Abstract
Introduction
Osteoporosis has placed a huge burden on the healthcare system in Hong Kong. Patients suffering from osteoporosis are associated with an increased risk of fragility fracture, leading to an increase in both morbidity and mortality rate. 1 Patients often lose their independence as well as their quality of life. There is a projected increase in life expectancy according to the latest Hong Kong Population Projections for 2017–2066. The average life expectancy is predicted to increase from 81.3 years for males and 87.3 years for females to 87.1 years and 93.1 years in 2066. 2 The prevalence of geriatric hip fractures is expected to be in an increasing trend due to the ageing population. 3
Patients suffering from fragility hip fractures are particularly vulnerable to physiological stress, from the injury itself and the subsequent surgical intervention due to their underlying comorbidities. Anaemia which further increases mortality, length of stay in the hospital and result in poor functional outcomes,4,5 is frequently noted in patients with hip fractures, with more significant blood loss in extracapsular hip fractures compared with intracapsular hip fractures. 6 There are previous studies advocating the benefit of the use of tranexamic acid (TXA) in hip fracture cases, with the aim to reduce blood loss.
In our centre, there was a change of guidelines regarding the management of hip fractures with the incorporation of the use of TXA infusion on 14 January 2019. This study is conducted to evaluate the effectiveness of TXA on the management of acute geriatric intertrochanteric hip fracture with proximal femoral nail antirotation performed, in terms of reduction of blood loss, needs of transfusion and the incidence of adverse effects of TXA.
Methods
This is a single-centre retrospective cohort study. Inclusion criteria were (1) patients with age ≥ 65 years old, (2) admitted to the United Christian Hospital for acute hip fracture from 1 January 2018 to 31 December 2019 and (3) received close reduction and proximal femoral nail antirotation as surgical intervention. Cases of pathological fractures, multiple fractures within the same admission, open fracture, non-acute fracture and surgical intervention requiring the use of a long cephalomedullary device or open reduction, presence of other sources of bleeding or with contraindication to intravenous TXA infusion were excluded (Tables 1 and 2).
Exclusion criteria.

TXA: tranexamic acid.
Contraindication to tranexamic acid (TXA).

Antiplatelets or anticoagulants other than aspirin were withheld at least 3 days before surgery. Patients were divided into the Controlled group and the TXA group. Patients in the TXA group were given 1 g of intravenous TXA on induction before surgical incision. The surgeries were performed by surgeons with different levels of expertise without the insertion of a drain.
Demographic data including age, gender, use of antiplatelets and anticoagulants and fracture pattern were analysed. The primary outcome was the amount of blood loss, which was classified into immediate blood loss and hidden blood loss. The immediate blood loss was defined as intraoperative blood loss, being estimated by visual scale by anaesthetists, nurses and surgeons. The hidden blood loss was assessed by the change in haemoglobin and haematocrit levels from admission to post-operation Day 0, and the need for transfusion. The secondary outcome included the length of stay and operative time. Post-operative thromboembolic events within 14 days and 30-day mortality rates were also evaluated.
Data were retrospectively retrieved from the Clinical Data Analysis and Reporting System (CDARS) of the Hospital Authority, Hong Kong.
We hypothesize that the use of TXA would help (1) reduce blood loss, (2) the need for transfusion and (3) without increasing the risk of thromboembolism.
Statistical analysis
Continuous variables are expressed as mean ± standard deviation. For categorical variables, chi-square test is used for analysis. For the significance of each group, unpaired t-test and Mann–Whitney U test are used, with p < 0.05 being statistically significant.
Results
Demographics
From 1 January 2018 to 31 December 2019, there were a total of 552 patients admitted to United Christian Hospital presenting with acute intertrochanteric fracture of femur. One-hundred eighty-eight patients were excluded according to the predefined criteria. Among the 364 patients included in this study, 213 patients and 151 patients were classified into the Controlled group and TXA group, respectively (Figure 1). The mean age was 85.2 ± 6.71 years; 72.0% were female. There was no significant inter-group difference in the baseline characteristics as shown in Table 3.
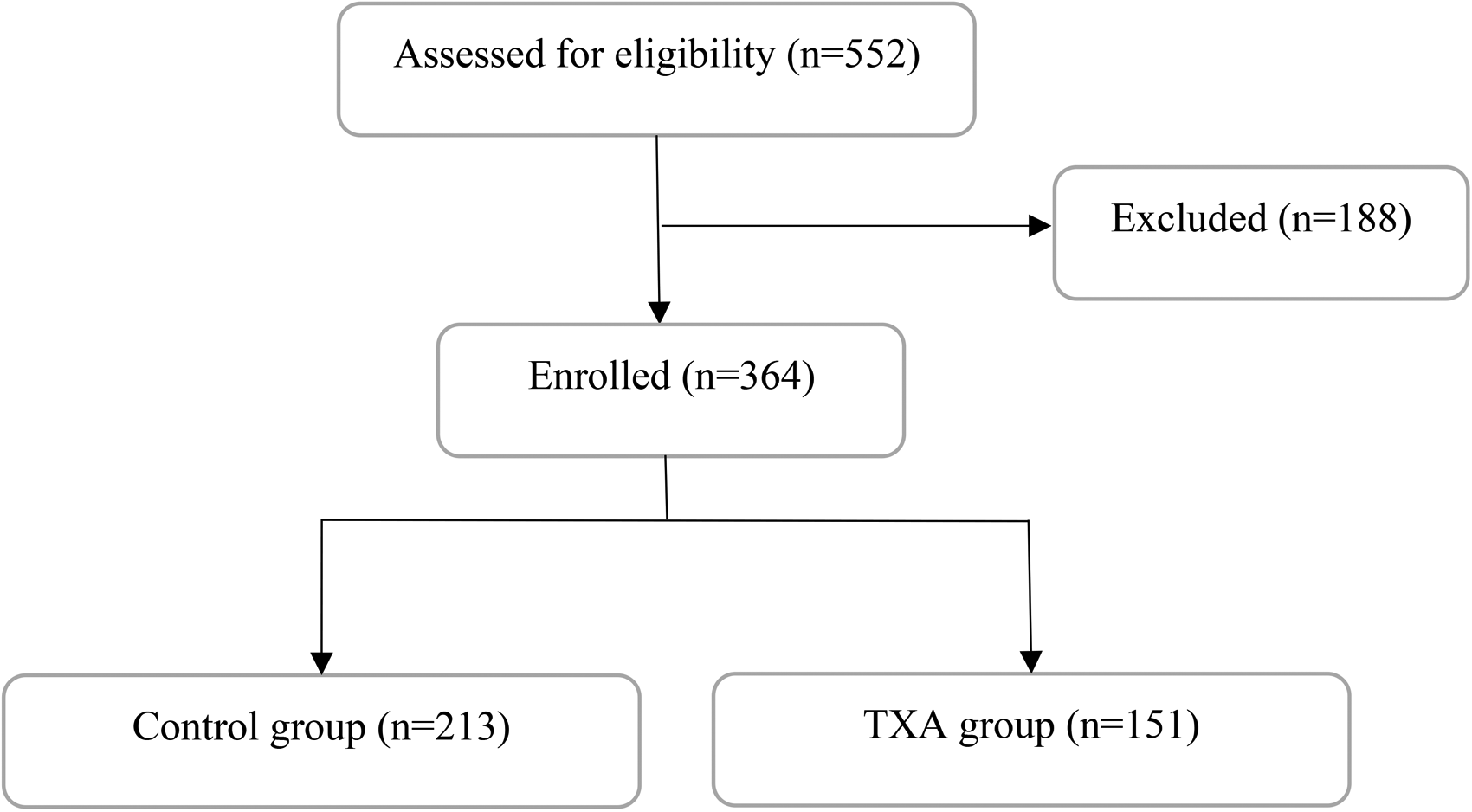
Flowchart of patient enrollment.
Baseline demographics.
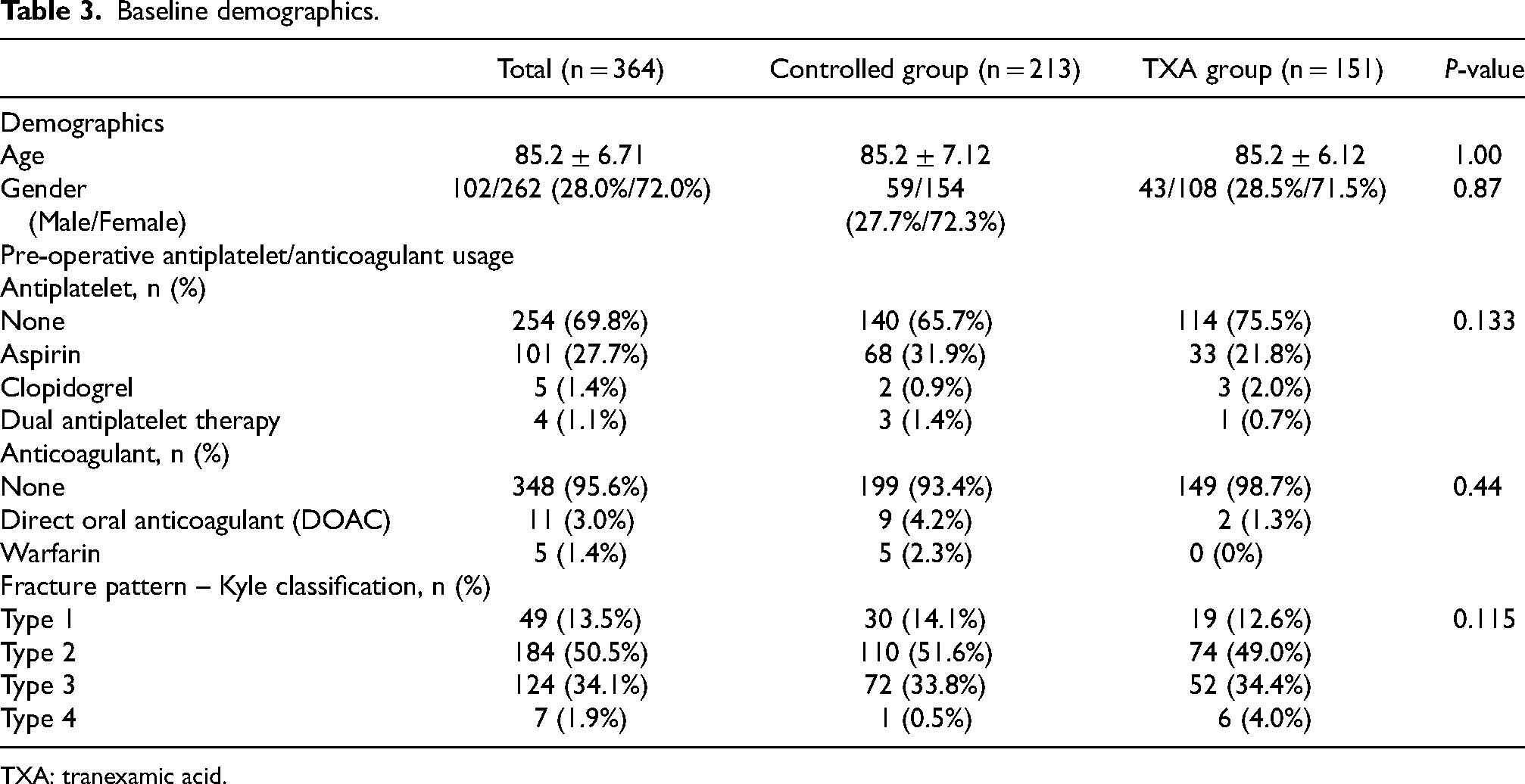
TXA: tranexamic acid.
Primary outcome
The primary outcome included intraoperative blood loss, post-operative haemoglobin and haematocrit change and post-operative blood transfusion. There was a reduction in intraoperative blood loss (Controlled group: 97.8 ± 67.7 ml; TXA group: 76.0 ± 71.4 ml; Difference: −22.3%, p-value: 0.0036), post-operative haemoglobin drop (Controlled group: 1.46 ± 1.33 g/dl; TXA group 1.25 ± 1.23 g/dl; Difference −14.4%, p-value 0.121), post-operative haematocrit drop (Controlled group: 0.04 ± 0.03 L/L, TXA group 0.03 ± 0.03 L/L; Difference −25%, p-value: 0.05) and post-operative blood transfusion (Controlled group 0.70 ± 0.95 unit; TXA group: 0.59 ± 0.81 unit; Difference −15.7%, p-value: 0.236) with the use of TXA. The reduction in intraoperative blood loss and post-operative haematocrit drop reached statistical significance (Tables 4 and 5).
Primary and secondary outcomes.
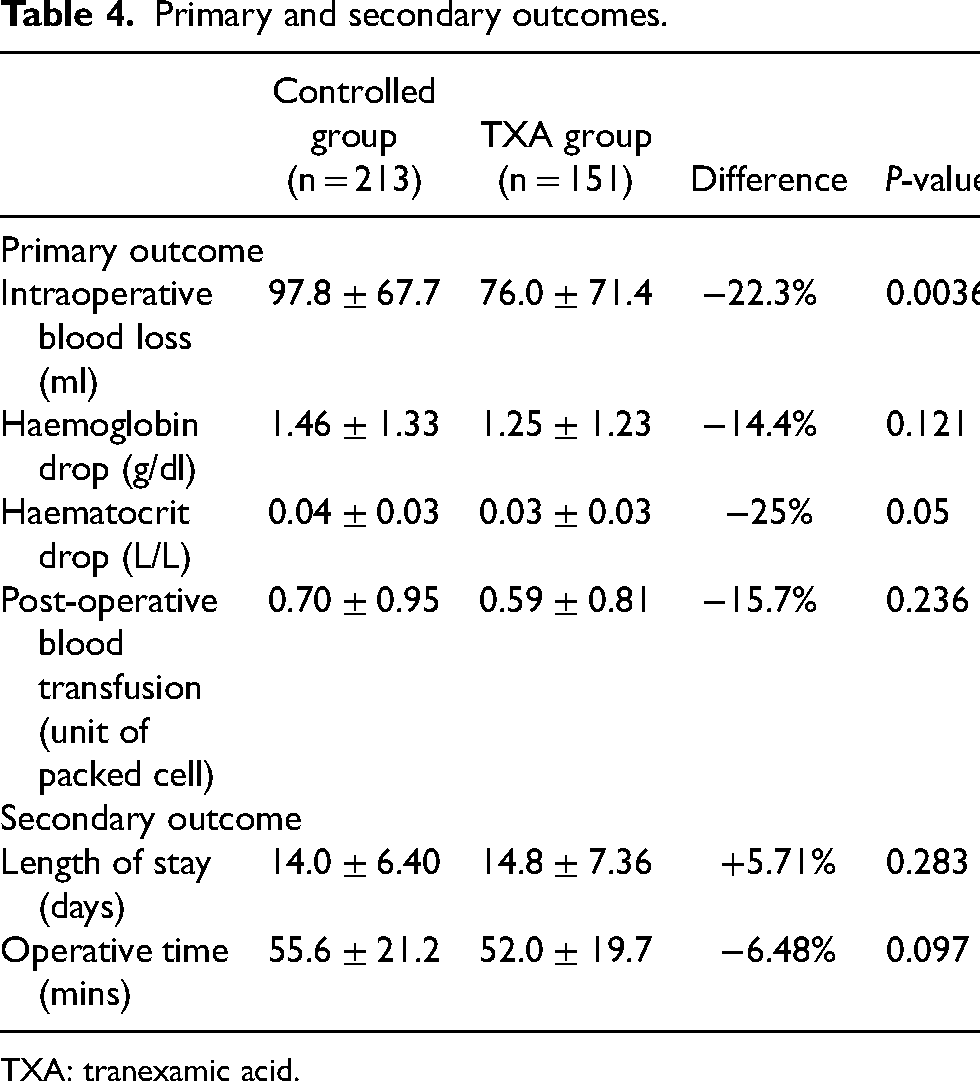
TXA: tranexamic acid.
Post-operative Thromboembolic events and 30-day mortality.
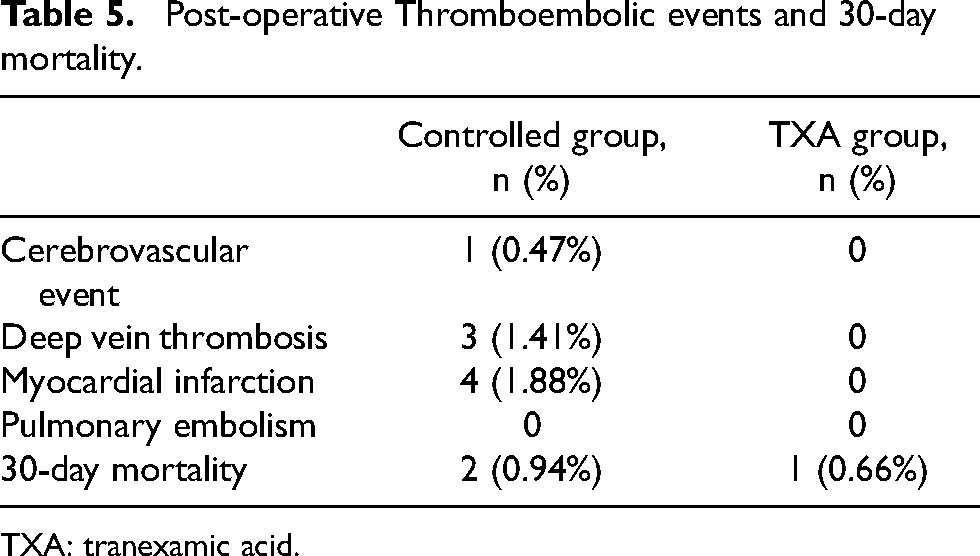
TXA: tranexamic acid.
Secondary outcome
There was a reduction in operative time (Controlled group: 55.6 ± 21.2 min; TXA group: 52.0 ± 19.7 min; Difference: −6.48%, p-value 0.097) in TXA group. There was an insignificant increase in the length of stay in TXA group (Controlled group: 14.0 ± 6.40 days; TXA group: 14.8 ± 7.36 days; Difference: + 5.71%, p-value: 0.283).
Post-operative thromboembolic events and 30-day mortality
Eight thromboembolic events occurred in the Controlled group whereas no thromboembolic occurred in the TXA group.
For the 30-day mortality, there were two cases in the Controlled group and one case in the TXA group.
Discussion
TXA is a synthetic lysine analogue which was first reported in 1962. It binds on the lysine binding site on the plasminogen which inhibits plasmin formation and displaces plasminogen from fibrin, which inhibits fibrinolysis and promotes clot stability. 7 It has been widely used in various orthopaedics surgery to reduce intraoperative blood loss.
There are various modes and dosages of TXA administration regarding the application in geriatric hip fracture cases. It can be administrated topically or intravenously. Previous studies revealed the effectiveness of intravenous TXA infusion with loading and maintenance doses to reduce total blood loss.8–9 Tengberg et al. demonstrated a reduction of total blood loss of 570.8 ml with the use of intravenous TXA infusion with the regime of 1 g TXA bolus prior to surgery, followed by post-operative 24-hour infusion of 3 g of TXA. 8 Tian et al. demonstrated a reduction in overt bleeding (50.59 ml), total blood loss (181.58 ml) and 20% lower transfusion rate with the use of intravenous TXA at the dosage of 10 mg/kg, 10 min preoperatively and 5 h post-operatively. 9
In our study, we would like to evaluate the clinical significance of the change in management protocol with the use of 1 g intravenous TXA infusion before surgical incision in patients with hip fractures. We demonstrated the reduction of intraoperative blood loss by 22.3% (97.8 ± 67.7 ml vs. 76.0 ± 71.4 ml, p-value: 0.0036) with the use of a single dose of TXA administration.
There was a reduction in haemoglobin drop by 14.4% (1.46 ± 1.33 g/dl vs. 1.25 ± 1.23 g/dl, p-value: 0.121), haematocrit drop by 25% (0.04 ± 0.03 L/L vs. 0.03 ± 0.03 L/L, p-value: 0.05), post-operative blood transfusion needs by 15.7% (0.70 ± 0.95 vs. 0.59 ± 0.81, p-value: 0.236) with the use of TXA injection in our study. A reduction in operative time by 6.48% (55.6 ± 21.2 min vs. 52.0 ± 19.7 min, p-value: 0.097) has been observed.
Lei et al. also advocated the use of 1 g intravenous TXA infusion prior to proximal femoral nail antirotation. 10 It is a single-centre randomized controlled trial. It showed a significant reduction in post-operative hidden blood loss (210.09 ± 202.14 ml vs. 359.35 ± 290.12 ml) and total blood loss (279.35 ± 209.11 ml vs. 417.89 ± 289.56 ml) in TXA group.
Patients on long-term use of antiplatelets and anticoagulant are predisposed to increase intraoperative blood loss. In our study, antiplatelet and anticoagulant therapy other than aspirin were withheld for at least 3 days prior to surgery. Ohmori et al. illustrated that the perioperative use of aspirin for patients undergoing operations for hip fracture would not result in significant blood loss. 11 The Perioperative Anticoagulation Use for Surgery Evaluation (PAUSE) study suggested that there would be only minimal or even no residual effect of direct oral anticoagulant (DOAC) at the time of surgery if they were interrupted for 1–4 days before the operation. The timing of interruption depends on the types of DOAC, bleeding risk of surgery and patients’ renal function. 12
TXA is an anti-fibrinolytic agent which could theoretically increase the risk of thromboembolic events. Previous studies demonstrated that TXA is safe to be administrated in various elective orthopaedics surgery, especially total knee or hip arthroplasty and spinal surgery.13–15 However, hip fractures more frequently occur in geriatric patients with multiple comorbidities. There were concerns about the safety of the usage of TXA in this population. Viberg et al. showed that the use of TXA in cases of hip fractures was not associated with an increased risk of mortality or thromboembolic events. 15 As in our study, there were no thromboembolic events in the TXA group.
This study supports the use of a single dose of TXA infusion to improve the clinical outcome of geriatric hip fractures. To our knowledge, this is the first study to evaluate the use of single-dose intravenous TXA infusion for surgical intervention of intertrochanteric fracture in Hong Kong.
There are some limitations in our study. It is a single-centre study without randomization. The surgeries were performed by surgeons with different levels of expertise involved, which might alter the clinical outcome. The intraoperative blood loss was measured by visual estimation. Gerdessen et al. suggested that this method would be subject to a high degree of bias, and the volume of blood loss might not be reflected accurately. 16 However, it is the most used method for estimating blood loss and there is still no golden standard for measuring intraoperative blood loss. Regarding blood transfusion, there is no standard protocol regarding the need for transfusion as it is subject to clinical judgement. A multi-centre randomized control study with a standard transfusion protocol would be able to further confirm the effectiveness of TXA.
Conclusion
In conclusion, we demonstrated that a single standard dose of intravenous TXA infusion can reduce intraoperative blood loss and post-operative haematocrit drop without the increased risk of thromboembolic events or post-operative mortality rate.
Intravenous TXA infusion helps to reduce blood loss in patients undergoing proximal femoral nail antirotation. It is safe to be used in geriatric patients. We recommend the usage of TXA infusion to improve the surgical outcome.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
