Abstract
Objectives
The present study aimed to investigate the association of disc degeneration with low back pain and the effect of ageing on disc degeneration in low back pain versus asymptomatic individuals.
Methodology
A total of 50 patients with chronic low back pain (Group A) were compared to 25 healthy controls (Group B). Both the groups were subjected to magnetic resonance imaging of the lumbar spine and the disc degeneration was measured by the Pfirrmann grading system. The study group (Group A) was further sub-grouped into A1 (lumbar spondylosis, n = 11), A2 (mechanical back pain, n = –13) and A3 (lumbar disc herniation, n = 26).
Results
There was a statistically significant difference in degeneration at the L4-L5 (p = 0.001) and L5-S1 disc levels (p = 0.001) between the two groups and contiguous disc involvement was more in low back pain patients. The subgroups of group A showed no statistically significant difference. Age showed a strong positive correlation (r > 0.5) at all the lumbar levels (except at the L5-S1, r = 0.487) with Pfirrmann grading in Group A (p = 0.001). There was a weak positive correlation (r = 0.414) between age and Pfirrmann grade in the controls (p = 0.04) at the L4-L5 level only.
Conclusions
Significantly higher Pfirrmann grading on magnetic resonance imaging was found at the L4-5 and L5-S1 levels in symptomatic patients suggesting higher involvement of these levels. Progressive disc degeneration is seen with ageing but in patients with low back pain, it is significantly accelerated.
Introduction
Low back pain (LBP) is a common clinical complaint for which many patients visit health care facilities. There is a lot of health care resources consumption involved in the evaluation of LBP patients by use in the form of computed tomography (CT) and magnetic resonance imaging (MRI) scanning, leading to economic burden.1–3 The standard treatment options commonly fail due to the absence of a definitive cause in most patients.
MRI is the most commonly used modality for the assessment of LBP patients and can specifically assess the intervertebral disc degeneration (DD) patterns. DD is seen as a reduction in the signal of the nucleus pulposus and the inner fibres of the annulus. 4 Many asymptomatic individuals with MRI scanning can have DD in their imaging, thus limiting the diagnostic importance of this finding.5–8 There is a widespread belief that DD has a role in the causation and chronicity of disease.9,10 However, it still continues to be a topic of immense controversy. Considering the high volume of patients having this finding even in the asymptomatic stage, it is pivotal to study the lumbar DD pattern and its association with respect to various pathological causes leading to LBP. This is of importance to health care providers as well as patients to know the importance of degenerative findings in MRI. The present study aimed to investigate the association of DD with LBP and the effect of ageing on DD in LBP versus asymptomatic individuals.
Methodology
This prospective study was conducted at the author's tertiary health care centre in the Department of Orthopaedics in collaboration with the Department of Radiodiagnosis from August 2017 to March 2018. Informed written consent was taken from all the study participants for inclusion in the study. The study was approved by the Institutional Review Board and the ethical committee. Fifty patients of either sex aged between 18 and 50 years with LBP of minimum 3 consecutive months duration (chronic LBP, CLBP) and who give consent to participate in the study were enrolled as a study group (Group A). Symptomatic patients who did not give consent for enrollment in the study, contraindications for MRI, and patients planned for the surgery for severe neurological deficit and neurological claudication were excluded. Patients included in this study were cases of lumbar spondylosis (lumbar canal stenosis, degenerative disc disease), mechanical back pain and lumbar disc herniation (LDH). The study group (Group A) was further subgrouped into A1 (lumbar spondylosis, n = 11), A2 (mechanical back pain, n = 13) and A3 (LDH, n = 26) on the basis of clinical examination and MRI. Patients included in the lumbar spondylosis (A1) subgroup had degeneration of vertebrae and/or discs radiologically and were clinically symptomatic. The mechanical back pain subgroup (A2) included patients where no specific cause of pain could be attributed clinico-radiologically and complained of non-radiating pain in the lower back aggravated by activity. The majority of the patients with LDH (A3) presented with both sciatica and back pain. This subgroup division of group A was done to evaluate the correlation of DD with the common spinal pathologies. A total of 25 healthy volunteers with no history of back pain were also enrolled randomly as a control group (group B).
Each patient was thoroughly examined clinically and MRI was done on a 1.5 T machine with the patient lying supine comfortably with knees and hips extended. Multislice sagittal and transverse sections were taken with the following MRI sequence:

Locations of vertebral disc levels for MRI were determined on a pilot parasagittal section. DD was graded with Pfirrmann classification of DD. 11
Statistical analysis
For normally distributed data, a t-test was employed for analysis. Mann–Whitney and χ2 tests were employed for categorical and ordinal data. Correlation between variables was assessed by Pearson's coefficient of correlation. Statistical software SPSS version 25.0 was employed for analysis. 12
Results
The demographic parameters of the groups are depicted in Table 1. Both groups (A and B) and subgroups of group A (A1, A2 and A3) were comparable in terms of age, sex, weight, height and body mass index. Patients of the study population (group A) presented with a mean duration of symptoms of 11.93 ± 12.62 months. The most common clinical sign was tenderness (n = 23, 46%) and paraspinal muscle spasm (n = 47, 94%) in group A. The maximum number of patients had symptoms for less than a year (n = 39, 78%).
Comparison of demographic profiles of the study group (group A) and control group (group B).
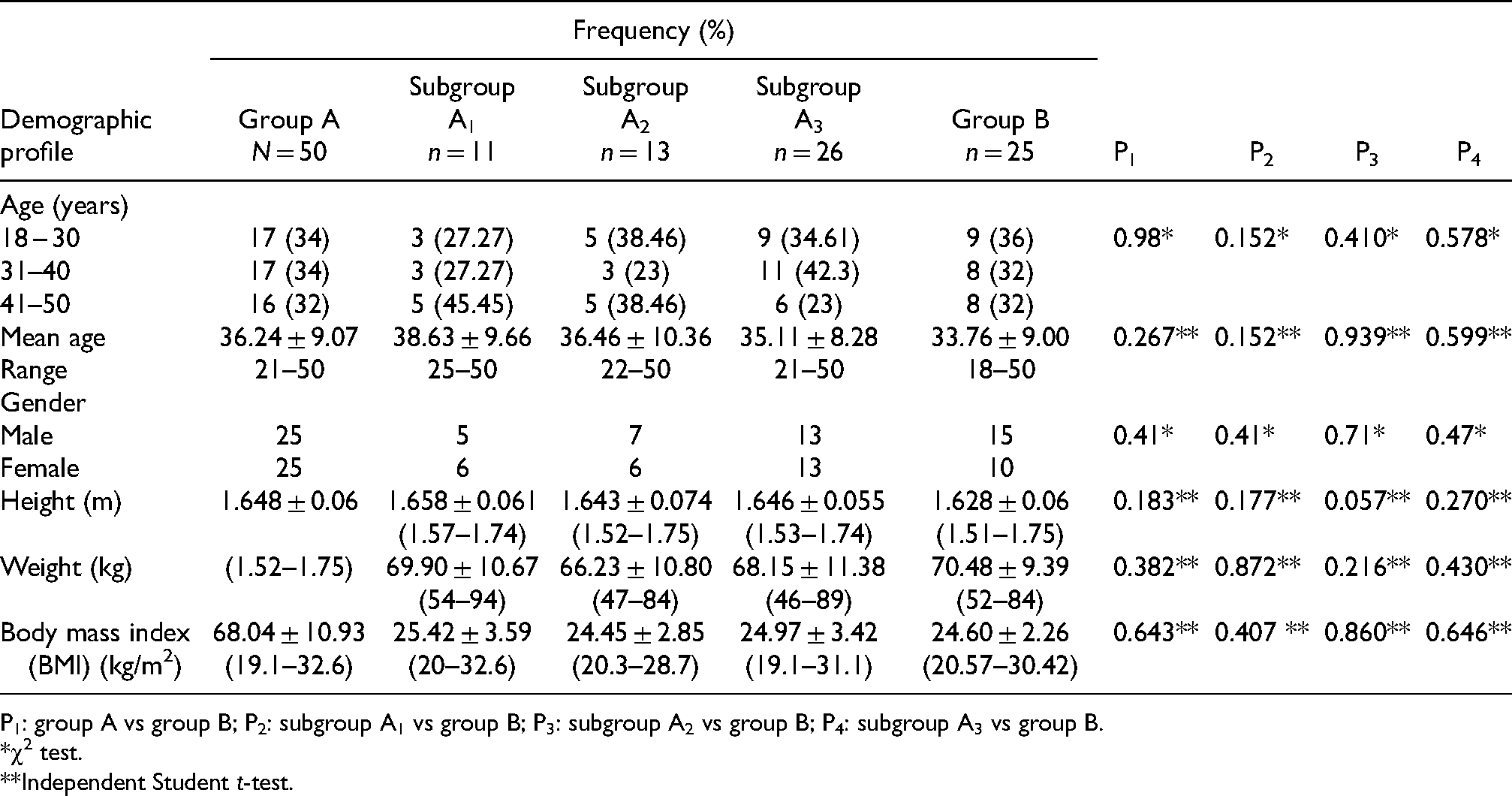
P1: group A vs group B; P2: subgroup A1 vs group B; P3: subgroup A2 vs group B; P4: subgroup A3 vs group B.
χ2 test.
Independent Student t-test.
Table 2 depicts the Pfirrmann grading of groups A and B, respectively, at various lumbar disc levels. Patients with LBP showed more DD. The comparison of MRI measurement of Pfirrmann grade at various lumbar spine disc levels between groups A and B is shown inTable 3. There was a statistically significant difference in degeneration at the L4-L5 (p = 0.001) and L5-S1 disc levels (p = 0.001) and contiguous multilevel disc involvement was more in LBP patients. Comparison of MRI measurement of Pfirrmann grade at various lumbar spine disc levels in subgroups of group A (A1, A2 and A3) showed no statistically significant difference (Table 4).
Pfirrmann grading at each lumbar disc level in the study (group A) population and in the control (group B).
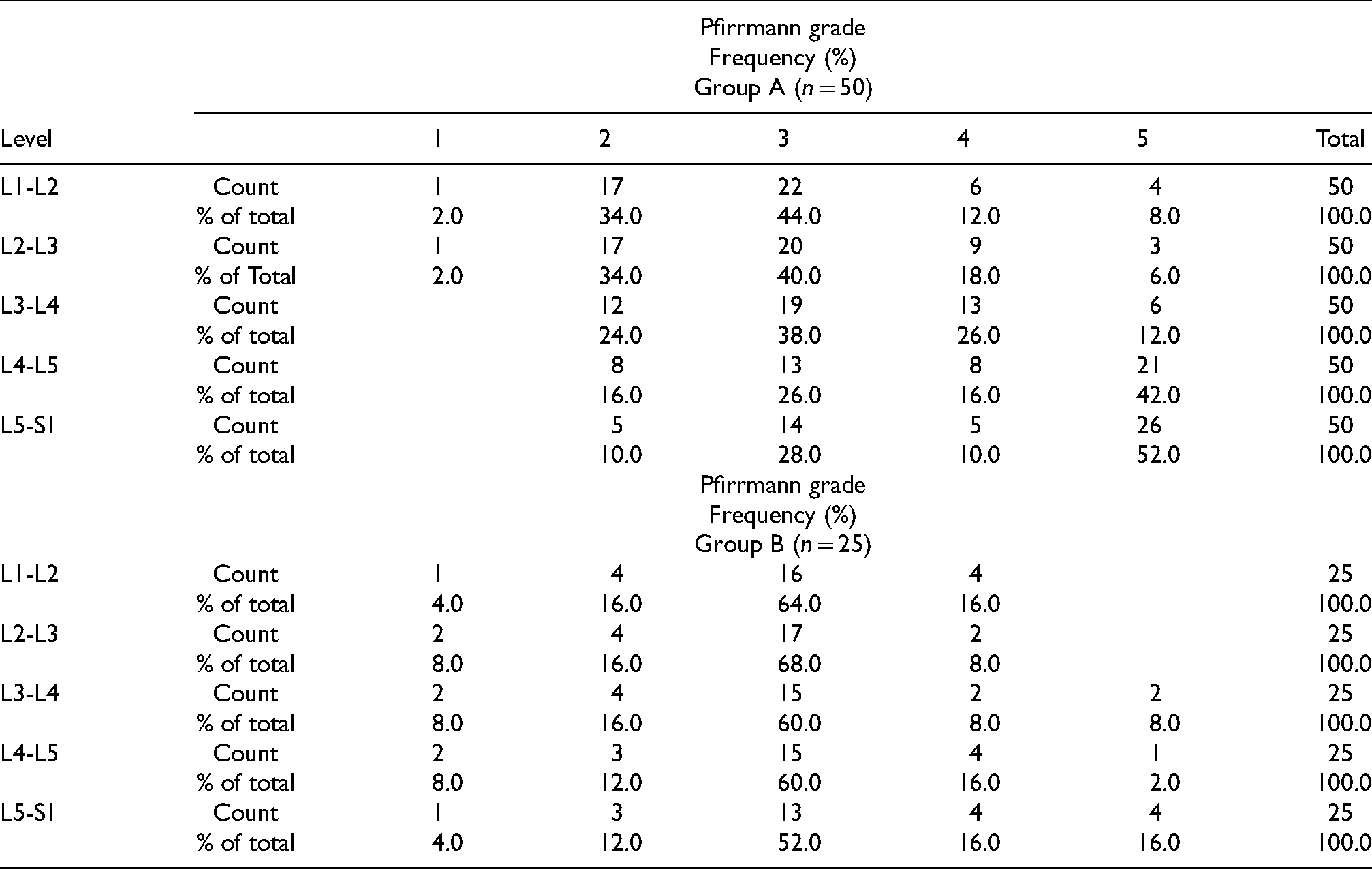
Magnetic resonance imaging measurements and comparison of average Pfirrmann grade at the L1-L2, L2-L3, L3-L4, L4-L5 and L5-S1 disc levels between groups A and B.
p value calculated from an independent t-test.
Magnetic resonance imaging measurements and comparison of average Pfirrmann grade at the L1-L2, L2-L3, L3-L4, L4-L5 and L5-S1 disc levels among subgroups (A1, A2 and A3) of the study population.

Calculated from independent t-test P1 = p value from independent t-test for subgroups A1 and A2.
P2 = p value from independent t-test for subgroups A2 and A3; P3 = p value from independent t-test for subgroups A1 and A3.
The correlation between age and Pfirrmann grade in both study and control population at each lumbar disc level is depicted in Table 5, which shows a strong positive correlation (r > 0.5) at all lumbar levels (except at the L5–S1 level where weak correlation was there, r = 0.487) between age and Pfirrmann grade in the study (group A) population (p = 0.001). There was a weak positive correlation (r = 0.414) between age and Pfirrmann grade in the control (group B) population (p = 0.04) at the L4-L5 level only.
Correlation between age and Pfirrmann grade in both study (group A) and control population (group B) at each lumbar disc level.
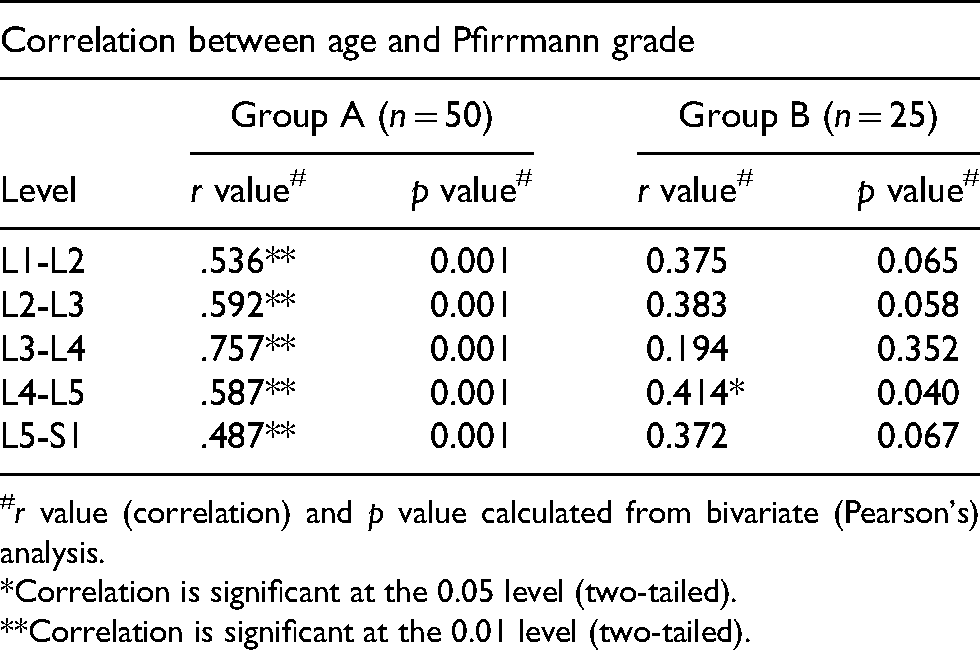
r value (correlation) and p value calculated from bivariate (Pearson's) analysis.
Correlation is significant at the 0.05 level (two-tailed).
Correlation is significant at the 0.01 level (two-tailed).
Discussion
Lumbar intervertebral DD is a dominant factor in the aetiology of LBP. DD is an age-related process and mechanical loading has been identified as a major extrinsic component in the onset and progression of intervertebral DD.13,14 The normal orientation and morphology of the lumbosacropelvic structure play an important role in the determination of shear and compressive forces applied on the anterior (corpus vertebrae and intervertebral discs) and posterior (facet joints) elements of the lumbar vertebral column. 15 In the present study, we evaluated DD to find their correlation with LBP and ageing.
Some studies in the literature do not support the correlation between DD and LBP.16,17 However, a recent systematic review and meta-analysis revealed that MR imaging evidence of disc bulge, degeneration, extrusion, protrusion, Modic 1 changes and spondylolysis were more prevalent in adults ≤ 50 years of age with back pain compared with asymptomatic individuals. A strong association of DD and LBP in individuals 50 years of age or younger was reported. More than 30% of asymptomatic individuals and >50% of symptomatic individuals had DD on MRI. 18 In the present study, also we noted a higher Pfirrmann grade of DD in the group A population (CLBP) compared to the control population (group B) specially at the L4-L5 and L5-S1 levels (p = 0.001). We agree with Zheng and Chen 10 that the fast dynamics of pain recurrence emerge in succession upon the foundation laid by the DD. DD should be considered as one of the potential causes of pain in the context of the patient's clinical condition. 18 However, there is a need to distinguish age-related degenerative changes from pathologic, pain-generating degenerative changes at a more advanced level of pain genetics.18,19
The prevalence of DD increased from ∼70% of individuals <50 years of age to >90% of individuals >50 years of age in a cross-sectional study having both symptomatic and asymptomatic participants. 20 We also noted progressive DD with increasing age in the form of Pfirrmann grade at all lumbar levels (strongly positive correlation, r > 0.5) in patients with CLBP (group A) (p = 0.001). There was also progressive DD by Pfirrmann grade with the increasing age at all lumbar levels in the control population (group B), but it was not statistically significant (p < 0.5) except at the L4-L5 level [weak positive correlation, r = 0.414, (p = 0.040)]. It is anecdotal that with ageing DD occurs, but the accelerated DD is observed in LBP patients. Leung et al. 21 reported progressive degenerative change in discs with increasing age. Buckwalter 22 also reported DD with progressive increase in age. The findings of the present study corroborate this, as there was a strong positive correlation (r > 0.5) of DD with ageing in patients with LBP than the age and sex-matched controls (weak positive correlation, r < 0.5) (Figures 1 and 2). The underlying mechanism of this differential effect of ageing in LBP patients needs to be elucidated. The mean Pfirrmann grade was higher showing increased DD in CLBP patients (group A) at almost all lumbar levels, but only statistically significant at the L4-L5 and L5-S1 (p = 0.001). Most of the patients with CLBP (group A) and control (group B) showed DD grade of Pfirrmann grade 3. Pfirrmann grade 5 DD was mostly present at the L4-L5 and L5-S1 levels in group A patients. L4-L5 disc had been reported to be the most degenerated disc, and the same had been observed in the present study. 23 Takatalo et al. 24 also using MRI and Pfirrmann classification reported an association between intervertebral DD and severity of LBP symptoms. They also reported that moderately degenerated discs were more likely than mildly degenerated discs to be associated with the most severe low back symptoms. The mean Pfirrmann grade was higher in lumbar spondylosis (group A1) among the subgroups at all lumbar levels, but it was not statistically significant. We agree with the literature findings that the mere presence of degenerative disc is not synonymous with pain. However, it is the specific degeneration patterns and severity that may foretell a pain risk profile, as had also been observed in the present study. 19 A study by Ogon et al. 25 had shown that the T2 values (quantification of moisture content and collagen sequence breakdown) for discs tended to be lower in the CLBP group than in the control group, and these values were significantly different within the posterior annular fibrosus. Forell et al. 26 reported that various patterns of DD throughout the lumbar spine may affect motion and soft-tissue structures as well that may have a bearing in the clinical pathway of pain generation.
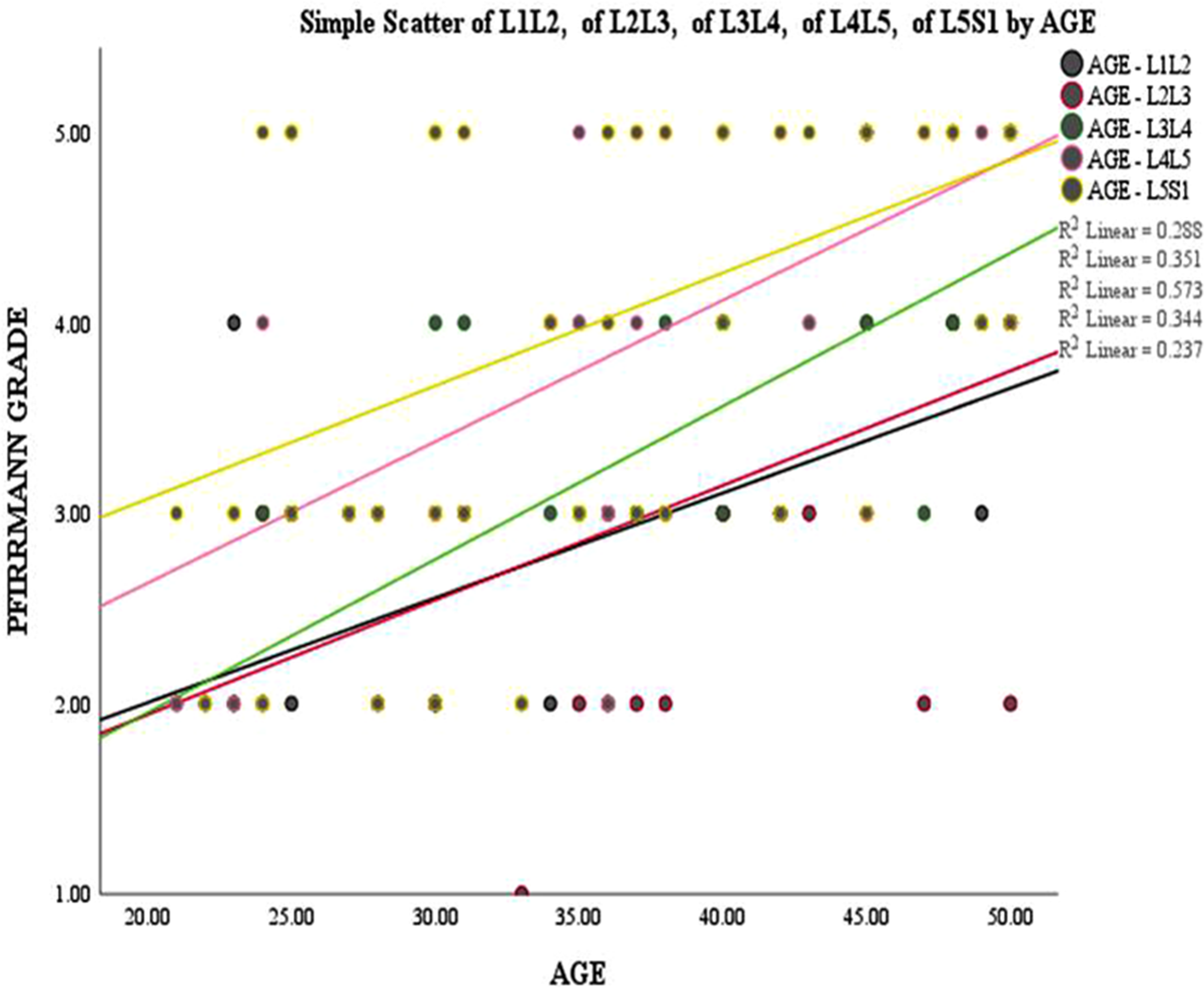
Correlation of age with Pfirrmann grade of study population (group A).
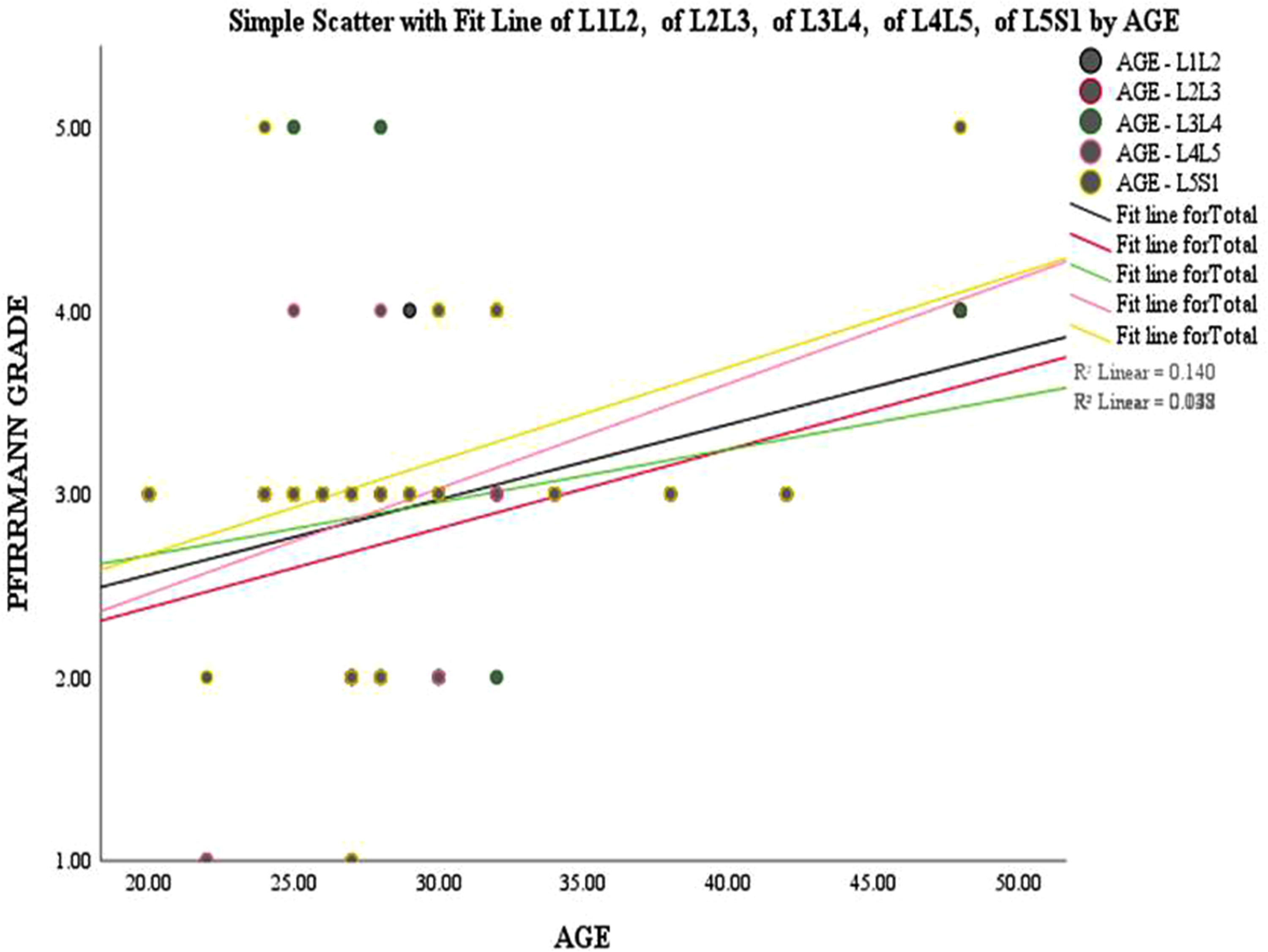
Correlation of age with Pfirrmann grade of control population (group B).
One of the salient findings of the present study was that there was no statistically significant difference in the Pfirrmann grading among common lumbar spine diseases except that the mean Pfirrmann grade was higher in lumbar spondylosis. It makes us believe that DD may be the common underlying cause leading to pain in different lumbar pathologies. However, the severity, pattern, and number of degenerated levels may be different in these diseases. The clinical relevance of the findings of the present study is that physicians, researchers and physiotherapists should work on the modalities to reduce this accelerated degeneration in LBP patients.
The present study has a limitation of a small sample size without power analysis. Statistically proven results and the unique nature of the study are the strength of our study
Conclusions
Significantly higher Pfirrmann grading on MRI was observed at the L4-5 and L5-S1 levels in symptomatic patients suggesting higher involvement of these levels. Progressive DD was seen with ageing but it was significantly accelerated in patients with CLBP. The current study's findings show that DD acts as a scaffold pathology in the spine, allowing numerous other CLBP-causing pathologies to build on top of it.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
