Abstract
Background
Periarticular multimodal drug injection (PMDI) is a safe and effective pain management technique after anterior cruciate ligament reconstruction (ACLR); however, adding morphine and epinephrine sometimes causes adverse effects. Therefore, we evaluated the efficacy of PMDI without morphine and epinephrine after ACLR.
Methods
This retrospective matched case-control study included patients who had undergone primary double-bundle ACLR with PMDI and were then matched one-to-one with a control group without PMDI based on sex, age, and body mass index using propensity-matched analysis. The following clinical outcomes were compared between the groups: visual analog scale (VAS) score, C-reactive protein (CRP) concentration, number of times the patients used additional analgesics, complication rate, and postoperative time to achieve straight leg raise (SLR).
Results
Twenty-nine patients with PMDI and 29 controls were enrolled. The VAS score at 1 day postoperatively was lower in the PMDI than the control group (1.93 ± 1.44 vs. 3.41 ± 1.75, respectively; P < 0.001). The CRP concentration at 1 and 3 days was lower in the PMDI than the control group (0.46 ± 0.47 vs. 1.00 ± 0.69 mg/dL, P < 0.001; and 1.93 ± 1.71 vs. 4.01 ± 2.55 mg/dL, P < 0.001, respectively). The average number of additional analgesics used was significantly lower in the PMDI than the control group. There were no significant differences in the frequency of occurrence of postoperative complications between the two groups. The number of patients who could achieve SLR within 1 day was 27/29 (93%) in PMDI group, which was significantly higher than the control group (12/29, 41%) (P < 0.001).
Conclusion
PMDI without morphine and epinephrine after ACLR reduced patients’ subjective pain level, objective inflammatory response without complications and enabled patients to achieve early functional recovery.
Keywords
Introduction
Anterior cruciate ligament reconstruction (ACLR) has been further developed during the past decade, and excellent results have been reported with respect to restoration of knee stability after ACLR.1–3 However, postoperative pain management after ACLR must be improved to facilitate early accelerated rehabilitation and increase patient satisfaction.4,5
Past studies have utilized various procedures for pain management after orthopedic hip or knee surgery.6,7 Although satisfactory pain relief can be achieved by continuous epidural anesthesia 6 and femoral and sciatic nerve block, 7 these techniques have disadvantages such as epidural hematoma formation and muscle weakness 8 that can delay postoperative recovery and rehabilitation. However, several studies have shown that local intra-articular injection 9 and periarticular injection10–12 of analgesics has higher safety and efficacy than other standard techniques, including epidural anesthesia, femoral nerve block, and intrathecal injection of morphine. In randomized controlled trials, periarticular multimodal drug injection (PMDI) has achieved better pain relief after ACLR than intra-articular injection 13 and femoral nerve block. 10 The addition of an injection of periarticular morphine is reportedly effective for pain relief; however, morphine often causes nausea as an adverse effect, and pain relief provided by PMDI without morphine was reported to be equivalent to that with morphine. 14 Some studies have also shown that local anesthesia with epinephrine can cause wound complications such as delayed healing or dermal necrosis due to ischemic adverse effects.15,16
Although previous studies have shown the advantages of PMDI for subjective pain relief after ACLR,10,11 few studies have analyzed the efficacy of PMDI without morphine and epinephrine for subjective pain relief, objective inflammatory changes, and functional recovery after ACLR. Therefore, this study was performed to (1) confirm the subjective efficacy of PMDI without morphine and epinephrine after ACLR and (2) investigate objective inflammatory changes and postoperative functional recovery. We hypothesized that PMDI can improve postoperative pain without adverse effects, reduce the inflammatory response and enable patients to achieve early functional recovery.
Materials and methods
Patients
This retrospective case-control study was performed to investigate the efficacy of PMDI. The study was approved by the IRB of the authors’ affiliated institutions. Among 148 skeletally mature patients who underwent ACLR from December 2013 to December 2019, those who underwent primary ACLR with a hamstring tendon autograft were included in this study. Continuous intravenous patient-controlled analgesia (IV-PCA) was generally performed for all patients, and PMDI with IV-PCA was started at the end of surgery from May 2018. The exclusion criteria were as follows: (1) ACL reconstruction with another type of graft (bone–patellar tendon–bone graft or quadriceps tendon graft); (2) ACL reconstruction with other knee ligament surgeries or osteotomy in the affected knee; (3) revision ACL reconstruction; (4) kidney or liver dysfunction, diabetes mellitus, or psychiatric conditions; (5) patients who did not receive or continue IV-PCA; (6) patients who received femoral nerve block; (7) cases that could not be retrospectively evaluated because of insufficient medical records. Demographic variables including sex, age, and body mass index (BMI) were used to calculate propensity scores derived with a logistic regression model. Propensity matching was then performed using neighbor matching with each patient who received PMDI matched to one control patient (a caliper width; 0.2). Fifty-eight patients (PMDI group, n = 29; isolated IV-PCA group (control group), n = 29) were investigated in this study (Figure 1).
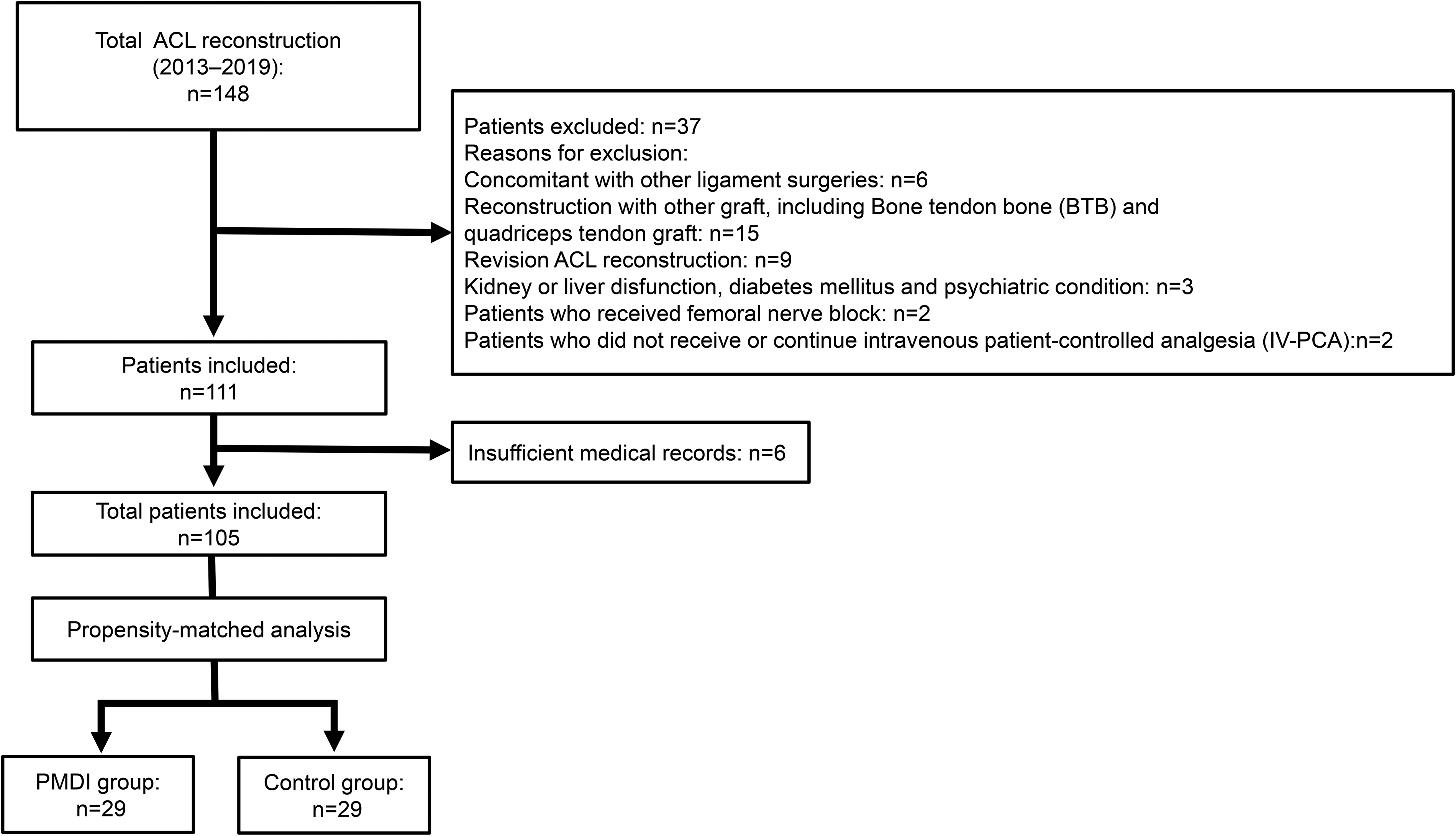
Study flow diagram. ACL, anterior cruciate ligament; PMDI, periarticular multimodal drug injection.
Surgical techniques and rehabilitation
All double-bundle ACL reconstructions were performed by a senior experienced surgeon (Y.H.). Two tunnels were created on the femoral side to the anatomical position of the insertion sites of each bundle by the transportal technique. Tibial tunnels were created with a tibial guide to the anatomical insertion site of the ACL tibial footprint. Hamstring autografts were harvested from the same leg and double-looped. Grafts were inserted in a retrograde manner through the tibial tunnels and secured with one type of fixation device (Endobutton CL; Smith & Nephew, Andover, MA, USA) on the femur and another type of fixation device (Double Spike Plates; Smith & Nephew) on the tibia at 20° of knee flexion. A suction drain was placed at the tendon harvest site and removed within 24 h after the operation. At 24 h postoperatively, the patients began a rehabilitation regimen that included isometric quadriceps muscle strengthening and a straight leg-raising exercise. The knee was immobilized and kept at 10° of flexion with a knee brace for 1 week postoperatively. Partial weight-bearing and knee range-of-motion exercises were allowed at 1 week postoperatively, full weight-bearing was allowed at 4 weeks, and participation in low-impact activities (including jogging) was allowed at 3 to 4 months. Vigorous sports activities were allowed at approximately 8 to 10 months postoperatively.
Postoperative pain management
For pain management, the PMDI group received a solution containing 30 mL of ropivacaine (10 mg/mL) (Anapeine; Aspen Japan, Tokyo, Japan), 0.5 mL of prednisolone (40 mg/mL) (Predonine; Shionogi Pharma, Osaka, Japan), and 2.5 mL of ketoprofen (20 mg/mL) (Capisten; Kissei, Matsumoto, Japan). The PMDI solution was injected into the infrapatellar fad pad, periosteum, fascia, and incision site around the tibial tunnel side and portal side at the end of surgery.
All patients underwent IV-PCA, which was composed of a 50-mL solution containing 10 mL of fentanyl (100 µg per 2 mL) (Daiichi Sankyo, Tokyo, Japan) and 0.5 mL of droperidol (2.5 mg/mL) (Droleptan; Alfresa, Tokyo, Japan). The IV-PCA solution was administered at 2 mL/h and 40 µg/dose with an additional 1 mL injection and lockout time of 20 min. Additionally, the patients received 200 mg of oral celecoxib (Celecox; Astellas Pharma, Tokyo, Japan) every 12 h from 1 to 14 days after surgery. The use of a 25-mg diclofenac sodium suppository (Nihon Generic, Tokyo, Japan) or 1-mL subcutaneous injection of pentazocine hydrochloride (15 mg/mL) (Sosegon; Maruishi Pharmaceutical, Osaka, Japan) was allowable as an additional pain medication.
Outcome measures
The clinical outcome included the visual analog scale (VAS) score, which was used to rate pain intensity and ranged from 0 to 10 (0, no pain; 10, worst pain). The VAS score at rest was measured in the same manner at 3, 6, and 12 h and 1, 2, 3, 7, and 14 days after surgery. In addition, Serum C-reactive protein (CRP) concentration (Hitachi Co., Ltd, Tokyo, Japan) was measured 1 day preoperatively and 1, 3, 7, and 14 days postoperatively. The postoperative maximum body temperature and the number of times patients used analgesics (diclofenac sodium suppository or subcutaneous injection of pentazocine hydrochloride) were also measured. Adverse effects related to PMDI were observed, including nausea, vomiting, gastric ulcers, deep vein thrombosis, or wound complications such as delayed healing or superficial or deep infection. With regard to functional recovery, the ability to perform straight leg raise (SLR) was evaluated every day by physical therapists. SLR was performed with the patient lying down on bed and they were instructed to raise the affected leg to the maximum hip flexion position.
With regard to clinical outcome, Lysholm score, IKDC score and the side-to-side difference of anteroposterior laxity measured by the KT-1000 arthrometer (MED metric Corp., San Diego, CA, USA) with manual maximum pull (KT measurement) were assessed pre and postoperatively at the latest follow up. Mean postoperative follow-up was 26.7 ± 5.4 months in PMDI group and 28.1 ± 9.1 months in control group, respectively.
Statistical analysis
Student's t test or the Mann–Whitney U test was used to compare continuous variables, and the chi-square test or Fisher's exact test was used to analyze categorical variables. All data are presented as mean ± standard deviation or range. A power analysis was performed with the power, α, difference and standard deviation set at 0.8, 0.05, 1.48, and 1.77, respectively, according to the VAS at 1 day postoperatively. The analysis revealed that a minimum of each 24 patients was required for the student's t test to detect a difference between the two groups. All statistical analyses were performed with EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), 17 which is a graphical user interface for R (The R Foundation for Statistical Computing, Vienna, Austria). More precisely, it is a modified version of R commander designed to add statistical functions frequently used in biostatistics. Values of P < 0.05 were considered statistically significant.
Results
The demographic and clinical characteristics of the overall patients and the patients in the PMDI and control groups are shown in Table 1. There was no significant difference regarding demographic and clinical outcome between two groups. The change in the VAS score over time is shown in Figure 2. The pain levels at 1 day postoperatively were lower in the PMDI than control group (1.93 ± 1.44 vs. 3.41 ± 1.75, respectively; P < 0.001) and there was no such difference after that. The CRP concentration at 1 and 3 days was lower in the PMDI than control group (0.46 ± 0.47 vs. 1.00 ± 0.69 mg/dL, P < 0.001; and 1.93 ± 1.71 vs. 4.01 ± 2.55 mg/dL, P < 0.001, respectively) (Figure 3). No patients in the PMDI group used pentazocine hydrochloride, and the mean frequency of use was significantly different between the two groups (Table 2). The average number of diclofenac sodium suppositories used at 1 day was significantly lower in the PMDI than control group (P = 0.018, Table 2). There were no significant differences in maximum body temperature (PMDI, 37.4°C ± 0.37°C vs. Control, 37.6°C ± 0.36°C, respectively; P = 0.09) and the frequency of occurrence of postoperative complications including nausea, vomiting, gastric ulcers, deep vein thrombosis (all were asymptomatic), or wound complications between the two groups (Table 3). Regarding functional recovery (Figure 4), the number of patients who could achieve SLR within 1 day was 27/29 (93%) in PMDI group, which was significantly higher than control group (12/29, 41%) (P = 0.0001).
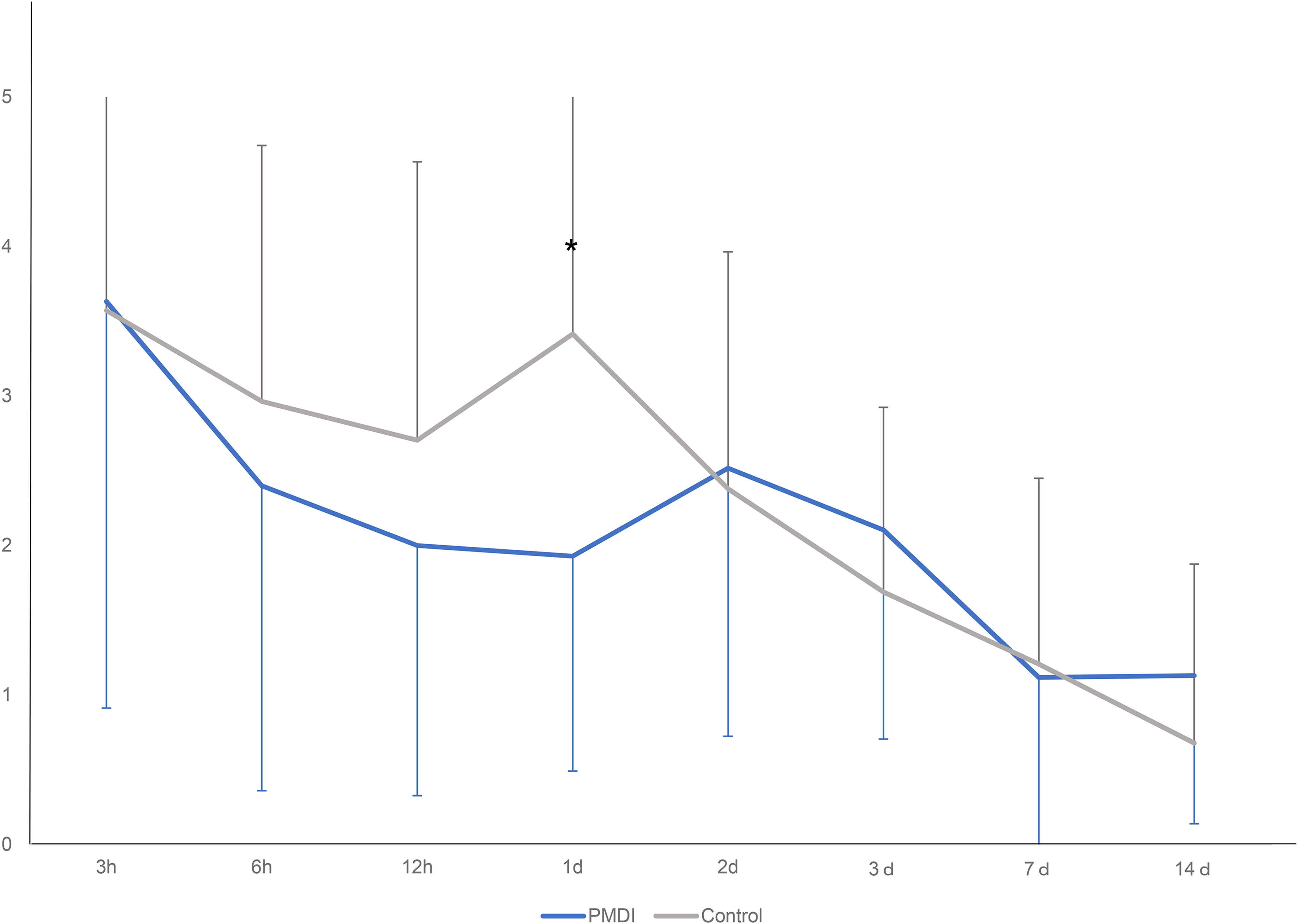
Change in VAS score. *Patients in the PMDI group had significantly less pain than those in the control group at 1 day after the operation (P < 0.001).
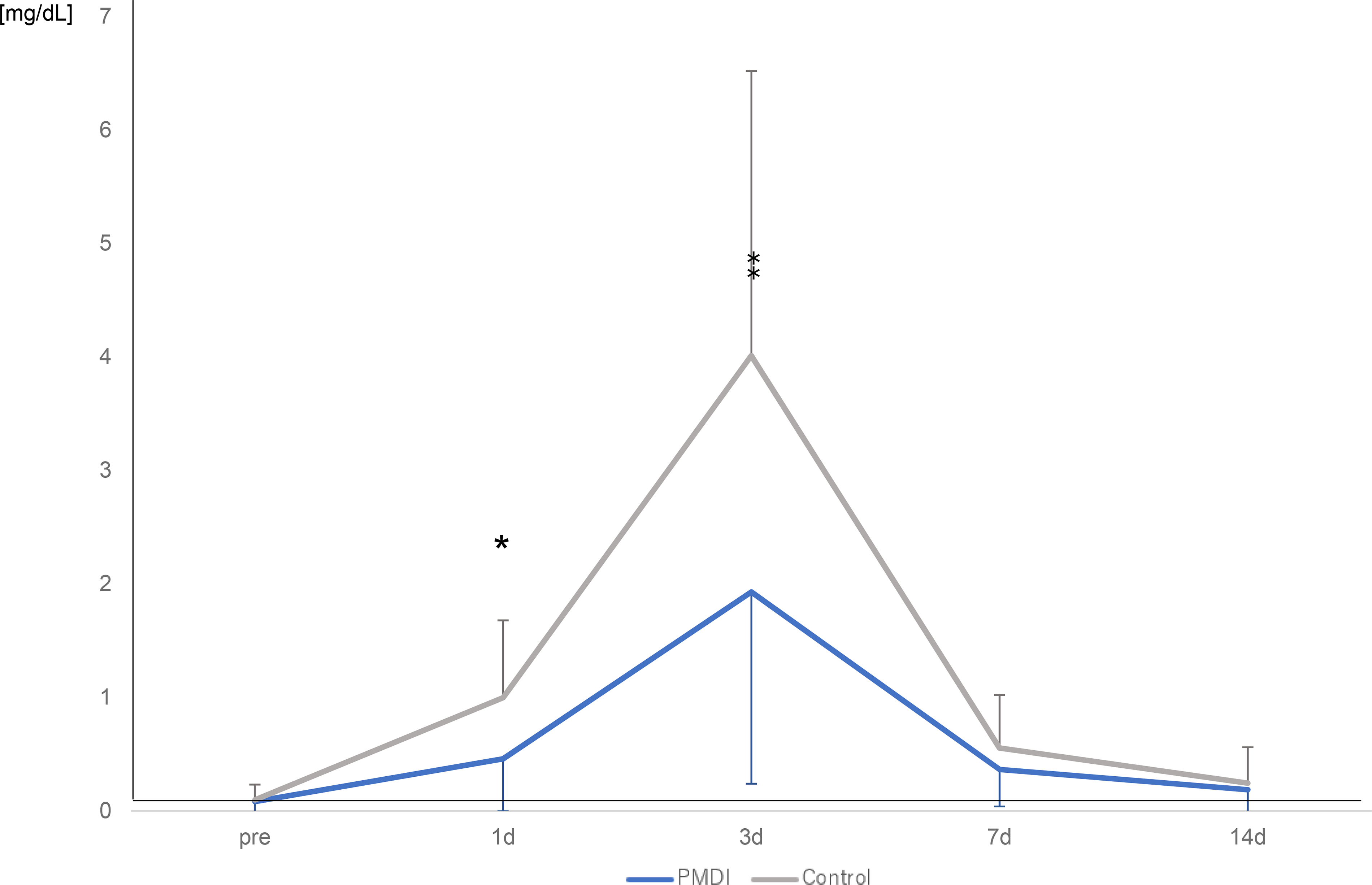
Change in CRP concentration. *, ‡ The CRP concentration at 1 and 3 days was lower in the PMDI than control group (P < 0.001 and P < 0.001, respectively).
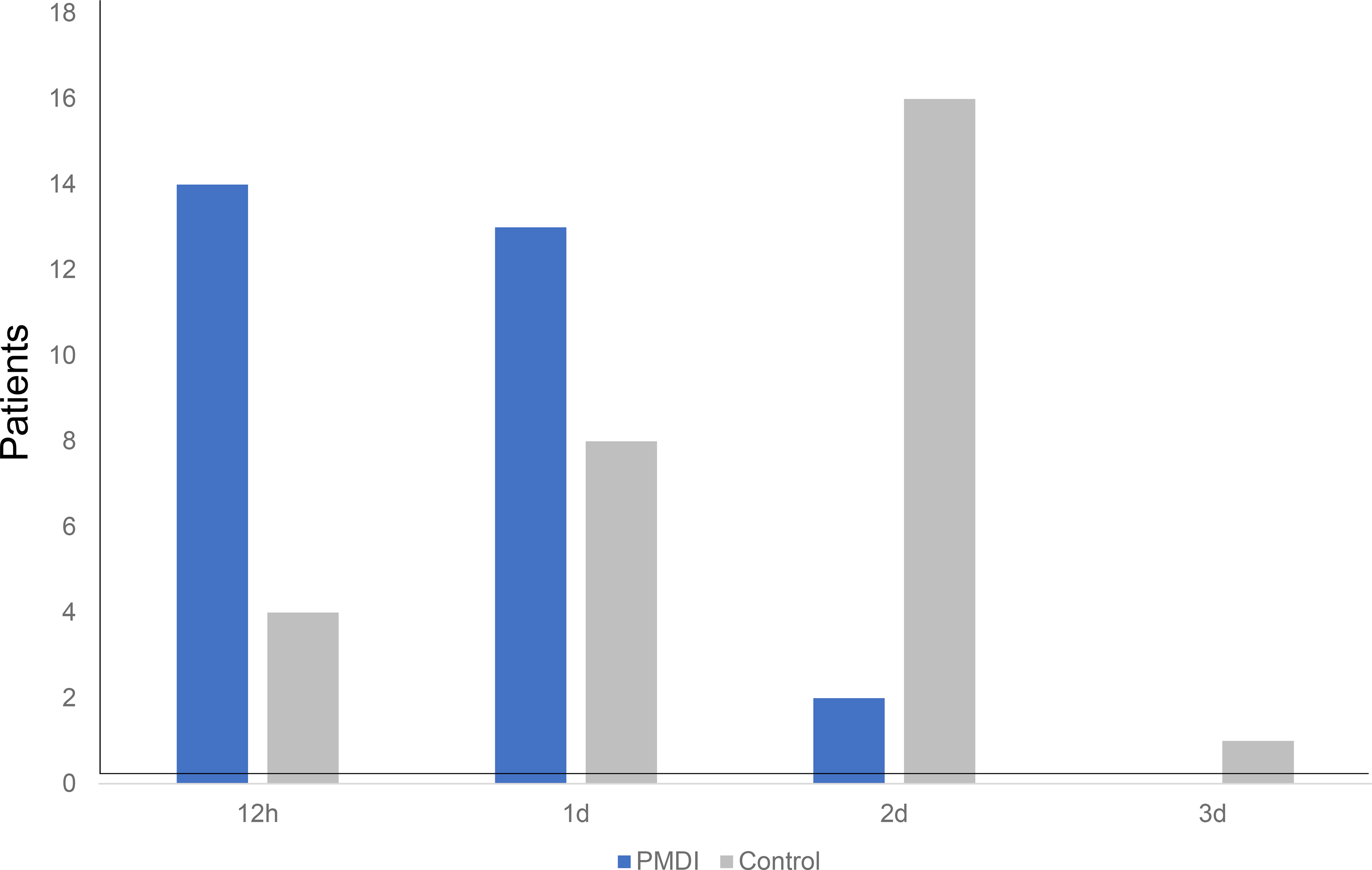
Postoperative time to achieve straight leg raise. The number of patients who could achieve SLR within 1 day was 27/29 (93%) in PMDI group, which was significantly higher than control group (12/29, 41%) (P = 0.0001).
Demographic and clinical characteristics of overall patients and patients in the PMDI and control groups.

Data are shown as mean ± standard deviation or n (%).
All variables were compared between the PMDI and control groups.
Postoperative clinical outcome was measured at the latest follow up.
Analysis of variance with Student's t test or the Mann–Whitney U test was used to analyze continuous variables, and the chi-square test or Fisher's exact test was used for categorical variables.
BMI, body mass index; PMDI, periarticular multimodal drug injection.
Mean number of additional additional analgesia uses in PMDI and control groups.
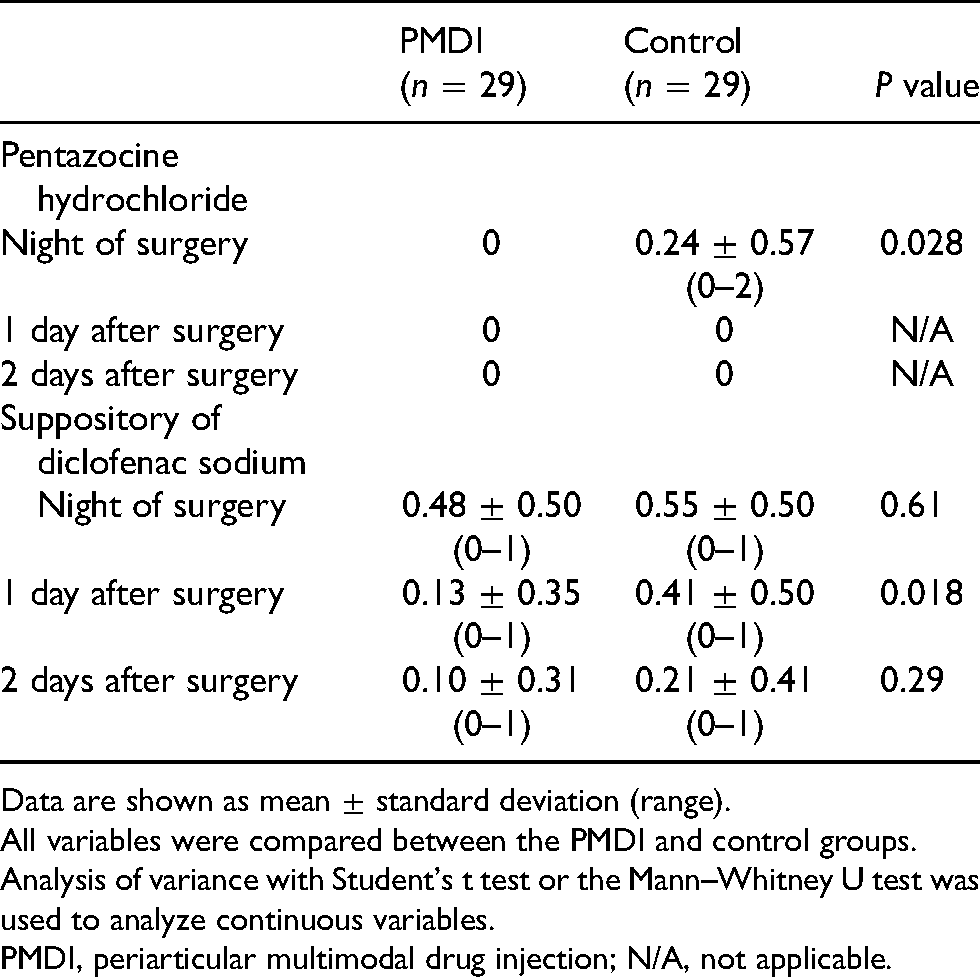
Data are shown as mean ± standard deviation (range).
All variables were compared between the PMDI and control groups.
Analysis of variance with Student's t test or the Mann–Whitney U test was used to analyze continuous variables.
PMDI, periarticular multimodal drug injection; N/A, not applicable.
Complications in PMDI and control groups.
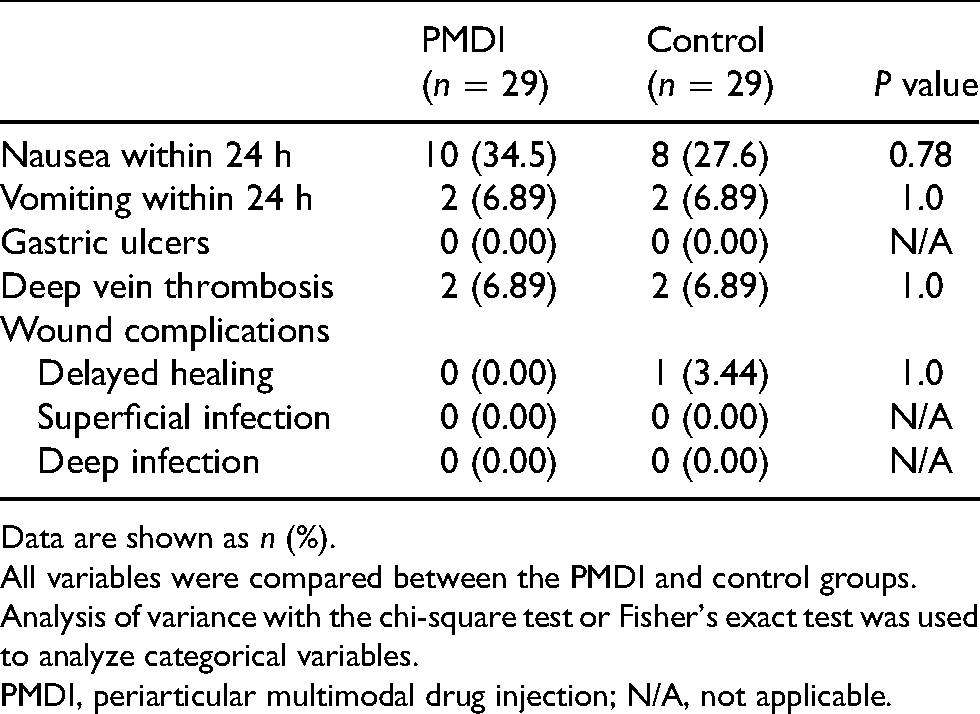
Data are shown as n (%).
All variables were compared between the PMDI and control groups.
Analysis of variance with the chi-square test or Fisher's exact test was used to analyze categorical variables.
PMDI, periarticular multimodal drug injection; N/A, not applicable.
Discussion
The current study showed that PMDI without morphine and epinephrine after ACLR improved patients’ subjective postoperative pain without early complications or the use of additional pain medications and enabled patients to achieve early functional recovery. The objective postoperative inflammatory response also decreased by adding PMDI.
Subjective pain at 1 day postoperatively and the frequency of additional pain medication use were significantly lower in the PMDI than control group. Previous studies have shown the efficacy of PMDI for pain relief after knee surgery.6,10,13,18,19 Koh et al. 13 performed a randomized controlled trial showing that PMDI after ACLR was associated with less pain on the first postoperative night than intra-articular injection or control treatment. Another recent randomized controlled trial showed that the VAS score and patient satisfaction level until 2 days after ACLR were better in the PMDI group than femoral nerve block group. 10 Another study demonstrated that PMDI had better effectiveness and certainty for pain relief up to 24 h postoperatively than femoral nerve block or adductor canal block. 12 In one systematic review, peripheral nerve block techniques such as femoral and sciatic catheter placement after total knee arthroplasty (TKA) demonstrated analgesic effectiveness equivalent to that of epidural analgesia and an improvement in adverse effects such as hematomas, drop foot, sphincteric disturbance, paresthesia, and numbness. 8 The review also showed that the incidence of inadequate or failed block was 3.0% in the peripheral nerve block group and 7.7% in the epidural group. 8 Another study showed that femoral nerve block caused postoperative temporary motor block (31.4%). 20 This adverse effect can lead to delayed early postoperative rehabilitation. In contrast, PMDI is a simple procedure that reliably blocks periarticular nerves and effectively improves postoperative pain without increasing adverse effects. The current study supports the findings of previous reports6,10,13,18,19 showing that PMDI provides adequate pain relief without using additional pain medication or increasing early complications compared with the control group.
Many different types of PMDI have been used in previous studies.21–24 Maheshwari et al. 23 first showed the efficacy of PMDI comprising bupivacaine, morphine sulfate, epinephrine, methylprednisolone, and cefuroxime after hip or knee arthroplasty. However, a randomized trial showed that PMDI with morphine caused increased vomiting 14 compared with PMDI without morphine, and the VAS scores did not differ between PMDI with and without morphine. In the current study, PMDI without morphine reduced patients’ pain more effectively than in the control group without an increase in complications such as nausea and vomiting. In addition, a few case reports described skin necrosis caused by local anesthesia that included epinephrine.15,16 Based on these reports, our regimen did not include epinephrine, and no wound complications occurred in the PMDI group. Kerr et al. 21 and Andersen et al. 22 demonstrated that PMDI including nonsteroidal anti-inflammatory drugs (NSAIDs) reduced postoperative pain and resulted in earlier readiness for hospital discharge. The current study included NSAIDs instead of morphine in the PMDI solution, and this have probably contributed to the reduced postoperative pain level.
With respect to steroids, Ikeuchi et al. 19 performed a randomized controlled trial showing that periarticular injection of anesthesia with dexamethasone after TKA significantly improved the increasing serum CRP concentration at 3 days postoperatively and the interleukin 6 concentration in the drain fluid sampled 24 h after surgery compared with periarticular injection of anesthesia without dexamethasone. Dexamethasone is a long-acting glucocorticoid with potent anti-inflammatory properties, and its anti-inflammatory effects, both locally and systemically, were confirmed by the interleukin 6 concentration in the drain fluid and the serum CRP concentration. Ng et al. 25 demonstrated that patients who received PMDI with a corticosteroid after unicondylar knee arthroplasty had significantly lower CRP concentrations on postoperative days 1, 2, and 3 than did patients who received PMDI without a corticosteroid. Additionally, active knee flexion was well correlated with the CRP concentration, so patients with better knee flexion tended to have lower CRP concentrations. 25 The current study showed that PMDI reduced the objective inflammatory response, and serum CRP concentration. PMDI with a steroid showed anti-inflammatory effects with rapid recovery of serum CRP concentration. However, wound infection is a risk associated with steroid use and is of particular concern to surgeons. A recent meta-analysis 26 of the complications of PMDI use with steroids showed no significant difference in either the total incidence of complications or the postoperative infection rate. This report 26 supports our findings regarding complications. However, compromised patients with diabetes mellitus and kidney or liver dysfunction were excluded from the current study; therefore, eligible patients must be selected to avoid an increased risk of infection.
Current study showed PMDI use enabled patients to achieve early functional recovery. Previous studies demonstrated early functional recovery such as early SLR, 19 range of motion18,19,24 with PMDI. The addition of PMDI can reduce pain in the acute postoperative period, which can lead to early functional recovery.
This study had several limitations. First, the current study focused solely on the postoperative pain relief after ACLR using PMDI. Other outcomes like postoperative range of motion could not be investigated since we used the conventional rehabilitation protocol. Several studies after TKA have shown that earlier range of motion or straight leg raising was achieved in the PMDI group.18,25,26 This is an important benefit supporting our recommendation for the use of PMDI. Second, this study was the retrospective study and had a small sample size. Multicenter randomized control trials are needed to confirm the efficacy and safety of PMDI after ACLR. Finally, the results might not be generalizable to the United States and Europe because the statistical population, operative procedure, and postoperative management in this study might not correspond to other regions.
Conclusion
PMDI without morphine and epinephrine after ACLR reduced patients’ subjective pain level, objective inflammatory response without complications and enabled patients to achieve early functional recovery.
Footnotes
Acknowledgements
Authors’ contributions
Changhun Han, M.D.: main author.
Yusuke Hashimoto, M.D., PhD: data analysis, comprehensive management.
Sunao Nakagawa, M.D.: data collection.
Shinji Takahashi, M.D., PhD: statistic data analysis.
Yohei Nishida, M.D.: data collection.
Shinya Yamasaki, M.D., PhD: comprehensive management.
Junsei Takigami, M.D., PhD: comprehensive management.
Hiroaki Nakamura, M.D., PhD: comprehensive management.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The study was approved by the hospital ethics committee and the internal review board of our institution (IRB number: 3072).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was obtained from all patients.
