C T ) of Acetaminophen and Its Standard Deviation (SD) in Aqueous Solutions of Sodium Dodecyl Sulphate (SDS) at Various Temperatures and the Relative Standard Deviation (RSD) Values for Repeated Measurements (Number of Data Points Is at Least 3).
The main advantages of the proposed setup are (1) temperature range and its uncertainty are adjustable by the user, (2) it is suitable for the solubility measurement of highly low-soluble drugs to very soluble drugs by employing various syringes, (3) the measured solubilities are very close to those determined using the common shake-flask method, (4) no chromophor for drug molecule is required because quantification is not based on UV measurement, (5) more repeatable results are obtained when compared with a previously reported setup, and (6) affecting parameters, such as stirrer rate, required time for equilibration, and so forth, could be adjusted by the user.
As a conclusion, an automated solubility determination setup was developed and validated. This setup could be used in the pharmaceutical industry and also in academia for faster solubility determination of drugs and/or drug candidates to speed up the drug discovery and development processes.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.