Abstract
The mechanical properties of cells have been shown to be useful markers of cell state by the biophysics community. Here, I highlight clinical and research problems that this label-free, potentially inexpensive cellular biomarker can address and discuss technical challenges to realize automated instruments to achieve robust and high-throughput mechanical measurements. Important features found in traditional fluorescence-based flow cytometry that can enable cytometry based on mechanical properties (i.e., deformability cytometry) are emphasized, especially the need for throughput, simple operation, multidimensional data visualization, and internal controls. Next-generation approaches to automate deformability measurements of cells are surveyed, and future directions are outlined that promise to bring low-cost mechanical measurements to medicine and biological research.
Introduction
The deformability of cells (i.e., compliance under an applied load) is a promising label-free biomarker indicative of underlying cytoskeletal or nuclear changes associated with various disease processes and changes in cell state. Examples include changes in red blood cell stiffness in cytoskeletal disorders such as spherocytosis,1,2 increased cell deformability of invasive cancer cells compared to benign or normal cells of the same origin,3,4 and changes in stiffness of leukocytes in response to activation with antigens or other signals. 5 An increased deformability has also been identified as a potential biomarker for pluripotent stem cells.6,7 In this review, I will discuss several of these areas of potential clinical importance as shown in Figure 1 . I will briefly address advantages of deformability as a label-free biomarker in reducing costs and yielding improved repeatability for classifying cells within these application areas. I will then discuss current techniques that are being used to measure cell mechanical properties, challenges to translate these approaches to clinical tools, and finally highlight next-generation, automated, and high-throughput techniques needed for improved adoption in clinical and research applications.
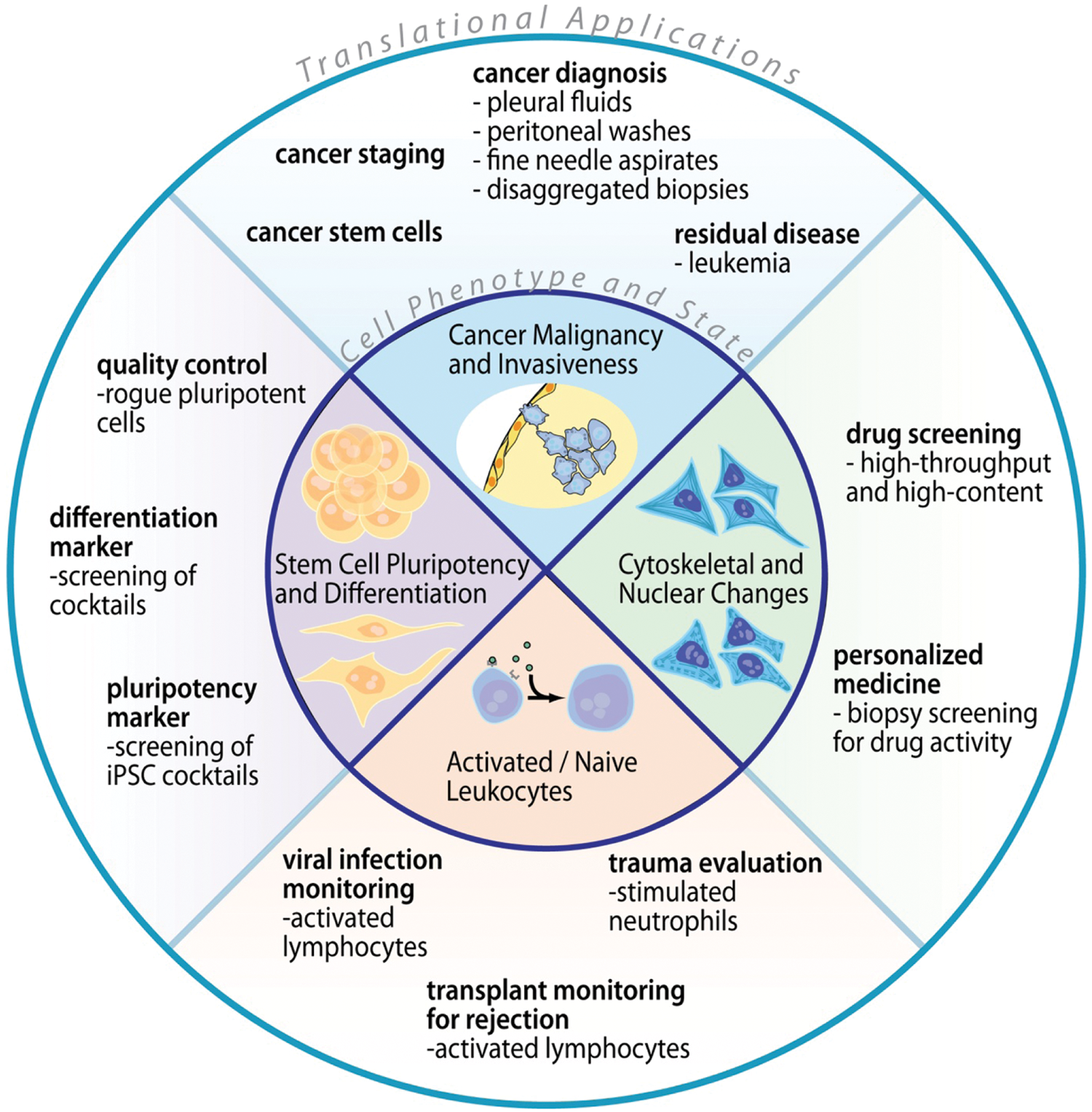
Impact of a mechanical biomarker in medicine. The chart shows changes in cell state that have been associated with cell mechanical changes in the inner circle. Corresponding biomedical applications enabled by measurement of those changes in state are arrayed in the outer circle. iPSC, induced pluripotent stem cells.
Mechanical Changes Accompanying Cell Processes and Anticipated Applications
Cancer malignancy and invasiveness
Cytoskeletal changes have been suggested to underlie mechanical differences observed in invasive cancer cells,3,8 which is consistent with a process of selection for cells that are able to squeeze into vessels (intravasate) and metastasize ( Fig. 1 ). Using optical forces to stretch adherent cells brought into suspension, Guck et al. 9 and Lincoln et al. 10 found significantly higher average stiffness for normal breast epithelial cells (MCF10A) when compared to benign breast carcinoma cells (MCF7). They found further decreases in stiffness for high-motility mod-MCF7 cells considered to have a malignant phenotype. Demonstrating the universality of these results, similar data have been obtained using atomic force microscopy (AFM). 11 Importantly, work by Remmerbach et al. 12 and Cross et al. 13 has extended these observations to primary cancer cells from human patients. Remmerbach et al. 12 observed that oral cancer cells from patients with histologically confirmed oral cancer were on average 3.5 times more deformable than normal oral epithelial cells obtained from healthy patients. In a separate study, Cross et al. 13 observed an approximately 4 times increase in cell deformability as measured by AFM for disseminated breast and lung cancer cells when compared to normal cells in pleural effusions. Furthermore, a link between increased cancer cell deformability and invasiveness, as measured by Matrigel invasion assays, was found by Swaminathan et al. 14 for patient-derived ovarian cancer cells. Taken together, these studies suggest the usefulness of a mechanical biomarker in diagnosing disease state in a label-free manner that is independent of current immunohistological methods.
Activated and naïve leukocytes
Previous work in the biophysical community has focused on the mechanical properties of neutrophils and monocytes and how these properties may change upon stimulation with chemokines associated with infection or stress.15–20 These previous studies have suggested that neutrophils and monocytes become stiffer upon stimulation and that this could be physiologically important in sepsis or trauma patients, in which these cells are observed to accumulate in narrow capillaries in the lungs. Conceivably, a measure of stiffness could also be used as a proxy for the level of stimulation of these cells and aid clinicians in evaluating recovery from conditions such as sepsis and posttraumatic stress. It should be noted that many of these studies used a technique that measures cell transit through pores under pressure to assay stiffness, in which cell size or surface adhesion may also play a role in the results obtained.
There is limited previous work examining how the mechanical properties of the subset of leukocytes involved in adaptive immunity—lymphocytes—change upon activation, although this is also diagnostically important. Contrary to increases in stiffness for stimulated neutrophils, Brown et al. 21 have suggested that a decrease in stiffness would be necessary for transmigration of lymphocytes through the vasculature, lymph nodes, and tissue to infection sites following activation, and this may be partly explained through changes in the localization of the intermediate filament vimentin. Preliminary results from my group support the notion that lymphocytes become softer upon activation (data not shown). There are significant changes in gene expression when lymphocytes are activated as well as structural epigenetic changes in chromatin distribution and the nuclear to cytoplasmic ratio 22 that also likely lead to differences in whole-cell mechanical properties. Activation of various lymphocyte subsets is associated with viral infection, autoimmune disorders, and transplant complications such that assaying activation may be an important clinical tool to diagnose these disease states or evaluate response treatment.23–25
Stem cell pluripotency and differentiation
Mouse and human embryonic stem cells (ESCs) and nuclei were found to be more deformable than cells and nuclei of their differentiated progeny using atomic force microscopy and micropipette aspiration, suggesting deformability may be a viable biomarker for pluripotency.6,7 Pajerowski et al. 6 found nuclei of human ESCs to be 6 times stiffer after being terminally differentiated, whereas Chowdhury et al. 7 found differentiated mouse ESCs to be 10 times stiffer than their undifferentiated counterparts. It remains to be seen what molecular changes are responsible for these differences in whole-cell mechanical stiffness upon differentiation; however, as discussed for activated leukocytes, the nuclear architecture and epigenetic state has been implicated. Pajerowski et al. 6 found that nuclear lamins A and C (intermediate filaments absent from pluripotent cells) contributed to measured increases in nuclear stiffness that occurred as cells matured, whereas the stiffer rheological character of the nucleus was dependent on tighter chromatin packing. In the process of differentiation, cells gain regions of condensed heterochromatin with which nucleoproteins are more closely associated, but these proteins are dynamically associated with a loose chromatin structure in undifferentiated cells. 26
Embryonic stem cells and induced pluripotent stem cells (iPSCs) are currently being explored for use in cell-based regenerative medicine. However, major translational hurdles exist involving populations of ESC- and iPSC-derived cells that do not fully differentiate pretransplantation and are tumorigenic in vivo,27,28 as well as ESCs that experience cytogenetic changes, similar to those that are responsible for cancer, when propagated in vitro.29–31 In fact, the standard measure of pluripotency is the ability of these cells to develop into teratomas when implanted in immune-deficient mice. Because of this risk, an inexpensive tool for quality control screening for “failed-to-differentiate” cells will be critical. Besides the potential use in characterizing differentiation state for clinical quality control, a label-free biomarker would facilitate high-throughput screens of conditions for differentiation and reprogramming.
Cytoskeletal and nuclear changes as an indicator of drug response
Single-cell mechanical measurements have the potential to aid a high-throughput but inexpensive approach to personalized medicine. Most tumors consist of a heterogeneous population of cells that will respond to anticancer drugs in a nonhomogeneous manner. Many drugs, such as paclitaxel, act by modifying cytoskeletal function, specifically microtubule dynamics, in actively dividing cells.32–35 Other anticancer drugs act by disrupting DNA synthesis and cell division mechanisms (e.g., doxorubicin, aurora-kinase inhibitors). Treated cells, ultimately, cannot divide successfully, leading to initiation of apoptosis and cell death. Some of these drugs also are expected to modify the viscoelastic properties of the cells due to their action on the putative “compressive” struts of the cytoskeleton (microtubules). 36 Similarly, dramatic intracellular changes accompanying an apoptotic pathway may also be mechanically distinct. Cells that are sensitive to these anticancer drugs are expected to display discernible differences in mechanical properties before and after treatment, whereas cells that are resistant to these drugs would have little variation in cell mechanical properties pre- and posttreatment. Resistance may arise through drug efflux pumps (e.g., P-glycoprotein, Pgp), intracellular degradation enzymes, or various mutations in drug binding sites. 32 Notably, all of these mechanisms of resistance are likely to result in a similar maintenance in the pretreatment mechanical properties. Genetic screening for these resistance mutations requires a priori knowledge of potential mutations. Furthermore, performing a mechanical measurement on single cells would also reveal heterogeneity within a population and aid in identification of rare resistant cells that can be masked when conducting genetic screening methods that act on bulk lysed populations. A label-free mechanical screen could also be conducted quicker, without having to wait days for signs of cell death in a viability screen while preserving a quantifiable, comparable measure. Thus, a simple technique for measuring viscoelastic properties of hundreds of thousands of cells may have clinical implications in screening cells from cancer biopsies for resistance and sensitivity to available chemotherapeutics in a new approach for personalized medicine. 37
Benefits of a Label-Free Mechanical Approach
Typical sample preparation procedures for classifying cellular samples by molecular markers using histology dyes or immunolabeling require multiple centrifugation steps for cell labeling and washing, which can be a time-consuming (>30 min), laborious, and costly process for diagnostics and research. A simple label-free assay in which cells in suspension (e.g., leukocytes from blood, or disseminated cancer cells from urine or pleural or peritoneal effusions) are directly assayed would have reduced costs associated with labeling reagents as well as reduced lab technician time and expense required to prepare samples. Reduced assay time and complexity may also allow for the test to be conducted in a format amenable to the point of care (e.g., Clinical Laboratory Improvement Amendments [CLIA] waived in the United States), with clear advantages in providing treatments earlier for time-sensitive conditions such as acute rejection of a transplant. A final advantage is a potential increase in the repeatability of the assay across clinical labs because it would be less dependent on manual sample preparation steps. In summary, a label-free mechanical measurement of cell state has the potential to help address the high cost of health care by reducing the labor burden of diagnostics and improve quality of life by increasing our diagnostic capabilities—leading to the identification of the correct treatment quickly.
Current Approaches to Measure Single-Cell Mechanics
Because of the increased interest in cell mechanics and its relation to disease, a range of approaches have been applied to measure the viscoelastic properties of individual cells.38,39 These approaches originate in diverse fields and include rheological measurements of storage and loss moduli (G′ and G″) as a function of frequency, as well as measurements of effective elasticity (Young’s modulus, E) and viscosity (µ) using AFM, micropipette aspiration, and optical stretching. Cumulatively, these approaches have yielded data on the mechanical properties of a variety of cell types. Nevertheless, the varied approaches, models, and limited sample size of data for each experiment have led to a lack of a consensus in cell mechanical properties, even for identical cell types. 38 The biological heterogeneity within a cell population and inherent variation present with point measurements on differently shaped adherent cells both are expected to contribute to this lack of consensus.8,40,41 Increased cell throughput is one approach to address the significant heterogeneity and is reported along with other key characteristics for the following techniques ( Fig. 2 ).
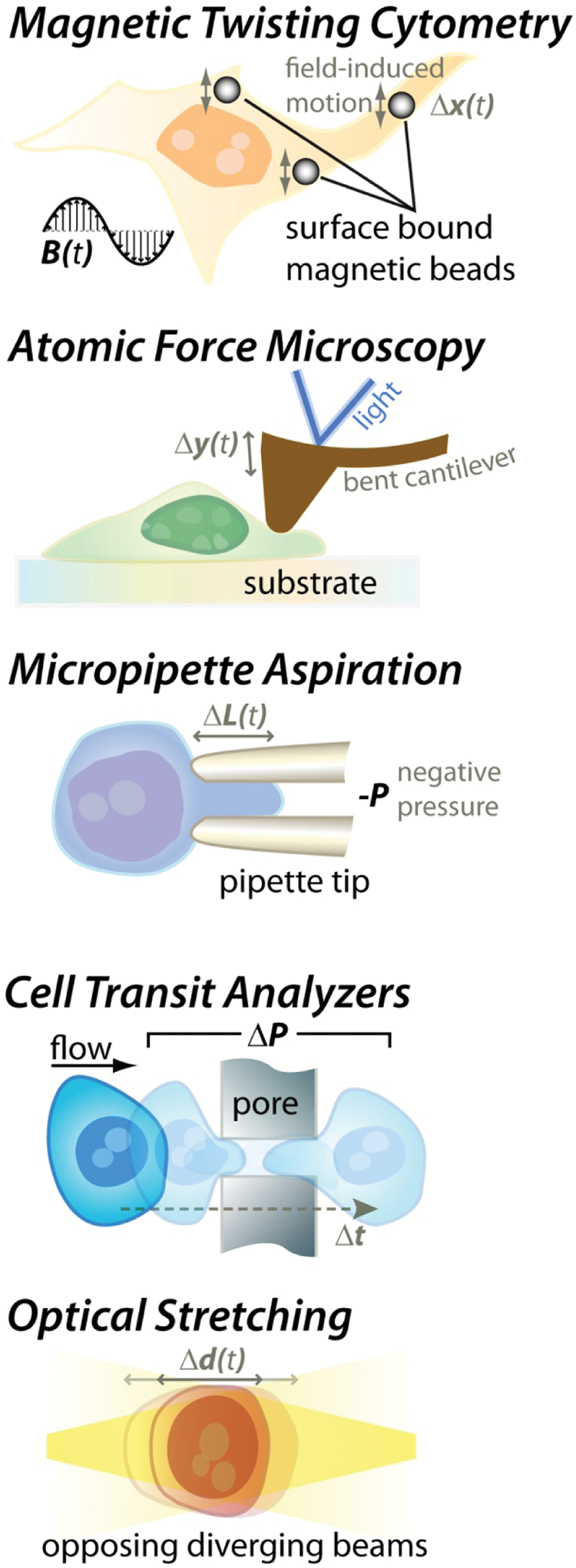
Techniques to measure the mechanical properties of cells. In magnetic twisting cytometry, the rocking motion (Δ
Cell rheology techniques for adherent cells
There are several classes of cell rheology techniques. Several approaches rely on observing the time-dependent fluctuations in internalized or surface-attached micro/nanoparticle positions. 38 These passive techniques convert this time domain information into mechanical response of the local cell material in the frequency domain, obtaining viscoelastic information (storage and loss moduli). Active techniques include magnetic twisting rheology, in which the response of a cell-attached magnetic bead is probed in response to a sinusoidally varying external magnetic field, again yielding elastic (in-phase) and viscous (out-of-phase) responses. 42 Overall, these techniques allow for local probing of the cell environment. However, this can be a disadvantage as the position of the probe can yield different mechanical properties that are difficult to standardize even among a single cell type. 8 Furthermore, these techniques require microscopic observation at high magnification for a period of time, such that throughput is low (~1 cell/min).
AFM and micropipette aspiration
Atomic force microscopy techniques rely on the deflection of a cantilever indented into a particular location of a cell 43 and usually are applied to measurements on adherent cells, where the position of indentation has a large impact on the value of the mechanical measurement. In contrast, micropipette measurements are usually applied to nonadherent cells, in which the location of applied force is less sensitive due to the largely spherical symmetry of cells in suspension. This has been widely used to study the mechanical properties of various leukocytes. 44 In the most common implementation of these techniques, the cell is aspirated into the micropipette with a known force, and the time-dependent extension into the pipette is recorded. Although precise, these techniques require skilled manual operation and are very low throughput (<1 cell/10 min).
Optical stretcher
More recently, a technique has been introduced that stretches cells with optical gradient forces.8,9,12 Time-dependent deformation is measured to obtain viscoelastic properties. This approach has been integrated with microfluidic delivery and operates on spherically symmetric cells in suspension—a significant advantage to obtain standardized measurements. The throughput of this approach remains ~1 cell/min and cannot be improved significantly because of the trade-off between higher optical force and the increased optical power required, leading to significant heating of the measured cells.
Cell transit analyzers
The cell transit analyzer and similar microfluidic implementations15,45–47 operate by evaluating the transit time as cells pass through microchannels, single or multiple pores. Measurement of the transit time can be obtained from electrical resistance changes or optical observations. An important advantage of these techniques is the ability to achieve a higher throughput than other single-cell measurement approaches (up to ~1 cell/s). However, changes in cell size or adhesiveness must be taken into account and decoupled from deformability effects (i.e., stiffer, larger, and stickier cells are all expected to have longer retention times). In addition, small pores are prone to clogging, and creating arrays of pores leads to issues with the simultaneous arrival of multiple cells leading to difficulties in interpreting overlapping resistive pulses. This issue necessitates lower dilutions of cells and leads to lower throughput.
Challenges for Clinical and Research Use
Several challenges remain to develop an automated instrument that can bring mechanical measurements to the clinic or widespread biological research. Current techniques have provided a strong biophysical foundation for the cytoskeletal, nuclear, and cortical underpinnings of cell mechanical properties and opened significant avenues for further exploration. It is clear that these tools, developed for basic biophysics research on tens of cells, are not expected to be directly translatable to diagnostic instruments that require limited manual intervention or operator expertise.
An ideal “deformability cytometer” will have similar characteristics to traditional fluorescence-based flow cytometers, which have been successfully applied commercially to clinical and research problems.
Throughput
Automation is critical to achieve a throughput similar to flow cytometers (>1000 cells/s). For most applications, a heterogeneous population of cells will be analyzed in which the cells of interest are previously unknown and in a background of other cells. A high throughput allows for effective sampling of a minority population that may be important clinically. An example where throughput is relevant is identifying malignant cells in pleural fluid—fluid in the cavity between the lungs and chest. In this case, cells of potential interest are usually in a large background of leukocytes and erythrocytes that may have varied sizes and physical characteristics (e.g., in cases where chronic or acute inflammation leads to fluid accumulation). By sampling only 10 to 100 cells with current approaches, low-abundance cancer cells may not even be selected for analysis, potentially leading to an incorrect negative diagnosis. As discussed above, throughput is a significant issue with current technologies such that the majority of studies to date have focused on pure populations of cells that were already isolated or required preselection through other means.
Ease of use
Similar to flow cytometry, operating on minimally handled samples without complex protocols can enable widespread use. Ideally, only two steps are required: The sample is collected and directly input into the instrument. The instrument should be fully automated following this point—not requiring user intervention to identify cells of interest, calibrate the system, align optics, and so on. For some applications (e.g., analysis of stem cell pluripotency or solid tumor biopsies), additional sample preparation steps will be required prior to analysis to bring adherent cells into suspension or disaggregate tissue. In these cases, similar to flow cytometry—in which surface marker fluorescence can change with trypsinization time—care should be taken to apply uniform conditions and times for suspension and disaggregation to avoid deformability changes. 48 Also of concern is the effect of sample freezing or fixation on deformability measurements. These treatments are often done to store molecularly labeled cellular samples. Further work should be conducted to evaluate whether these treatments should be avoided or are viable approaches that act uniformly to maintain mechanical properties over all cells—since this would allow storage or shipment of samples.
Data visualization and management
The simple, intuitive scatter plots and gating approaches used ubiquitously for flow cytometry should be a model for how deformability data are presented and analyzed. Mechanical measurements often yield several independent forms of data, including elastic properties but also time-dependent viscous information and cell size. Two-dimensional scatter plots based on a combination of parameters for each individual cell can provide improved intuition and understanding of data, as well as uncover heterogeneous populations for research and clinical applications. Unlike for basic biophysics studies, real physical parameters such as Young’s modulus, or viscosity, are not required for most clinical or research applications where relative changes suffice, as long as measurements can be repeated. Similar to fluorescence intensity measurements in flow cytometry, mechanical measurements can use normalized arbitrary units—which can reduce data analysis times—and still be useful. Also, using the standardized data visualization and file structure developed for flow cytometry can allow use of the myriad of previously developed tools and software to analyze data, including multidimensional gating methods where percentage of cells within an N-D space of interest is identified. An example in which multidimensional data analysis could be of use is in characterizing the mechanism of action of a drug—it may be possible to characterize drugs acting directly or indirectly on different cytoskeletal or nuclear structural elements with predominantly elastic or viscous behavior through variation in whole-cell elastic versus viscous properties.
Internal controls
Beads with a highly controlled number of fluorescent molecules are often used in flow cytometry systems to calibrate measurements across instruments with different lasers, detectors, and/or optical loss. To advance as a clinically useful tool, instruments measuring cell mechanics can take a similar approach to calibration—by first measuring a population of uniformly manufactured cell-like particles/droplets or a set of these particles with different mechanical properties. This method of calibration is advantageous in that abnormal results with these known particles can then flag the system as malfunctioning rather than making incorrect predictions with potentially unacceptable consequences in a clinical situation. This self-contained approach is important for broad use of an instrument because the technical expert will not be present to identify errors as in the research environment.
Next-Generation Tools for Single-Cell Mechanical Measurements
To address these challenges, there has been a flurry of recent activity in academic research labs to develop new approaches to assay single-cell mechanics. Many of these new approaches take advantage of cell deformation in microfluidic flows where large shear rates are possible. Alternative approaches attempt to parallelize and automate traditional mechanical measurement tools. A few approaches also attempt to measure changes in cell size after osmotic shock (e.g., due to membrane poration). I will briefly discuss these approaches and the promise to address the challenges above.
Mechanical measurements in microflows
Several approaches are being developed that move beyond measurements of transit time through microchannels/structures, where position in a microchannel reports information about cell mechanical properties. One approach takes advantage of the well-known cell margination effect49,50 in which stiffer white blood cells are known to migrate to the periphery of a blood vessel or microchannel due to interparticle interactions. Hou et al. 51 show that this effect allows for separation or measurement of stiffer malaria-infected red blood cells (RBCs), which localize to streamlines near the channel wall, from healthy RBCs. This approach proved useful for enriching the infected RBC population with yields of >75% and appreciable throughput due to continuous flow. Challenges remain to implement this approach as a more general deformability measurement tool because the position of the stiffer cells of interest overlaps with that of healthy cells. Therefore, without further labeling, identification of a population of interest becomes inaccurate.
My research group has developed an approach in which position within the cross section of a straight rectangular channel indicates a combination of size and deformability for flowing cells in dilute solutions. 52 This approach takes advantage of a balance between fluid dynamic lift forces in a simple high-aspect ratio channel. Inertial lift forces 53 and deformability-induced lift forces, which are dependent on the cells’ deformed shape,54,55 balance at a dynamic equilibrium position within the microchannel cross section. We showed that less viscous oil droplets migrated to a focusing position closer to the channel centerline, and more invasive (and deformable) cancer cells also followed this trend. The readout of this approach is through high-speed imaging of cells in the flow, yielding both cell position and size information after automated image analysis. These data are necessarily collected at high throughput (>100 cells/s) since inertial lift forces are necessary for reaching a dynamic equilibrium, and 2D scatter plots can be assembled with these two parameters. Disadvantages of this approach include that size affects focusing position to a large extent and may confound a potential measurement (e.g., a large, stiff cell may have a similar position as a small, deformable cell). Furthermore, cells are deformed only a small amount in the current implementations of this method, which may lead to insensitivity to many important mechanical properties—such as chromatin compactness—that are only visible at large strain.
Beech et al. 56 and Quek et al. 57 have also shown that position in a deterministic lateral displacement (DLD) filtration system is deformability dependent. Briefly, DLD is a steric method of continuous filtration in which particle size—and now deformability—is shown to affect the flow path of particles in arrays of obstacles with gaps larger than the particle size. 58 Unlike previous filtration-based approaches, transit time is not measured, and cells do not squeeze through mechanical gaps prone to measure cell adhesiveness as well—which are significant advantages. Although further results are becoming available, it appears that cells become deformed, change shape, and are deflected or not based on this deformed shape. This can be a continuous-flow and high-throughput technique, where strain can be tuned by flow rate, leading to useful multidimensional data sets that can be visualized in 2D scatter plots of exit path distribution versus flow rate. Like the other fluid dynamic techniques, size and deformability can both affect these distributions such that obtaining mechanical information independently remains a challenge. In addition, it is unclear what level of strain would be achievable using these approaches since as flow increases, further inertial effects will become important and likely change the flow path behavior.
To access measurements of very large strains, we have also recently introduced a separate fluid dynamic approach in which cells are sequentially stretched upon reaching a microfluidic junction.59,60 Cells are initially inertially focused 61 to a narrow set of streamlines to achieve more uniform pressure fields upon reaching the junction. The junction consists of two channel flows with average velocities >1 m/s meeting head on. This sets up an extensional flow region62,63 where cells rapidly decelerate and are stretched as fluid around the cell accelerates in the perpendicular direction. Cells are imaged through this process and upon leaving the junction using high-speed microscopy. Importantly, greater than 1000 cells/s can be deformed and imaged in this manner—approaching throughputs of traditional flow cytometers. Images are then analyzed to extract cell size and deformation parameters. Large strains (the long over short axis of the cell) greater than 3 can be achieved, and multidimensional scatter plots have been demonstrated with unique characteristics for separate cell types and clinical states. As in the DLD approach, multiple levels of strains are achievable by tuning the flow rate of the device. Using oil droplets with known viscosity to calibrate our system, we also determined that there was a trend for increasing deformability with droplet size, such that for this approach as well, size should be measured independently to decouple its effect from deformability.
Parallel contact-based measurements
Another approach to increase throughput and ease of use for clinically relevant single-cell mechanical measurements simply involves parallelizing techniques traditionally used for biophysical studies (i.e., AFM, magnetic bead rheology, and micropipette aspiration). For AFM, one parallelization approach 64 employs arrays of cantilevers and interferometric signals reporting the height of each cantilever imaged with a CMOS camera. Simultaneous force measurements from multiple fields/cells can be achieved in this way. Still, standardization of probing position will be important to obtain more repeatable measurements with small probe tips. Larger flat probe tips that apply force over the entire cell area may address this issue. In general, sample delivery and registration can be more complex than with microflow-based approaches since cells need to be held in defined positions over an array to optimize readout. Magnetic bead rheology approaches can also be parallelized, 14 but compared to passive approaches, a “labeling” step—attaching beads to cells—is still required, reducing ease of use. Data collection following magnetic actuation over an array of cells is still limited by the slow response to the applied low-magnitude forces. Parallel mechanical measurement with micropipette aspiration is also possible using microfluidic approaches,65,66 in which many parallel microchannels could be used to aspirate multiple cells with the same pressure differential. An advantage of this approach (as well as the other approaches in this section) is that mathematical analysis and extraction of physical parameters has already been established in the biophysical community for single-probe versions and should be transferrable. Like the other static approaches, it will still be important to address the limited throughput for cells imaged in one field of view, and refreshing the system with new cells leads to similar disadvantages as described for cell transit analyzers. Importantly, for all of these techniques, multiparameter data sets can be obtained by extracting time-dependent responses (or adding electrophysiological data 66 ), but this increased information content comes at the cost of decreased throughput.
Deformation after electrical or electrochemical stress
Oscillating electric fields are known to cause Maxwell stresses within a cell and accompanying deformation. 67 There has been recent work in using this electrodeformation effect with microelectrode systems to analyze single-cell mechanical properties.68–70 Measurements are currently somewhat low throughput given that cells must be positioned precisely in the vicinity of electrodes and analyzed with microscopic imaging. However, parallelization may be possible considering wide field-of-view imaging approaches that are becoming available. 71 Automated cell positioning is also potentially feasible using parallel microfluidic delivery. 72
Recently, another approach to measure cell mechanical properties has been introduced that increases the electric field magnitude further than for electrodeformation and reports the timescale of osmotic swelling upon poration of the cell membrane with this higher field-induced membrane tension. This approach has been demonstrated in a continuous-flow system in which the electric field was concentrated in a channel constriction to yield repeatable exposure to uniform fields.73,74 Bao et al.73,74 achieved throughputs of ~10 cells/s with this approach, which may be limited by the slower kinetics of the swelling process. An advantage of the approach is the simplicity of actuation with a DC field, imaging-based readout and larger and more easily measured changes in cell size compared to electrodeformation. It should be noted that electric field–induced poration is also dependent on cell size, with larger cells being more easily porated, suggesting a potential dependence of the timescale measurement on cell size. A similar approach to cell timescale measurements has also been suggested in a static system in which cells are introduced into a chamber in which electrochemically generated hydroxide leads to poration. 75 Another future challenge would be developing relevant internal controls that respond to an electric field or chemicals in a similar manner to cells—which may be more difficult than for direct force-induced deformations. In general, poration-based approaches should obtain information on membrane resistance to poration and cortical cytoskeletal strength but should be less dependent on intracellular cytoskeletal or nuclear elements. It will be important in the future to be able to decouple these contributions; otherwise, a cell that is resistant to poration but deformable might yield a similar timescale of deformation as an easily porated but stiff cell.
Future Directions
There are exciting developments on all fronts, from instruments to applications, suggesting the imminent arrival of mechanical measurements to broader biological research and the clinic. Until now, most work has focused on proof-of-concept demonstrations of new instruments with improved capabilities using cell lines. There are now a myriad of developing application areas to explore with these next-generation systems. To be successful and prove usefulness over a large data set, instruments will still need to be improved with increased throughput, automation, and robustness. As discussed above, increased throughput approaching flow cytometry is becoming a reality with continuous-flow fluid-applied strains. For automation, a key point is that a majority of approaches discussed require image analysis—which is the bottleneck in obtaining data. It will be important to develop fast software and hardware approaches in the community that are specific to analysis of cell images. My group, along with Kastner et al., 76 has reported some initial strategies for hardware-assisted cell analysis that should be useful to the community. The ever-increasing shutter speeds and frame rates of high-speed cameras should reduce motion blur and increase robustness for all imaging-based approaches as well. As discussed above, representative and stable cell analogues are also important to properly normalize measurements and advance the field, and there should be dedicated efforts toward developing these. Additional commercial development is likely to aid in and improve ease of use for many of these approaches that are ready to move beyond the academic environment and become productized. These early products will then make their way back into the lab to help acquire even more data, faster.
A critical mass of data will be needed to convince the wider cell biology and clinical communities of the merits of a deformability biomarker and is necessary for wider adoption beyond its biophysics origins. Key experimental designs that are likely to accelerate this process include combined fluorescent/molecular and deformability measurements for the same single cell. This will tie molecular biomarkers that biologists are comfortable with directly to mechanical biomarkers and therefore should be a high priority for future research. In addition, although initial hints are emerging, clearly identifying molecular, nuclear, and other epigenetic underpinnings of particular mechanical readouts will go a long way in reaching biologists as well as suggesting new applications connected to these underpinnings. Particular attention is now being paid beyond the “traditional” cytoskeleton to wider epigenetic modifications. If mechanical measurements are shown to provide a new window into the wider epigenetic world of the cell, biologists and clinicians are likely to embrace these new tools and never look back.
Footnotes
Acknowledgements
The author thanks Daniel R. Gossett and Henry T. K. Tse for useful comments and additions to the manuscript.
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author received financial support for the research, authorship, and/or publication of this article: This work is partly supported by a Defense Advanced Research Projects Agency (DARPA) Young Faculty Award #N66001-11-1-4125 and a Packard Fellowship in Science and Engineering.
