Abstract
The formation of a pseudoaneurysm or ‘false’ aneurysm is a rare vascular complication of arthroscopic surgery. Its occurrence is even more uncommon in the background of a clotting disorder. We present a case of a recreational athlete with a history of haemophilia, who developed a pseudoaneurysm over an arthroscopic portal site following arthroscopic-assisted anterior cruciate ligament reconstruction. The lesion was treated successfully with image-guided thrombin injection. We discuss the association between haemophilia and pseudoaneurym formation as well as prevention methods and treatment options.
Keywords
Introduction
Arthroscopies are widely performed orthopaedic procedures. One of the rare complications is the formation of a pseudoaneurysm. This can occur following arthroscopy of any joint. When there is a breach in vessel wall, localized extravasation may occur. Treatment can be either radiological or surgical, depending on the size of the lesion. However, the management of this uncommon condition may be further complicated by a pre-existing clotting disorder.
Case report
A 23-year-old male with a known history of haemophilia A suffered a left knee sprain during a recreational soccer match. His left knee was subject to a sudden pivoting force, resulting in an audible pop, followed by swelling, bruising and persistent instability for 3 weeks.
On physical examination, his left knee showed no effusion. Active range of motion was 0–130°. Lachman’s test and anterior drawer test were positive with no end point. Pivot shift test was positive. There was no posterior sagging, and the medial and lateral collateral ligaments were stable. McMurray’s test was negative.
Magnetic resonance imaging of his left knee was performed, and a low-grade partial tear of the anterior cruciate ligament (ACL) was reported. There was also a complex tear of the medial meniscus.
In view of the patient’s signs and symptoms being consistent with an ACL-deficient left knee, arthroscopic-assisted ACL reconstruction was offered. Haematology was consulted for perioperative optimization of the patient’s clotting disorder. An infusion of 2500 U of factor VIII prior to induction was suggested, followed by 1500 U every 12 h for a minimum of 5 days.
Standard blood tests were carried out preoperatively, which included a clotting profile. At the time, the patient’s activated thromboplastin time (APTT) was 61.7 s (reference range 27.4–37.4 s), international normalized ratio was 0.95, prothrombin time was 10.4 (reference range 9.5–12 s) and platelet count was 230 × 109 L−1. An upper limit for preoperative serum APTT was not specified by the haematologists. The patient proceeded to surgery with the recommended perioperative factor VIII regimen.
Intraoperatively, a complete tear of the ACL was found. The reconstruction was performed with a single bundle hamstring graft. Intraoperatively, a posterior horn longitudinal tear of the lateral meniscus was noted and repaired with an all-inside technique. Meticulous haemostasis was ensured throughout the procedure. At the end of the procedure, the tourniquet was deflated after dressing and crepe bandaging was applied. The patient was discharged on post-operative day 5 and only after completion of a course of factor VIII infusion. Upon discharge, there was no knee pain or ecchymosis.
At the post-operative 2 weeks’ follow-up, his surgical wounds had healed with no signs of infection. There was mild effusion of his left knee, and his range of motion was 10–100°. We removed the sutures and continued his physiotherapy according to the standard ACL rehabilitation protocol.
At the post-operative 6 weeks’ follow-up, a non-tender scar was noted at the anteromedial portal site. The range of motion of his knee had improved to 0–130°.
At the post-operative 10 weeks’ follow-up, a firm nodule approximately 1 cm in size was in the place of the previous anteromedial portal scar (Figure 1(a) to (c)). The nodule was non-tender and initially resembled a calcified nodule. Plain radiographs revealed no calcifications. An ultrasound (Figure 2(a) and (b)) and contrast computed tomography (Figure 3(a) to (d)) revealed a 1.3-cm vascular pulsatile lesion at the anteromedial knee joint with overall features suggestive of a small pseudoaneurysm with slow flow. The feeding vessel was suspected to arise from the branches of the medial inferior geniculate artery.
In view of these findings, the patient was treated with ultrasound-guided thrombin injection at post-operative 24 weeks (Figure 4).
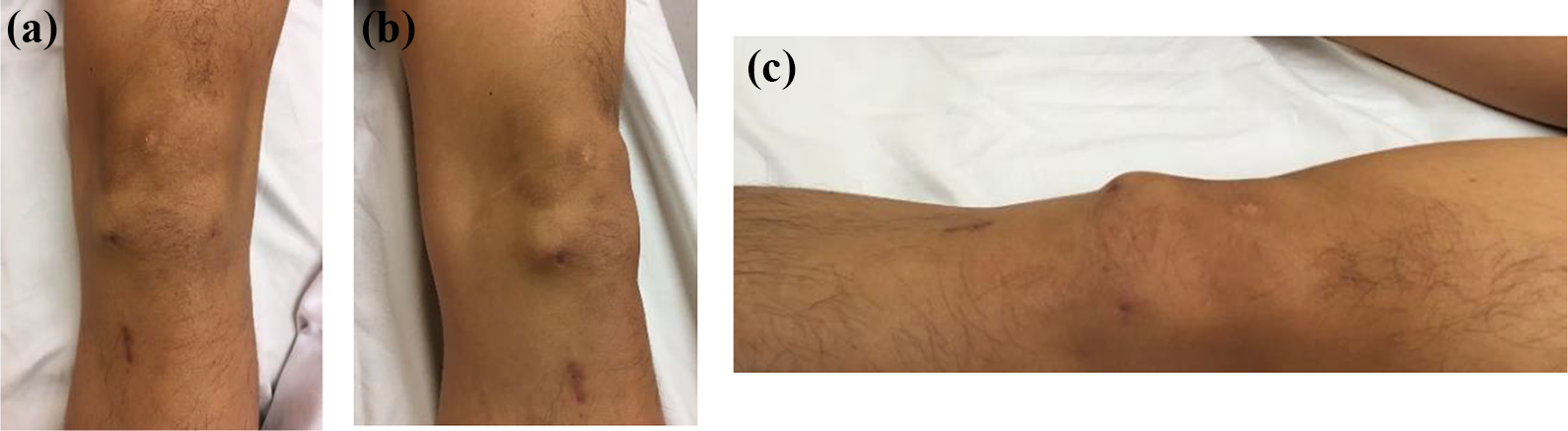
Clinical photographs showing the (a) frontal view, (b) oblique view and (c) lateral view of the pseudoaneurysm over the anteromedial portal scar of the left knee prior to treatment.
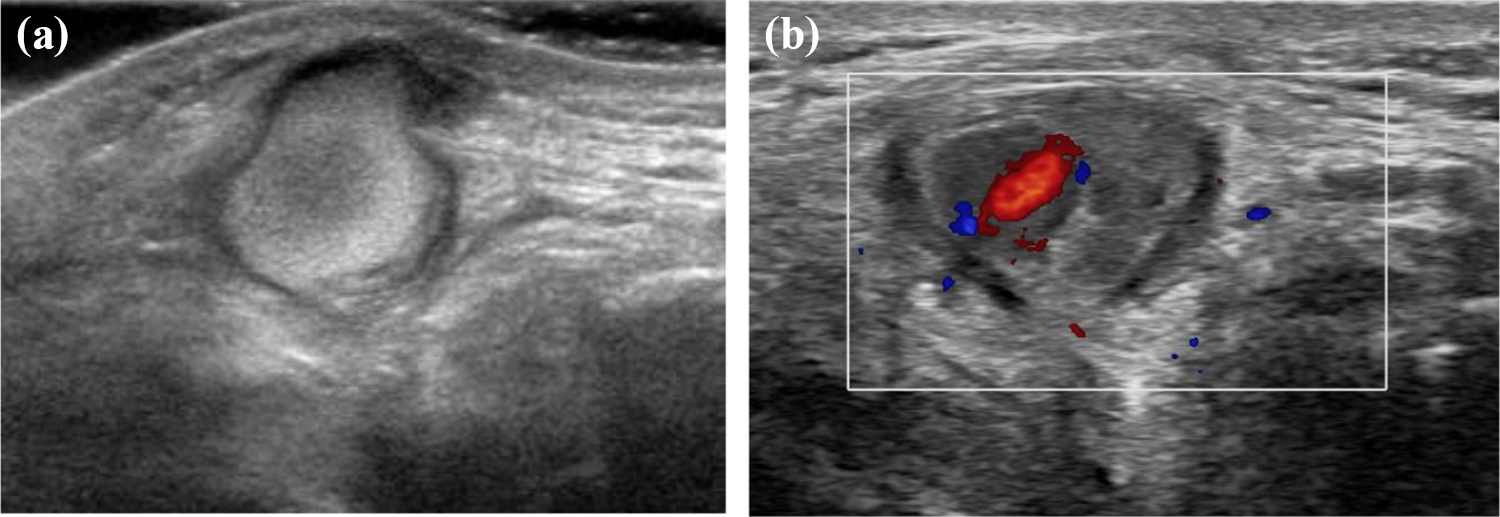
(a) B-mode and (b) Doppler ultrasonography of the pseudoaneurysm. In B-mode, the aneurysm sac is isoechoic due to the slow turbulence flow within the sac. The small focal ‘yin-yang’ sign in colour mode within the slow flow indicates bidirectional flow due to swirling of blood within the aneurysm.
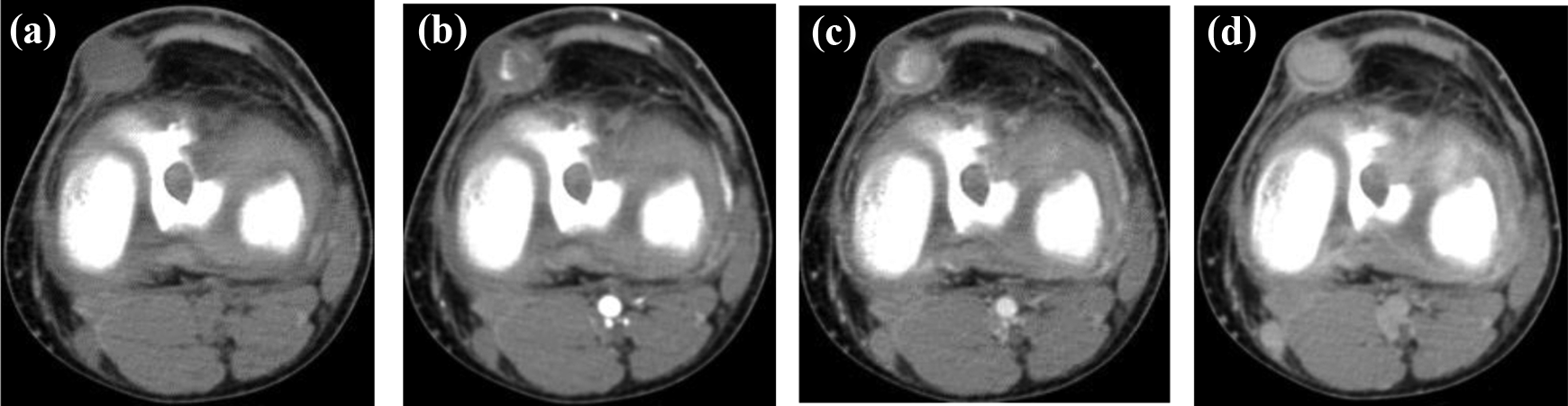
Axial computed tomography with contrast showing the (a) non-enhanced phase, (b) early arterial phase, (c) late arterial phase and (d) delayed phase. The patent portion of the lumen of the aneurysmal vessel demonstrates increased attenuation, due to contrast enhancement, whereas a part of the lumen shows non-filling due to mural thrombus.
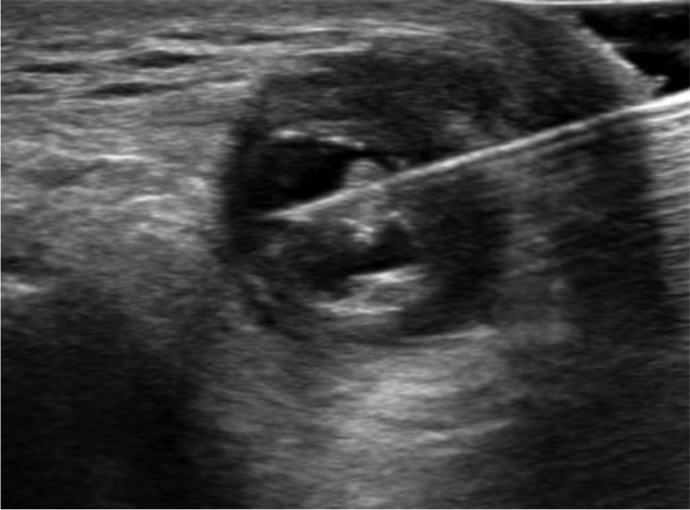
Ultrasound image at the time of the first thrombin injection.
However, 3 weeks after the thrombin injection, a 1-cm swelling was visible over the anteromedial scar. The patient reported that this swelling had initially subsided after the injection but subsequently reappeared in the following days. A recurrence of the pseudoaneurysm was clinically suspected and confirmed by a follow-up ultrasound that showed a residual slow flow cavity present at the deeper part of the pseudoaneurysm (Figure 5).
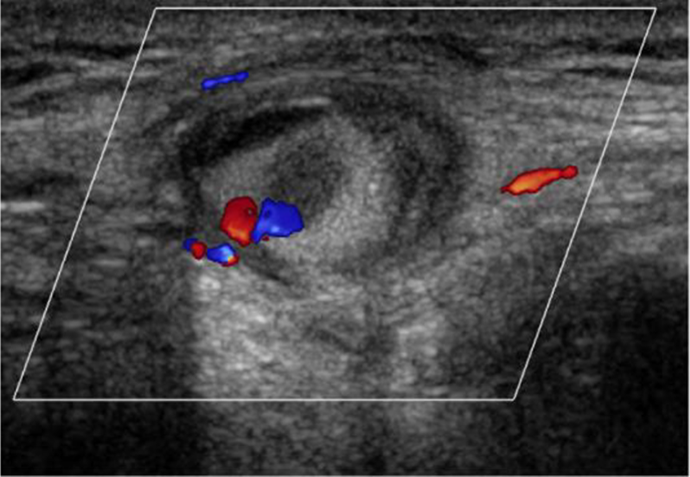
Doppler ultrasound after the first thrombin injection, showing persistence of the small ‘yin-yang’ sign close to the neck while remaining part of the sac is thrombosed.
The patient was inclined to repeat the procedure, and thus, a second ultrasound-guided thrombin injection was performed 6 weeks after the initial injection. As the patient suffered from only mild haemophilia, he has been trained to self-administer factor VIII intravenously when needed (e.g. in the event of minor gum bleeding). Routine monitoring of post-administration APTT levels was not required. Therefore, prior to the second thrombin injection, the patient was instructed to self-administer his regular dose of 1000 U of factor VIII intravenously to ensure the sufficiency of clotting factors in his blood. Afterwards, 1000 U of thrombin were injected into the residual cavity of the pseudoaneurysm (Figure 6). To facilitate haemostasis, crepe bandaging was applied post-procedurally, as was done after the first thrombin injection. No additional factor VIII was given immediately afterwards, and subsequent self-administration followed the patient’s normal protocol. Follow-up ultrasonography demonstrated a thrombosed sac with only a small stalk remaining (Figure 7).
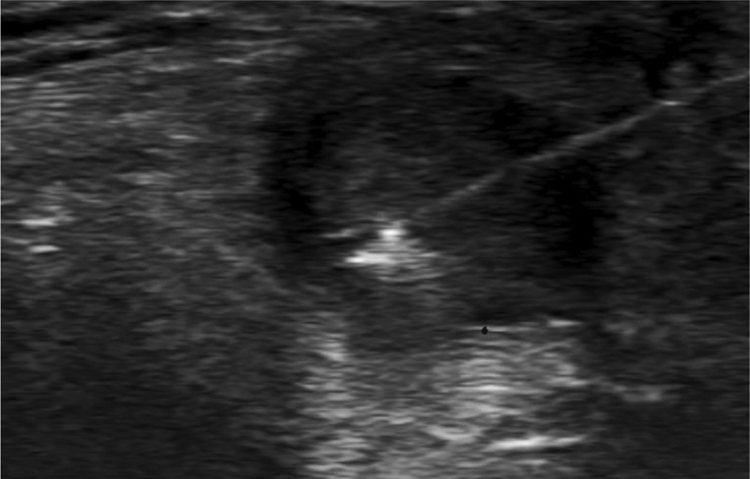
Ultrasound image at the time of the second thrombin injection.
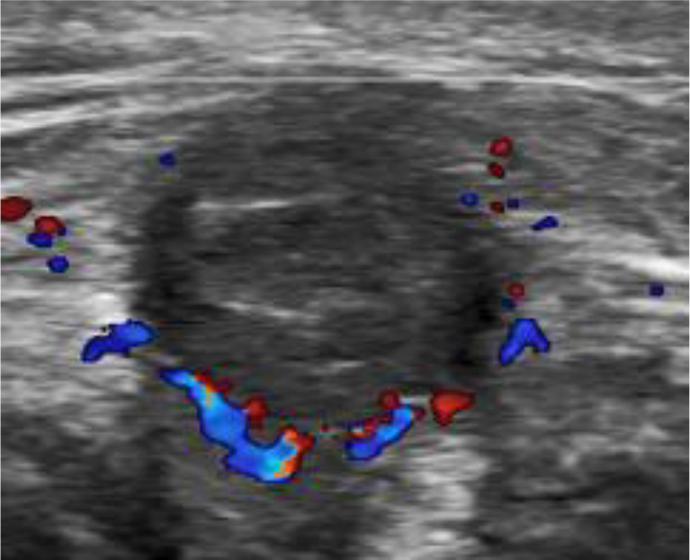
Interval Doppler ultrasound after the second thrombin injection, showing an absence of the ‘yin-yang’ sign or any flow within the sac.
By post-operative 35 weeks, the clinical swelling over the anteromedial portal had resolved.
Discussion
Pathophysiology
The formation of a pseudoaneurysm, or a false aneurysm, is caused by a perforation of all three layers of the vessel wall, leading to extraluminal extravasation of blood that is contained within a fibrous capsule. 1 The aneurysm is ‘false’ because unlike a true aneurysm, it does not consist of the three layers normally found in vessel walls. The factors contributing to the formation of pseudoaneurysms can be broadly divided into those that incite endothelial damage and those that promote the extravasation of blood.
Endothelial damage in the form of penetrating surgical trauma may be brought on by the initial stab wound incisions, dissection with forceps, or from the introduction of the trocar. Since the branches of the geniculate arteries have a small cross-sectional diameter, it is more common that surgical trauma results in complete transection of the branches with ensuing ecchymosis or haematoma. When there is a partial transection, an aneurysmal dilatation may occur. 2
Haemophilia A is a clotting disorder characterized by a deficiency or dysfunction of factor VIII. Factor VIII is involved in the intrinsic pathway of coagulation, which results in the prolongation of the APTT. Factor VIII is closely associated with the von Willebrand factor (vWF), which is involved in the stabilization of factor VIII, protecting it from rapid degradation in the plasma. Clinical manifestations depend on severity of this deficiency and range from prolonged bleeding after trivial injury to spontaneous haemorrhage. Treatment involves intravenous factor VIII replacement. Haemophilia and other clotting disorders predispose a patient to pseudoaneurysms by increasing the likelihood and ease of extravasation. Furthermore, it has also been proposed that a dysfunction in vWF production is linked with a deficiency in collagen III production, an essential component in the structural integrity of vascular basement membranes. 3
Incidence
Arthroscopy is generally considered a safe procedure with few complications. Vascular complications including portal pseudoaneurysm formation are uncommon. Two large North American series involving 118,590 and 375,000 arthroscopies reported incidences of pseudoaneurysm formation at 0.56% 4 and 0.80%, 5 respectively.
Portal pseudoaneurysms specifically following knee arthroscopy have also been reported, with lesions arising at various sites of the knee vasculature. There have been 22 cases arising from the popliteal artery and its branches, 10 occurring at the medial inferior genicular artery, 3 from the medial superior genicular artery, 3 from the lateral inferior genicular artery, 2 from the perforating artery of the medial gastrocnemius, 2 from the sural artery, 2 from the descending genicular, 1 from the recurrent tibial artery and 1 from the lateral superior genicular artery. 6
Post-operative pseudoaneurysms occurring in the background of haemophilia are rare. The incidence of post-operative bleeding complications for haemophiliac ranges from 3% to 6%, although the specific incidence of pseudoaneurysms in haemophiliac has not been reported. 7 In the literature, there have been at least 14 pseudoaneurysms reported in patients with haemophilia. 8 Nine of these cases were of musculoskeletal origin, of which, four had occurred in the knee. The majority of those occurring in the knee followed total knee arthroplasties. 8
Prevention
Haemophilia patients undergoing major surgery require factor infusions during the perioperative period. The general recommendations involve a preoperative bolus of recombinant factor VIII, followed by a course of 12-hourly infusions.
Preoperative assay of factor VIII, vWF titers and detection of inhibitors may also be of benefit with the addition of intravenous DDAVP (desmopressin) in the presence of low vWF levels. 9 Some authors also advocate continuous factor infusion with repeat monitoring of factor levels to achieve a factor VIII plasma level of 1 IU mL−1 until 2–3 days after surgery while omitting pharmacological thromboprophylaxis. 8 The risk of vascular injury may also be minimized preoperatively with duplex ultrasonography for arterial mapping. 10 Furthermore, it is prudent to ensure meticulous haemostasis intraoperatively and careful dissection of portal entry sites prior to trocar introduction to reduce the risk of post-operative haemarthrosis and vascular complications.
Treatment
In patients without clotting deficiencies, smaller pseudoaneurysms (<3.0 cm in size) have been reported to resolve spontaneously by thrombosis. 11 In other cases, the approach to intervention can be radiological or surgical. Radiological treatment offers an alternative to open surgery and its use is increasing in popularity. 11 There are percutaneous measures, such as ultrasound-guided compression or ultrasound-guided thrombin injection. There are also endovascular procedures, such as coiling or stent grafts. 11 These methods are generally less invasive, safer and often effective for selected cases. In the case presented, spontaneous thrombosis was unlikely to occur in the background of haemophilia, thus, ultrasound-guided thrombin injection was the treatment of choice. However, the first injection failed to control the lesion. This is because thrombosis by extrinsic thrombin hinged on having sufficient intrinsic clotting factors. Therefore, it may be postulated that the lack of pre-procedural replenishment of deficient clotting factors was the reason for failure of the first injection. The addition of prior factor VIII administration leads to the success of the second thrombin injection.
For the treatment of pseudoaneurysms in haemophiliac, it is advisable to first employ methods that are less invasive as there is always a risk further bleeding with more invasive techniques in a patient that is already susceptible. When minimally invasive radiological options have failed or when there is concomitant infection, ischaemia, or rapid expansion of the lesion, then surgical modalities such as ligation, excision or bypass grafting are indicated. 12
Of the 47 portal pseudoaneurysm cases following knee arthroscopy reported in the literature, the majority (75%) were treated by surgical excision, which remains the gold standard of treatment. 2,6 However, the lesions of the reported cases were generally larger in size and none of those patients had coexisting haemophilia. 2,6 The latency to diagnosis ranged from 1 to 180 days post-operatively, with a median of 11 days. 6 Moreover, in cases which involved higher calibre vessels such as the popliteal artery, additional grafting was required. 2 Therefore, given the size, location and nature of the lesion described in this report, our method of treatment had the advantage of being minimally invasive and tailored to the underlying clotting deficiency.
In summary, a case of a post-knee arthroscopy pseudoaneurysm in a patient with haemophilia was treated successfully with ultrasound-guided thrombin injection.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
