Abstract
Study Design
Systematic review of clinical studies.
Objective
To identify neurological, anatomical, and technical predictors of failure in closed cranial traction (CCT) for traumatic cervical facet dislocations (CFD) in adults, and to synthesize evidence to guide early surgical decision-making.
Methods
A systematic search was conducted across five databases: PubMed, PubMed Central (PMC), SciELO, Scopus, and Web of Science, for studies published from January 2000 to May 2025. Eligible studies included patients ≥16 years with traumatic CFD managed initially with CCT, reporting both success/failure rates and predictive variables. Data extraction focused on demographics, injury patterns, reduction techniques, and outcomes.
Results
Eight studies met the inclusion criteria, encompassing 631 patients. Overall, the success rate of closed reduction was 73.3% (463/631), ranging from 56% to 92%. Consistently reported predictors of failure included complete neurological deficit (ASIA A–B; four studies), absence of a contralateral perched facet, involvement of the C7–T1 level, inferior endplate fracture, and attempts without general anesthesia. When open reduction was required after failed CCT, posterior approaches achieved higher success rates than anterior approaches (100% vs 45%).
Conclusions
In adults with traumatic cervical facet dislocations, CCT is more likely to fail with complete neurological deficits (ASIA A–B), C7–T1 involvement, absence of a contralateral perched facet, and awake traction protocols; GA-first strategies showed higher success in available cohorts. Unlike prior technique-focused overviews, this review consolidates predictors of CCT failure and proposes a practical algorithm to triage patients for early open reduction.
Keywords
Introduction
Traumatic cervical facet dislocations (CFDs) represent a severe form of subaxial spinal injury, commonly resulting from high-energy mechanisms such as motor vehicle collisions or falls from height. These injuries are characterized by disruption of the posterior elements of the cervical spine, most frequently at the C5–C7 levels, leading to instability and risk of spinal cord compromise (Fischgrund, 1997). 1 CFDs can be unilateral or bilateral, often accompanied by perched (subluxated) or locked (luxated) facets, and may or may not be associated with fractures or herniated discs. The prompt restoration of spinal alignment is crucial to mitigate secondary neurologic damage (Fehlings, 2012). 2
In clinical practice, closed cranial traction (CCT) is widely used as the initial maneuver to achieve realignment. When successful, it may obviate the need for emergency open surgery and allow for staged anterior, posterior, or combined stabilization in a scheduled procedure (Liu, 2023). 3 The rationale is based on decompressing the neural elements before irreversible ischemic changes occur, restoring spinal biomechanics, and stabilizing the patient before definitive surgical fixation (Wilhelmy, 2025). 4
However, the reported success rates of closed reduction vary considerably across the literature, ranging from as low as 56% to over 90%, depending on the population, technique, and institutional protocol (Oae, 2023) 5 ; (Branche, 2018) 6 ; (Sousa, 2022). 7 Multiple factors have been hypothesized to influence the likelihood of failure: complete spinal cord injury (ASIA A–B), absence of contralateral facet mobility, presence of comminuted fractures, delayed intervention, and attempts performed in awake patients without muscle relaxation (Anissipour, 2017) 8 ; (Miao, 2018). 9 Yet, these factors have not been consistently validated across prospective or multicenter series.
From a clinical standpoint, failed closed reduction has several implications: prolonged instability, demanding more complex surgical procedures, delayed surgical decompression, increased risk of neurologic deterioration, and extended ICU and hospital stay (Potgieter, 2019). 10 Moreover, multiple traction attempts may provoke patient discomfort or complications such as worsening cord edema or iatrogenic displacement. For this reason, identifying pre-reduction features that predict failure can support earlier conversion to open surgery, particularly posterior approaches, and improve neurological outcomes (Fehlings, 2012). 2
Despite advances in spinal injury classification systems such as the Subaxial Injury Classification System (SLIC) and AOSpine modifiers, there remains a translational gap between morphologic descriptors and therapeutic decisions regarding reduction strategy (Schnake, 2017) 11 ; (Vaccaro, 2016). 12 Therefore, a critical clinical question persists: can specific clinical or imaging features reliably predict failure of closed traction?
We center this review on predictors of failed closed cranial traction (CCT), standardize a primary failure endpoint across studies, and translate consistent signals into a decision algorithm indicating when to bypass traction and proceed to posterior open reduction.
This systematic review addresses that question. We aim to synthesize existing clinical evidence to identify consistent predictors of failed closed reduction in adult patients with traumatic cervical facet dislocations. Our goal is to contribute a clinically actionable framework for early decision-making and resource optimization in acute spinal trauma.
Methods
This systematic review was conducted in accordance with the PRISMA 2020 guidelines (Page, 2021). 13 Its primary objective was to identify original clinical studies that evaluated predictive factors for failure of closed cranial traction in the management of traumatic cervical facet dislocations in adults. The core clinical question guiding the review was: “What are the risk factors for failure of cranial traction in reducing an acute traumatic cervical facet dislocation?”
Research question • Population: Adults (>16 years) with acute traumatic cervical facet dislocations • Intervention: Closed reduction with cranial traction • Comparison: Success vs failure of reduction • Outcome: Clinical, radiological, or procedural factors associated with failure
Search Strategy
A systematic search was conducted across five databases: PubMed, PubMed Central (PMC), SciELO, Scopus, and Web of Science. The search window covered articles published from January 1, 2000, to May 30, 2025, in English or Spanish. No filters for study design were initially applied. A combination of MeSH terms and free-text keywords was used, including: cervical facet dislocation, closed reduction, cranial traction, cervical spine injury, failure, predictive factors, neurological deficit, and adult. 14
A representative PubMed query was:
(“cervical facet dislocation” [Title/Abstract]) AND (“closed reduction” [Title/Abstract] OR “cranial traction”) AND (“failure” [Title/Abstract] OR “unsuccessful”) AND (“adult” [MeSH Terms]) AND (2000:2025[pdat])
Inclusion Criteria
• Original clinical studies (Oxford levels of evidence I–III) (CEBM, 2009)
15
• Adult patients (≥16 years) with traumatic cervical facet dislocations • Evaluation of the success or failure of closed reduction via cranial traction • Sample size ≥10 patients • Description of at least one associated predictive variable • Availability of full text or extended abstract
Study Selection
Titles and abstracts were screened independently by two reviewers (JT, AFJ), followed by full-text screening for eligibility. Disagreements were resolved through discussion with a second senior reviewer (AFJ). The entire process was documented in a PRISMA flow diagram (Page, 2021).
13
Figure 1. PRISMA 2020 flow diagram of study selection. PRISMA 2020 flow diagram for the updated search. We identified 691 records from databases. Before screening, 326 duplicates were removed (no automation-based or other removals), leaving 365 records screened. 336 were excluded at the title/abstract. We sought 29 full-text reports, all retrieved, and assessed 29 for eligibility. 20 reports were excluded (non-original studies, n = 6; absence of quantitative data, n = 5; irrelevant pathology, n = 3; inadequate methodological design, n = 6). 8 studies (8 reports) were included. Abbreviation: PRISMA = preferred reporting items for systematic reviews and meta-analyses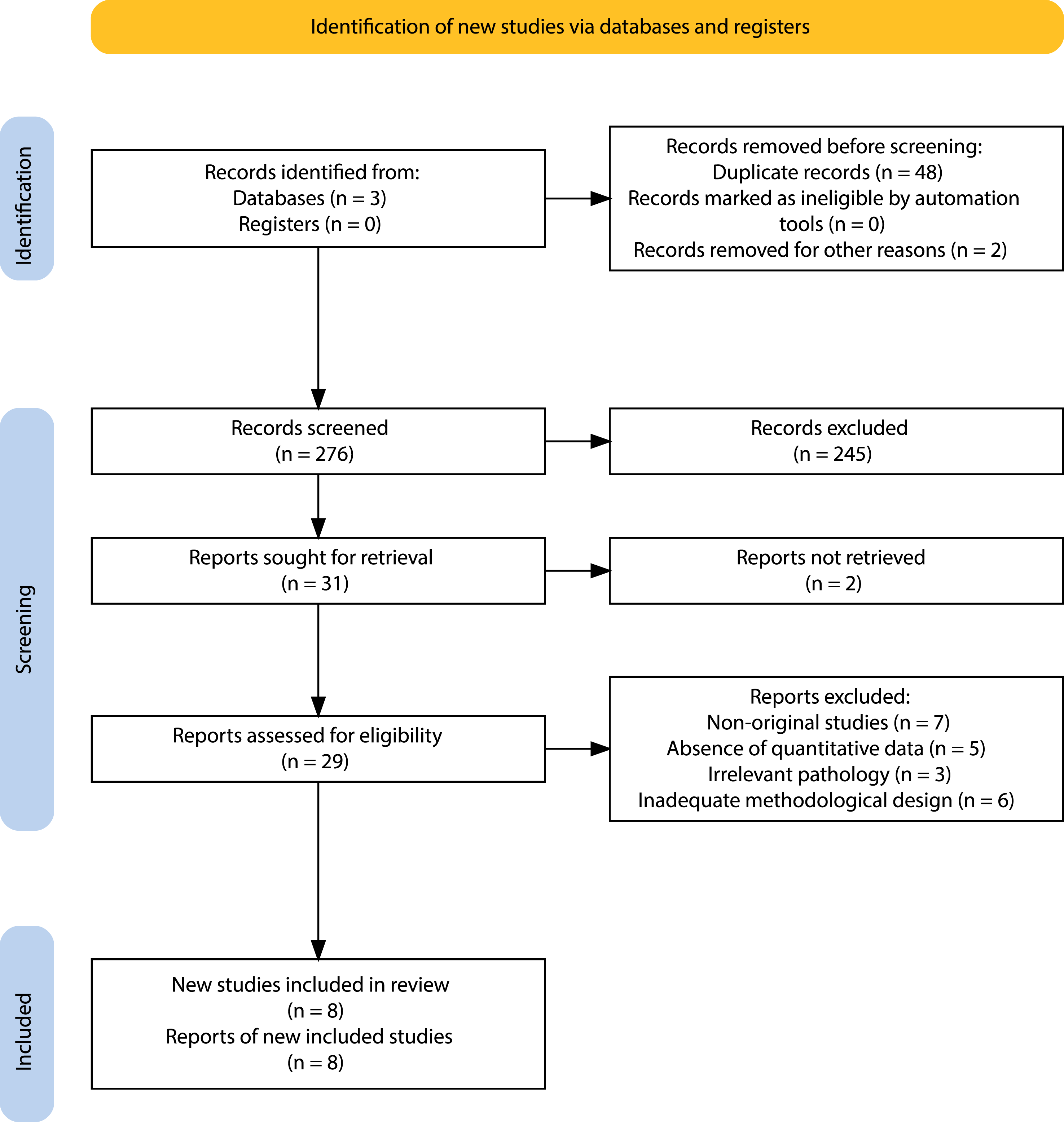
Data Extraction
A standardized data collection sheet was developed to extract the following items from each study: • First author, publication year, country, study design • Number of patients, age, sex distribution • Type and level of dislocation • Reduction protocol (awake vs under anesthesia) • Patient state (awake vs general anesthesia), • Loading schema (incremental vs rapid), • Device/halter type (eg, cranial tongs, halo, halter). • Success or failure of closed reduction • Reported clinical or imaging predictors • Neurological status ASIA grades (complete vs incomplete) • Surgical approach after failure • Imaging predictors • Oxford evidence level • Methodological quality (NOS)
Assessment of Methodological Quality
All included studies were evaluated using the Newcastle-Ottawa Scale (NOS) for non-randomized studies (Wells, 2013). 16 The NOS assesses three domains: selection, comparability, and outcome. Total scores range from zero to nine, with classifications as follows: high quality (7-9), moderate (5-6), and low (<5). This scale has been validated in trauma research and has been used in previous reviews of spinal trauma (Zhou, 2020). 17
Synthesis Strategy
Given the considerable heterogeneity in study designs, sample sizes, and definitions of “closed reduction failure,” a structured narrative synthesis was conducted. The results were grouped into three domains: (1) clinical predictors (neurologic status, timing), (2) radiological predictors (facet morphology, fracture pattern), and (3) technical predictors (type of traction, use of general anesthesia). The primary harmonized endpoint was failure = need for open reduction after CCT.
When numerical data were comparable across studies (ie, number of failures and successes per variable), we evaluated the feasibility of a meta-analysis of proportions using the DerSimonian–Laird random-effects model (Higgins, 2021). 18 However, due to methodological heterogeneity, no pooled analysis was ultimately performed.
Due to inconsistent MRI/CT reporting across studies, no cross-study morphometric analysis could be undertaken. Imaging findings were therefore synthesized narratively at the study level rather than pooled.
No adjusted meta-analytic modeling of confounders (eg, injury-to-reduction time, mechanism, polytrauma, comorbidities) was feasible given study-level data limitations; findings are interpreted as associations, not causal effects.
Given substantial heterogeneity across traction protocols and operational definitions of “failed closed reduction,” we retained each study’s own definitions without imposing cross-study reclassification. Protocol descriptors (awake vs GA, loading schema, device/halter) were recorded only when explicitly reported and otherwise noted as not reported. No cross-imputation or standardization was attempted. Accordingly, findings are synthesized narratively at the study level rather than pooled across protocol variants.
Results
Characteristics of Clinical Studies Evaluating Closed Reduction Outcomes in Traumatic Cervical Facet Dislocations
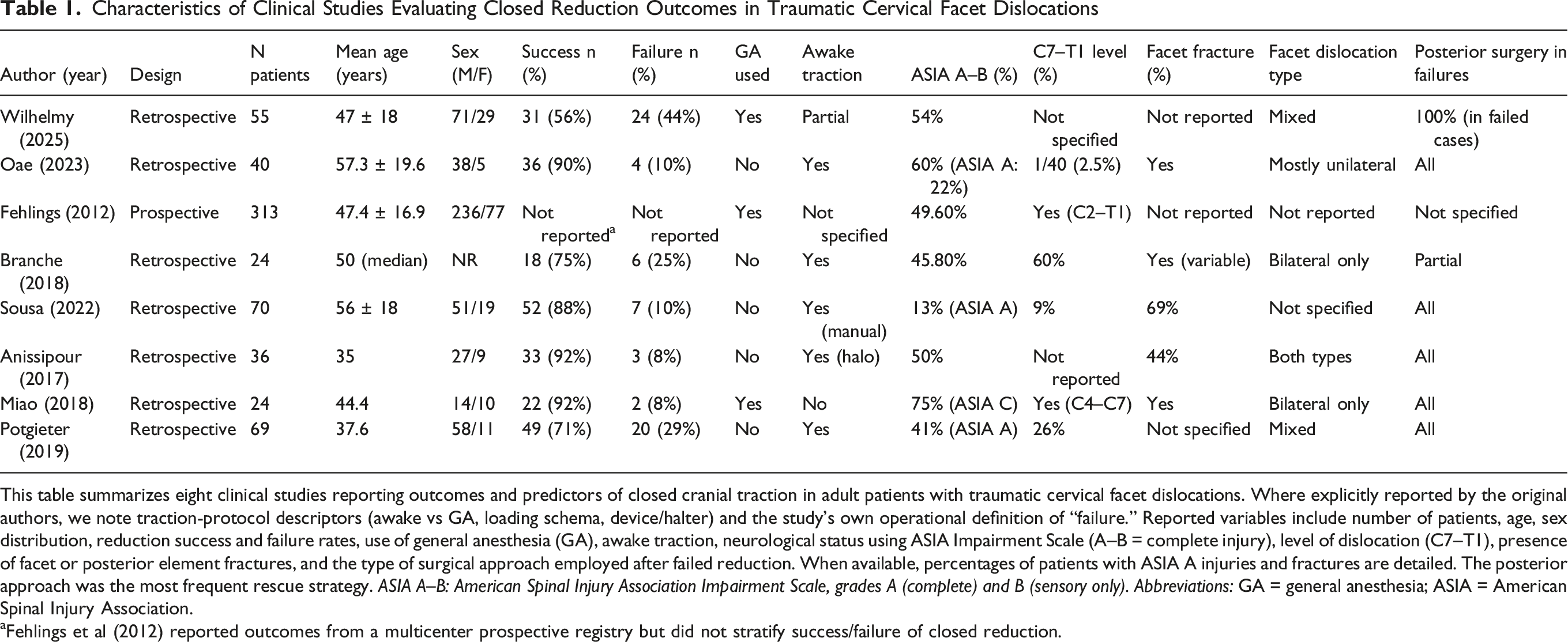
This table summarizes eight clinical studies reporting outcomes and predictors of closed cranial traction in adult patients with traumatic cervical facet dislocations. Where explicitly reported by the original authors, we note traction-protocol descriptors (awake vs GA, loading schema, device/halter) and the study’s own operational definition of “failure.” Reported variables include number of patients, age, sex distribution, reduction success and failure rates, use of general anesthesia (GA), awake traction, neurological status using ASIA Impairment Scale (A–B = complete injury), level of dislocation (C7–T1), presence of facet or posterior element fractures, and the type of surgical approach employed after failed reduction. When available, percentages of patients with ASIA A injuries and fractures are detailed. The posterior approach was the most frequent rescue strategy. ASIA A–B: American Spinal Injury Association Impairment Scale, grades A (complete) and B (sensory only). Abbreviations: GA = general anesthesia; ASIA = American Spinal Injury Association.
aFehlings et al (2012) reported outcomes from a multicenter prospective registry but did not stratify success/failure of closed reduction.
Across the included studies, a total of 631 patients were reported, of whom 463 underwent successful closed reduction and 168 required open surgical intervention. Reported success rates varied substantially among individual studies, ranging from 56% to 92%. No formal pooled analysis was performed due to methodological heterogeneity. The type of reduction varied: some studies employed awake protocols exclusively (Oae, Branche, Potgieter), others used general anesthesia (GA) either selectively or systematically (Wilhelmy, Miao), and one applied manual halo traction (Sousa).4-7,9,10
Global Analysis of Reduction Outcomes
Across all 631 patients: • 463 achieved successful closed reduction (range across studies: 56-92%) • 168 patients failed closed traction and required open reduction surgery (range: 8-44%) • The highest failure rate was observed in the Wilhelmy cohort (44%), which also had the most rigorous multivariate analysis (Wilhelmy, 2025)
4
• Lowest failure rates were seen in studies using general anesthesia from the outset (Miao, 8%; Anissipour, 8%) or with strict awake protocols but low C7–T1 involvement (Oae, 10%)
This confirms a wide inter-study variability, likely driven by differences in inclusion criteria (unilateral vs bilateral), traction protocols, use of neuromonitoring, and timing post-trauma.
Comparative Synthesis
Given the heterogeneity and limited number of studies, no formal pooled effect estimates were calculated; comparative trends were summarized narratively at the study level to highlight recurrent predictors of failed closed reduction.
Predictive Factors of Closed Reduction Failure
To facilitate interpretation, the predictive variables were grouped into three major domains: clinical, radiological, and technical. While not all studies performed statistical tests, several reported strong descriptive patterns, and two included adjusted odds ratios.
Clinical Predictors
Neurological Status (ASIA Score)
Six of the eight studies reported neurological status at presentation using the ASIA Impairment scale. Because ASIA grading granularity varied, analyses were harmonized to complete vs incomplete categories when finer strata were unavailable. The presence of complete spinal cord injury (ASIA A–B) was consistently associated with increased likelihood of failure: Wilhelmy (2025): Neurological injury of any degree (AIS A–D) reduced odds of successful reduction by 21-fold (OR = 0.05, P = 0.003).
4
Potgieter (2019): 41% had ASIA A; among them, the success rate was significantly lower (not numerically stratified).
10
Miao (2018): Among patients with ASIA A–C, failure occurred in 2/24 (8%), though reduction was performed under GA with neuromonitoring.
9
Sousa (2022): Only 13% had ASIA A injuries, but all failures occurred in patients with at least incomplete deficits.
7
These findings suggest a robust negative association between neurologic severity and the likelihood of closed reduction success. Damage to cord function may reflect more severe instability or impaction, impeding mechanical repositioning.
Timing of Reduction
Only three studies reported explicit timing between trauma and traction:
Although formal meta-analysis was not performed, earlier traction (<12 h) seems to be associated with improved outcomes.
Age
Reporting of age was inconsistent across studies. Three cohorts provided overall mean ages (Miao, 44.4 years; Wilhelmy, 47 ± 18 years; Potgieter, 37.6 years), but none stratified outcomes by age group or compared success vs failure subgroups. No statistical analyses were available to assess age as an independent predictor of closed reduction failure. As a result, the role of age remains indeterminate in the current evidence base.
Radiological Predictors
Injury Level (C7–T1)
Lower cervical injuries were more difficult to reduce: Sousa (2022): 100% of closed reduction failures occurred at C7–T1, despite only 9% of the sample having lesions at that level.
7
Potgieter (2019): 26% of cases were C7–T1, with high failure rates (not isolated numerically).
10
Wilhelmy (2025): did not isolate level but noted trends toward failure in lower subaxial segments.
4
Perched Facet (Contralateral)
The presence of a contralateral perched facet was among the strongest positive predictors of success:
Mechanistically, a perched facet may facilitate realignment by providing mechanical guidance for distraction, compared to a “locked” facet that resists engagement. Only two studies (Miao 2018; Bose 2023) explicitly included pre-reduction MRI in their protocol, representing 22% of the analyzed series. This quantitative finding underscores the limited imaging evidence available before traction maneuvers and highlights a major gap in current literature.
Imaging Overview
MRI and CT variables were reported heterogeneously. While several cohorts described the level of injury and facet/periarticular fractures, MRI-based disc/PLC assessments and CT morphometry were inconsistently documented and rarely linked to closed-reduction outcomes. As a result, imaging predictors are presented descriptively as reported by each study, without cross-study morphometric pooling.
Facet or Endplate Fractures
Anissipour (2017): 44% of patients had facet fractures, and failures were more frequent in this subgroup, though not quantified statistically.
8
Sousa et al (2022) identified vertebral endplate fractures in 19% of patients and facet fractures in 78%. Treatment failure occurred in 43% of those with endplate fractures vs 0% (0/29) without them (P = 0.004) Miao (2018) documented posterior element fractures in failed cases, though the sample was too small for statistical inference.
9
The presence of bony disruption, particularly involving the inferior vertebral endplate, may reduce the ability of traction to achieve controlled reduction.
Notably, no study stratified closed-reduction success by MRI-confirmed disc herniation or PLC disruption, and CT morphometric measures were insufficiently standardized to permit comparative analysis.
Facet Dislocation Laterality (Unilateral vs Bilateral)
Across studies that reported facet laterality, failure rates for unilateral and bilateral dislocations appeared broadly similar, suggesting that laterality alone is unlikely to determine irreducibility. Clinical context, neurological status, injury level (particularly C7–T1), and facet/endplate fractures seem to carry greater prognostic weight.
Technical Predictors
Use of General Anesthesia (GA)
One of the most impactful procedural variables: • Wilhelmy (2025): In a cohort of 55 patients with unilateral locked facet dislocations, closed skeletal traction (CCT) under general anesthesia demonstrated improved success compared to awake protocols. Specifically, success was achieved in 48% (13/27) of patients using awake CCT, 61% (11/18) with cervical traction under GA overall, and 83% (10/12) when GA was used as the initial strategy. Success rate with awake traction: 48% Success rate with GA: 61% Upfront GA (without prior awake attempt): 83% No failures after awake traction were rescued by subsequent GA
4
• Miao (2018): GA + neuromonitoring yielded 92% success, even in patients with ASIA C or worse
9
This supports the hypothesis that muscle relaxation and controlled distraction under GA improve biomechanical response and reduce patient resistance.
Neuromonitoring
Only Miao (2018) explicitly used intraoperative neuromonitoring (IONM) with somatosensory and motor evoked potentials. 9 No adverse events or neurological worsening were observed, even in patients with incomplete deficits.
Methodological Quality of Included Studies
Scores Reflect Selection (0-4), Comparability (0-2), and Outcome (0-3); Totals Range 0-9. Studies Scoring ≥7 Were Rated High Quality (5-6: Moderate; ≤4: Low). For Single-Arm Series, the “Non-Exposed Cohort” Item was Not Awarded (Maximum Selection = 3)
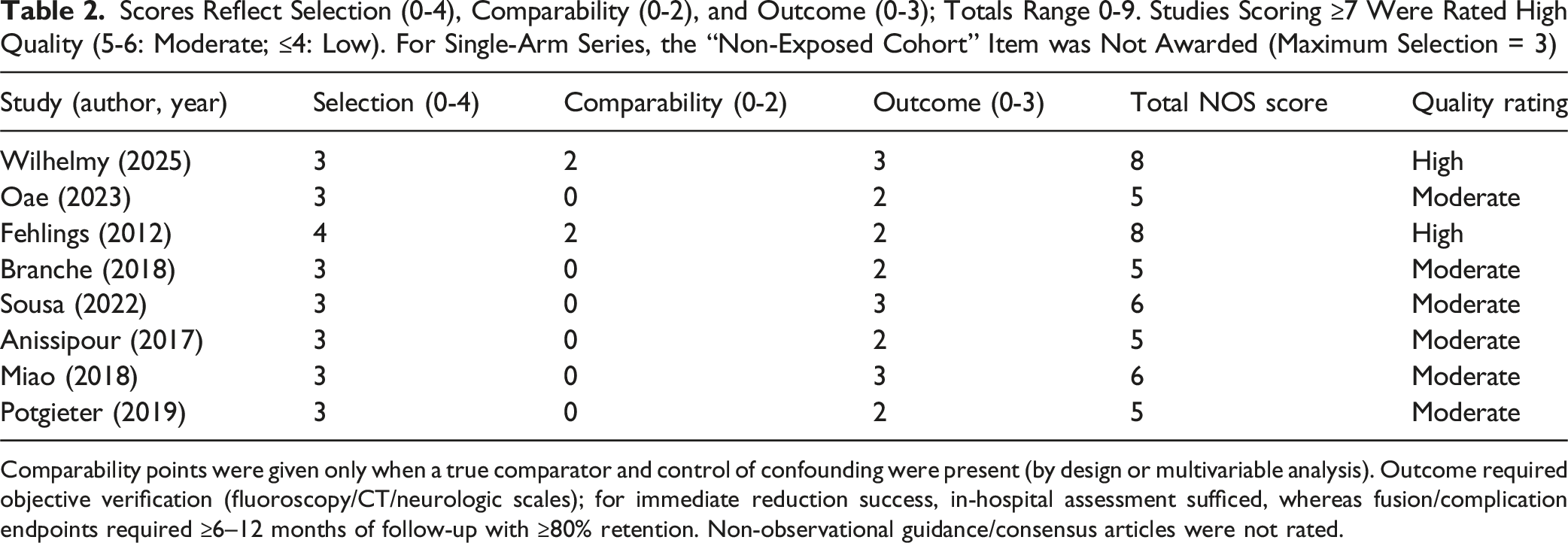
Comparability points were given only when a true comparator and control of confounding were present (by design or multivariable analysis). Outcome required objective verification (fluoroscopy/CT/neurologic scales); for immediate reduction success, in-hospital assessment sufficed, whereas fusion/complication endpoints required ≥6–12 months of follow-up with ≥80% retention. Non-observational guidance/consensus articles were not rated.
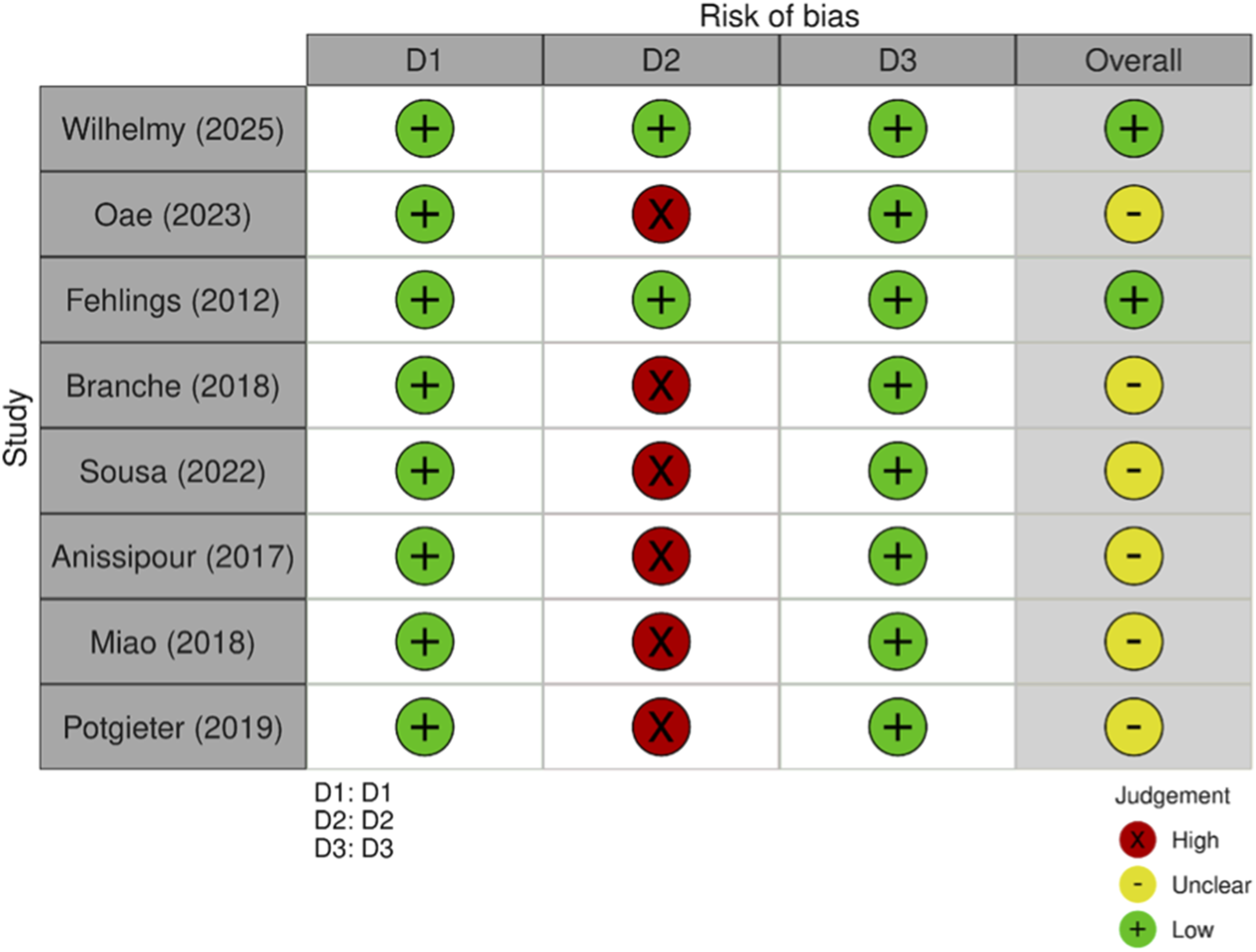
Risk-of-bias assessment using the Newcastle–Ottawa Scale (traffic-light display). Each study was judged across three domains, D1 Selection (0-4), D2 Comparability (0-2), D3 Outcome (0-3), and mapped to standardized categories: Low risk (green “+”), High risk (red “ × ”), Unclear/Moderate (yellow “–”). The Overall column summarizes domain-level judgements (overall low only if all domains are low; overall high if any domain is high; otherwise unclear/moderate). Abbreviation: NOS = Newcastle–Ottawa Scale
Final Narrative Integration
Synthesis of Prognostic Variables Reported Across the Included Studies
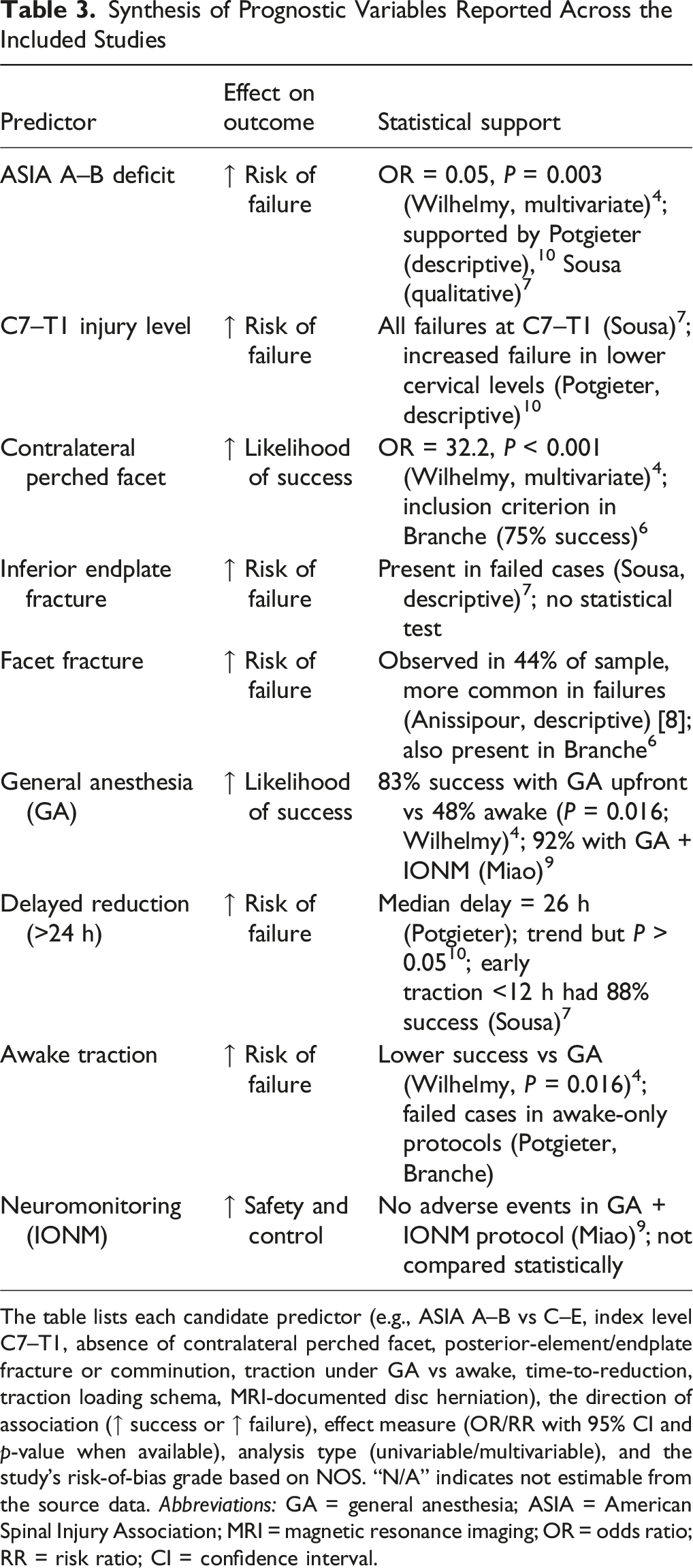
The table lists each candidate predictor (e.g., ASIA A–B vs C–E, index level C7–T1, absence of contralateral perched facet, posterior-element/endplate fracture or comminution, traction under GA vs awake, time-to-reduction, traction loading schema, MRI-documented disc herniation), the direction of association (↑ success or ↑ failure), effect measure (OR/RR with 95% CI and p-value when available), analysis type (univariable/multivariable), and the study’s risk-of-bias grade based on NOS. “N/A” indicates not estimable from the source data. Abbreviations: GA = general anesthesia; ASIA = American Spinal Injury Association; MRI = magnetic resonance imaging; OR = odds ratio; RR = risk ratio; CI = confidence interval.
Moreover, all studies that used awake traction protocols alone had higher failure rates when neurological deficits or lower cervical levels were present. Conversely, protocols incorporating GA with or without neuromonitoring demonstrated higher success, with no adverse neurological events reported in any included series.
This table summarizes the main clinical, radiological, and technical variables evaluated across the eight included studies, categorized by their directional effect on the outcome (ie, increased success or failure of closed reduction). The statistical support column specifies whether the association was based on adjusted odds ratios, P-values, or descriptive observations. Only two studies (Wilhelmy and Miao) conducted inferential analysis. The most robust predictors of failure included complete neurological deficits (ASIA A–B), C7–T1 level injury, absence of a perched facet, and awake traction. The presence of general anesthesia and early traction attempts was associated with greater success.
Discussion
This systematic review identifies and consolidates key clinical, radiological, and technical factors associated with the failure of closed cranial traction in adult patients with traumatic cervical facet dislocations. Across eight studies and 631 patients, we found that complete neurological deficit (ASIA A–B), absence of a contralateral perched facet, lower cervical level involvement (C7–T1), and awake traction protocols were consistently linked with failure. Conversely, use of general anesthesia and early traction was associated with higher success rates. These findings underscore the need for individualized decision-making strategies rather than universal traction attempts. Between-study heterogeneity in traction protocols and in the operational definition of “failure” precluded meaningful standardization; therefore, we retained study-specific definitions and synthesized findings narratively rather than pooling across protocol variants.
Our results are aligned with prior theoretical models of facet dislocation biomechanics. In classical descriptions, a unilateral dislocation may be amenable to reduction by distraction and extension alone, but bilateral locked facets often require realignment of articular surfaces that are mechanically interlocked (Allen, 1982). 19 The presence of a perched facet, rather than a fully locked one, appears to reflect a state of partial engagement that facilitates guided reduction (Wilhelmy, 2025). 4 This finding is supported by the remarkable odds ratio (OR = 32.2) observed by Wilhelmy et al, the only study to perform multivariate modeling with prospective traction protocol stratification.
The neurological status emerged as a strong clinical predictor. In the STASCIS cohort, early surgery was shown to improve neurological outcomes in SCI patients, but the effect of injury grade on mechanical reduction was not assessed (Fehlings, 2012). 2 In our review, complete spinal cord injury (ASIA A–B) predicted failure with high consistency: Wilhelmy et al reported an OR of 0.05 for successful reduction in patients with any AIS grade A–D injury, 4 and both Potgieter and Sousa observed that most failures occurred in those with neurological compromise.7,10 This may reflect the greater force required to overcome joint impaction and the severity of the injury. When spinal injury occurs, the degree of impaction is more severe than when the spinal cord is preserved, justifying this finding.
The anatomic location of injury also mattered: the C7–T1 level was disproportionately associated with failed reductions. This junctional zone presents unique biomechanical and surgical challenges due to its transitional anatomy, increased facet orientation, and often incomplete imaging on trauma CTs (Kepler, 2011). 20 Sousa et al. explicitly noted that all failures in their cohort occurred at C7–T1, even though this level was involved in only 9% of their patients. 7 Potgieter similarly observed higher resistance to traction in injuries at or below C7. 10 These findings suggest that lower cervical dislocations should be approached with caution, and perhaps with a lower threshold for early open reduction. Potentially, the effect of cranial traction in the lower segments of the cervical spine is also less evident than in the cranial levels. Differences in weight load may also influence these results, as well as more powerful ligaments involved in the cervicothoracic junction and a long distance arm between the cranium (tractioned) and the site of cervical dislocation.
Beyond anatomy and neurology, technical variables played a significant role. The use of general anesthesia (GA) with muscle relaxation was associated with improved outcomes, particularly when used as the initial approach. Wilhelmy et al. observed a dramatic difference: 83% success in patients who underwent GA-first reduction vs 48% in those who started awaking. Critically, no failed awake cases were rescued by subsequent GA traction. 4 Miao et al, who used GA and neuromonitoring systematically, achieved 92% success with no neurologic deterioration, even in patients with incomplete ASIA injuries. 9 These results suggest that GA not only enhances mechanical conditions for reduction but may also prevent patient distress, muscle resistance, and movement-related complications.
While this exploratory comparison suggests that failure rates were higher in patients managed under GA, this likely reflects selection bias, since GA was often reserved for more complex or refractory cases. Nevertheless, when used proactively, as in Wilhelmy’s cohort, GA appeared to enhance the likelihood of successful reduction. Future prospective studies are needed to clarify whether anesthetic modality independently influences the success of closed reduction.
Thus, while the aggregate data suggest higher failure rates with GA, this likely reflects selection bias with GA reserved for more complex or refractory cases. When used proactively, GA may enhance success, particularly in patients at high risk of failure.
The role of neuromonitoring remains underexplored. Only one study (Miao, 2018) used motor and somatosensory evoked potentials, documenting safety in all patients. 9 While not analyzed statistically, this reinforces the notion that neuromonitoring could enable safer closed reduction in high-risk patients, eg, those with incomplete deficits or difficult anatomy, though more data are needed to define its impact.
Although bilateral dislocations have traditionally been considered more unstable from a biomechanical standpoint, the results of this analysis do not show a significant difference in closed reduction failure rates based on laterality. In a subset of five studies with verifiable or inferable data on facet alignment patterns, failure occurred in 23.3% of unilateral dislocations and 25.4% of bilateral dislocations (χ2 = 0.05, P = 0.82), with no statistically significant difference. This suggests that laterality alone does not decisively determine the outcome of closed reduction maneuvers, and that other factors such as neurological status, injury level, and facet morphology should take precedence in prognostic evaluation and early management decisions. Of note, unilateral dislocation with a contralateral perched (subluxated) facet may reduce more consistently when there is unilateral dislocation without a contralateral subluxation. Further studies about this are necessary.
Across the included studies, complications directly attributable to closed skeletal traction were rare. Only Oae et al (2023) 16 reported three cases of transient neurological worsening (eg, increased neck pain or sensory symptoms) during traction, all of which resolved spontaneously with flexion repositioning. No permanent neurological deterioration occurred. Similarly, Miao et al (2018) 9 and Sousa et al (2022), 7 both employing continuous clinical or neurophysiological monitoring, reported zero cases of neurological aggravation during traction. Potgieter et al (2019) 10 also noted no worsening and documented neurological improvement in five patients post-reduction. In contrast, Fehlings et al (2012) 4 described five cases of neurological decline, though it was not specified whether these occurred during closed reduction or subsequent surgery. Other studies, such as Anissipour et al (2017) 8 and Branche et al (2018), 6 included closed reduction attempts but did not document procedural complications, likely due to design limitations or lack of systematic reporting.
Another variable considered in earlier literature but not consistently captured in the included studies is the presence of a disc herniation or PLL disruption. While Vaccaro et al (2007) and the AO Spine Classification incorporate these elements into decision algorithms (Vaccaro, 2007) 12 ; (Schnake, 2017), 11 none of the included articles in this review stratified outcomes based on disc-ligamentous integrity. Given the known risk of neurological worsening during distraction in the presence of a herniated disc, this represents an important gap that should be addressed in future prospective studies with MRI correlation.
Despite the consistency of key findings across diverse settings, this review must be interpreted in light of several methodological and clinical limitations. First, no randomized controlled trials (RCTs) were identified. All included studies were observational, with inherent risks of selection bias and confounding. Only one study (Wilhelmy, 2025) applied a prospective design with multivariate adjustment, and most used retrospective data collection methods. Second, definitions of “failure” were heterogeneous; some defined it as the need for open surgery, others as incomplete anatomical alignment. This variability complicates direct comparison of rates and effect sizes.
Third, only two studies (Wilhelmy, Miao) applied structured traction protocols with specified weights, durations, and escalation thresholds.4,9 In most cohorts, the traction technique was operator-dependent, limiting reproducibility. Additionally, imaging modalities and timing varied: some studies used CT immediately post-traction, others relied on intraoperative fluoroscopy or MRI. Without standardized assessment of post-reduction alignment and neural decompression, outcome classification remains vulnerable to measurement bias.
Despite these limitations, the internal coherence of the findings is notable. Across multiple studies, regardless of region or sample size, the same risk factors, ASIA A–B, absence of a perched facet, C7–T1 involvement, and awake traction emerged repeatedly as associated with failure. That convergence strengthens the external validity of these predictors and supports their integration into early decision-making.
Clinical Implications
From a practical standpoint, our findings suggest that some patients are at higher risk of failure to close traction, and surgery may be considered as first-line treatment or to avoid unnecessary extensive CCT. In particular, the following conditions appear to predict a high risk of failure: Complete neurological deficit (ASIA A–B) Involvement of the C7–T1 level Presence of comminuted facet or inferior endplate fracture Traction is attempted in awake patients without relaxation
In these scenarios, prolonged or repeated traction attempts may delay definitive care and expose patients to unnecessary discomfort or neurologic risk. Rather than adopting a “traction-first” default, clinicians may consider proceeding directly to open reduction, especially when posterior approaches are available.
Our proposed decision algorithm. Figure 3 integrates the most consistent predictors of failure of closed cranial traction (CCT) into a pragmatic, safety-oriented framework that reflects both the strengths and limitations of the available literature. Given the heterogeneous traction protocols, variable definitions of “failure,” and the predominance of observational data, we intentionally adopted a conservative structure. High-risk profiles, particularly those involving complete neurological deficit, C7–T1 injuries, or inferior endplate fractures, are funneled toward either a single brief reduction attempt under general anesthesia (GA) or direct surgery, avoiding prolonged or repeated maneuvers that have shown little incremental benefit and may delay definitive stabilization. In contrast, patients without identifiable risk factors may undergo controlled progressive traction with stepwise load increases and short-interval reassessment, preserving the potential benefits of CCT without compromising safety. Importantly, the algorithm formalizes the option of a limited, structured retry using increased weight or conversion to GA if initial attempts were performed awake, acknowledging that these adjustments may enhance success while still imposing a strict limit on procedural persistence. Overall, the algorithm provides a unified, clinically actionable pathway that prioritizes neurological integrity, minimizes unnecessary delays to surgery, and harmonizes evidence-derived risk factors into a reproducible decision tool for real-world practice. Simplified decision algorithm for management of adult traumatic cervical facet dislocation. Patients are stratified according to the presence of risk factors for failure of closed cranial traction (CCT). If risk factors are present, only one brief reduction attempt (preferably under general anesthesia, GA) or direct surgery is advised. Prolonged or repeated attempts should be avoided to minimize neurological risk. If no risk factors are present, a controlled closed reduction can be maintained with progressive traction load and intermittent reassessment under fluoroscopic and clinical monitoring. In case of initial failure, a brief retry may be considered with increased load and/or conversion to GA, followed by prompt reevaluation. If the reduction remains unstable or incomplete, direct surgical management is indicated. Abbreviations: CCT = closed cranial traction; GA = general anesthesia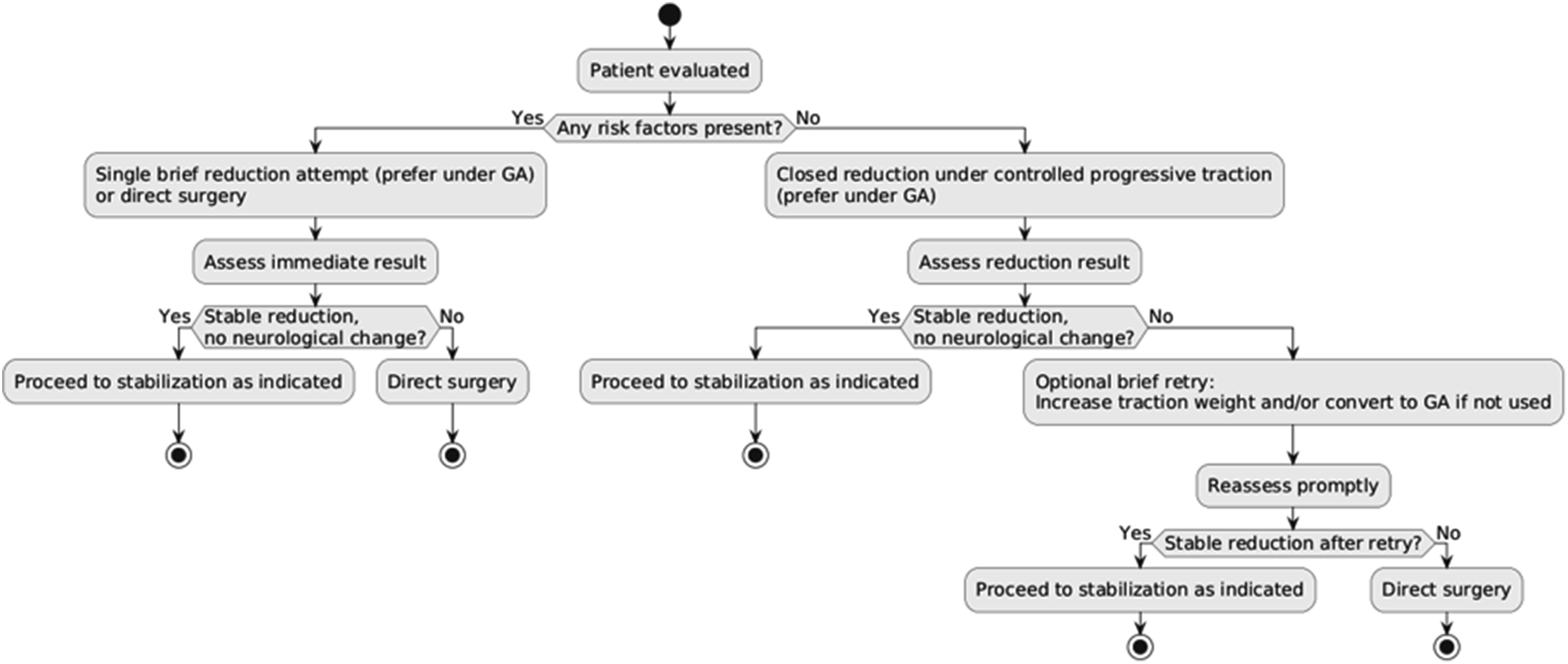
The association between neurological severity and failed reduction may be explained by the underlying mechanical complexity of these injuries. Severe cord injury likely reflects a greater degree of facet interlocking, axial load, or vertebral translation, all factors that compromise the effectiveness of traction. As such, neurological status may serve not only as a prognostic marker but also as a surrogate indicator of biomechanical irreducibility.
This perspective is aligned with trauma systems that prioritize early decompression and stabilization, such as those implemented in the STASCIS trial (Fehlings, 2012). 2 Moreover, the widespread availability of intraoperative neuromonitoring and modern imaging may reduce reliance on closed traction as a diagnostic or therapeutic tool in complex cases.
Limitations of This Review
This review is also subject to inherent limitations. First, the lack of individual patient data (IPD) restricted our ability to perform pooled risk estimates. Second, most studies lacked MRI data, preventing stratification by disc herniation or ligamentous injury. Third, possible publication bias cannot be ruled out, as many failed reductions may not be formally reported. Finally, language restrictions to English and Spanish may have excluded relevant data from other regions. Most cohorts were retrospective, with inherent risks of selection bias, missing data, and non-uniform outcome ascertainment. While the NOS appraises study-level risk of bias, retrospective designs limit internal validity for prognostic inference; as such, our synthesis emphasizes associations rather than causal effects. No meta-analysis was performed; comparative findings were synthesized narratively.
Several cohorts were small and single-center from high-volume tertiary units, potentially limiting generalizability to broader trauma systems. We therefore avoided pooled estimates and interpreted signals of association with appropriate caution.
Heterogeneous and incomplete MRI/CT reporting precluded standardized morphometric analysis across studies. Confounding by timing, mechanism of injury, associated polytrauma, and comorbidities could not be systematically addressed and may influence observed associations. Publication bias is likely, as failures or complicated reductions may be underreported. With a small number of heterogeneous studies, formal small-study effect assessments (eg, funnel plots, Egger’s test) are not appropriate; thus, the true magnitude of associations may be over- or under-estimated.
Language restrictions to English and Spanish may have excluded additional relevant cohorts; future updates will consider broader language inclusion to minimize this bias. No randomized or adequately powered prospective studies were identified; hence, the predictors synthesized herein represent associations, not determinants.
To advance this field, prospective multicenter registries should collect standardized data on reduction attempts, neurological status, injury morphology, and outcomes. In addition, trials comparing awake vs GA traction and imaging-based predictors (eg, perched facet angle, disc herniation volume) are urgently needed. Integration of predictive algorithms into trauma protocols could enhance early triage and reduce delays to definitive care.
Conclusion
Closed reduction remains a widely used initial approach for managing cervical facet dislocations, but its failure may delay decompression, prolong instability, and expose patients to neurologic risk. This systematic review of 631 patients from eight clinical studies identified several reproducible predictors of failure: complete neurological deficits (ASIA A–B), absence of a contralateral perched facet, C7–T1 level involvement, comminuted posterior element fractures, and the use of awake traction protocols.
Conversely, the success of reduction was higher when performed under general anesthesia and with early timing post-injury. These findings suggest that a selective, criteria-based approach rather than routine traction in all cases may optimize outcomes. We advocate for early recognition of high-risk patterns and prompt transition to open posterior reduction when appropriate. Future research should validate predictive algorithms through prospective, multicenter studies and consider the role of neuromonitoring, MRI-based predictors, and standardization of traction protocols.
Adopting evidence-informed criteria for initiating or omitting closed traction can reduce unnecessary delays, improve neurologic outcomes, and bring greater precision to the acute management of cervical spine trauma.
Accordingly, identified predictors should be interpreted as associative signals to guide early risk stratification, pending validation in prospective, multicenter studies.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
