Abstract
To transfer large cargo into mammalian cells, we recently provided a new approach called a photothermal nanoblade. Micron-sized membrane pores generated by the nanoblade are surprisingly well repaired with little cell death, suggesting rapid membrane-resealing dynamics. Here, we report the resealing time of photothermal porated mammalian cell plasma membranes using an electrical impedance sensor. Cell membrane pores were generated by high-speed cavitation bubbles induced by laser pulsing of metallic microdisks on a pair of transparent indium tin oxide electrodes. Electrical responses from the sensor electrodes were obtained with a signal voltage of 500 mV and a frequency at 500 kHz. Real-time impedance measurements show that membrane resealing and impedance recovery take a surprisingly long 1 to 2 min after laser pulsing. A nonrecovering impedance shift is also detected for cells after high-energy laser pulsing. This impedance response is also confirmed by a separate experiment in which thin-film gold electrodes are used to trigger cavitation bubbles for opening transient membrane pores on cells cultured on electrodes. Overall, our study platform provides new insight for micron-sized membrane defect repair dynamics to maintain cell viability.
Introduction
Technologies that enable membrane pore opening and material delivery into live mammalian cells have broad applications across biomedical fields. Methods include electroporation, sonoporation, optoporation, microinjection, and other chemical and biological approaches.1–10 The size of delivered cargo can vary by several orders of magnitude, from small molecules such as DNA, molecule probes, quantum dots, proteins, and microbeads up to micrometer-sized intracellular pathogens, such as bacteria. 11 Technologies that allow large-size cargo delivery into live cells are highly sought but rare. For example, the ability to deliver live bacteria into cells and bypass the endocytosis process allows biologists to study how pathogens infect a cell, duplicate inside a cell, and spread to other cells to form plague. 12 The ability to deliver quantum dots conjugating with tubulin allows labeling of the cytoskeleton structure of live cells. 13 Large cargo delivery has been difficult because it requires not only the opening of a large transient pore on a cell membrane but also an efficient mechanism to push large and slow-diffusing cargo into the cell interior before the membrane opening reseals. Among current delivery approaches, the photothermal nanoblade has shown its effectiveness in delivering large cargo into live cells with high viability and high efficiency.11,12,14 The photothermal nanoblade uses a metallic nanostructure to harvest energy from short optical pulses, typically at a laser fluence of ~180 mJ/cm2, and converts this energy into localized and patterned nanocavitation bubbles. When a mechanically soft and fragile cell membrane is near or in contact with the metallic nanostructure, an explosive cavitation bubble rapidly punctures the adjacent lipid bilayer via high-speed and localized fluid flows. This rapid motion produces little mechanical perturbation to the rest of the cell membrane and can generate a micron-sized delivery port with high cell viability. By integrating a metallic nanoscale thin film on a conventional microcapillary pipette, pressure-driven active delivery, instead of passive diffusion, can be synchronized with the transient pore-opening process to push large cargo into cells. Cell viability is enhanced over traditional microinjection techniques because the micropipette never enters the cell, which reduces mechanical trauma. A critical parameter of this delivery process is the resealing time of the transient membrane pore after laser pulsing. Cargo delivery must be complete before the membrane pore closes. Several studies have shown the dynamic cell membrane responses after optoporation, 2 sonoporation, 15 and electroporation. 16 However, there is currently no direct measurement of the resealing time of membrane pores opened by photothermally induced cavitation bubbles on metallic nanostructures. Here, we present a novel device that integrates metallic microdisks with transparent indium tin oxide (ITO) electrodes to achieve cell membrane opening by laser-induced bubbles and simultaneous real-time monitoring of electrical impedance responses of cells being pulsed. Fluorescent dyes were used to confirm cell viability and the opening of transient membrane pores after laser pulsing.
Materials and Methods
Working Principle
Figure 1a illustrates the working principle of the metal microdisk-integrated electrical impedance sensor. Metal microdisks are patterned on a pair of transparent ITO electrodes to monitor the response of a target cell (stage 1). By laser pulsing the microdisks, cavitation bubbles with a lifetime of a few hundred nanoseconds are generated to puncture a contacting cell membrane (stage 2). When the pore size is reparable, a cell reseals the pore over time and remains viable (back to stage 1); however, when a cell membrane is severely damaged by large cavitation bubbles, it dies immediately. Schematics of equivalent circuit models of stage 1 and stage 2 processes are shown in Figure 1b and Figure 1c , respectively. The electrical impedance near a membrane pore is dominated by the resistance term after laser pulsing, whereas before laser pulsing, it is dominated by the capacitance term because an intact plasma membrane is not electrically conductive and can be considered as a capacitor, as shown in Figure 1b . Therefore, the electrical impedance can be used to monitor the dynamics of the cell membrane opening and resealing processes.
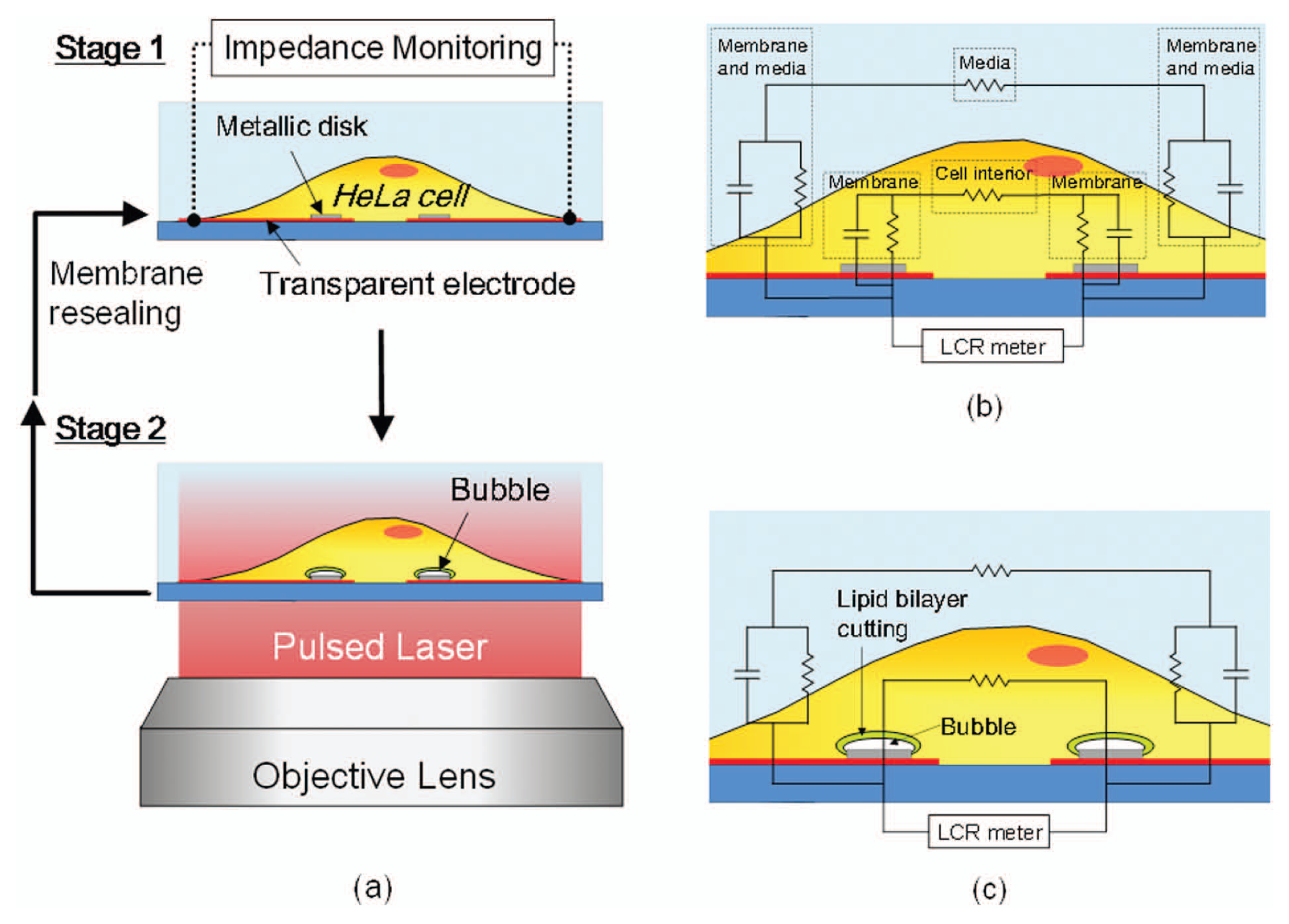
Metallic microdisk-integrated electrical impedance sensor. (
Figure 2a shows the top view of a microfabricated sensor that includes a pair of transparent ITO electrodes and metallic microdisks. The cross-section view of the sensor is schematically shown in Figure 2b . Fabrication of this device starts with a commercial ITO glass substrate. A 30 nm thick ITO coating was patterned by photolithography and dry etching techniques. A 400 nm thick SiO2 coating was deposited on the entire sensing area. A 200 nm thick Ti film was coated and patterned on top of the SiO2 layer to create metallic microdisks with a diameter of 2 µm. These Ti disks were also used as DRIE etching masks to selectively remove the SiO2 and expose the ITO electrodes for impedance monitoring. A second 200 nm thick SiO2 layer was deposited and patterned by a lift-off approach to provide electrical isolation in the area outside the monitored regions to minimize the electrical noise from background. Finally, a polydimethylsiloxane (PDMS) well with a height of 20 mm and an area of 3 cm2 was attached onto the device to build a reservoir for holding cell culture media. Metal wires feeding measured impedance signals to an external LCR meter were soldered on the large contact pads located outside of the PDMS reservoir.
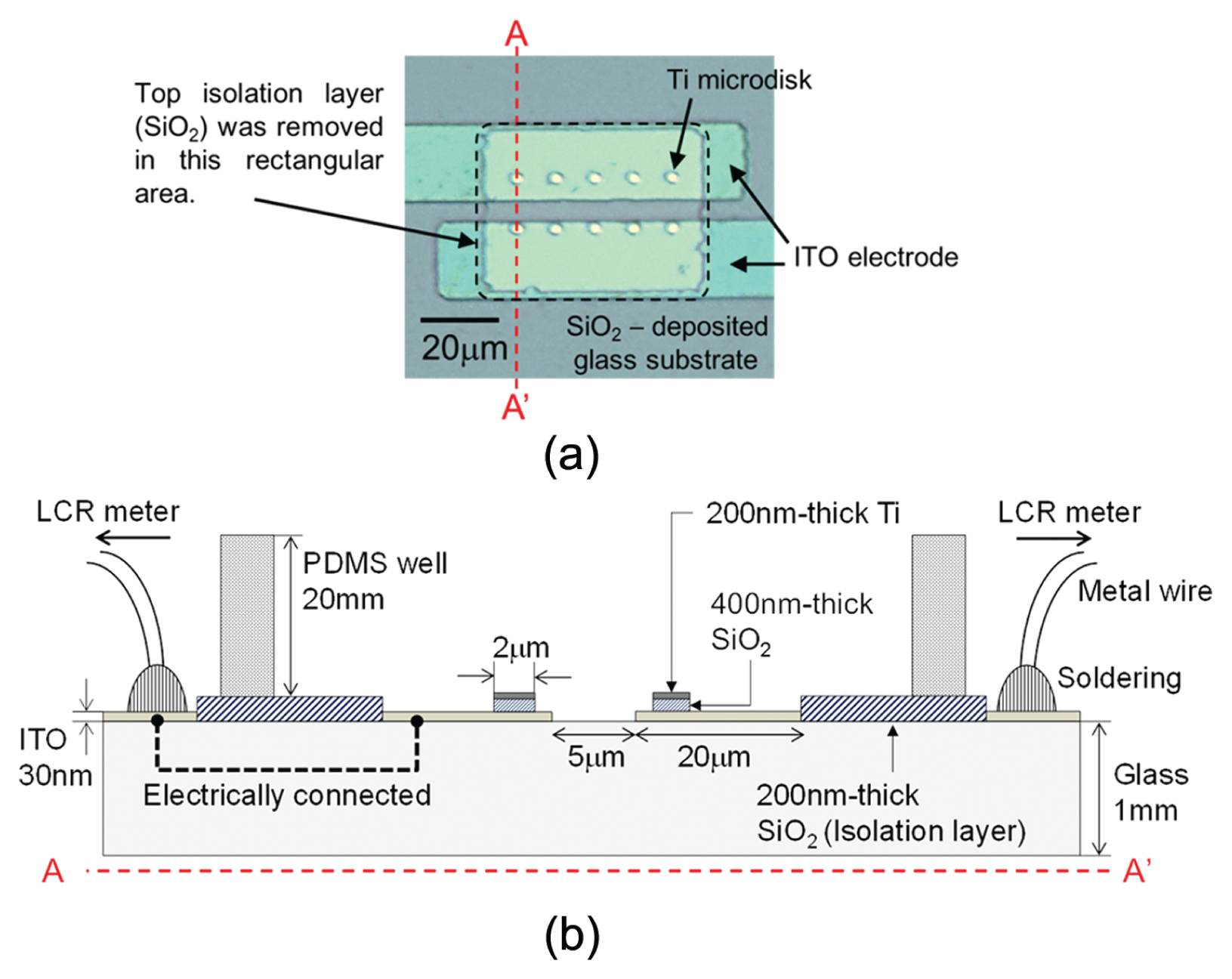
Structure of a microdisk-integrated impedance sensor. (
System Setup
The experimental setup is shown in Figure 3 . A Q-switched, frequency-doubled Nd:YAG laser (Minilite I; Continuum, Inc., Santa Clara, CA) with a wavelength at 532 nm and a 6 ns pulse width is used to trigger cavitation bubbles. The light beam is divided by a beam splitter into two arms, one for laser pulsing metal disks on the sensor and the other for providing illumination light to a custom-built time-resolved imaging system. Laser pulses for triggering cavitation bubbles are guided into an inverted microscope (AxioObserver; Carl Zeiss, Inc., Oberkochen, Germany) and passed through an objective lens (40×) to generate a 260 µm wide laser spot on the sample plane. A linear polarizer is inserted in the laser beam pathway before the inverted microscope to adjust pulse energy. The microdisk-integrated impedance sensor was fixed on the microscope stage at the center of the field of view. To observe the rapidly explosive and transient cavitation bubbles induced by laser pulsing the metal microdisks, a time-resolved imaging system was constructed using an intensified charge coupled device camera (PI-MAX2; Princeton Instruments, Trenton, NJ). This imaging system uses one arm of the laser beam split by a beam splitter as an illumination light source. The laser beam was focused by a convex lens into a fluorescent dye cell (LDS 698; Exciton, Dayton, OH) to generate incoherent fluorescent light pulses (center wavelength is 698 nm) that are collected by a multimode optical fiber and delivered into the microscope condenser to provide a flashlight illumination to the area that will be imaged. The length of the optical fiber controls the delay time, from a few to hundreds of nanoseconds, between laser ignition of cavitation bubbles and the arrival of flashlight. An LCR meter (4284A; Agilent Technologies, Inc., Santa Clara, CA) is used for real-time monitoring of the electrical responses from the impedance sensor with a sensing voltage set at 500 mV and frequency at 500 kHz. The impedance data are recorded by a LabVIEW (National Instruments Corp., Austin, TX) program on a PC connected to the LCR meter via a GPIB interface.
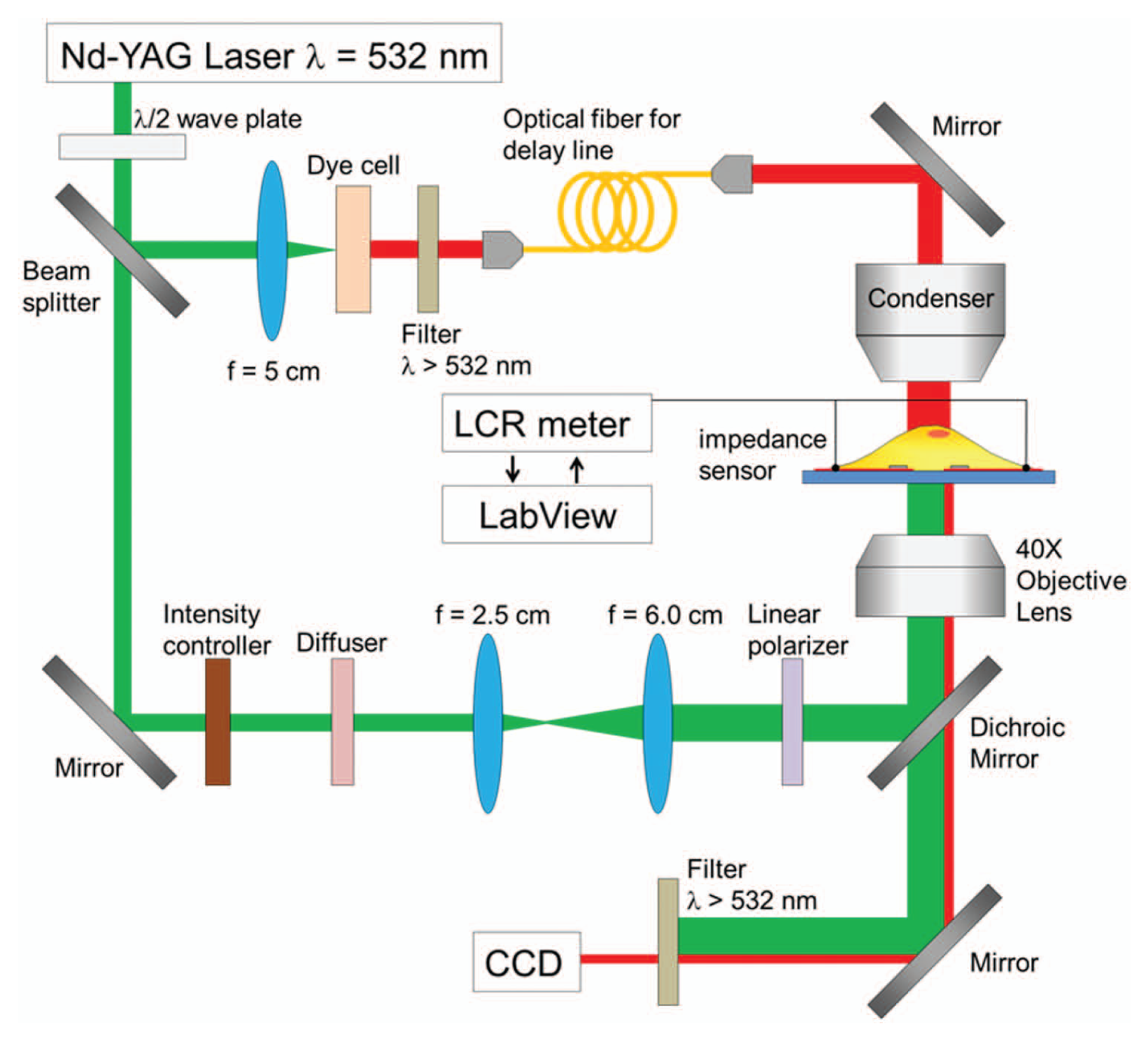
Experimental setup with laser pulsing for triggering cavitation bubbles, time-resolved imaging for monitoring the generation of bubbles, and real-time electrical impedance measuring of photothermal porated single cells.
Cell Culture
HeLa cells were cultured in the developed PDMS reservoir integrated with the ITO electrodes and microdisks using Dulbecco’s modified Eagle’s medium (DMEM) for 2 days beforehand. DMEM was then replaced with phosphate-buffered saline (PBS), pH 7.4, before the experiment of photothermal poration and impedance measurements. The electrical conductivity of PBS solution is 16 mS/cm. For a control experiment without cells, we used PBS as a solution in the PDMS reservoir.
Results and Discussion
Cavitation Bubbles on Ti Microdisks
A major difference between the nanoblade 14 and the current microdisk configuration is the heat dissipation pathways. In the nanoblade, heat from the hot spots diffuses along a micropipette ring, whereas on a microdisk, heat diffuses across the surface from hot spots at the edges to the center of the surface. Cavitation bubbles generated on the Ti microdisks were captured by a time-resolved imaging system, as shown in Figure 4 . Each image was captured at a 20 ns time delay after laser pulsing. The minimum threshold pulse energy required for triggering cavitation bubbles on the microdisks was 28 µJ, equivalent to a fluence of 50 mJ/cm2. No bubbles were observed if the pulse energy was below this threshold.

Cavitation bubble images of a Ti microdisk captured at 20 ns after laser pulsing. (
Impedance Measurement without Cells on Electrodes (Control)
Before measuring the impedance responses of cells on electrodes, a control experiment was performed to measure the impedance without cells on the electrodes. Figure 5 plots the impedance over time with laser pulsing on Ti microdisk-integrated electrodes. Both the magnitude and the angle of the measured impedance indicate that there is no significant change of impedance due to laser pulsing alone. The background signal drift is small. The drift magnitude and the angle of the complex impedance measured over 7.5 min were approximately 0.1% of the average impedance and 0.0003°, respectively.
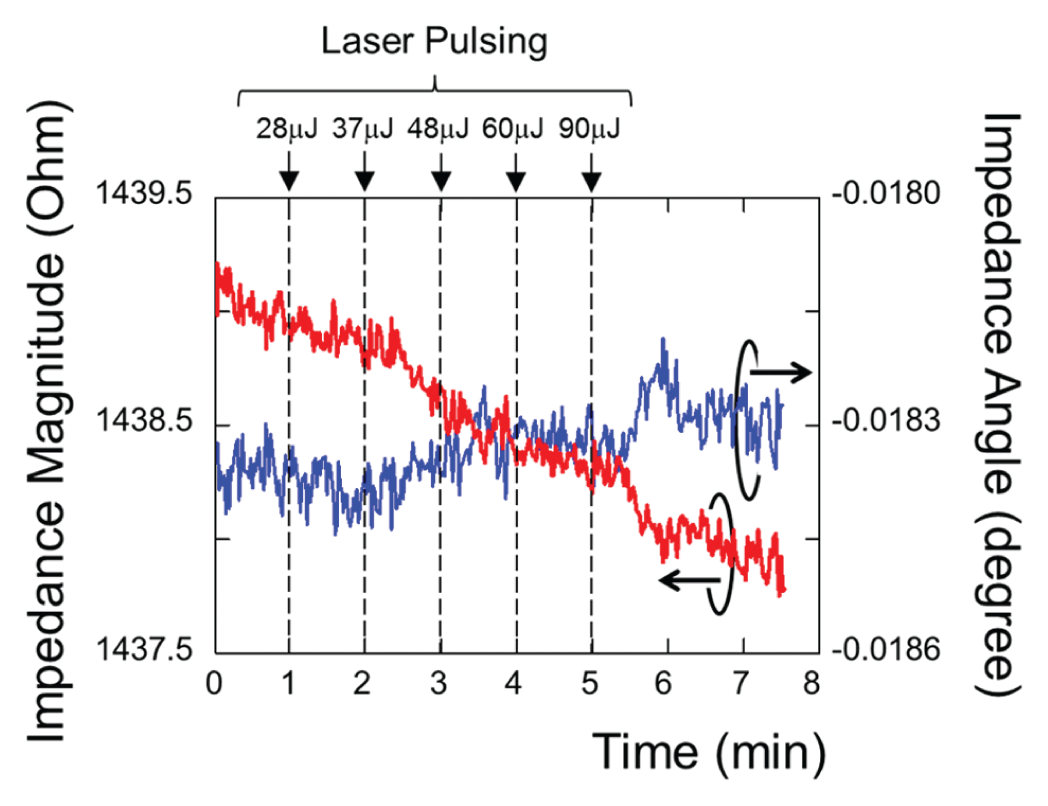
Impedance responses of laser pulsing Ti microdisks on indium tin oxide electrodes without cells.
Impedance Measurement of Severely Damaged Cells
Figure 6 shows the measured dynamic impedance of cells severely damaged by a large cavitation bubble excited by a high-energy (90 µJ) laser pulse. After pulsing, the magnitude and the angle of the impedance were measured over 8 min. The impedance magnitude shifted by 1.26% of the average impedance, 10 times larger than the case without cells on electrodes, and by 0.0017°, respectively. As shown in Figure 7 , these cells were permanently damaged after photothermal poration, and their impedance did not recover 90 min after pulsing. Propidium iodide (PI) fluorescent dye was added to the culture media 90 min after laser pulsing to confirm the death of these cells.
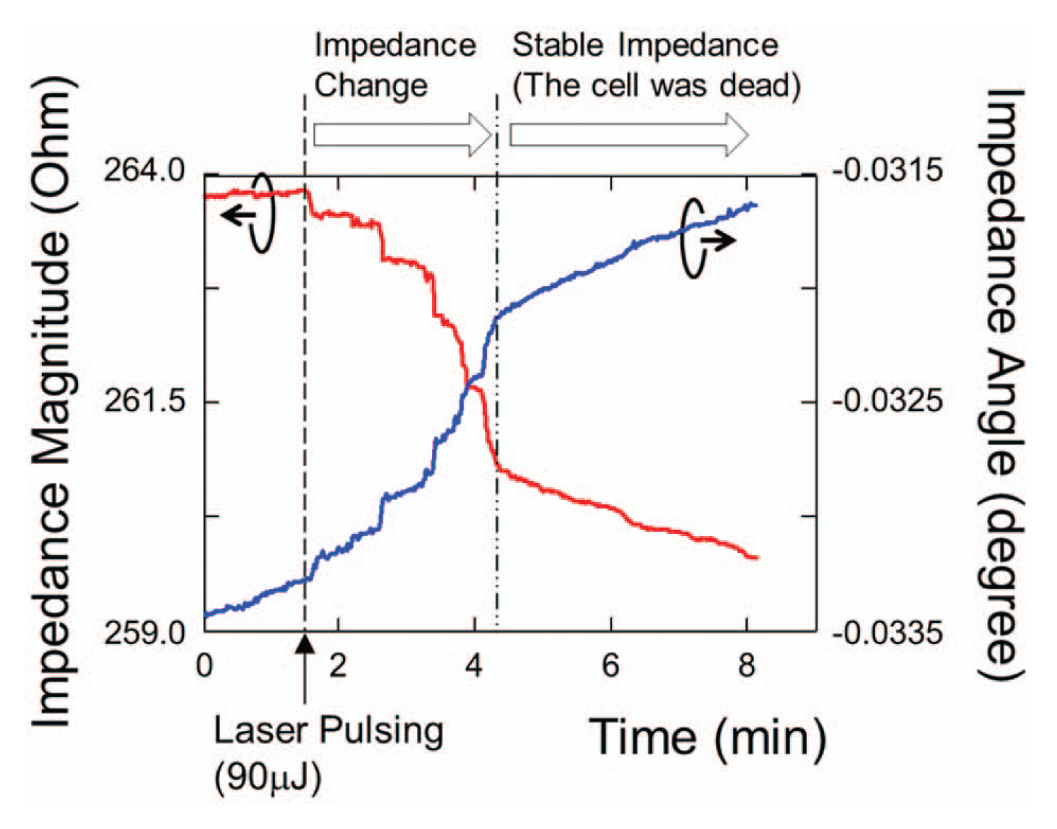
Impedance responses of a severely damaged cells irradiated by a 90 µJ laser pulse. The cells were dead after pulsing, and the impedance value did not recover after 90 min.
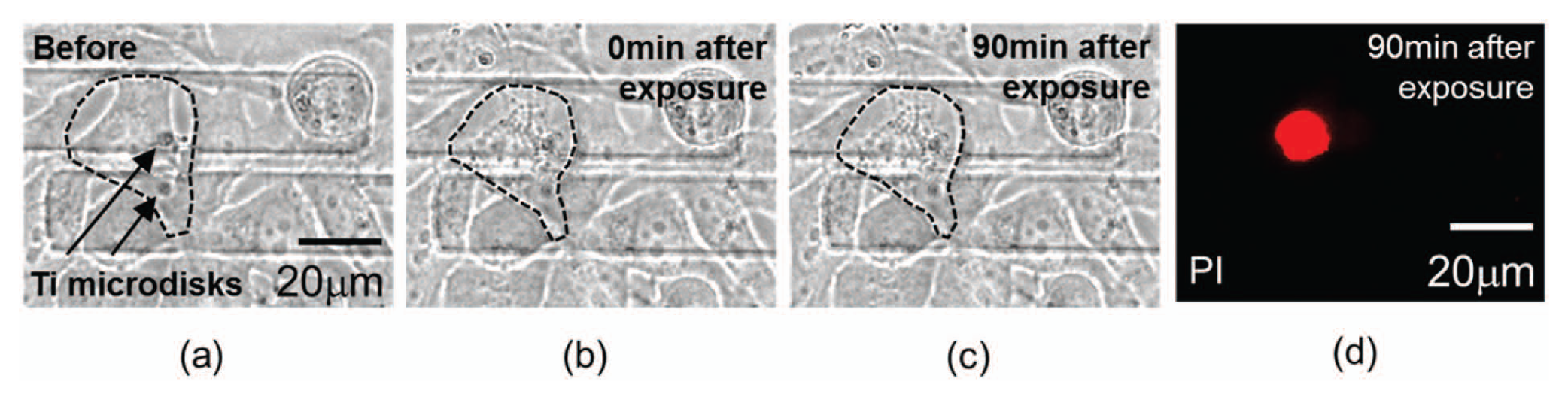
Irreversibly damaged cells caused by large-sized cavitation bubbles. (
Impedance Measurement of Viable Cells after Photothermal Poration
By using laser pulses at appropriate energy levels, photothermal poration with high cell viability can be reproducibly achieved, as shown in our prior work. 11 Figure 8 plots the impedance responses of cells remaining viable after photothermal poration. Both the magnitude and the angle of the impedance showed rapid changes immediately following laser pulsing. The impedance signal recovered in approximately 2 min back to the initial state. Figure 9 shows the cell images before and after laser pulsing under bright field and fluorescence modes. The culture media used in the experiment contained membrane-impermeable calcein green fluorescent dye to indicate successful pore opening on cell membranes. Cells with transient pores showed green fluorescence because the small-molecule dye diffuses across the pores into the cell cytosol before the pores resealed. After laser pulsing, calcein was washed away and replaced with fresh media. Figure 9d shows that cells remained viable and retained the calcein dye for longer than 90 min after laser pulsing. This indicates that the cell recovered fully from photothermal poration as small molecules such as calcein cannot diffuse across the intact cell membrane. If the cells do not fully reseal the membrane pores, the calcein dye will diffuse out to the fresh media, and cells cannot maintain their green fluorescence.
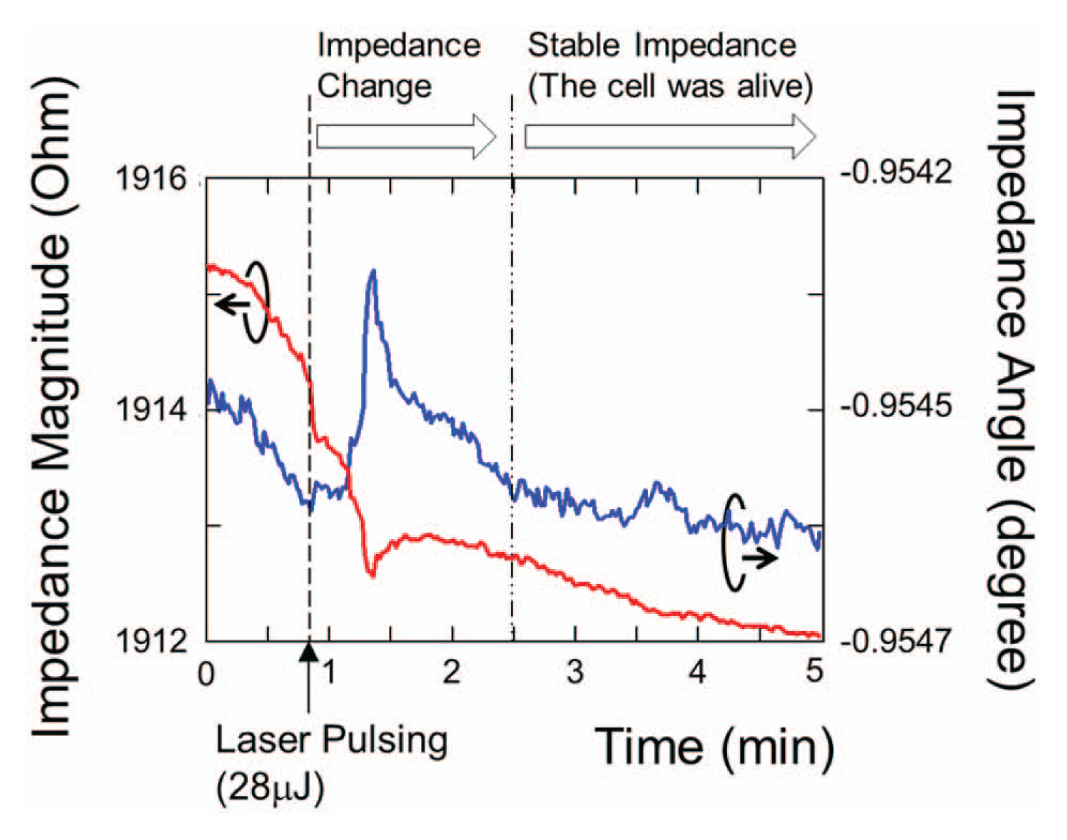
Impedance responses of living cells irradiated by 28 µJ laser pulses. The opened cell membrane resealed in approximately 2 min.
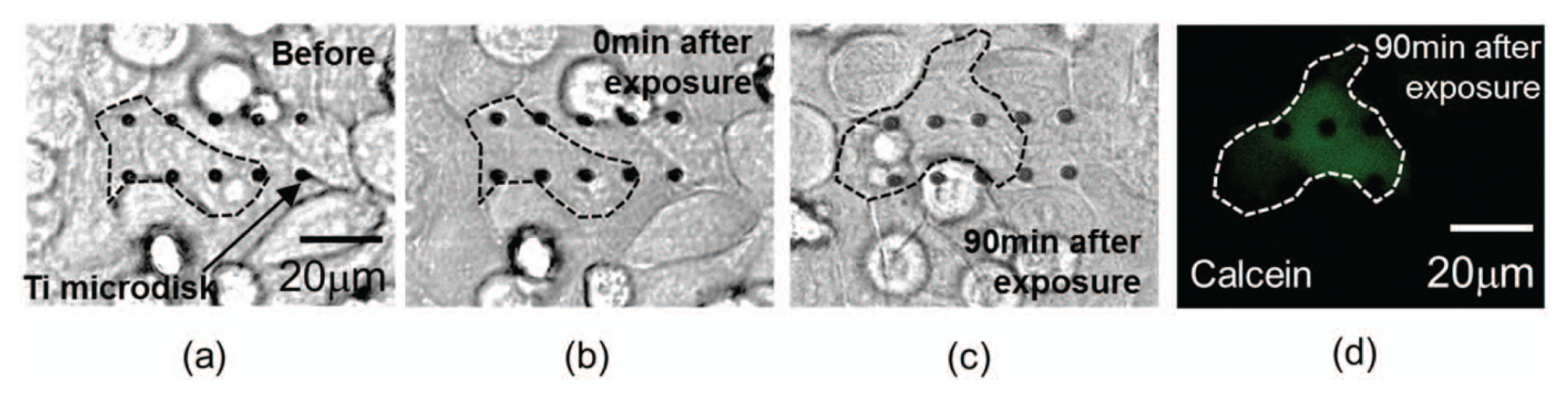
Photothermal porated cells on Ti microdisks and viability analysis. (
Impedance Measurement in Frequency Domain
We have also examined the dynamic impedance responses of cells after laser pulsing using a different approach, in which cells are cultured on thin gold electrodes. Compared with patterned Ti microdisks on ITO electrodes, gold electrodes are not patterned. Under laser pulsing, cavitation bubbles are generated randomly on electrodes. By properly controlling the pulse energy, good cell viability can also be maintained. An advantage of using gold electrodes is the high electrical conductivity of gold, which reduces a potential voltage drop across the measurement electrodes. This is important especially when conducting electrical measurements in physiological buffers that have high electrical conductivity. Using gold electrodes also enables an increased number of cells cultured on the electrode to increase the signal-to-noise ratio in electrical measurements.
In this independent approach, a pair of thin-film (100 nm in thickness) gold electrodes with cells cultured on top is connected to an impedance analyzer (4294A; Agilent Technologies, Inc.), as shown in Figure 10a . A PDMS isolation layer is patterned on the electrodes to minimize the background electrical noise. By exposing the electrodes to a laser pulse, cavitation bubbles were generated on the electrodes in a random fashion. A laser pulse with 40 µJ energy was used to irradiate the gold electrode from the bottom of the device. Cell viability and dye uptake of irradiated cells was monitored through fluorescence imaging ( Figure 10b ). The results indicate that the cell membrane can be successfully punctured to create transient pores while maintaining viability. At the same time, an impedance analyzer was used to monitor the frequency-versus-impedance characteristics of these cells ( Figure 11 ). The graphs plotted the measured impedance magnitudes at the frequency bandwidth from 499.5 kHz to 500.5 kHz. The impedance data were manually captured at different time frames (interval ~15 s) and superimposed. In Figure 11 , the last graph shows the impedance at the final steady state obtained at 75 s after laser exposure, and this impedance value was the same as the initial state in the first graph. The cell membrane resealing time after laser pulsing was thus estimated to be about 1 to 2 min. This membrane resealing time scale, despite random bubble generation, confirms the data measured using an LCR meter, as shown in Figure 8 . For cells that were severely damaged by cavitation bubbles and died, the impedance dropped after laser pulsing and never recovered back to its initial value, and the PI staining confirms the death of these cells, as shown in Figure 12 .
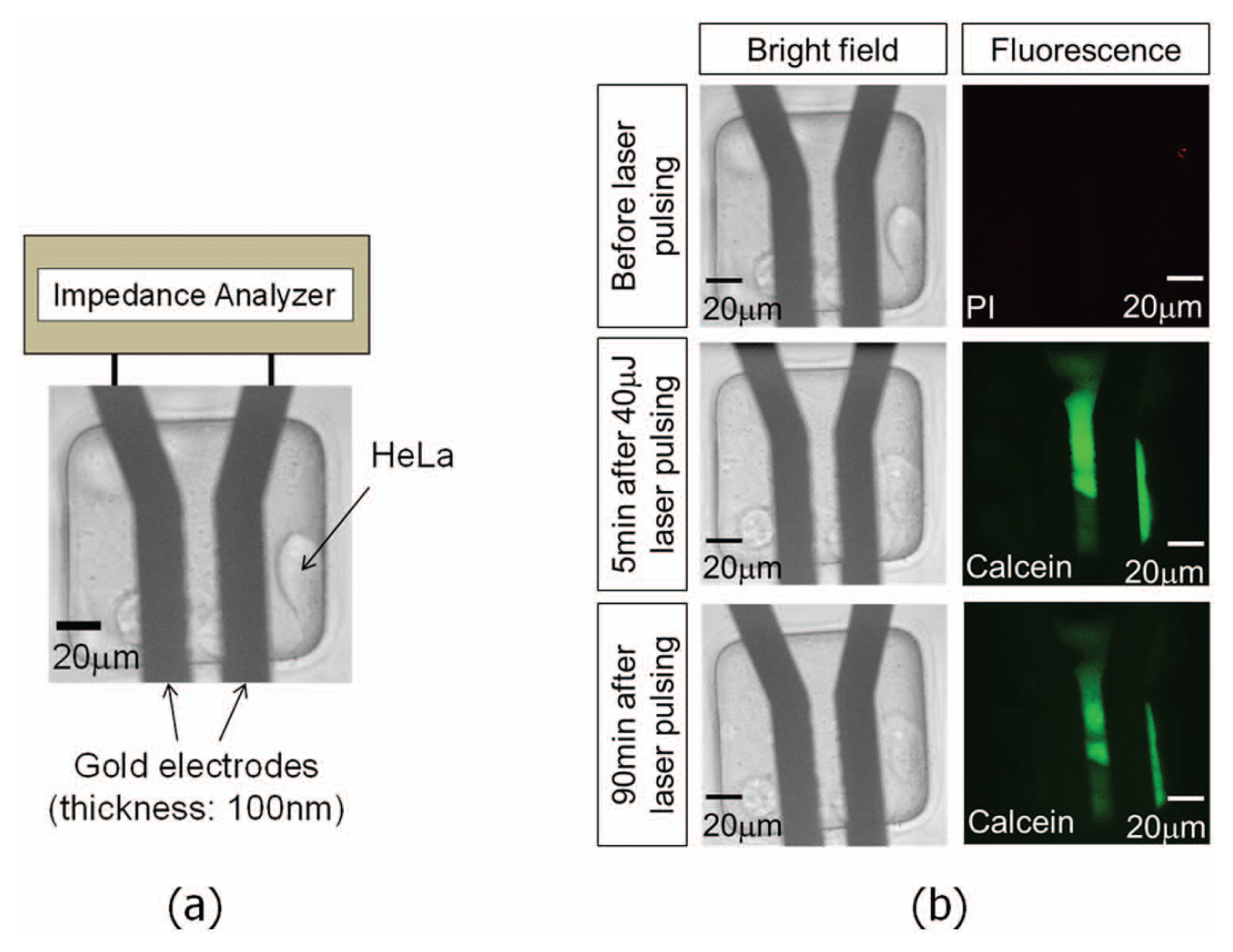
An impedance sensor with thin gold films (100 nm in thickness). Laser pulsing generates cavitation bubbles in a random fashion to permeablize cells cultured on electrodes. (
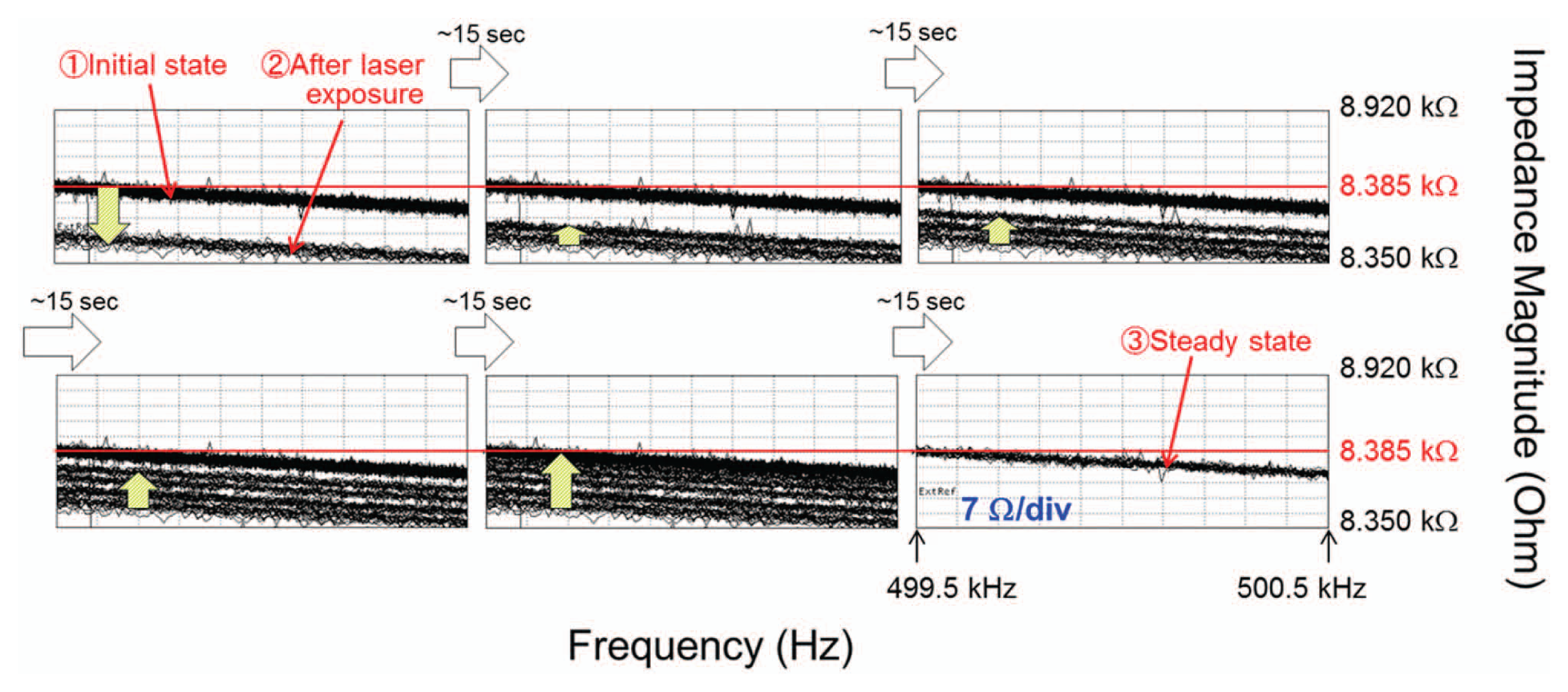
Frequency-versus-impedance data obtained by using the device, as shown in Figure 10 . Each frame was manually captured on the display of an impedance analyzer. The measured impedances at different time points were superposed sequentially. The last graph plots the steady-state impedance values observed at about 75 s after laser irradiation. These data show that cells that remained healthy after laser pulsing can reseal their membranes and that the transient impedance shift goes back to the initial value before laser pulsing.
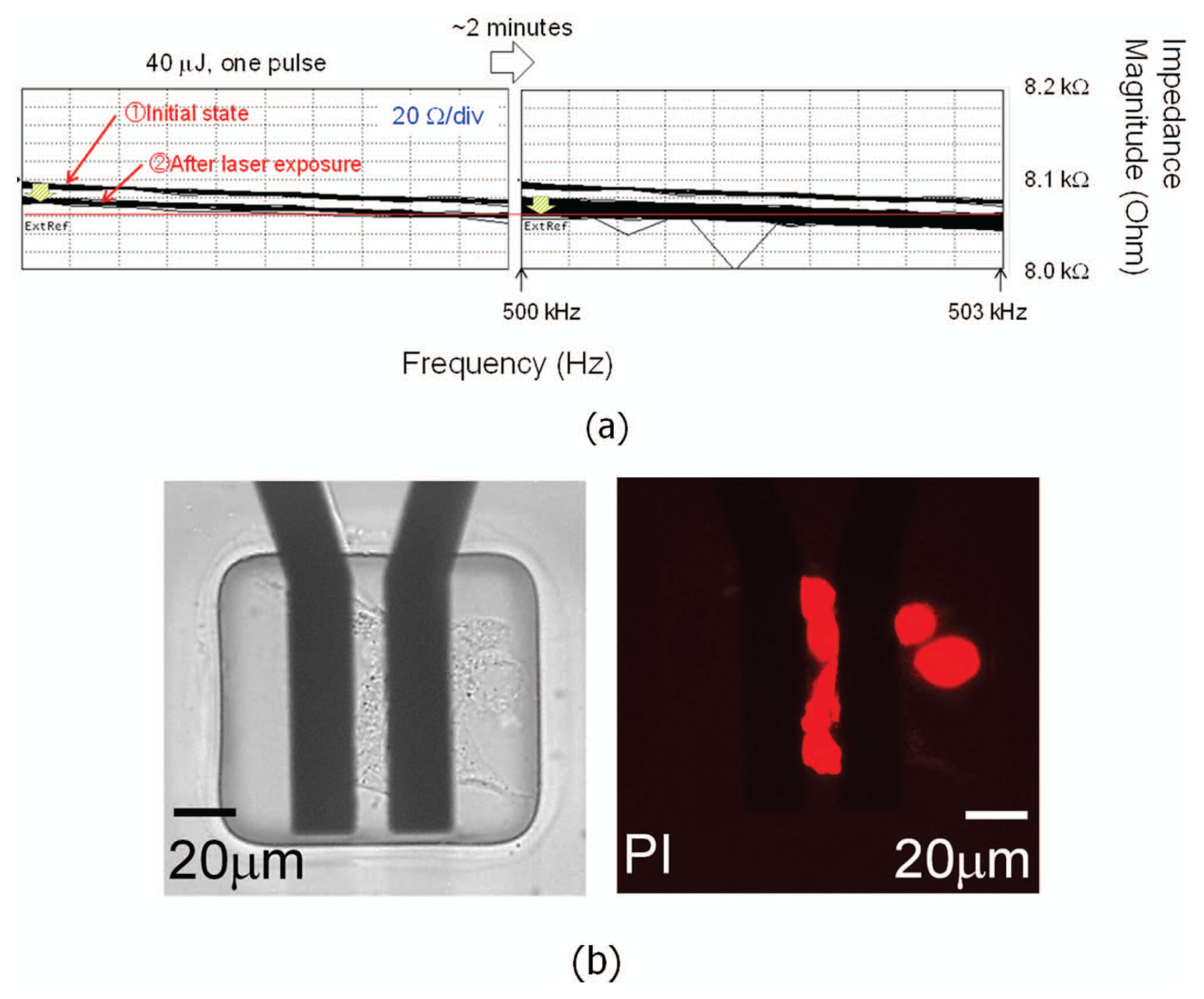
(
Comparison with Resealing Time of Transient Pores Opened by Other Approaches
Other studies have reported cell membrane resealing times over a wide range, from milliseconds to minutes, depending on the mechanism of membrane poration, pore size, cell type, and environmental factors, such as the extracellular Ca2+ concentration. A cell membrane depolarization time of 60 ms was measured when the membrane pore opening was induced by irradiation with high-repetition-rate femtosecond laser pulses for 30 ms. 2 Using a single-shot ultrasound wave, cell membrane repair takes ~5 s for a 1 µm sized pore. 15 After electroporation of multiple cells in suspension, a membrane-resealing time up to 20 min was observed. 17 In the current study, the membrane-resealing time was approximately 2 min after laser pulsing cells cultured on 2 μm titanium microdisks, as quantified by impedance responses.
Conclusion
We have developed a metallic microdisk-integrated electrical impedance sensor to measure the resealing time of photothermal porated cell membranes. The device was realized by patterning thin-film Ti microdisks on transparent ITO electrodes. By controlling the laser pulse energy, we have successfully detected that the membrane recovery time is ~2 min after laser pulsing HeLa cells cultured on Ti microdisks. The obtained time scale was confirmed by comparing frequency-versus-impedance characteristics measured by using thin-film gold electrodes producing cavitation bubbles in a random fashion to create transient membrane pores on multiple cells. We also note that cells irradiated by high-energy laser pulses, generating large cavitation bubbles with excessive shear force, experience rapid death, and the impedance never recovers. This platform is capable of studying real-time membrane-resealing dynamics under well-defined photothermal nano- and microstructure conditions and offers insight on cellular response dynamics due to photothermal poration.
Footnotes
Acknowledgements
The authors thank Dr. Kayvan Niazi and Dr. Shahrooz Rabizadeh (California NanoSystems Institute) for helpful discussions and support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project is supported by the NSF ECCS 0747950, ECCS-1232279, and DBI1256178, a UC Discovery/Abraxis BioScience Biotechnology Award (#178517), the NIH Roadmap for Medical Research Nanomedicine Initiative (PN2EY018228), NIH R21EB014456, the Broad Center for Regenerative Medicine and Stem Cell Research at UCLA, the Prostate Cancer Foundation Challenge Award, and the Japan Society for the Promotion of Science.
