Abstract
Introduction
Morton’s neuroma (MN) is a neuropathic metatarsalgia that causes pain in the plantar aspect of the forefoot generally between the third and fourth metatarsal heads. Treatment can be nonoperative or surgical. Among nonoperative procedures, alcohol injections are still commonly used as considered simple, relatively safe and well-tolerated treatment. However, they present transient and minor complications.
Methods
Two hundred patients with a diagnosis of MN underwent ultrasound-guided injections with a 47.5% alcohol solution between 2013 and 2020. We reviewed the current literature to highlight the known complications of this treatment, comparing them to the complications developed by our patients.
Results
Three patients out of 200 patients, developed necrosis of skin and subcutaneous tissue not described in previous studies about MN.
Conclusions
Our study focuses attention on the complications subsequent to the alcohol injection therapy for MN. Patients and surgeons should be aware that in a small number of cases this therapy can be burdened by necrotic complications of the skin.
Introduction
The term Morton’s neuroma (MN) refers to a neuropathic metatarsalgia generally affecting the third, and rarely the second, common plantar digital nerve. The condition was first anatomically described by Civinini in 1835 and subsequently clinically described by Thomas Morton in 1876.1,2 It mostly affects women between 30 and 50 years old and can represent up to 9% of patients seen in a medical clinic during a week.3,4 Classically, an intense neuralgic pain, often during a walk, is localized in the plantar aspect of the third interspace between the third and fourth metatarsal heads and the opposite surfaces of the third and fourth fingers; it can be associated with burning and hypoaesthesia in the forefoot. High heels closed and narrow-toe shoes worsen the symptoms, while sandals or slippers relieve their intensity and frequency. 5 The pathogenesis is still unclear, but four main theories have been proposed: the chronic trauma theory (the position and anatomical characteristics of the nerve make it more susceptible to repeated trauma), the ischaemic theory (the symptoms would depend on circulatory disorders and fibrous degeneration of the blood vessels supplying the nerve), the intermetatarsal bursitis theory (a chronic inflammation of the intermetatarsal bursa would cause fibrotic processes in the tissues surrounding the nerve) and the entrapment theory (during the walk the nerve remains trapped and repeatedly pressed between the plantar aspect of the foot and the anterior edge of the intermetatarsal ligament, causing irritation and development of a neuroma). 6 The diagnosis is generally clinical (indicative anamnesis and positivity to the semiological manoeuvres, in particular to the Mulder’s sign, with a palpable click, and to the squeeze test). The main imaging examinations are sonography and magnetic resonance imaging (MRI), which have a sensitivity of 96% and 88%, respectively, but cannot be separated from an accurate clinical examination.7,8 The MN appears as a well-demarcated low/intermediate signal intensity ovoid mass in the T1-weighted MRI images and as a focal hypoechoic nodule in the sonographic examination between the metatarsal heads of the interested intermetatarsal space. 9
The current treatments can be divided into nonoperative and surgical ones (the latter consist of the excision of the neuroma and decompressing the adjacent intermetatarsal space, usually in case of failure of conservative therapy). Among nonoperative treatments, modification of footwear or pronation and supination orthoses, radiofrequency ablation and therapeutic corticosteroid injection or alcohol neurolysis has been proposed.10–14 Alcohol is used not only in different diseases such as ganglionic cysts, but also in liver metastases, when this is not possible surgery is required.15–17 Currently, the therapeutic alcohol injections for MN are universally considered a simple, effective and safe procedure, with transient and minor complications.
In this study, we report a cutaneous complication never mentioned in the literature about the MN, a deep necrosis of skin and subcutaneous tissue after injection of therapeutic alcohol solution.
Materials and methods
Two hundred consecutive patients who underwent ultrasound-guided alcohol injections in the third interspace between January 2013 and January 2020 have been included in this study. Demographic characteristics were in line with the literature in terms of gender and age. The clinical and sonographic or MRI diagnosis of MN, the lesions of at least 7 mm diameter, patients who underwent ultrasound-guided alcohol injections, the period time between January 2013 and January 2020, and third interspace neuromas were considered as inclusion criteria. Only third interspace neuromas were included in this study because of the greater number of cases and because the few cases present in other localizations had no minimum size of 7 mm or indication of this kind of treatment.
The therapeutic scheme for every patient consisted of a total of three injections of a solution of 0.5 ml of 95% ethyl alcohol diluted in 0.5 ml of lidocaine (47.5% alcohol solution), with a 14-day interval between the first and the second injection and a 28-day interval between the second and the third injection. Three senior surgeons skilled in foot and ankle pathology (LM, IB and AV) performed under direct sonographic control every injection, after disinfection of the injection site. During the procedure the surgeon identifies the lesion by using a sonographic probe that he or she holds in one hand, and reaches the MN holding the needle in the other hand, assisted by a nurse who modulates the injection of 1 ml of solution through a 23-gauge needle (Figure 1).
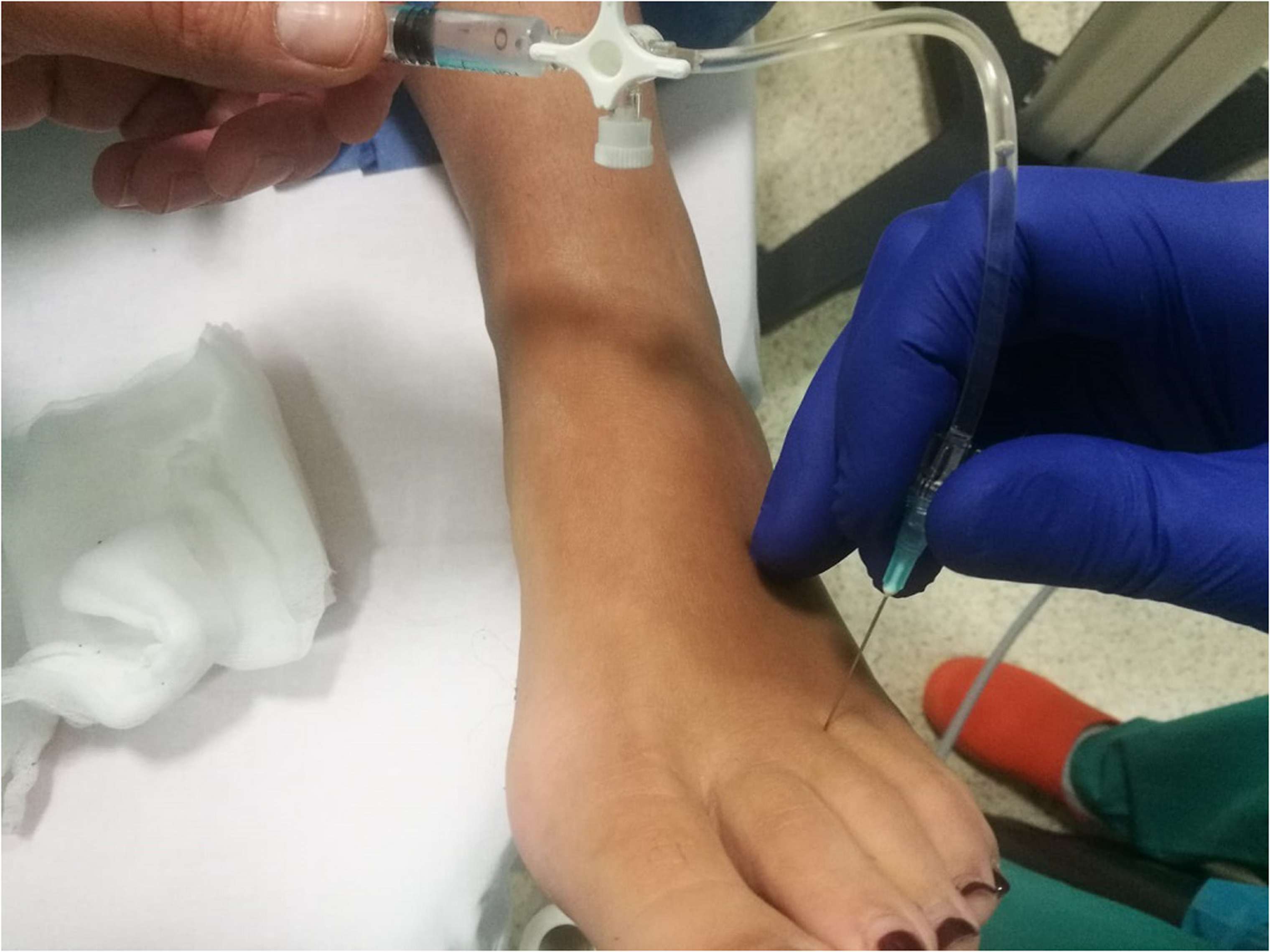
The procedure of ultrasound-guided alcohol injection consists of a variable number of infiltrations (three in our series of patients) of 1 ml of a 47.5% ethyl alcohol solution diluted with lidocaine in the intermetatarsal space interested by Morton’s neuroma.
We investigate the previously described complications related to the sclerosing therapeutic alcohol injections for MN, and we compared them to the complications developed by our own patients. All of them underwent careful clinical follow-up. Kept under close observation for the first 12 h, then checked at 7–30–90–180 days.
Results
Among 200 consecutive treated patients, 180 declared exacerbation of pain at the time of the first injection and a transient ‘pebble feeling’ between the third and fourth metatarsal heads during the subsequent 48 h; 17 patients did not communicate any post-procedural problems; in 3 patients we observed skin necrosis.
The first patient, a 65-year-old female, with no relevant health problems, after initial erythema at the site of injection in the left foot, developed necrosis of skin and subcutaneous fat 7 to 10 days after the first injection in August 2017 (Figure 2). The second patient, a 35-year-old male, affected by ulcerative colitis and multiple allergies, came to our attention in January 2019: he developed after the first injection an initial burn-like lesion with red edges and blisters just distally to the needle insertion site in the right foot, later complicated with deep necrosis of skin and subcutaneous tissues, with peritendinous exposition (Figure 3). The third patient, a 52-year-old female, with no relevant health problems, developed an initial bruising, which evolved into an eschar and deep necrosis of skin and subcutaneous tissues at the site of injection in the left foot just after the second injection in December 2019 (Figure 4). None of the three patients who developed necrotic complications was affected by peripheral circulatory or neurological disorders or diabetes and none showed any sign nor a symptom of local or general infection. Every patient stopped the treatment after the development of the complication, in agreement with the surgeon who performed the procedure.
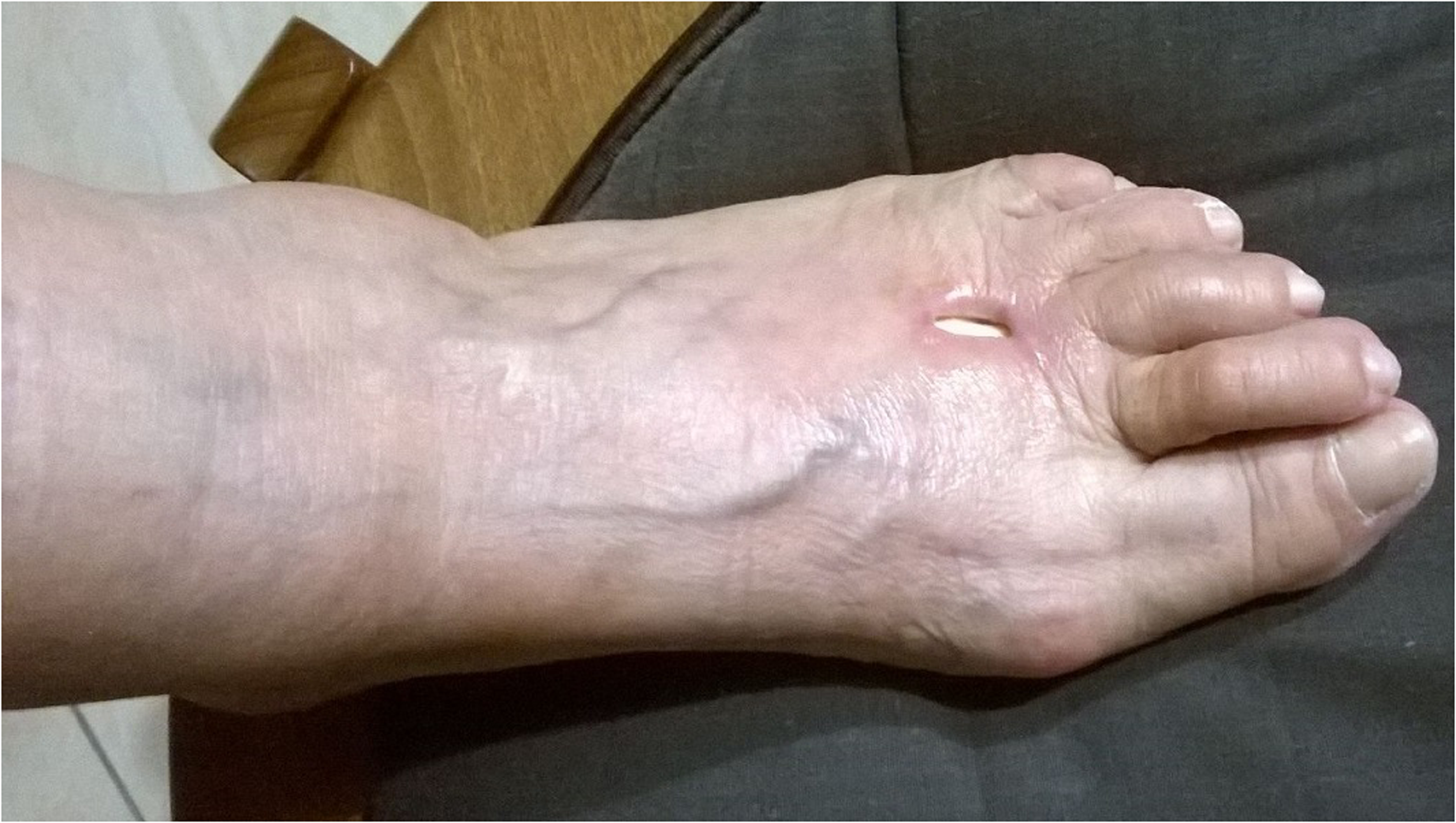
In the first patient, we observed the necrosis of skin and subcutaneous fat at the site of injection 10 days after the beginning of treatment.
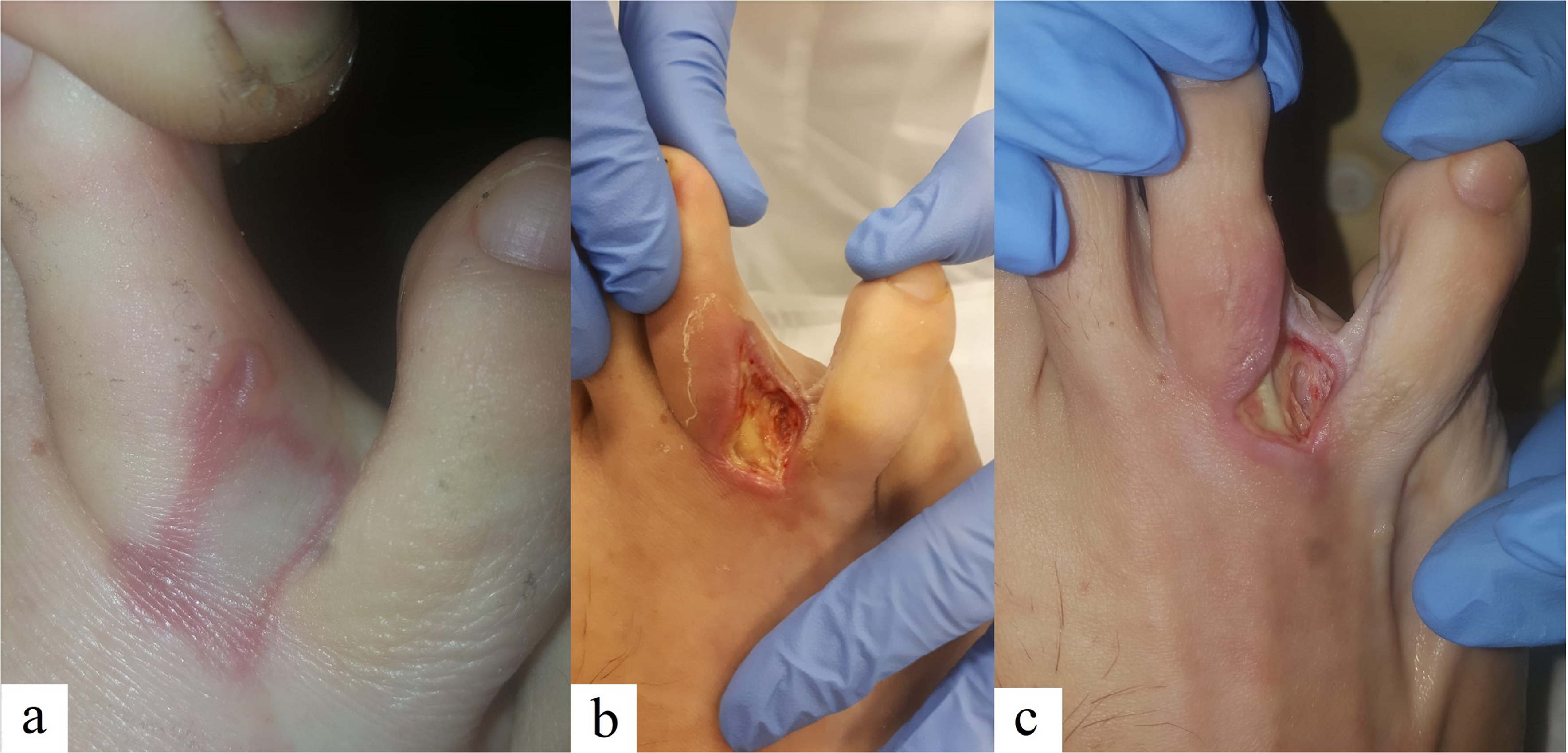
In the second patient, the damage developed from a burn-like lesion just distal to the site of injection after 7 days from the beginning of the treatment (a) to a deep necrosis of skin and subcutaneous tissues with peritendinous exposition after a further 7 days (b, c).
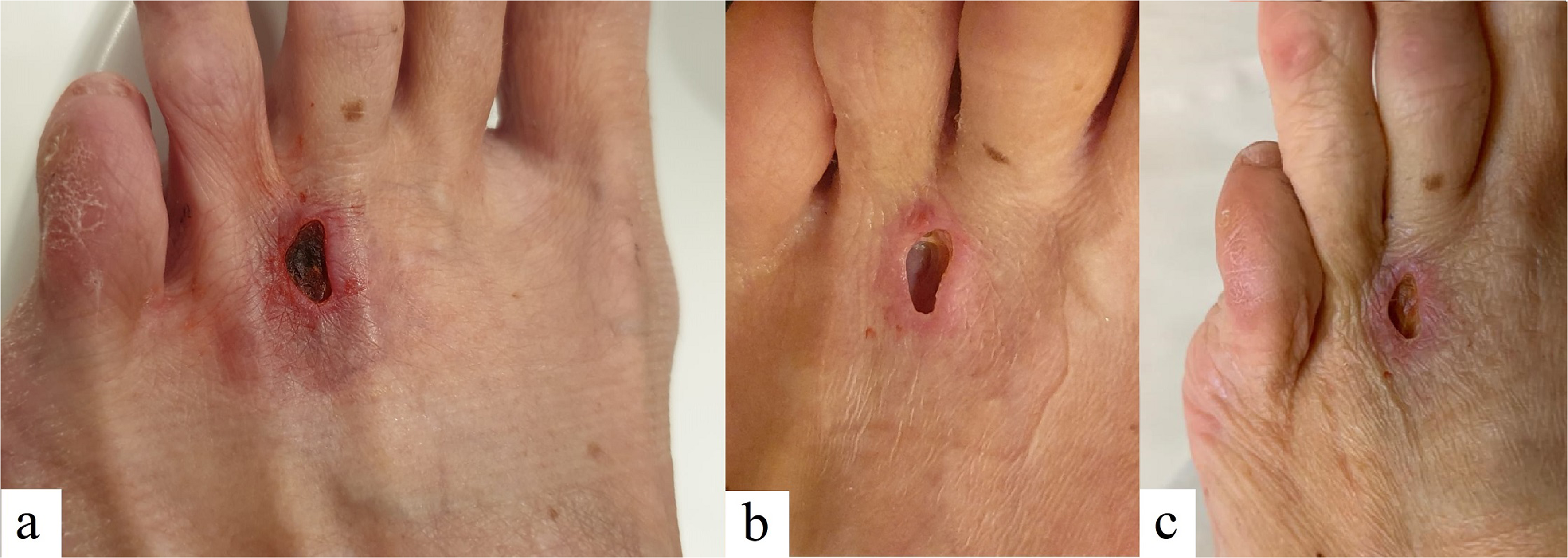
The third patient developed an eschar at the site of injection at 18 days from the second injection, after an initial bruising (a) and skin and subcutaneous fat necrosis, respectively, at 40 and 50 days after the second injection (b, c).
The first patient underwent a conservative treatment consisting of observation, medications and silver dressing until the complete resolution. The second patient was referred to a consultant plastic surgeon but was lost at the current follow-up. The third patient underwent to open neurectomy in February 2020.
Discussion
In our series, with 1 ml of a 47.5% alcohol solution through a 23-gauge needle, similar to solutions safely used before in bigger cohorts of patients, 3 of 200 patients (1.5%) developed skin necrosis with an involvement of subcutaneous tissue and peritendinous exposition, a known effect of alcohol therapy but not previously reported for ultrasound-guided alcohol injection for MN.
There is no univocal opinion in the literature on the concentration of alcohol to inject. Nevertheless, in his systematic review, Santos reports that the highest post-alcohol injection adverse events occurred at alcohol concentrations of 30% and 50%. 18 Although it is well reported that corticosteroid injections may lead to dorsal skin hypopigmentation, skin atrophy and plantar fat pad atrophy, no similar complications are recorded in treating MN with alcohol sclerosing injections.19–22 Skin necrosis and subcutaneous cellulitis were only described in a case report treating chronic toe neuritis with a 3 ml solution of 1% lidocaine with 98% dehydrated alcohol. There is not a note of correlation between the severity of the complications and the concentration of alcohol injected in this study (47.5%) or the literature (4%–50%). Least of all, of the frequency of the treatment, on average 3 or more infiltrations 14 days away.
Alcohol neurolysis has been widely used in treating neuropathic pain conditions such as incoercible trigeminal pain or pain related to cancer infiltration in these cases, if the patient does not respond to the therapy, surgical treatment is necessary.23–26 There are fewer studies about using alcohol sclerosing injections in treating neuropathic pain caused by peripheral nerves, such as lateral femoral cutaneous nerve and intercostal nerves.27,28
High concentrations of alcohol (up to 98%) have been used to induce endothelial damage in cystic disease and portal hypertension, leading to fibrosis.29–33
When injected around a nerve, alcohol produces chemical neurolysis by dehydration, necrosis, denaturation of proteins and precipitation of protoplasm, inducing Wallerian degeneration and inhibiting neurotransmission.24,33,34 In histological examinations of primarily excised MNs, previous studies found fibrosis of perineural and epineural tissue with degenerative vascular changes, axonal demyelination and arrested axonal nerve endings.3,35–37 Histological findings in surgical specimens of non-responder MNs after alcohol sclerosing injection revealed adhesions between neuroma and subcutaneous tissue, intra and perineural fibrosis, reduced cellularity, presence of sclerosis with hyalinosis, pyknosis, clear cytoplasm and apoptosis with ghost cells, demonstrating a partial damage of the nervous tissue caused by the previous alcohol infiltration.38–40
However, Mazoch et al., 41 in an experimental study conducted on the rat sciatic nerve used as a model for MN, did not find any significant histological changes in the nerve, nor perineural and muscular tissue, after injections of solutions of 4%, 20%, and 30% alcohol mixed with a local anaesthetic, when compared to a marcaine control injection group, and raised questions about the efficacy of alcohol injections. Contrary to findings from previous studies, recently DeHeer et al. 42 showed histologic changes in perineural and intraneural fibrosis in MN with the use of a 4% ethanol sclerosing injection.
The alcohol sclerosing injection therapy in treating MN gained popularity since the study of Dockery in 1999, in which 100 adult patients affected by MN received from 3 to 7 injections of 0.5 ml of a 4% alcohol sclerosing solution through a 27-gauge needle in the intermetatarsal space involved at intervals of 5 to 10 days, with 89% success rate, without any evidence of complication, but a temporary sensation of increased symptoms after the first injection occurred in the first 48 h and decreased sharply thereafter. 14
In 2004, Fanucci et al. 34 described, in a series of 40 patients, moderate pain that disappeared after 3–4 days, at the site of anaesthesia in 23 cases and a transitory plantar pain due to leakage of fluid in 12 cases, with no major complications, using 0.5 ml of a 30% ethylic alcohol solution through a 22-gauge needle. In 2007 equally, Hughes et al. 40 reported that 17 of 101 patients complained of plantar pain, which decreased after a mean of 4.5 days (from 2 days to 3 weeks post-injection), using 0.5 ml of a 20% ethylic alcohol solution. In the same year, Mozena and Clifford 43 declared that 3 of 42 patients had minor complications after treatment with 0.5 ml of a 4% dehydrated alcohol solution: 2 complained of pain resolved within the first 24 h and 1 had erythema around the injection area, resolved within 2 days.
In 2012, Musson et al. 44 referred that 3 of 92 patients treated with 0.5 ml of a 20% ethylic alcohol solution through a 23-gauge needle reported complications related to the injections: 1 patient developed symptoms suggestive of an allergy, 1 patient referred unbearable pain and stopped the course after one injection and 1 patient had prolonged pain and swelling until 1 week after the third injection.
In 2013, Gurdezi et al., 45 resuming Hughes’s work of 2007, reported that of 60 patients, 9 patients reported immense pain at the time of injection, 3 had bruising at the injection site, 2 complained of ongoing numbness of the toes, concluding that, despite the idea of injection therapy as a less traumatic option, one-third of their patients referred intense pain and should be well informed of this.
In 2019, Samaila et al. 46 referred that of 115 consecutive patients affected by MN and treated with injections of 3 ml of a 6% phenol solution, 1 patient showed osteonecrosis of the third metatarsal head 2 years after the last alcoholization, with unclear correlation with the treatment, and no major complication or skin necrosis were observed.
Other studies reported a few complications in line with literature with intolerable transient pain, 47 mean local inflammatory reactions and short-living pain at the moment of injection.38,39
Finally, we recommend maximum attention during the injection procedure, especially when removing the needle; the use of small-scale diameter needles and the injection of the minimum effective amount of solution may also help to prevent the above complications. The necrosis of the superficial tissues at the site of injection after the procedure could depend on excessive intra and perilesional resistance. The last could have led to the need for greater injection pressure, with subsequent retrograde extravasation of solution and damage to the dorsal tissues of the foot. Furthermore, ineffective timing during the procedure could lead, especially when the needle is being removed, to extravasation of the solution and linkage effect.
Conclusions
This current study calls into question the fact that alcohol sclerosing injection therapy for MN can determine only minor complications. In our experience, according to the literature, the most common referred complication is temporary pain at the site of injection, usually resolved within the first 48 h. In most cases, it led to a momentary exacerbation of pain at the site of injection and to a ‘pebble feeling’ between the third and fourth metatarsal head, which spontaneously receded in a few hours; however, in a small number of patients, it was burdened by a ‘major’ complication. Although it is a rarity, patients and surgeons should be aware not only of the risk of transient increased pain immediately following the injection but also of the risk to develop necrotic complications. We believe in the importance to share our experience with this severe complication with the surgeons who approach this treatment.
Footnotes
Authors contribution
All authors have made substantial contributions to the conception and design of the study, acquisition of data, analysis and interpretation of data, drafting of the article and revising it critically for intellectual content, and final approval of the version to be submitted.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
