Abstract
Treatable diseases continue to exact a heavy burden worldwide despite powerful advances in treatment. Diagnostics play crucial roles in the detection, management, and ultimate prevention of these diseases by guiding the allocation of precious medical resources. Motivated by globalization and evolving disease, and enabled by advances in molecular pathology, the scientific community has produced an explosion of research on miniaturized integrated biosensor platforms for disease detection. Low-cost, automated tests promise accessibility in low-resource settings by loosening constraints around infrastructure and usability. To address the challenges raised by invasive and intrusive sample collection, researchers are exploring alternative biomarkers in various specimens. Specifically, patient-generated airborne biomarkers suit minimally invasive collection and automated analysis. Disease biomarkers are known to exist in aerosols and volatile compounds in breath, odor, and headspace, media that can be exploited for field-ready diagnostics. This article reviews global disease priorities and the characteristics of low-resource settings. It surveys existing technologies for the analysis of bioaerosols and volatile organic compounds, and emerging technologies that could enable their translation to the point of care. Engineering advances promise to enable appropriate diagnostics by detecting chemical and microbial markers. Nonetheless, further innovation and cost reduction are needed for these technologies to broadly affect global health.
Introduction: Airborne Biomarkers for Diagnostics in Global Health
Diagnostics play crucial roles in the detection, management, and, ultimately, prevention of diseases by guiding the allocation of limited medical resources. In the developed world, diagnostic technology has typically been engineered for primary to central health care facilities in high-resource settings where infrastructure and staffing are adequate for costly or sophisticated procedures. On a global scale, this strategy has neglected the majority of the affected population who lack access to well-equipped clinics.
Despite advances in therapy, diseases that are largely treatable continue to exact a heavy burden worldwide. Markedly, preventable infectious causes like pneumonia, meningitis, diarrhea, malaria, and AIDS accounted for 64% of the 7.6 million deaths in children younger than age 5 years in 2010. 1 Interventions exist to significantly reduce mortality, but they are not available to those most in need. 2 Thus, point-of-care (POC) testing can meet the challenges of addressing accessibility in remote settings, resource-poor facilities, scarce expertise, 3 and the need for broader public health strategies like emergency response. 4
Motivated by globalization and the evolution of disease, and enabled by rapid scientific advances in molecular pathology, researchers are leveraging emerging technologies to translate existing diagnostics from the developed world into low-resource settings. The scientific community has experienced an explosion of research to integrate miniaturized biosensors on microfluidic platforms for disease detection. Low-cost, automated tests promise accessibility in low-resource settings by circumventing logistical constraints regarding infrastructure and highly trained operators. Most miniaturized tests, however, require a blood specimen, the traditional medium for chemical, proteomic, and genomic analyses. A blood draw is invasive, so specimen collection still involves trained personnel and significant noncompliance obstacles.
To overcome challenges associated with blood specimens, researchers are exploring the availability and utility of biomarkers in alternative specimens. Specifically, airborne biomarkers suit minimally invasive to noninvasive collection and automated analysis. Although several disease states of global priority are associated with aerosolized microbes or distinct profiles of volatile organic compounds (VOCs), scientists have yet to realize field-ready diagnostics that can rapidly detect these airborne disease markers.
This work reviews the potential for aerosol and VOC disease markers in a number of priority global health conditions, existing technologies for the collection and analysis of aerosol and VOC biomarkers, and emerging technologies that could enable their translation to the point of care. Technological advances promise to enable appropriate diagnostics by detecting chemical markers in odor and breath, and aerosolized microbes generated by the patient. The noninvasiveness of this type of diagnosis method facilitates more frequent sampling, which in turn leads to a promise of early disease detection, the holy grail of disease diagnosis and prognosis.
Requirements for Point-of-Care Testing
Disease Burdens and Priorities
Disease burden is often quantified in units of disability-adjusted life years (DALYs) to account for extrapolated loss of livelihood due to mortality and disability. 5 In the developing world, infectious diseases are the major cause of lost DALYs. By contrast, noncommunicable diseases like cardiac ischemia and unipolar major depression account for the highest burden in developed countries. A projection of global mortality and burden of disease, 6 however, indicates that several such conditions, now prevalent in higher-income countries, will become priorities in developing countries by 2030.
The following discussion presents several diseases with high global burdens for which a new POC diagnostic test would have significant impact and for which aerosol or volatile biomarkers have been explored.
Tuberculosis
Among infectious diseases, tuberculosis (TB) is second only to HIV/AIDS in mortality. In 2011, World Health Organization (WHO) estimates of its global burden indicated 1.4 million deaths among 8.7 million new cases, almost 40% of which were accounted for in India and China alone. 7 The burden is especially great in the Africa region, the location of the world’s highest per-capita rates of tuberculosis transmission and death.
Although TB is treatable with rather effective therapy (successful treatment of 85% of diagnosed patients was achieved in 2006), the global detection rate of new cases was just 67% in 2011. Delayed diagnosis not only worsens patient outcomes but also propagates transmission of the disease. 8 The likelihood of rapid progression is yet increased by HIV co-infection. Nonetheless, front-line screening methods remained largely unchanged for decades.
Many countries rely on microscopy of sputum smears to detect Mycobacterium tuberculosis, an outdated technique that, although low in cost, possesses only modest sensitivity and cannot identify drug-resistant strains. Where facilities are accessible, bacterial culture provides a gold-standard diagnostic method. Its multiweek turnaround time, however, holds back its clinical effectiveness. Limited availability or patient access to adequate health services, compounded by the chronic nature of the disease and slow diagnostic methods, obliges patients to make frequent visits throughout time for complete treatment. Therefore, a rapid POC diagnostic test that reduces infrastructure requirement or patient loss to follow-up visits could save hundreds of thousands of lives annually. 9
Nucleic-acid amplification tests (NAATs), theoretically able to detect M. tuberculosis with high sensitivity and specificity, have been realized in a number of commercial systems. In 2010, WHO approved the Xpert MTB/RIF, a cartridge-based assay for TB and rifampicin resistance that analyzes minimally treated sputum using the GeneXpert system 10 (Cepheid, Sunnyvale, CA). Although this is a breakthrough in TB testing, the cost of the test cartridge and the GeneXpert system precludes unaided widespread accessibility to low-resource countries. Therefore, global support and concessional pricing were required to roll out the test, which has been deployed to 83 countries as of March 2013. 11
Researchers such as Pavlou 12 rightly observe that a need still exists for a lower-cost POC diagnostic test for low-resource settings. Like other species of Mycobacteria, tuberculosis produces specific VOCs that have been discerned in culture, sputum headspace, and breath. Distinctive volatile biomarkers include methyl p-anisate, methyl nicotinate, and derivatives of naphthalene, benzene, and cyclohexane.13,14 Thus, analysis of VOC signatures has the potential to detect TB from sputum in a simpler way than NAATs, without dependence on high-cost instrumentation.
Under natural circumstances, pulmonary TB infection requires airborne transmission. Coughing and sneezing, or even simple exhalation 15 by an infected host, can aerosolize viable bacteria in droplet nuclei that can be inhaled by a new host and deposited in the distal lung. 16 Furthermore, opportunistic infection of the gastrointestinal (GI) tract can occur when TB bacilli are swallowed in sputum. When the bacilli are subsequently shed in feces (diarrhea is a symptom of GI TB), excrement aerosols pose a further risk of airborne transmission. 17 Thus, in theory, pathogens and their derived biomarkers can be detected in patient-generated aerosols.
Given the variety of possible airborne TB biomarkers, and seeing as the most common clinical diagnostic tests rely on direct detection of the pathogen, it seems that testing of breath could enable rapid and noninvasive identification of patients with active TB.
Malaria
Malaria, although preventable and treatable, resulted in an estimated 219 million cases and up to 971,000 deaths in 2010. 18 This mosquito-borne disease is most widespread in the Africa region, where about 90% of malaria deaths occur. Especially vulnerable are young children, who can progress to severe malaria in mere hours. 19
Owing largely to a scale-up of interventions like insecticidal nets, indoor spraying, new therapies, and rapid diagnostic tests (RDTs), related mortality rates fell by 26% in the decade leading up to 2010. Malaria is, however, still endemic in 104 countries. Surveillance systems are essential to treatment and control, but they lack the necessary effectiveness; the global case detection rate is only 10%.
Acute fever cases are commonly treated presumptively without parasitological confirmation, a practice that could accelerate drug resistance. Thus, universal diagnostic testing is needed to reduce the requirement for antimalarial treatment. Microscopic examination of blood slides remains the gold standard for diagnostic testing, although the deployment of RDTs has risen substantially.
Microscopy is, however, a lengthy procedure requiring trained personnel, and any blood-based test raises the risk of accidental transmission of infectious disease. The invasive sampling procedure faces additional difficulties with noncompliance among young children and cultures with blood taboos. To overcome such obstacles, researchers have sought alternative diagnostic media like saliva 20 and urine. 21
The blood-borne Plasmodium parasite is naturally transmitted among humans via the Anopheles mosquito. Although no route of aerosol transmission is known, at least one study has confirmed the presence of Plasmodium DNA in feces. 22 As malaria symptoms can include vomiting and diarrhea, it may be possible to detect parasite biomarkers in host-generated aerosols.
On the other hand, an investigation by Wong et al. of VOCs in the headspace of in vitro P. falciparum cultures did not identify unique malarial-specific compounds. 23 Despite this result, the authors reasoned that future in vivo study of malaria patients with large enough parasite loads may yet reveal specific VOCs. It is also possible that indirect VOC signatures from breath, skin, or fluid headspace may arise from the physiological sequelae of Plasmodium infection.
Acute Respiratory Infections
Acute respiratory infections (ARIs), including pneumonia, meningitis, influenza, and severe acute respiratory syndrome (SARS), collectively accounted for 3.9 million deaths in 2002. 24 In fact, just acute lower-respiratory infections (ALRIs) of viral or bacterial bronchiolitis or pneumonia are the primary cause of death among young children in developing countries. The Child Health Epidemiology Reference Group (CHERG) connected ALRIs to the deaths of more than 2 million children younger than 5 years old annually between 2000 and 2003. 25
In resource-limited countries, ARIs are typically diagnosed with clinical algorithms based on abnormal respiratory symptoms. Overuse of antibiotics is common and accelerates bacterial drug resistance, leading to a reduced likelihood of effective treatment and various societal costs. Modeling of the potential benefits of new diagnostic interventions 26 has indicated that 405,000 deaths among young children annually would be prevented by a test for bacterial ALRIs with 95% sensitivity, 85% specificity, and minimal infrastructure requirement.
ARIs are commonly caused by Streptococcus pneumoniae and Haemophilus influenzae (type b) but also by viruses and fungi. Pathogens can spread in droplets originating from the respiratory tract. Detection of pathogens is thus in principle possible in exhaled aerosols. Indeed, aerosols have been associated with increased risk of SARS transmission.27,28 In addition, volatile molecules in breath have been used to accurately predict multiple respiratory diseases in patients.29–31
Diarrheal Diseases
Diarrheal diseases, the fourth leading cause of mortality in 2010, caused 1.4 million deaths (89.5 million DALYs). 32 Along with ALRIs, diarrheal disease is a dominant killer of neonates, accounting for 17.4% of deaths among infants between 1 and 11 months old.
An effective treatment regimen for diarrhea depends strongly on the accurate identification of its etiology as the disease can be caused by any of a number of bacterial, viral, and parasite pathogens. 33
Cholera causes the most severe of diarrheas. Conventional culturing of stool samples in central laboratories remains the gold standard for diagnosis of Vibrio cholerae, the bacterium responsible for cholera. Besides its requirement for infrastructure and training, the procedure is inconsistent. 34 A laboratory RDT produced a useful predictive value during the 2010 outbreak in Haiti, 35 but the need still exists for a diagnostic for individual patients. The pathogen is present in feces and has been transmitted in aerosol form among hogs. 36 Furthermore, exploratory study of volatile biomarkers identified two compounds in fecal headspace that could be used in the diagnosis of cholera. 37
Surface contamination associated with virus-associated diarrhea is a major transmission route. 38 Rotavirus, which can remain infectious even on sanitized surfaces, has been detected from sewage, 39 fomites, and areas surrounding toilets, 40 apparently deposited from aerosols. Rotavirus stools also appear to have a characteristic smell detectable by human olfaction. 41 Airborne biomarkers could be viable research areas for these and other diarrheal pathogens.
Noncommunicable Diseases
Granted the priority of infectious disease, noncommunicable diseases (NCDs) have a large and growing global burden, even in the developing world. In the decade prior to 2010, deaths from NCDs rose by almost 8 million and account for two-thirds of all deaths. 32 Cancer, cardiovascular disease, and chronic respiratory diseases are primary causes.
In 2010, cancer caused 8 million deaths. About one-fifth of these cases (3.2 million DALYs 42 ) affected the lower respiratory tract (trachea, bronchus, and lung), and the upper respiratory regions accounted for 9%. Cancers of the breast, liver, and GI tract were among the other most prevalent cases. Accordingly, analysis of VOCs in breath has revealed signatures of lung cancer, 43 breast cancer, melanoma, colorectal cancer, 44 and bladder cancer—and this list continues to grow.
Cardiovascular and circulatory diseases also exact a severe worldwide toll, accounting for 11.8% of DALYs in 2010. 42 Ischemic heart disease was the primary global cause of death and, collectively with stroke, claimed 12.9 million lives. Breath analysis research has achieved notable advances identifying biomarkers for the diagnosis and management of cardiovascular disease. 45
Chronic respiratory diseases constituted 4.7% of global DALYs in 2010. Chief among these were chronic obstructive pulmonary disease (COPD) and asthma, which together made up approximately 85% of the total. Although the two conditions share clinical features, molecular profiling was able to distinguish them from each other and control subjects. 30 In addition, exhaled breath condensates show potential for monitoring of respiratory disease. 46
Physiological responses to lung diseases apparently produce airborne markers that express in exhaled breath. Many of the technologies presented in this chapter promise to enable new diagnostics for the POC.
Requirements for Point-of-Care Use
The WHO Sexually Transmitted Diseases Diagnostics Initiative has captured the ideal characteristics of a diagnostic test for resource-limited settings in a simple acronym, ASSURED: 47
It may be that in many cases, no technical solution can fulfill all ASSURED criteria. These guidelines touch on factors well beyond the performance of a particular technology. For any given clinical application, they also imply constraints on the medical status quo, including staffing practices, supply-chain logistics, informatics, and even behavioral bias.48,49 Although criteria for a POC diagnostic test generally differ in the developed world, some methodologies developed for niche settings may be transferrable to low-resource settings.
In modern hospitals, live culture remains the gold standard for the confirmation of many infectious agents. Demand for throughput has driven the development of increased automation and faster turnaround. On the individual level, trends in self-directed health management are producing point-of-care testing to safely bridge some capabilities of the centralized laboratory to the physician’s office and the patient at home. Furthermore, governments have developed military technologies to monitor the health status of personnel in the field. The U.S. government in particular invests heavily in portable medical equipment and detection systems for early warning of biological attacks. 50 In similar ways, emergency response relies on POC testing for evidence-based triage to guide time-critical rescue efforts. 4 Telemedicine networks linking disaster victims, relief teams, and regional hospitals have been proposed to optimize emergency patient care.
Many constraints in the preceding contexts overlap those faced by emerging diagnostic technology for the developing world. Therefore, the scientific community might benefit from strategies therein that could be applied to global health priorities.
Technologies for Sampling and Detecting Bioaerosol Disease Markers
The scientific investigation of atmospheric bioaerosols has been ongoing in some form or another for about 160 years. 51 In the 1800s, the science of atmospheric micrography fueled the Golden Age of aerobiology with an explosion of research for “germs” that “floated about in the atmosphere,” especially in connection with the spread of disease. 52 , 53 Indeed, the term malaria (from the medieval Italian mal aria, “bad air”) conveys the once-held belief that the febrile disease was actually caused by odorous marsh air. 54
The prevalent scientific paradigm eventually shifted to the belief that diseases that are communicable via the respiratory tract are transmitted through direct contact with contaminated surfaces or close-range exposure to large droplets, a view exemplified by Charles Chapin’s influential 1910 volume The Sources and Modes of Infection. 55
However, scientists like Wells have since pointed out that the settling time and, thus, infective range of human-generated droplets are significantly greater than expected because evaporation forms droplet nuclei that can remain suspended in air currents. 56 In fact, the population density and interconnectedness of modern urban infrastructure have since proven to create favorable conditions for bioaerosol epidemics, as in the 2003 transmission of SARS via virus-laden aerosol plumes in an apartment complex. 28
In modern times, bioaerosol research has experienced a resurgence with specific interests in agriculture, 57 urban air pollution, and biodefense 50 in addition to disease transmission. In regard to how each of these areas affects human health considerations, much research has sought to understand how inhaled particles are deposited in the respiratory tract.
Each human or environmental source generates aerosols with characteristic size distribution ( Figure 1 ). Aerodynamic size is the most important single factor in determining aerosol penetration into the respiratory tract.58,59 Bacteria and viruses are readily carried in aerosols that are of sizes that can enter the upper and lower respiratory tract, where they are deposited by gravity, inertia, diffusion, and electrostatic forces. Besides bioaerosols that penetrate the respiratory tract, those emanating from an infected host can be sampled to detect indicators of disease.
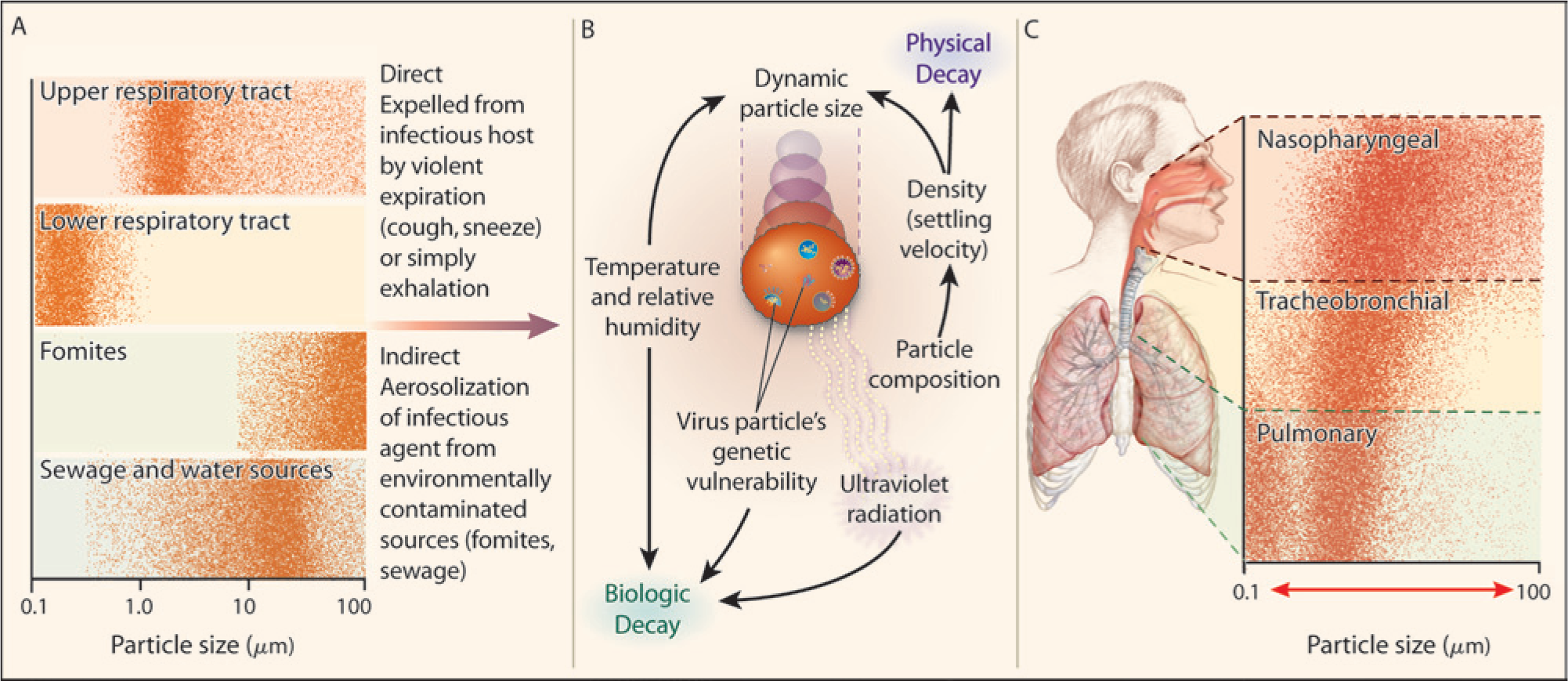
The aerobiological pathway for the generation and transmission of communicable respiratory disease. Bioaerosols emanating from an infected host can in principle be sampled to detect indicators of disease. (
Mechanisms of Aerosol Generation, Collection, and Analysis
For the present consideration, a bioaerosol is an airborne suspension of disperse liquid or solid small particles that contain living organisms or their fragments. In humans, these biological particles include resident viruses and bacteria, and cellular debris containing protein and genetic material. Collectively, the aerosolized biomass generated from breath, skin, and excrement provides an information-rich resource that can be exploited for diagnostics.
Droplets in exhaled breath can carry viable microorganisms in addition to diverse nonvolatile species like peptides and cytokines. The size distribution of exhaled droplets is greatest in the submicron range, although larger droplets can be generated by coughing and sneezing. 15 Smaller bioparticles such as viruses, protein, and microbial byproducts are rarely found as discrete individual particles but rather as components of larger organisms or within liquid droplets. It is important to note that for such small bioaerosols, droplet size is determined by the liquid medium and the mechanism of nebulization rather than individual bacteria, viral particles, or protein. 60 Exhaled aerosol can be either inhaled by another host, and thus transmit biological material among persons, or collected and analyzed for clinical use. Not surprisingly, then, exhaled breath condensate (EBC) 61 contains a spectrum of biomarkers that can enable noninvasive assessment of pulmonary and systemic pathobiology.
The body continuously sheds skin cells in the process of normal epidermal growth. On average, 500 million cells (approximately one gram) per day are shed via desquamation. 62 Skin cells about 5 to 15 µm in size 63 can remain airborne for up to days. In fact, the information contained in skin debris is sufficiently high that researchers have proposed biochemical fingerprinting 64 based on aerosolized skin particles.
Excrement is a third potential source of information-rich bioaerosols. The fecal microbiome has been an area of active investigation for health and disease states, 65 but relatively limited research has elucidated its frequent aerosolization by flush toilets. 17 A toilet bowl contaminated with enteric pathogens after an episode of diarrhea or vomiting could aerosolize viable microbes even after multiple flushes. 66 Consequently, their droplet nuclei could remain adrift in air currents.
Exhalation, desquamation, and excretion are all regular activities in healthy people, although their abnormalities may well be symptomatic of disease. This characteristic suggests that human-generated bioaerosols are an attractive medium for noninvasive monitoring and diagnostic testing, provided that the technology is adequate for their collection, processing, and analysis.
The ideal bioaerosol sampler collects aerosol in a specified size range without altering its physical or biological properties. Accordingly, three major efficiency metrics guide the design of samplers: (1) the inlet sampling efficiency quantifies the ability to capture a representative fraction of the ambient environment; (2) the removal efficiency indicates the ability to extract aerosols from the sample airstream and deposit them onto the collection medium; and (3) the biological sampling efficiency measures the ability to preserve the viability and biological characteristics of collected particles. Whereas high-volume environmental samplers enable a large throughput, miniature personal samplers are ideal for precise localized measurements.
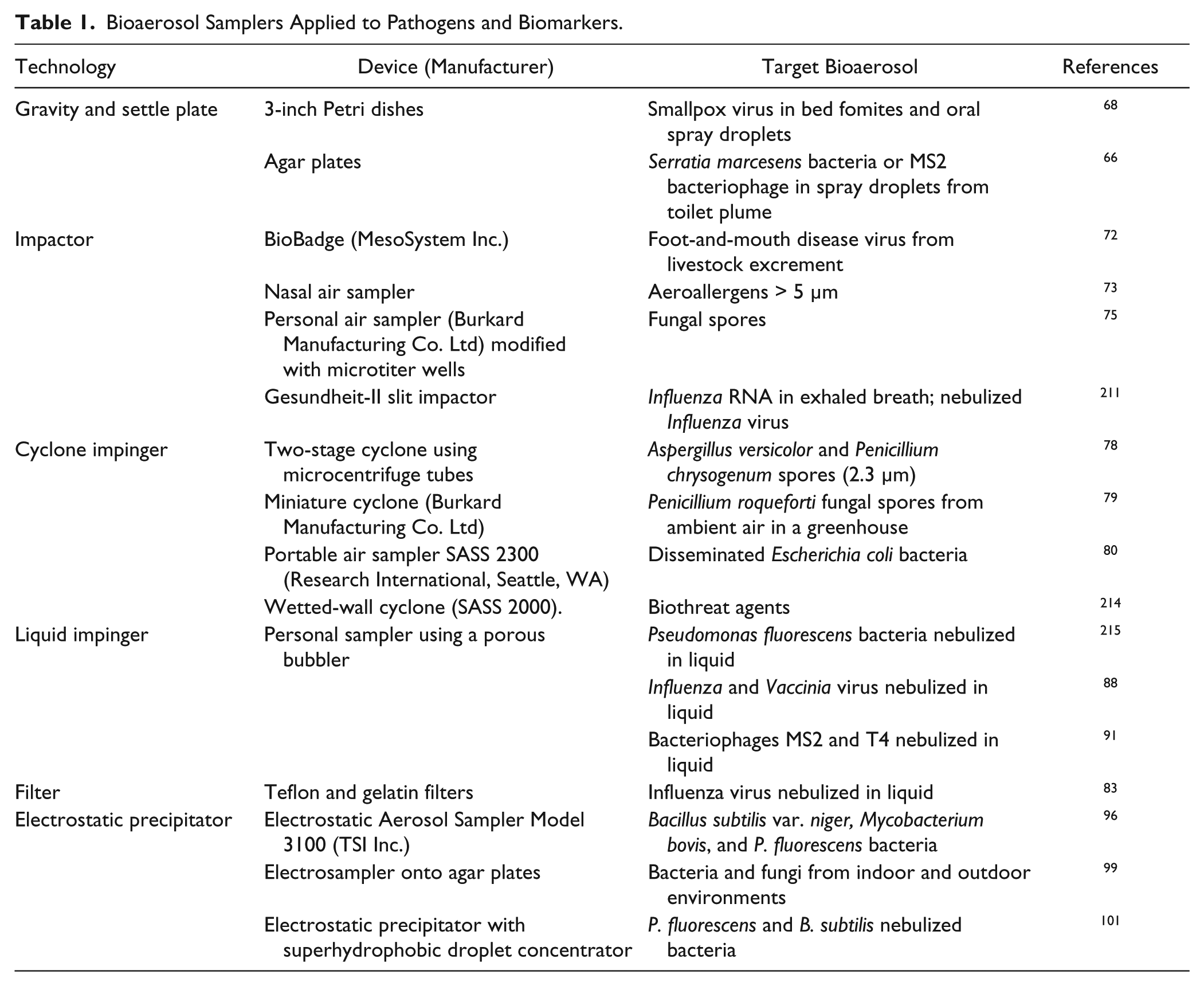
In practice, principle methods of bioaerosol sampling largely reflect the mechanisms at work in the human respiratory tract: gravity, impaction, impingement, filtration, and electrostatic precipitation. The following discussion examines the most common sampling modes for bioaerosols with representative demonstrations of their application to disease ( Table 1 ). Microscopy and culturing have traditionally been the most common methods of analysis, but integration with biochemical assays is leveraging powerful advances in chemical, proteomic, and genomic techniques.
Bioaerosol Samplers Applied to Pathogens and Biomarkers.
Gravity Sampling
The simplest of aerosol-sampling techniques, gravity sampling exposes a coated microscope slide or an agar plate to the atmosphere for a controlled time. These so-called settle plates passively sample airborne particles that fall by force of gravity onto the collection surface. The technique is especially useful for identifying viable airborne microorganisms as the samples may be cultured in situ and directly observed by microscopy. The low-cost technique is sufficiently simple for consumer use in household-mold test kits from numerous vendors.
During the global effort to contain and eradicate smallpox, much research was undertaken to understand airborne transmission of the variola virus, particularly in smallpox hospitals. 67 For instance, Downie et al. compared settle plates with impinger samples for the recovery of smallpox virus in proximity to hospitalized patients. 68 Impinger samples near the patients’ mouths and bedclothes yielded little virus compared to that recovered by plates. The different sample-size selectivities of these two techniques led the researchers to conclude that airborne transmission near smallpox patients was due not to smaller droplets in their breath, but large dust particles from bedclothes (i.e., fomites) that had been contaminated by respiratory, oral, and lesion secretions.
In a more recent public health study, Barker and Jones used a combination of agar settle plates and sieve impactor samples to discern the spatial distribution of bacterial and viral aerosols resulting from a toilet flush. 66 They measured the effects of different seeding conditions as models of pathogen shedding during acute diarrhea. Repeated flushes produced sustained air counts and surface contamination on the toilet. The study concluded that the common toilet flush posed a risk of infection by persistent enteric pathogens via inhalation or the surface-to-hand-to-mouth route.
Gravity sampling by design is biased toward heavier particles like large fungal spores, dust, and spray droplets. Smaller particles tend to be underrepresented in the sample. Furthermore, the results are strongly affected by convective air currents. Due to these characteristics, gravity sampling is useful for simple qualitative measurements that need not be volumetric. As in the above studies, it is usually used in conjunction with other sampling methods and has thus found little application in modern quantitative disease diagnostics.
Impaction
Impaction samplers separate particles from an airstream using particle inertia. The airstream is directed at a solid collection surface and forced to bypass the surface by turning a sharp angle. Particles with sufficient inertia exit the bending airstream and deposit onto the surface.
Commercial environmental systems commonly use rotating-arm impactors and suction impactors to sample pollen and spores. In a variation designed for culturable specimens, sieve impactors split the airstream among an array of nozzles to distribute samples over a culture plate. Under high flow rates, however, biological recovery can be compromised by mechanical and osmotic stress. 69 Furthermore, captured particles are typically stained or cultured for microscopic enumeration, a slow and laborious process. Thus, for POC, a need exists for compact systems capable of automatically and specifically identifying impacted particles.
The BioBadge is a wearable, battery-operated personal sampler that samples airflow using a rotating impactor and impeller. 70 Although the compact unit (approximately 7 × 14 cm) pulls an impressive flow rate (35 L min−1), it collects the particles dry and requires separate elution in liquid for analysis. Kesavan et al. characterized the sampling efficiencies of the BioBadge unit for three kinds of aerosols between 1 and 8 µm in size. 71 The sampling efficiency increased with aerosol size in the range of interest. For 8-µm nebulized oil droplets, efficiencies from 66 to 74% were achieved. Single 1-µm Bacillus globigii spore particles resulted in 4 to 9% efficiency. In separate studies, the BioBadge successfully detected airborne viruses, but as noted by Ryan et al., field utility would require integration with portable analysis instrumentation. 72
The nasal air sampler reported by Graham et al. is an innovative, highly compact means of measuring personal exposure to aerosols. 73 Worn just inside the nasal passages, it harnesses the user’s own inhalation to impact particles on a protein-binding membrane. Thus, no outside power source is necessary. Rig tests using latex particles showed a particle cutoff diameter of about 5 µm. The impactor substrate has also been processed for immunostaining by laboratory methods. 74 Although developed and tested for aeroallergens, this approach of nasal air sampling might be combined with analysis and adapted to measure personal exposure to and generation of infectious aerosols.
Researchers have indeed connected impactor sampling with more powerful molecular assays. Kennedy et al. demonstrated rapid immunodetection of fungal plant pathogens using a microtiter immunospore-trapping device. 75 This device adapted a compact (590 g) personal air sampler (Burkard Manufacturing Co. Ltd, Hertfordshire, UK) to aim nozzles directly at the base of microtiter wells. Impacted particles were then assayed for target analytes using colorimetric enzyme-linked immunosorbent assay (ELISA). A subsequent study experimented with a variety of well coatings to improve the retention of spores and the sensitivity of ELISA. 76 Such devices indicate that inexpensive portable systems can be combined with conventional immunoassays. Nonetheless, POC applications would require shortened assay time with higher levels of integration and automation.
For example, Schmitt et al. reported immunofluorescence recognition of airborne particles in a gel-based biochip within 1 h. 77 This device used a two-stage impactor to select for polystyrene beads between 0.8 and 10 µm and deposit them on freeze-dried porous cyclic olefin copolymer (COC) hydrogel containing fluorescently tagged immunoglobulin G (IgG) antibodies. The gel was then hydrated to reconstitute salts and IgG, which bound to surface antigens on impacted particles. Unbound IgG was removed by electrophoretic separation using integrated electrodes flanking the hydrogel. Labeled particles were detected by automated fluorescence imaging. The approach by Schmitt et al. shows the potential for integrated sampling, preparation, and detection to reduce labor and assay time. These authors anticipate further testing of the detection system using antibodies developed against Aspergillus fumigatus spores.
Cyclone Impaction
The cyclone is a subclass of impactor in which streamlines follow a circular path. A jet of air enters tangentially near the top of a cylinder with a conical bottom. As it swirls downward, high-inertia particles impact on the cylinder wall by centripetal force, and sufficiently large particles collect at the bottom of the cone. Below the cylinder, the airstream enters the conical section, where it reverses direction, ascends along the cyclone axis, and exits through the top of the cylinder, carrying with it particles smaller than the cutoff size.
Cyclone samplers have been made on a portable-size scale to probe personal breathing zones. Lindsley et al. developed a two-stage cyclone sampler to collect bioaerosols directly into 1.5-mL microcentrifuge tubes. 78 The unit, measuring approximately 68 mm × 97 mm and weighing 86 g, collected 92% of aerosolized spores from Aspergillus versicolor and Penicillium chrysogenum. Samples in the tubes could then be suspended in saline for biological assay, but analysis was not reported in this study.
Various methods of DNA extraction and PCR were investigated by Williams et al., who collected Penicillium roqueforti fungal spores in 1.5-mL tubes using a Burkard miniature cyclone. 79 The spores were then suspended in water containing Nonidet P-40 (a nondenaturing, non-ionic detergent) before undergoing a test matrix of DNA-extraction methods and PCR assays: untreated spores, mechanical disruption of spores using glass impact beads, and disruption followed by DNA purification; and single-step PCR, nested PCR, and PCR with Southern blotting. The authors reported that nested PCR was the most sensitive assay method. Disruption yielded single-spore detection regardless of DNA purification. Purification was, however, essential for specific detection when air samples contained high background concentrations of other aerosols. These results illustrate the importance of sample preparation and the characterization of nontarget aerosols.
Antibody-based sensing can achieve sensitive and specific detection of bioaerosols without the need to extract genetic material. Moreover, immunochemical sensing is compatible with several low-cost transducers that could enable a portable format. Such was the approach of Skládal et al., who coupled the ImmunoSMART electrochemical sensor (Smart, Brno) to a wetted-wall cyclone sampler (SASS 2300, Research International, Woodinville, WA) for detection of the model aerosol bioagent E. coli DH5α. 80 Combining cyclonic impaction with liquid impingement, a wetted-wall cyclone sampler pulls a liquid curtain up the cylinder wall so that aerosols are directly deposited into this capture liquid. This system collected aerosols in 5 mL of phosphate-buffered saline (PBS) with a 5 min sampling interval and 20 min analysis cycle. The entire system was battery operated and remotely controlled, and it demonstrated a 150 CFU (colony-forming unit) L−1 limit of detection, a promising result for further tests and field testing.
Hindson et al. reported a fully integrated autonomous pathogen-detection system (APDS) capable of aerosol sampling, sample preparation, multiplex immunoassay, and confirmatory PCR. 81 Designed to monitor for covert releases of bioagents in urban centers, the APDS collected aerosols using a high-volume virtual-impactor preconcentrator coupled to a wetted-wall cyclone (SASS 2000). Periodic aliquots of the collection fluid underwent automated sample preparation and analysis by multiplex fluorescence microsphere immunoassay. In parallel, liquid samples were also analyzed by real-time flow-through PCR. The strategic use of these orthogonal biomolecular assays significantly improved the specificity of the detector. In a characterization study of the APDS with oleic acid and polystyrene particles, Mainelis et al. 82 measured a collection efficiency of around 85% and cutoff sizes of 1.1 to 1.6 μm and 1.5 to 2.0 μm for the two sample types, respectively. Thus, the APDS was well suited to detect low concentrations of airborne pathogens.
Liquid Impingement
Impingers extract particles from the sample airstream by depositing them into a collection liquid. This medium not only is convenient for downstream biochemical analysis but also can preserve microbial viability better than dry-collection methods do. 83 The flow rate, volume, and composition of collection fluid influence the collection efficiency for a given particle type. Impingers like the All-Glass Impinger 30 (AGI-30; Ace Glass Inc., Vineland, NJ) rely on particle inertia by directing the airstream through a nozzle outlet positioned slightly above the liquid surface. Similarly, as in impactors, high-inertia particles (typically larger than 0.4 mm) that cannot follow the sharp turn in streamlines enter the collection liquid.
With such inertial impingers, the collection of submicron particles like viruses requires high-speed airflow (e.g., 300 m s−1), which causes violent bubbling and evaporative loss after prolonged sampling. This limitation has been addressed by bubbling impingers and the swirling BioSampler (SKC Inc., Eighty Four, PA), 84 which can perform longer-term sampling at lower airflow rates. The combination of turbulence and diffusion during bubbling traps nanoparticles in the collection liquid. The production of very small bubbles by means of a fritted nozzle 85 or porous medium further enhances particle diffusion to the air–liquid interface in the bubbles.
In addition to the challenge of evaporation, liquid impingement can compromise biological recovery efficiency. The effect on viability should be characterized for individual viruses; 86 however, it apparently can be mitigated with appropriate selection of the collection fluid. Using influenza, measles, and mumps viruses, Agranovski et al. were able to reduce viral inactivation during 4 h of bubbling by collecting in virus maintenance fluid instead of sterile water. 87
Conventional laboratory analysis of collection liquid that requires several days of incubation is not amenable to rapid testing. To address the need for a rapid analytical procedure, Agranovski et al. combined a porous impinger with real-time PCR.88,89 They sampled aerosolized Vaccinia virus (300 nm particles in 1 to 2 µm droplets) and isolated viral DNA from the collection liquid. Using a plaque assay and PCR, they achieved minimum detectable concentrations of airborne viruses of 125 × 103 PFU m−3 (plaque forming unit per m3) with 1 min of sampling time, and 10 × 103 PFU m−3 with 12.5 min of sampling.
Pyankov et al. augmented the system using real-time PCR to identify airborne influenza virus in a contaminated natural environment. 90 The system used 4-h sampling and an average of 2.5 h of analysis time. Most recently, Usachev et al. demonstrated a portable system by integrating the impinger with a duplex real-time PCR technique (Taqman variant) without nucleic acid purification. 91 The elimination of laborious sample purification was particularly essential to miniaturization of the device. Bacteriophage suspensions at 109 PFU mL−1 were aerosolized and detected using a single fluorescent dye with as short as 10 min of sampling and approximately 32 min of thermal cycling before the threshold cycle. The collection efficiency for viruses larger than 20 nm was higher than 90%.
Filtration
Filtration sampling traps aerosols by passing an air stream through a porous solid-phase medium. A variety of filter materials with specific pore-size and cutoff characteristics is available to match the target aerosol. Filter retentate can then be extracted into liquid for bioanalysis.
Fabian et al. collected aerosolized influenza (H1N1) virus using four aerosol samplers, including a 2-µm pore Teflon filter and a 3-µm pore gelatin filter. 83 The gelatin filter could not endure sampling longer than 15 min without dessicating and cracking. Although the Teflon filter efficiently captured total virus RNA, it preserved viability poorly compared to collection in PBS. The authors attributed the loss of infectiousness to the drying of the aerosol on the filters under continued airflow.
Zhao et al. envisioned a lab-on-a-chip aerosol-monitoring system enabled by a novel integration of a microfilter membrane with microdroplet manipulation by electrowetting-on-dielectric (EWOD) actuation. 92 The EWOD digital microfluidic technique uses electrodes coated in dielectric and hydrophobic layers to attract an overlaying droplet by reducing its interfacial tension. 93 The application of voltage sequences over an electrode array can thus “drag” individual droplets along programmed, even intersecting, paths. Zhao et al. proposed embedding a perforated filter with an EWOD electrode array such that retained particles are swept up by moving droplets. The droplets could then be transported by EWOD to an adjacent on-chip unit for analysis. Using particles manually deposited onto a microfilter membrane and water droplets approximately 500 nL in volume, the system collected 8-µm glass particles with over 95% efficiency and 8-µm polystyrene particles with over 85% efficiency. Based on these promising preliminary results, the authors anticipated subsequent demonstration of a more complete model system, including the air-suction filter unit and real airborne particles.
Electrostatic Precipitation
Efficient collection of submicron aerosols by inertia-based methods requires high-speed airflow, which may increase damage to microbes colliding with the collection medium. 94 However, electrostatic precipitation of charged particles, which has been used for dust control in indoor environments since the early 1900s, has proven especially effective to separate submicron aerosols from an airstream. 95 Therefore, electrostatic precipitation sampling (EPS) was developed as a means to deposit bioaerosols onto a substrate with a “soft landing” (i.e., a perpendicular speed on the order of 1 m/s or lower).
Mainelis et al. investigated the feasibility of EPS for viable microorganisms using a modified electrostatic aerosol sampler originally designed for nonbiological aerosols. 96 Testing three bacterial species on agar, water, and a filter as collection media, the authors observed relatively high removal efficiency but widely varying biological recovery. The authors reasoned that some of the more sensitive species could have been injured during the collection process by stress from corona discharge, ozone, and a strong electric field.
The researchers subsequently developed a new EPS system that could charge incoming particles with ionizers and then apply an adjustable precipitating electric field to deposit them onto agar plates along the flow axis.97,98 The sampler removed close to 80% of charge-neutralized fungal spores and vegetative cells, most of which were confirmed to be CFUs. Moreover, the experiments revealed that the aerosolized spores carried a net negative electric charge even after treatment by a Kr-85 charge neutralizer, a finding that raised the question of whether airborne microorganisms could be collected based on their native charge.
Yao et al. tested this hypothesis by collecting bacteria and fungi using a new electrosampler with no charging of incoming particles. 99 Indoors, the electrosampler achieved a collection efficiency around 30 to 40% but measured up to 9 times higher culturable bacteria and 25 times more culturable fungi compared to a BioStage impactor (SKC Inc.). The data indicated that the microorganisms under test carried a net charge that could be leveraged in an EPS collector without the initial charging stage. Such a device promised not only high biological recovery but also simplified design for field use.
In a separate study, 100 the electrosampler was used to analyze airborne allergens and toxins, nonculturable bioaerosol components whose biological integrity might not be affected by charging. Dust allergens (DER p 1 and Der f 1) and toxins (endotoxin and (1,3)-β-d-glucans) were precipitated into water in 96-well plates for analysis with ELISA and limulus amebocyte lysate (LAL), respectively. The electrosampler obtained up to 11 times higher aerosol concentrations compared to the BioSampler impinger.
Effective integration of sampling with microfluidic analysis requires that aerosols be collected and concentrated into small volumes of liquid. In a significant step toward automated detection using EPS, Han and Mainelis combined the electrostatic collection mechanism with a superhydrophobic electrode surface that produced efficient bacteria collection (as high as 72%) and concentration (factors of 105 to 106) in water droplets as small as 5 μL.101,102
Tan et al. designed an electrostatic sampler with a hemispherical electrode to collect and concentrate aerosolized bacteria in a microliter-scale liquid reservoir. Designed to be easily integrated with microfluidic analysis, the system collected 70% to 90% of indoor particles of size 0.3 to 2 µm. Formats such as those of the two preceding samplers could prove most suitable for quantitative PCR.
Technological Challenges at the Point of Care
Bioaerosols form a rich sample medium for the noninvasive measurement of human disease states. As human bioaerosols may span a wide range of size and morphology—from relatively large skin particles to submicron droplets in exhaled breath—a variety of sampling methods should be leveraged to suit specific purposes. Although some of the sampling methods described in this article were originally developed for environmental applications, they may yet be adapted for direct sampling of bioaerosols from a human sample.
Compact aerosol collectors could harness the strengths of modern analytical techniques by serving as front-end samplers to lab-on-a-chip systems. Fully integrated biochips can realize tremendous optimization for niche applications to deliver reduced processing time, lower cost, ease of use, and miniaturization. 103
This vision, however, comes with its share of challenges. Liquid-phase analysis requires air-to-liquid transfer prior to sample preparation. For this reason, dry matrix-type filters and impactors are difficult to couple to autonomous systems. Impingement, wetted-wall cyclone sampling, and EPS into a liquid medium better suit downstream microfluidic analysis.
Furthermore, the sample pre-concentration required to detect a dilute aerosolized analyte implies a high-volume air flow coupled to a low volume of collection liquid. Therefore, compact personal sampling pumps may be an appropriate starting point for portable systems, in which the pump technology faces tight constraints of size, power consumption, and cost. Stationary applications, such as bedside monitoring for nosocomial infections, might impose less stringent constraints.
For robust field performance of a bioaerosol-based POC diagnostic, technology developers must quantify and mitigate the effects of nontarget aerosols. Depending on the use case, exposure to the ambient environment during collection should be carefully controlled. In the case of EBC analysis, standardization of instruments and techniques is required to advance the field of knowledge. 104
Finally, because sampler performance depends on a complex interaction of inlet, removal, and biological efficiencies, each system must be thoroughly characterized for its intended analyte as surrogates may not be valid models.
Disease Detection Using Volatile Organic Compounds in Breath and Odor
Noninvasive diagnosis of disease through measurement of volatile biomarkers has long been of special interest in clinical practice. It is widely accepted that any disruption in the normal functions of an organ or organ system is reflected at the interface of the human body with its surroundings. Consequently, disease biomarkers produced deep in the body are eventually distributed via circulation and excreted through a number of pathways, such as urine, breath, sputum, saliva, and sweat ( Figure 2 ). Many of the excreted biomarkers are volatile, creating a premise for their use in noninvasive disease diagnosis.
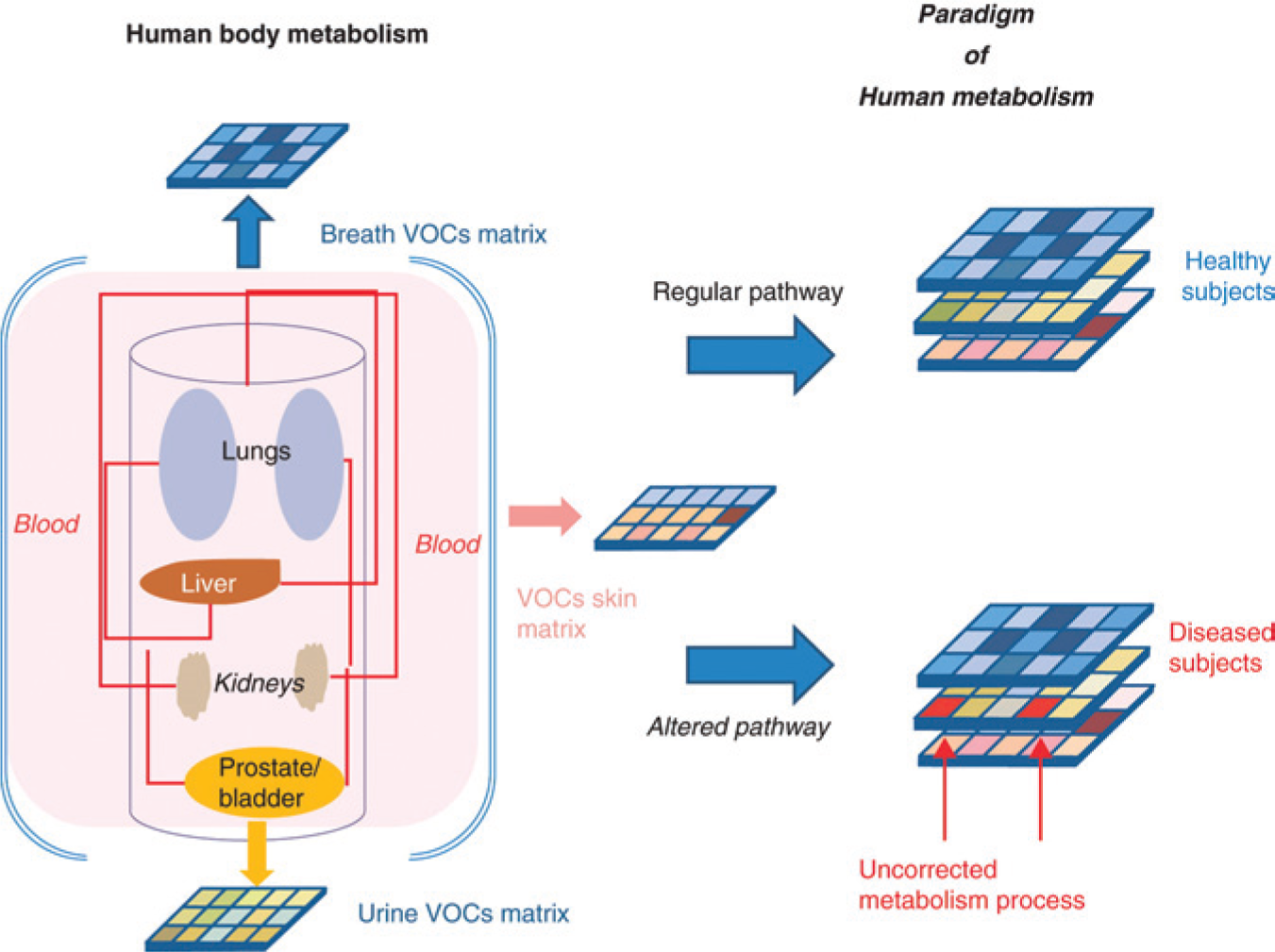
Volatile organic compounds as a paradigm of human metabolism. Physiological processes deep in the body are reflected through a number of pathways, such as urine, breath, sputum, saliva, and sweat. A. D’Amico et al., Expert Opinion on Medical Diagnostics, 2012; 6 (3): 175–185, copyright © 2012, Informa Healthcare. Reproduced with permission of Informa Healthcare. 213
Volatile biomarkers, particularly those resulting in a change of odor that is perceptible to the human olfactory system, have been used for years in disease diagnosis. Diagnosis of disease by placing human sputum on hot coals to generate odors dates back as early as 400 BC. 105 Numerous examples of disease detection by human-perceptible odors include the smell of rotting apples or acetone in breath as a marker for diabetic ketoacidosis 106 and the foul, putrid smell of sputum and breath as a marker for lung abscess. 107
With the goal of increasing sensitivity and discrimination, clinicians even trained animals such as domesticated dogs to indicate human disease states. In particular, several studies successfully demonstrated the use of dogs for cancer diagnosis. 108 A systematic review by Moser and McCulloch examined the feasibility of human cancer detection by animals and concluded that breath represents the best sample type for early diagnosis of cancer. 109
A growing number of biomarkers and biomarker mixtures have been linked to disease endpoints.37,110–112 Most of these studies have been conducted using gas chromatography–mass spectrometry (GC-MS), currently a gold standard for gas analysis. Because of its high sensitivity and ability to characterize gas mixtures, this method is ideal for comprehensive studies of biological samples and the initial identification of disease biomarkers. However, many barriers hinder the use of this technique for clinical diagnosis in POC settings: high-cost, time-consuming sample preparation, and the requirement of extensive training and expertise for technical maintenance and operation. Finally, the mechanism for complex sample mixture analysis is not ideally suited for POC settings. To be able to effectively resolve mixture components, samples must be passed through a gas chromatography pre-separation step prior to detection by mass spectroscopy, resulting in sequential analysis of individual mixture components rather than the true mixture analysis.
A more direct and accurate analysis of volatile biomarker samples examines disease signatures. This type of analysis assesses all sample components at once without explicitly selecting specific chemicals for investigation, resulting in signatures of specific disease states unique to the specific biomarker sample mixtures. By calibrating the result of such analysis to disease presence and severity, a more reliable relationship linking the biomarker mixture to disease endpoints can be derived. In other words, the relationship between biomarkers and disease can be corrected to account for the presence of any protective factors (e.g., therapeutic drugs or host-response molecules). Also, because most of the sensor array devices can detect changes in the relative levels of different biomarkers in a sample mixture, these devices can be used to measure subtle physiological changes. This type of measurement thus represents a holistic view of the condition for which the device has been calibrated.
Sensor Array Technology
A sensor array, commonly referred to as an e-nose, is an alternative method for biomarker-mixture analysis that is gaining in popularity due to its applicability to POC settings. Commonly composed of sensors with low specificity but a range of selectivity toward different compounds, sensor array devices enable the analysis of complex sample mixtures by coupling sampling with instantaneous analysis without the need for pre-separation. As a result, the combined response of all sensors can be used to differentiate among samples without the need for high specificity from each of the sensor elements.
The selection of sensors with complementary cross-sensitivities, along with suitable feature extraction and pattern-recognition algorithms for sensor data analysis, allows for effective discrimination between different volatile biomarker mixtures. This type of multiplexed detection can be very sensitive to subtle changes in disease progression, because even small changes in physiological processes and metabolic pathways can modify the relative levels of biomarker-mixture components. The ability to distinguish sample mixtures that differ only by the relative levels of their components also enables the identification of various bacterial infections. Even though different species might share metabolic pathways, they usually generate metabolites in different proportions, leading to sufficient differences in volatile mixtures for resolution by the sensor array.
The use of sensor arrays for medical applications has been reviewed by Thaler et al. 113 In a recent review, Mahmoudi explored how developments in sensor technology could support disease control and management in a global context. 114 The use of sensor arrays for early disease diagnosis, with a focus on microbial diseases, has been the subject of another review. 115 Sensor array operation mechanisms as well as the feasibility of using sensor arrays in medicine were assessed in depth by Wilson and Baietto. 116
The large and growing selection of transducer technologies and materials further increases the range of biomarkers that can be detected, and advances in signal-processing and pattern-recognition algorithms enable complex mixtures to be reliably analyzed.113,114,116–118 Coupling these algorithms with the appropriate calibration of sensor outputs to specific disease outcomes allows the compilation of libraries of disease signatures. This work has been a focus of several groups that are building data-warehouse systems for volatile biomarker sampling and pattern recognition. 119 These systems can be used to generate reference libraries of detectable volatile biomarkers as well as standard operating procedures for volatile-biomarker sampling and detection. As more biomarkers are discovered and as these libraries grow, sensor array diagnosis will become more specific and useful. As the number of different instruments and analysis methods grows, more effort will need to be directed toward standardization of sampling, measurement, and analysis methods, as well as associated terminology. 120
For a given application, selecting the most suitable type of sensor technology depends on factors such as the type of sample (e.g., breath, urine, and skin), physical characteristics of the sample (e.g., temperature and humidity), physiochemical characteristics of target biomarkers, concentration of biomarkers of interest, and complexity of the mixture (i.e., the number of compounds that need to be resolved and their physiochemical similarities). 121 Characteristics used for evaluating sensor performance include sensitivity, selectivity, stability, as well as response and recovery times. Types of sensor technologies used for the development of biomedical devices include chemical, physical, and biological ones. They differ in the process by which detection and signal transduction occur.122–124 Because chemical sensors are most frequently used in biomedical sensor array systems, they will be the focus of this review.
The major chemical sensor technologies include optical,125–129 electrochemical,130–137 conductive polymer,138 –143 semiconductor,144–146 and mass sensitive (piezoelectric)147 –151 technologies. Due to their sensitivity and selectivity, optical and electrochemical devices are often the technologies of choice for monitoring inorganic compounds (e.g., NO, CO2, and H2), which are often associated with cardiovascular conditions, asthma, and COPD. Optical and electrochemical instruments, however, are not yet optimized for low-cost production. In addition, due to their higher sensitivity and selectivity toward specific inorganic molecules, they are seldom used in a sensor array format. Thus, the present sensor technology overview focuses on conductive polymer, semiconductor, and piezoelectric technologies, which are most commonly used in commercial sensor arrays for biomedical applications due to their potential for miniaturization and lower-cost manufacturing processes ( Table 2 ).
Commercially Available Multisensor Array Devices That Have Been Used for Healthcare Applications.
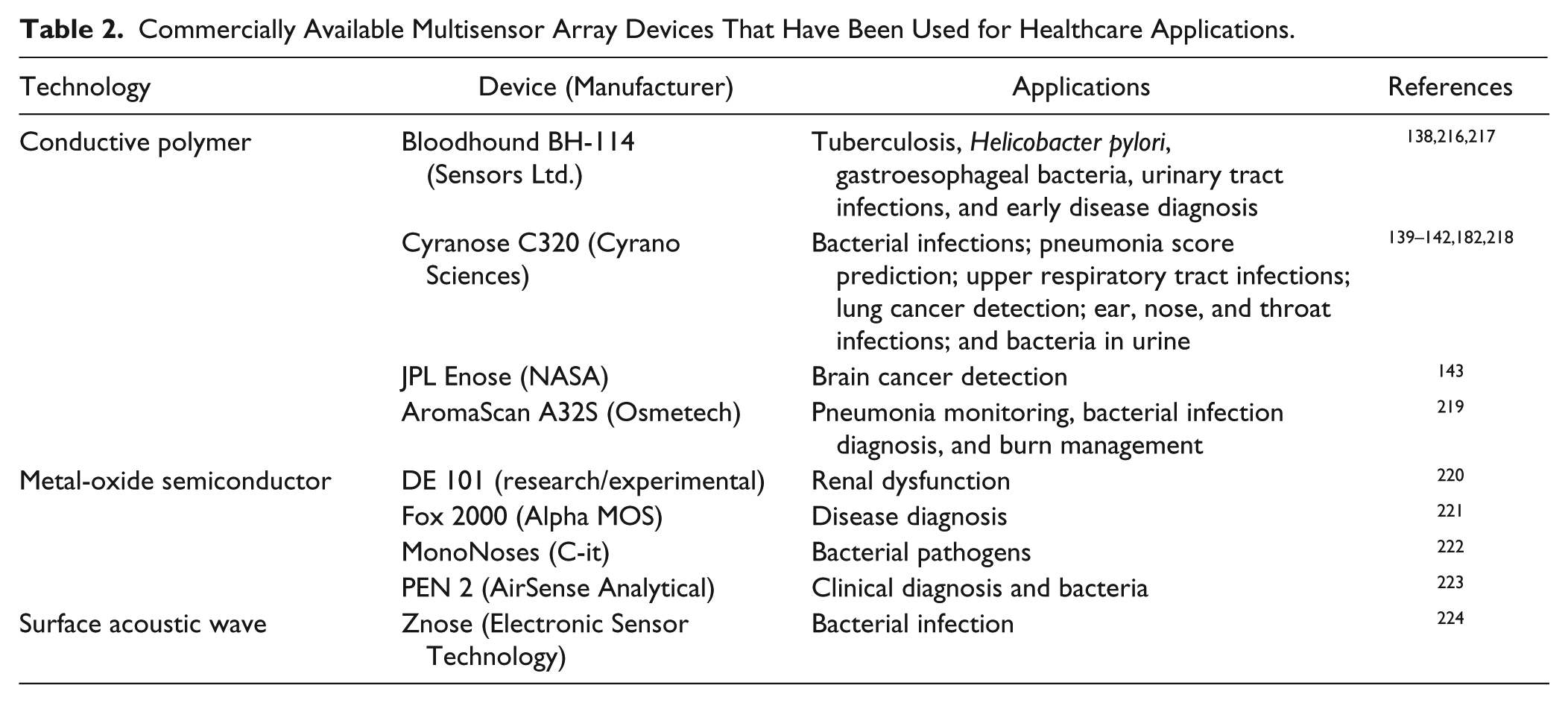
Conductive polymers and metal-oxide semiconductors are two of the most frequently used sensor technologies for volatile biomarker analysis. Conductive polymers are suitable for applications that require low power consumption. They exhibit fast and reversible response at ambient temperature and are sensitive to a range of different compounds, particularly ketones, alcohols, esters, and fatty acids. The rate of compound adsorption to a polymer surface modulates the polymer conductivity and determines the extent of the sensor response. Selectivity and sensitivity can be controlled by engineering the morphological and chemical properties of the polymer.
Metal-oxide semiconductors, like conductive polymers, are also conductance transducers. They exhibit large, fast, and reversible changes in conductivity in response to a range of different compounds, such as alcohols, alkanes, NOx, CO, NH3, H2S, H2, and SOx. However, they require high temperatures for operation (~300–400°C), which results in high power consumption. They are composed of a metal-oxide thin film (e.g., SnO2) heated by a ceramic pellet. The metal-oxide layer acts like a semiconductor, the conductivity of which changes as a result of volatile analytes adsorbing to its surface. The selectivity and sensitivity can be altered by doping and control of the operation temperature. With well-developed manufacturing processes, these sensors can now be manufactured at low cost with a small form factor, resulting in their popularity in research and commercial applications.
Piezoelectric sensor technology has been gaining in popularity recently because of its low power consumption, high sensitivity and selectivity for a range of VOCs, stability, and reproducibility. These sensors are composed of a quartz crystal oscillator coated with a membrane or film that determines the selectivity of the sensor. 152 The mass change caused by adsorption of volatile molecules to the membrane shifts the quartz crystal resonant frequency. Differential sensor output is common, in which the reference sensor signal is used to compensate for temperature, humidity, and pressure effects. The two major types of piezoelectric sensors—surface acoustic wave (SAW) and bulk acoustic wave (BAW) sensors—differ in the layer through which acoustic waves travel (i.e., surface vs. bulk). SAW sensors are generally operated at higher frequencies, often resulting in improved sensitivity. Piezoelectric sensors are not yet abundant in commercial devices due to the complexity of interface electronics and the manufacturing process, factors that often result in higher instrument cost.
To make the most out of the multivariate response of sensor array devices, a fast and reliable signal-processing and pattern-recognition algorithm must be selected. Sensor array data-analysis algorithms can be classified as supervised and unsupervised. Supervised techniques, such as discriminant function analysis, are used when some information on sample composition is available and when analysis aims to differentiate a population group with disease and controls. Unsupervised techniques, such as cluster analysis, are used to classify a population without prior knowledge about sample composition. Principal-components analysis (PCA) is another example of an unsupervised technique that is commonly used for finding and visualizing relationships among samples. For nonlinear sensor technologies and fast analysis, neural network analysis is usually preferred. Assuming a relatively large training data set, this analysis technique can be very robust for predicting specific disease outcomes, even in the presence of sensor drift and nonlinear sensor response.
Until recently, the sensitivity of most commercially available sensors was insufficient for detection of the low levels of volatile biomarkers that is required for disease diagnosis. Improved manufacturing methods and innovation in materials engineering in the past few decades have improved detection properties while reducing device manufacturing costs. Target characteristics of a multisensor device for disease diagnosis have been reviewed by Payne et al. 153 To become ubiquitous in POC environments, however, device cost needs to be further reduced and instrument operation simplified.
Both of these goals can be achieved by designing application-specific instruments. A popular strategy of sensor array instrument development is to make the instrument capable of detecting as wide a range of volatile compounds as possible. While this strategy makes the instrument more versatile, it also increases its complexity, cost, and time to market. An alternative strategy is optimizing the instruments down to the essential components and sensors needed for discrimination of specific disease states. This strategy often has an added advantage of better reliability and accuracy for the specific disease(s) it was designed to detect, while reducing instrument cost, increasing portability, and simplifying the operation.
The list of possible applications and needs for this type of instrument in clinical settings is growing beyond early detection and monitoring; these include telemedicine, 154 contamination prevention, and detection of environmental health hazards. 155 The use of sensor arrays in telemedicine, in particular, is promising and has been used for discriminating patients remotely in Africa. 154
In Vitro Off-Line Volatile-Biomarker Detection
The detection of volatile biomarkers using sensor arrays is now possible in a number of human samples, including breath, 110 sputum, 138 saliva, 156 ocular fluids, 157 urine, 158 feces, 37 sweat, 159 and skin. 160 Most early studies involving the measurement of volatile biomarkers were accomplished by taking samples of volatile biomarkers with analysis follow-up rather than by combining sampling and analysis. This offers an opportunity to collect a more concentrated sample facilitating the analysis. This also introduces new challenges, however, such as optimizing biomarker capture and preservation because of the high volatility of many of the biomarkers.
To ensure reliable analysis, the sampling procedure and the exposure of the sensor array to the sample should be consistent. To avoid sample contamination, materials for the instrument and sampling system should be carefully selected, with preference given to materials such as glass, stainless steel, Teflon, and brass. Plastics and rubber should be avoided because of the off-gassing effect. Sample flow rate, temperature, and humidity should be standardized to avoid confounding effects as well as drift. In fact, water vapor is known to deplete water-soluble volatile organics and is a major problem for samples with high relative humidity, such as breath and urine headspace. Cryogenic 161 and adsorbent 162 techniques have been used to remove water samples, but they should be used with care because they can result in significant loss of analytes as well. 163 Appropriate standards should also be developed for sensor array devices to maintain consistent performance throughout the long term.
Due to the low concentration of volatile biomarkers present in many of the biological samples, sample pre-concentration is often a necessity. A variety of techniques for sample collection, pre-concentration, and preservation was developed, including chemical trapping, cryogenic trapping, and adsorption trapping. The chemical pre-concentration method exploits the chemical reactivity of the analyte, which gets bubbled through a solution and extracted as a result.164,165 This method is associated with a number of challenges, such as long reaction time, memory effects, and analyte loss.
Cryogenic trapping involves the capture of volatile compounds by cooling. It was first developed by Pauling for sampling human urine volatiles with liquid nitrogen–chilled cold traps and human breath with stainless-steel tubes cooled by an isopropyl alcohol dry-ice bath. 163 This method is often challenging to implement in low-resource or field settings, and it is also problematic due to the freezing of water and carbon dioxide in addition to volatile organics in the sample.
Adsorptive trapping is the most convenient method to implement in the field and is the most common currently used method of volatile compound sampling and pre-concentration. In this method, volatile compounds get adsorbed to porous media such as polymers, graphitized carbon, activated charcoal, and carbon molecular sieves, all of which are designed to trap compounds of specific size ranges and with specific chemical-physical properties. Before a specific adsorbing agent is selected, the collection and recovery efficiency of VOCs needs to be carefully evaluated to avoid sample breakthrough and memory effects as well as to minimize the possible impact of sample characteristics, such as high water content. Often, different types of sorbents are combined in a single system to take advantage of their different ability to adsorb various types of compounds as well as protect the system from saturation and humidity impact.166 –168
Solid-phase micro-extraction (SPME)169 –171 and its derivative, low-temperature glassy carbon (LTGC) 169 SPME, are popular techniques for sampling and concentrating volatile samples. These techniques are popular because of their greater extraction efficiencies and lower detection limits. However, they are limited in the number of compounds that can be adsorbed. 172
Another interesting technique that combines sampling and pre-concentration, membrane extraction with sorbent interface (MESI), has been used for breath sample collection. 173 Finally, because the sorbent-collected volatile samples need to be desorbed by heating, it is important to ensure that this will not result in any reactivity or decomposition of either the sorbent material or the collected sample.
Most early analyses of volatile biomarkers were conducted in vitro using bacterial plate cultures and tissue samples. 174 Even though these types of analyses are inherently off-line, sensor array devices can be used to significantly speed up the analyses, which can take 24–48 h using conventional techniques. Dutta et al. later used volatile organic biomarker signatures to differentiate different strains of ear, nose, and throat bacteria using the Cyranose 320 conducting-polymer multisensor device with 99.69% accuracy. 139 Gardner et al. used a six-sensor metal-oxide semiconductor device to classify the growth stages of common pathogenic bacteria with 81% accuracy. Cyranose 320 was used by Boilot et al. to detect and discriminate among six different species of bacteria associated with eye infections. The group also compared six different classification algorithms and found that the variants of neural network algorithms worked best for the task. 175
As sampling methods improved, in situ analysis of human samples as well as on-line sampling followed by real-time analysis became possible. This resulted in more targeted sensor array devices designed for use in clinics and other POC facilities. One of the key features that is important for widespread acceptance of volatile-biomarker sensor array devices at the POC is noninvasiveness. Not only is this important for patient comfort, but also it reduces the chance of transmission of infection and reduces the cost of the setup. Addressing this factor, the following sections describe the systems based on the least invasive sampling methods for in situ volatile-biomarker analysis: breath, skin surface, and urine headspace.
Diagnosis Using Volatile Biomarkers in Breath, the Upper Respiratory Tract, and Sputum
Breath analysis is one of the most popular types of sample that is used with volatile-biomarker sensor arrays due to its diversity and abundance of physiologically relevant compounds. Up to 600 different compounds have been identified in human breath.61,172,176,177 Although it is one of the least invasive procedures, breath analysis does require careful design of sampling systems as well as some effort on the part of the patient to ensure sample reproducibility. Exogenous variables such as environmental exposure and food and drug intake strongly influence what compounds are present in breath.166,172,178 Breath sampling procedures, therefore, have to be carefully controlled to collect the most representative and biomarker-rich sample.
Only the last 350-mL fraction of the breath—alveolar air—is biologically relevant, for it exchanges substances with the blood. Because of reduced dilution, selective sampling of this air fraction, as opposed to the total breath, allows 2–3 times higher concentration of the endogenous biomarker to be measured and avoids interference from external compounds. Interpersonal and even intrapersonal variation in compound levels within the breath necessitates that an average of several breath samples is measured and that collection efficiency is estimated by measuring CO2, which is generally considered stable in resting healthy subjects. Several different exhaled-air sampling devices have been proposed to optimize the quality of sample collected. 179
Breath analysis has been used to diagnose diseases such as upper respiratory tract infection, 180 diabetes, 181 and ventilator-associated pneumonia.29,182,183 Diabetes has been diagnosed using breath samples in a number of studies. Ping et al. developed a sensor array for accurately measuring breath acetone, a key marker for diabetes. 181 Guo et al. evaluated the effect of confounding factors such as the time since food intake as well as the presence of other health conditions on the ability of a sensor array to distinguish between people with and without diabetes. 184 They found that with correct timing of sampling, diabetes patients were discriminated from nondiabetes patients with 100% accuracy.
Cancer is commonly diagnosed using breath samples. It was shown that some alkanes, such as hexane, are found in larger concentrations in patients with lung cancer compared to healthy controls. 185 Di Natale et al. used an array of eight piezoelectric quartz microbalance (QMB) sensors coated with different metalloporphyrins to identify lung cancer patients in a population with healthy controls and lung cancer patients who had undergone surgical treatment. 186 D’Amico also used a QMB sensor array to distinguish lung cancer from other types of lung disease and healthy controls with greater than 85% accuracy. 187 These findings were validated by Chen et al., 188 who used an array of different types of SAW piezoelectric sensors to validate 11 VOCs as markers of lung cancer. A Cyranose 320 was used by Machado et al. to create a predictive model for bronchogenic carcinoma based on sensor output. 141
Gas-sensor array technology is also promising for asthma diagnosis. A Cyranose 320 was used by Dragonieri et al. to diagnose asthma and distinguish among different levels of severity. 111 Seeking to improve accuracy, Montuschi et al. compared different types of sampling protocols and exhaled air fractions. 189 They found that alveolar air resulted in better diagnostic accuracy compared to airway air. The sensor array–based diagnosis also outperformed (87.5% accuracy) a combination of standard techniques such as exhaled iron nitric oxide and spirometry. The sensor array was also used to discriminate between asthma and COPD, conditions that often have overlapping symptoms. 30 Asthma patients, COPD patients, and healthy people were successfully distinguished by the sensor array response, even under the confounding effect of smoking.
Detection of TB using gas sensor array technology is especially attractive given the global burden and time-consuming diagnosis of the disease. In the first report of mycobacteria detection using a rapid gas-sensing system, Pavlou et al. used cultures of pre-diagnosed TB samples to optimize data-processing algorithms for an array of 14 polymer gas sensors. The system then correctly diagnosed 9 of 10 sputum samples and distinguished among different causative Mycobacterium species. 12 Fend et al. obtained similar success rates in a larger-scale study with 91% specificity and 89% sensitivity among 330 tuberculosis patients. 138
Phillips et al. successfully isolated TB-associated VOC compounds in breath to distinguish hospitalized patients from normal controls, and to isolate TB-positive patients from other patients. 13 Despite achieving 100% sensitivity and specificity, the study had insufficient statistical power to rule out potential confounding factors such as concomitant HIV infection. A follow-up study involving 279 patients from 3 different countries identified active pulmonary TB in patients with 80% accuracy using a 6 min POC breath test. 190
Pneumonia is another respiratory condition that is notoriously difficult to diagnose, which is part of the reason for the absence of a gold standard diagnostic method. A Cyranose 320 was used to correctly differentiate patients with pneumonia from healthy controls. 29 Similar results were obtained by Hanson and Thaler. 183
Even though most studies using sensor arrays for breath diagnosis are conducted by sampling air coming deep within the lungs, a few studies have investigated upper-airway and nasal out-breath. This approach has been particularly useful for the diagnosis of chronic rhinosinusitis as shown by Mohamed et al. 191 Bruno et al., using a commercially available ZNose instrument based on SAW technology, analyzed the volatile organic signature of the same condition to identify six marker compounds for the disease. 192
Diagnosis Using Volatile Biomarkers at the Skin Surface
Skin analysis is an especially interesting application of VOC-based diagnostics due to its potential for long-term monitoring. Biomarkers reflecting changes in metabolite patterns can be useful for early detection of disease onset. Skin biomarker detection faces significant challenges from the high dilution rate of volatile biomarkers as well as the high potential for interference from exogenous compounds in ambient air. Therefore, sample pre-concentration is often necessary.
Volatile skin biomarkers can be monitored directly from the skin surface, from swab samples, or by sampling sweat. Di Natale et al. analyzed VOCs at the surface of human skin and analyzed the feasibility of using them to diagnose diseases. 193 Boilot et al. used the skin-swab method combined with a commercially available sensor array instrument to distinguish six species of bacteria commonly associated with eye, nose, ear, and throat infections with around 97% accuracy. 194 A frequent application for monitoring skin volatile biomarkers is assessment of volatile microbial metabolites released from wounds. 207 Byun et al. used a sensor array composed of commercially available metal-oxide semiconductor sensors to diagnose wound infection. 195 Witt et al. discovered that volatile gases from sweat change as a result of liver dysfunction and liver cirrhosis associated with alcohol abuse. 160 Using a sensor array combined with an in-house applicator to monitor transpired dermal gases at the skin surface, they distinguished healthy people from patients addicted to alcohol and those with liver cirrhosis. D’Amico used a QMB sensor array to diagnose melanoma using VOC signatures associated with skin lesions. 196
Diagnosis Using Volatile Biomarkers in Urine Headspace
The high biomarker content of urine makes it one of the most common diagnostic media in clinical settings. Gas sensor array devices show great promise as fast, low-cost analyzers for relatively noninvasive analysis of biomarkers from urine headspace, in which a number of different VOCs have been identified and linked to conditions relevant to POC settings, such as tuberculosis. 197
Several groups have used volatile metabolites in urine to successfully diagnose urinary tract infections. Aathithan et al. used the Osmetech Microbial Analyzer, combining a headspace sampler with an array of four conductive-polymer sensors, to analyze volatile biomarkers of bacteriuria, achieving greater cost reduction, sensitivity, and selectivity for diagnosis than conventional culture methods. 112 This is a promising result, and it could be especially useful for POC facilities in developing countries.
Pavlou et al. evaluated the feasibility of using a gas sensor array to diagnose a range of kidney diseases. The device was used to identify traces of blood in urine and to distinguish among three different bacteria species with high accuracy. 198 Lin et al. used a gas sensor array to characterize a volatile biomarker signature associated with uremia. 199 Kodogiannis et al. used the Bloodfound BH-114, composed of 14 conducting polymer sensors, to diagnose urinary tract infection. Barnabei et al. used an array of eight QMB sensors to differentiate urinary tract cancer from prostate cancer and healthy controls. 200
Technological Challenges at the Point of Care
Clearly, the capability for comprehensive analysis, the portability, and the lower cost of sensor arrays could be invaluable in POC settings. Most commercially available sensor array devices are the size of a laptop and are battery powered, making them already suitable for clinics in low-resource settings. Improvements in manufacturing technology and new materials are enabling further miniaturization of these instruments to handheld and even wearable dimensions.201–206 By the same token, the instrument cost is also rapidly decreasing as these devices can be manufactured using standard screen printing and chemical vapor deposition techniques. Sensor arrays based on printable conductive polymers and dyes are especially promising for the development of low-cost and even disposable devices.207,208 The current cost range for commercial sensor arrays, which is between $1000 and $3000, could be significantly reduced by optimizing sensor arrays for specific applications. Many sensor array devices integrate with on-line functions and do not require consumables or sample transportation, all attractive features for next-generation diagnostic tests.
Certain barriers, however, must be overcome for clinical uptake of the technology. From a research and development perspective, long-term repeatability and reproducibility of the sensor devices need to be characterized. Given the complexity of biological samples and the differences among experimental techniques used by different research groups, discrepancies among the research results of different groups have been unavoidable. Sampling and test protocols need to be standardized to ensure data reliability and equivalence among devices. Comprehensive standardized databases of sensor response signatures must be developed, and transferability of these databases needs to be evaluated.
The clinical performance of sensor array devices must be further evaluated. Most early sensor array devices were developed for general research, so operating protocols in most cases have been developed by end users. To gain adoption by medical personnel, sensor array devices need to be optimized for specific applications with rigorous contextual evaluation. A promising strategy to this end is the integration of sensor array technology within existing medical processes and procedures such as wound dressings 209 and endoscopy. 210
Conclusion
Airborne specimens of volatile compounds and bioaerosols form an information-rich sample medium for the noninvasive measurement of human disease states.
Microbes and cellular debris emanating from breath, skin, and excrement have long been studied as possible modes of disease transmission, but they have not been exploited as biomarkers for diagnosis. This dearth of research presents a significant opportunity for new explorations of noninvasive POC tests.
Selection of the method of aerosol collection should consider the size of the target particles and requirements of downstream analysis, including any need to preserve biological characteristics. Assays that rely on the infectiousness or viability of the target microorganism most likely require liquid collection media, whereas dry collection may be applied for nonviable bioassays like PCR and ELISA.
Compact aerosol collectors that achieve efficient air-to-liquid transfer in a small volume of collection liquid could harness the strengths of modern analytical techniques on lab-on-a-chip systems. In the literature, the most promising developments toward this goal have been demonstrated using impingers, wetted-wall cyclones, and electrostatic precipitators. Still needed, however, is a new field-ready sampler that can efficiently collect submicron human-source aerosols while preserving the integrity of airborne infectious agents in a manner compatible with lab-on-a-chip analysis. Such systems, perhaps integrated into living spaces or already-familiar medical form factors such as respirators, 211 would be especially promising for the diagnosis of infections in the respiratory and GI tracts.
Noninvasive diagnosis of disease by measuring volatile biomarkers has a long clinical history. For example, distinctive patient odors detected by human olfaction have been traditionally linked with specific health conditions. In modern biomedicine, many volatile biomarkers from breath, sputum, skin, and excrement have been associated with disease outcomes using isolation and identification by GC-MS. An even more reliable relationship between a biomarker mixture and health state can be derived without pre-separation using an e-nose sensor array.
Portable sensor array systems are particularly attractive for their independence from consumable reagents and sample transportation. Application-specific sensors optimized for usability and low cost could meet the demands of field use if connected to on-line response signature databases. Among the conditions of global priority, tuberculosis, respiratory infections, diarrheal infections, and cancer have been accurately predicted using portable sensor arrays. In contrast, little to no research has examined the volatile compounds or signatures associated with malaria and HIV/AIDS, two of the world’s leading causes of death. Sensor arrays are well positioned to advance much-needed diagnostic solutions.
Technology for aerosol and volatile biomarkers alike shows significant potential in the challenge of meeting the ASSURED technical constraints for low-resource settings. Nonetheless, further innovation and cost reduction are needed before these technologies find effective niche applications in health care systems. Comprehensive development programs under which multiple disciplines work together with standardized methodology and clinical validation can realize the potential of airborne biomarker analysis to address unmet needs for the world’s most pressing disease burdens.
Footnotes
Acknowledgements
The authors are grateful to Aterica Health Inc. for support received during the preparation of this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
