Abstract
Study Design
Retrospective Study.
Objectives
Spinal cord injury (SCI) has become a major health threat, and existing diagnostic tools such as MRI and CT have limitations. This study aims to investigate the expression changes of miR-200a-3p and its target PTEN in SCI and explore their potential diagnostic and therapeutic values.
Methods
A total of 148 SCI patients were enrolled. An in vitro SCI model was established using PC12 cells treated with LPS. The levels of miR-200a-3p and PTEN were measured using qRT-PCR. The diagnostic value of both for SCI was evaluated using ROC curve analysis. The targeting relationship between miR-200a-3p and PTEN was validated through dual-luciferase reporter assays, and RNA pull-down experiments. Cell viability and apoptosis were analyzed using MTT and flow cytometry. ELISA was used to measure pro-inflammatory cytokines. Levels of ROS, CAT, and SOD were determined using respective kits.
Results
miR-200a-3p was significantly decreased, while PTEN was upregulated in SCI patients. Both miR-200a-3p and PTEN could distinguish SCI patients from individuals with normal neurological function, as well as complete vs incomplete SCI. Furthermore, miR-200a-3p directly targets and suppresses PTEN; overexpression of miR-200a-3p enhanced cell viability, reduced inflammation and oxidative stress, and inhibited apoptosis. Conversely, PTEN overexpression reversed these protective effects.
Conclusions
miR-200a-3p and PTEN have certain diagnostic value for SCI, and miR-200a-3p exerts neuroprotective effects by targeting PTEN to reduce inflammation and oxidative stress. This study provides promising biomarkers and therapeutic targets for the early diagnosis and intervention of SCI.
Introduction
Spinal cord injury (SCI) is a severe damage to the nervous system, 1 usually resulting in the loss of motor and/or sensory functions in the lower and/or upper limbs, significantly impacting patients’ quality of life. 2 According to statistics, there are hundreds of thousands of new cases worldwide each year. 3 SCI not only causes physical trauma but also has profound psychological and social impacts. 4 The current treatment options mainly include surgical intervention, pharmacotherapy, and rehabilitation training. 5 Early diagnosis and assessment are crucial for subsequent treatment and prognosis evaluation in SCI patients. The main diagnostic methods for SCI primarily rely on imaging techniques such as MRI and CT, 6 which can evaluate the extent of spinal cord structural damage. However, these methods have limitations, including high costs and exposure to ionizing radiation. 7 Therefore, identifying novel biomarkers to improve early diagnosis and prognostic assessment of SCI, achieving more non-invasive, convenient, and cost-effective detection, has become one of the current research hotspots. MicroRNAs (miRNAs) as emerging molecular biomarkers show potential applications in neurological diseases. 8
MiRNAs are a type of non-coding RNA, typically consisting of about 18 to 25 nucleotides.9,10 These molecules are involved in a variety of biological activities, such as promoting cell proliferation, guiding cell differentiation, inducing apoptosis, and mediating inflammatory reactions.11,12 In recent years, an increasing number of studies have shown that miRNA is involved in musculoskeletal diseases such as osteoarthritis and tendon injuries.13,14 More importantly, its expression changes in SCI are closely related to the extent of injury, nerve function recovery, and clinical prognosis. 15 This makes them potential biomarkers. Numerous clinical and experimental studies have shown that specific miRNAs change significantly in expression levels following SCI. For example, miR-21, miR-10a, and others have been demonstrated to be upregulated or downregulated in spinal cord injury models.16,17 The observed expression patterns are closely linked to inflammatory responses, cell apoptosis, and tissue regeneration following SCI, serving as key markers for predicting recovery of neural functions. miR-200a-3p has also been found to exhibit differential expression in adult zebrafish after SCI 18 and is downregulated in chronic constriction injury (CCI) rats. 19 Moreover, overexpression of miR-200a-3p can alleviate mechanical allodynia and thermal hyperalgesia in CCI rats, thereby reducing neuropathic pain. 20 Additionally, studies by Shakespeare et al indicated that normal astrocytes release miR-200a-3p, which exerts neuroprotective effects by downregulating MKK4. 21 These findings support the potential application of miR-200a-3p in neuroprotection; however, there are currently no reports on the clinical significance and mechanisms of miR-200a-3p in SCI.
Based on the above research, we selected miR-200a-3p as the study target by assessing the levels of miR-200a-3p and its targets in the serum of SCI patients, as well as constructing an in vitro SCI cell model, to explore the diagnostic value of miR-200a-3p and its potential role mechanisms involved in SCI. The goal is to provide new perspectives and evidence for the exploration of potential therapeutic targets and biomarkers for SCI.
Methods
Clinical Samples
This study was approved by the Ethics Committee of Zhucheng People's Hospital (No. 2018-0010). All participants signed informed consent forms. A total of 148 SCI patients treated at Zhucheng People's Hospital from May 2018 to October 2021 were enrolled. The SCI patients were divided into three groups according to the American Spinal Injury Association (ASIA) impairment scale of the international standard for spinal cord injury neurofunction grading: Normal neurofunction group (ASIA-E, control group, n = 40); Incomplete SCI group (ASIA-B, C, D, n = 64); Complete SCI group (ASIA-A, n = 42). Inclusion criteria: (1) Age ≥18 years; (2) Diagnosed with SCI confirmed by clinical physical examination, CT, MRI, and other methods; (3) Admission within 24 hours post-injury. Exclusion criteria: (1) History of spinal cord or brain trauma; (2) Acute or severe brain injury; (3) Malignant tumors; (4) Severe cardiovascular diseases, hepatic or renal dysfunctions, acute or chronic infections, autoimmune diseases; (5) Pregnant or lactating women. Venous blood samples were collected from all study participants upon admission (within the 24-hour window post-injury); serum was separated by centrifugation at 4°C. Demographic and clinical data were recorded.
Cell Culture and Treatment
Considering the widespread application and good reproducibility and representativeness of PC12 cells in neural injury and neuroprotection studies, we chose PC12 cells and established an in vitro SCI model. 22 PC12 cells were cultured in DMEM medium containing 10% fetal bovine serum, 100 U/mL penicillin, and 100 μg/mL streptomycin at 37°C with 5% CO2 humidification. For in vitro SCI model construction, PC12 cells (5 × 103 per well) were seeded into 96-well plates and treated with 100 ng/mL LPS for 4 h.
Cell Transfection
To regulate molecular expression, synthetic miR-200a-3p mimic and negative control mimic NC, pcDNA-3.1-PTEN vector and negative control pcDNA3.1 empty vector were transfected into PC12 cells on the basis of experimental grouping using the Lipofectamine 3000 transfection kit. 48 h of incubation was used for subsequent analysis.
MTT Cell Viability Assay
PC12 cells were seeded at a density of 2 × 102 cells per well in 96-well plates. 5 mg/mL MTT solution was added to each well and incubated at 37°C for 4 h. The supernatant was then aspirated, and 150 μL DMSO was added to dissolve the formazan. Absorbance was measured at 540 nm using a plate reader.
Apoptosis Analysis
Cell apoptosis was analyzed using an Annexin V FITC/PI apoptosis detection kit. PC12 cells were washed with PBS, then stained with 5 μL Annexin V-FITC and 5 μL PI, followed by incubation at 37°C in the dark for 30 min. Quantitative analysis was performed by flow cytometry to identify early and late apoptotic cells.
RNA Extraction and Quantitative Real-Time PCR (qRT-PCR)
Total RNA was extracted from serum and PC12 cells using TRIzol® reagent according to the manufacturer’s instructions. Reverse transcription was performed using the RevertAid First Strand cDNA Synthesis Kit. qRT-PCR was carried out using SYBR Green I Master Mix and the 7300 Real-Time PCR System. GAPDH served as the internal control for PTEN, while U6 was used as the internal control for serum and cellular miR-200a-3p. The 2−ΔΔCt method was used to calculate relative expression levels.
Bioinformatics Analysis
Potential target mRNAs of miR-200a-3p were predicted using five online databases: TargetScan, TarBase, miRDB, miRWalk, and EVmiRNA. The intersection of results was obtained using Venn diagrams. Protein–protein interaction (PPI) network analysis was performed with the STRING database. Interactome hubs were the top 10 nodes in the network in terms of the number of interaction partners (PPI degree).
Dual-Luciferase Reporter Assay
TargetScan predicted the binding sites between miR-200a-3p and PTEN. For validation, wild-type (wt) and mutant (mut) PTEN sequences were cloned into the pmirGLO vector to generate reporter plasmids. PC12 cells were co-transfected with PTEN-wt/mut plasmids and miR-200a-3p mimics or negative control (NC) using Lipofectamine 3000. After 48 h, luciferase activity was measured using a dual-luciferase reporter assay system.
RNA Pull-Down Assay
PC12 cells transfected with miR-200a-3p were lysed, and the lysates were incubated with biotin-labeled negative control (Bio-NC), biotin-labeled miR-200a-3p (Bio-miR-200a-3p), or biotin-labeled mutant miR-200a-3p (Bio-miR-200a-3p MUT). The complexes were pulled down with RNeasy Mini Kit, RNA was extracted, and quantified via qRT-PCR.
ELISA
Pro-inflammatory cytokines released by LPS-treated PC12 cells were measured using ELISA kits for IL-1β, IL-6, and TNF-α. Cells were seeded in 6-well plates, and cytokine levels in the culture supernatant were measured per the manufacturer’s instructions.
ROS Level Evaluation
ROS levels in PC12 cells were assessed using DCFH-DA probe. Cells were incubated with DCFH-DA in serum-free medium at room temperature in the dark for 30 min, then resuspended in PBS. ROS levels were quantified with flow cytometry.
Catalase (CAT) and Superoxide Dismutase (SOD) Activity Detection
CAT and SOD activities in cell lysates were measured using respective activity kits. Cells were collected and resuspended in 0.3 mL PBS, sonicated for 1 minute, centrifuged, and the supernatant was used for enzyme activity analysis. Total protein concentration was determined with a BCA assay.
Statistical Analysis
Data were analyzed using SPSS 22.0 and GraphPad Prism 9.0. All data are presented as mean ± SD. The Shapiro-Wilk test was used to verify the normality of the data. Comparisons between two groups were performed using independent-samples t-test, and multiple groups were compared via one-way ANOVA. The correlation between miR-200a-3p and PTEN was evaluated using Pearson’s correlation coefficient. The efficacy of miR-200a-3p and PTEN in distinguishing controls from SCI patients, and complete vs incomplete SCI, was assessed by ROC curves. A P-value <0.05 was considered statistically significant.
Results
Baseline Characteristics of Subjects
Clinical Characteristics of the Study Subjects
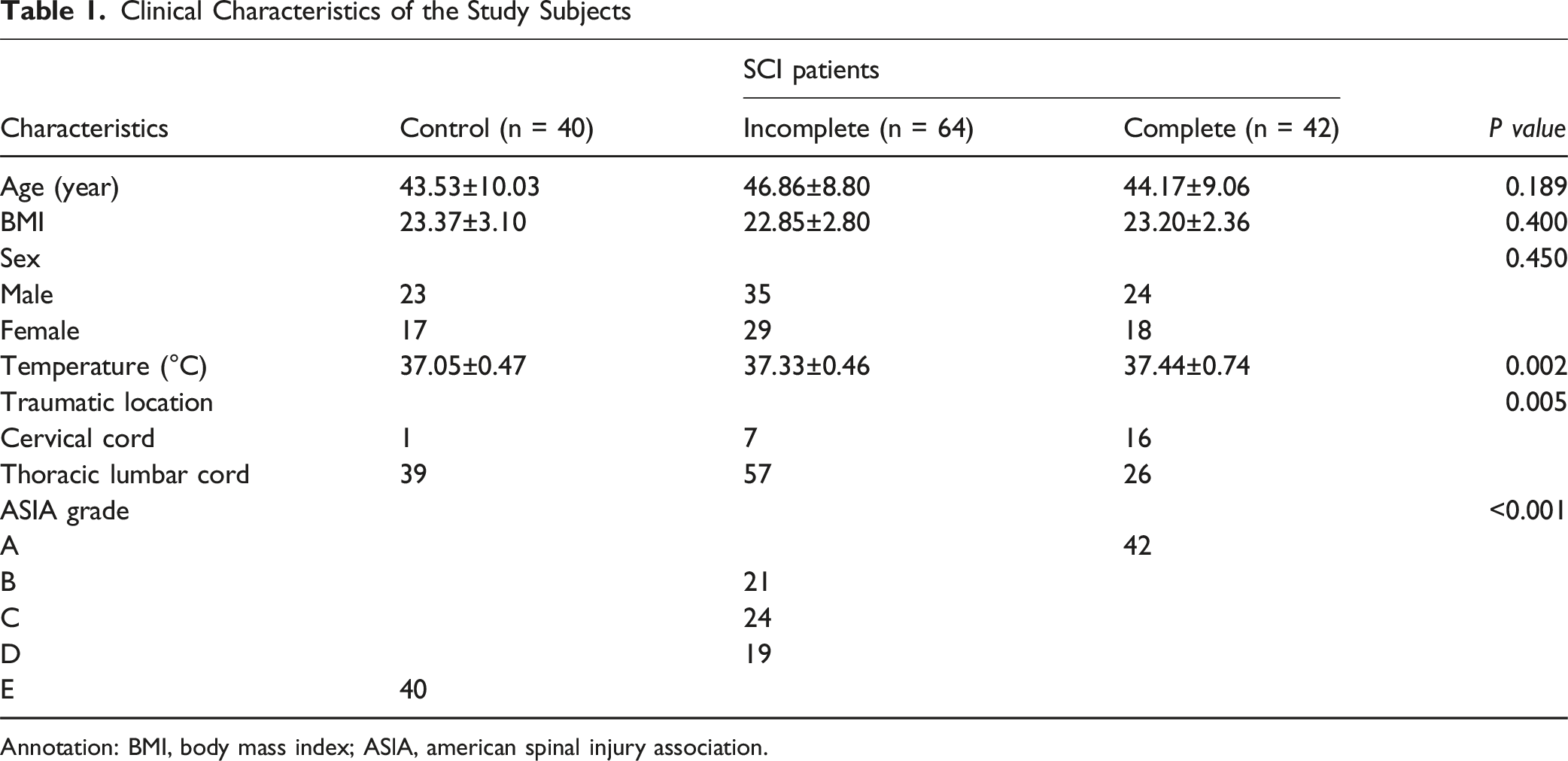
Annotation: BMI, body mass index; ASlA, american spinal injury association.
Decreased miR-200a-3p and Increased PTEN in SCI Patients
qRT-PCR analysis revealed that miR-200a-3p levels were considerably lower in SCI patients than in controls (P < 0.001, Figure 1A), and patients with complete SCI had lower levels than those with incomplete SCI (P < 0.001, Figure 1B). Conversely, PTEN levels were significantly elevated in SCI patients compared to controls (P < 0.001, Figure 1C), with higher levels in complete SCI than in incomplete SCI (P < 0.001, Figure 1D). Levels of miR-200a-3p and PTEN in Serum From SCI Patients. Compared to the Neurologically Normal Group, miR-200a-3p Levels Decreased in SCI Patients (A), while PTEN Increased (C). Additionally, Compared to the Incomplete SCI Group, miR-200a-3p Levels Were Also Reduce and PTEN Levels Were Elevated (D). ***P < 0.001, ****P < 0.0001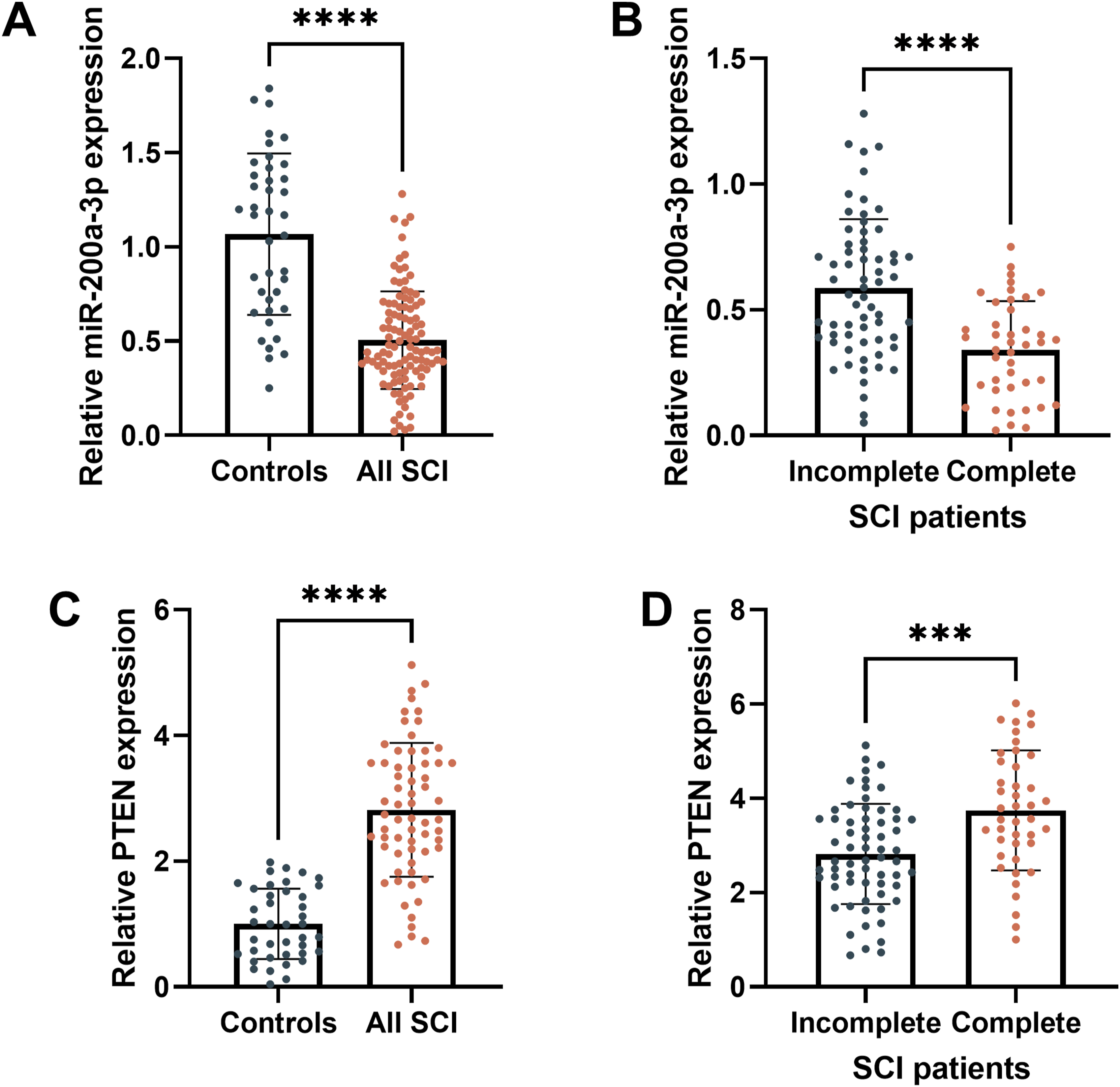
miR-200a-3p Directly Targets PTEN
Predictions from various databases identified 101 target genes of miR-200a-3p (Figure 2A). PPI network analysis revealed a network with 120 connections, with an average connectivity degree of 2.38 per node; PTEN emerged as the node with the greatest number of interactions (Figure 2B). The online databases also predicted the binding sites between miR-200a-3p and PTEN (Figure 2C). Dual-luciferase reporter and RNA pull-down assays confirmed the interaction between miR-200a-3p and PTEN (Figure. 2D and E). Furthermore, serum levels of miR-200a-3p and PTEN in SCI patients showed a considerable negative correlation (r = −0.717, P < 0.001, Figure 2F). miR-200a-3p Targets Binding to PTEN. Venn Diagram of miR-200a-3p Target mRNAs (A). PPI Network Analysis (B). Binding Sites of miR-200a-3p and PTEN (C). Dual-Luciferase Reporter Assay (D). RNA Pull-Down Assay (E). Correlation Analysis Between miR-200a-3p and PTEN Levels (F). *P < 0.05, ****P < 0.0001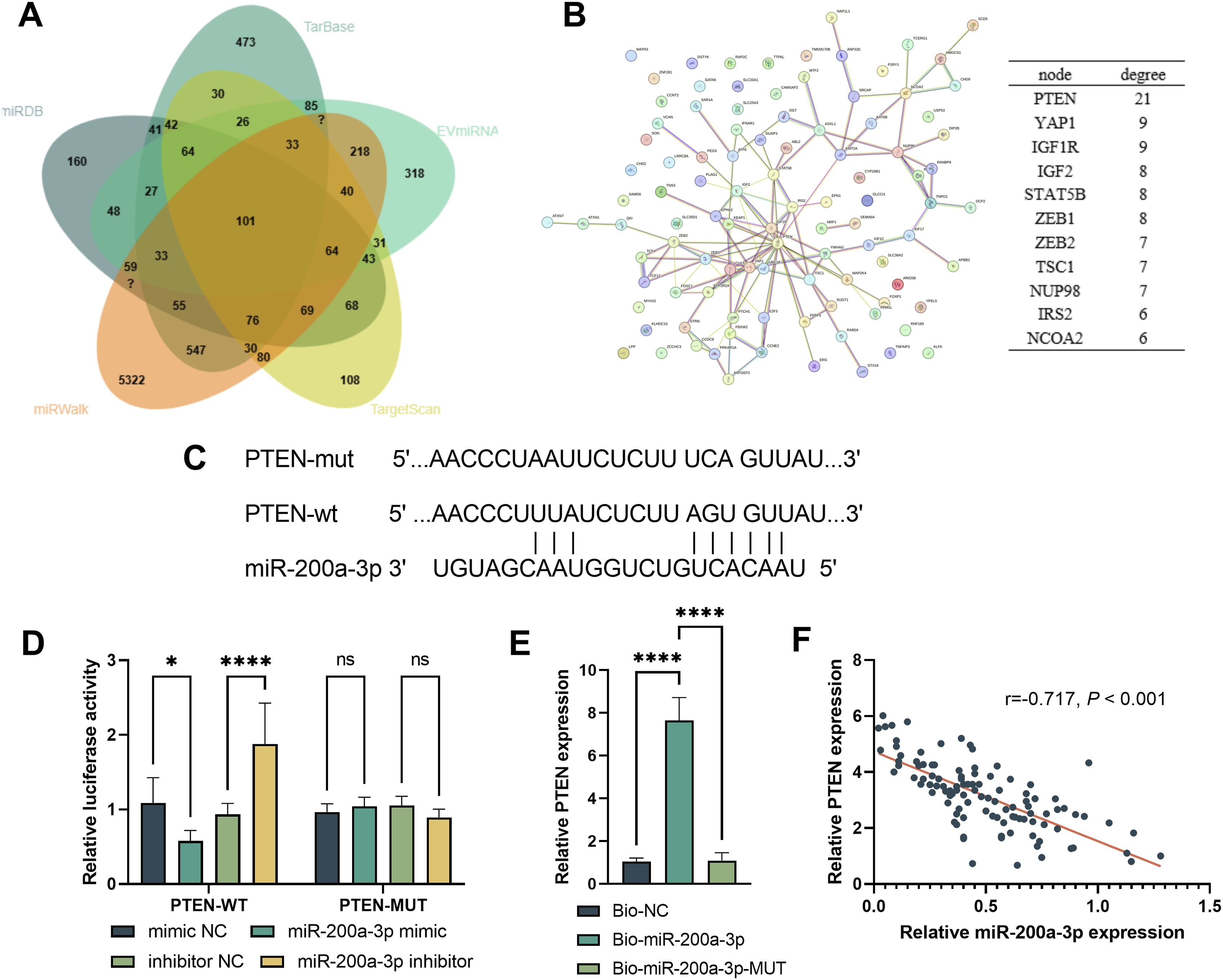
Clinical Value of miR-200a-3p and PTEN in SCI
ROC analysis demonstrated that miR-200a-3p could effectively differentiate SCI patients from individuals with normal neurological function, achieving an AUC of 0.863, sensitivity of 84.91%, and specificity of 72.50% (Figure 3A). It also demonstrated potential in differentiating incomplete from complete SCI, with an AUC of 0.761, sensitivity of 71.43%, and specificity of 68.45% (Figure 3B). Similarly, serum PTEN levels could distinguish SCI patients from controls with an AUC of 0.923, sensitivity of 79.69%, and specificity of 97.50%, and differentiate between incomplete and complete SCI with an AUC of 0.795 (Figure 3C and D). Diagnostic Value Assessment of the miR-200a-3p/PTEN axis. miR-200a-3p and PTEN can Distinguish SCI Patients From Normal Individuals (A and C) and Also Differentiate Complete SCI From Incomplete SCI (B and D)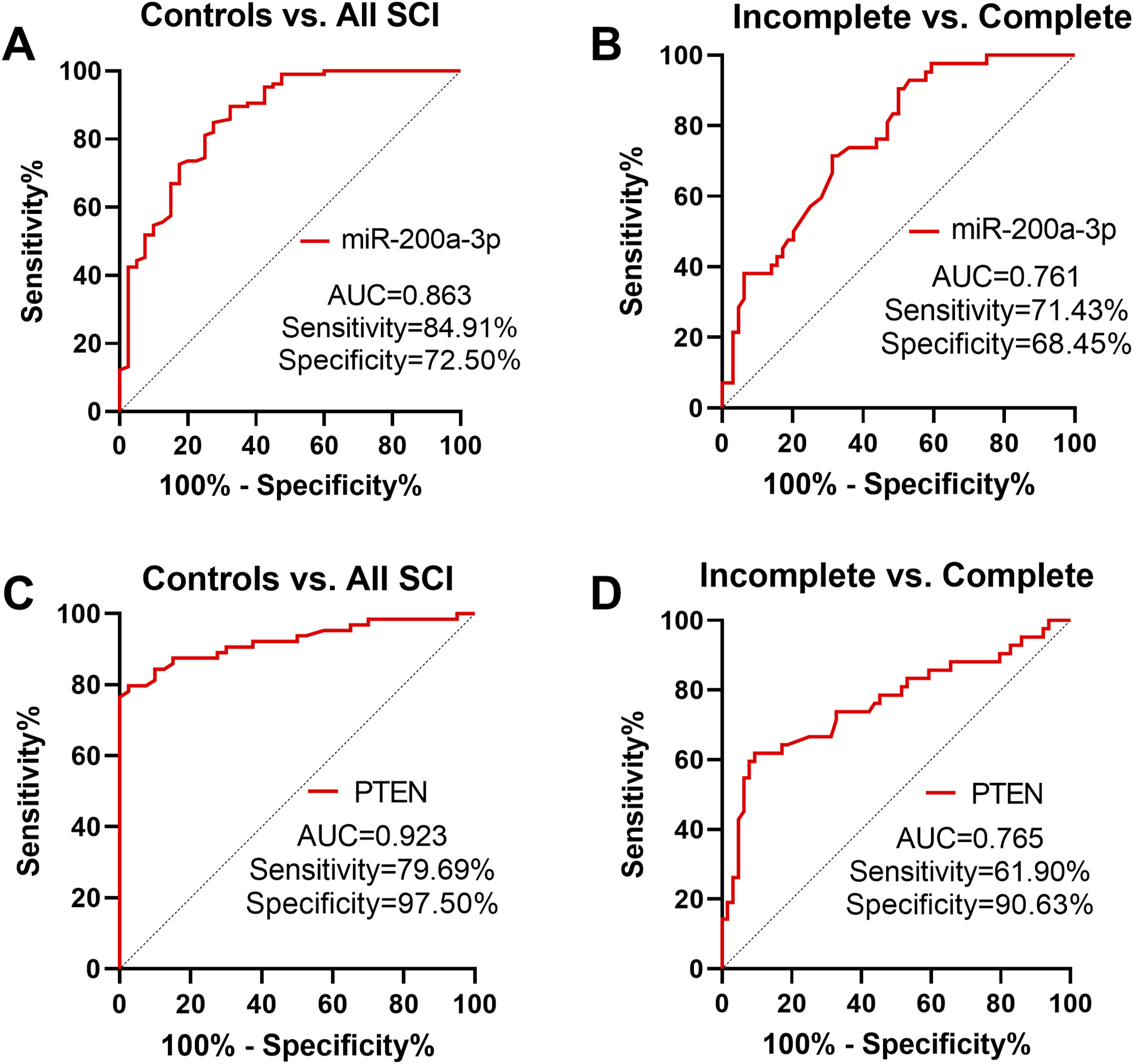
Effects of miR-200a-3p/PTEN Axis on LPS-Induced Cell Viability and Apoptosis
As shown in Figure 4A, LPS treatment significantly decreased miR-200a-3p levels in PC12 cells, which were restored after transfection with miR-200a-3p mimics (P < 0.001). LPS also significantly increased PTEN levels, which were decreased upon miR-200a-3p mimic transfection, and further increased after co-transfection with pcDNA-3.1-PTEN (P < 0.001, Figure 4B). LPS markedly reduced cell viability; overexpression of miR-200a-3p alleviated this reduction, while overexpression of PTEN decreased viability again (P < 0.05, Figure 4C). Similarly, miR-200a-3p overexpression suppressed LPS-induced apoptosis, whereas increased PTEN levels promoted apoptosis (P < 0.01, Figure 4D). Effects of the miR-200a-3p/PTEN Axis on LPS-induced Cell Viability and Apoptosis. Transfection With miR-200a-3p Mimic and pcDNA-3.1-PTEN in LPS-Treated PC12 Cells Affects miR-200a-3p Levels (A), PTEN Levels (B), Cell Viability (C), and Apoptosis Rate (D). *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001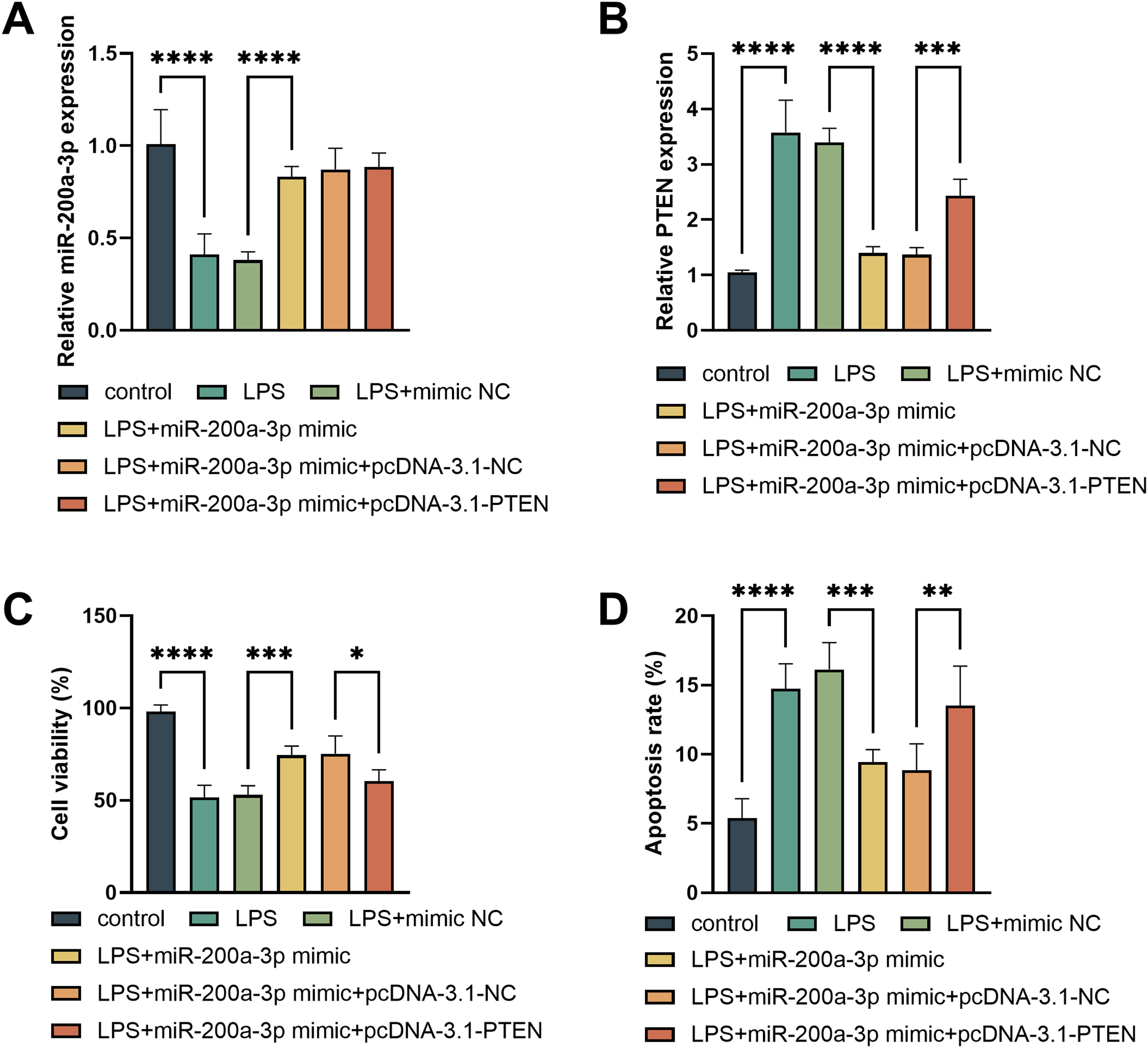
Effects of miR-200a-3p/PTEN Axis on LPS-Induced Inflammation and Oxidative Stress
LPS triggered significant increases in IL-6, IL-1β, and TNF-α levels in PC12 cells; overexpression of miR-200a-3p reduced pro-inflammatory cytokine release, while PTEN overexpression attenuated this effect (P < 0.05, Figure 5A). ROS levels elevated by LPS were suppressed by miR-200a-3p overexpression and stimulated further by PTEN overexpression (P < 0.001, Figure 5B). LPS treatment decreased SOD and CAT activities; overexpression of miR-200a-3p increased these antioxidant enzyme activities, while PTEN overexpression reduced (P < 0.05, Figure 5C). Effects of the miR-200a-3p/PTEN Axis on Inflammation and Oxidative Stress in LPS-induced PC12 Cells. miR-200a-3p Mimic and pcDNA-3.1-PTEN Influence Pro-inflammatory Cytokines (A), ROS (B), CAT, and SOD (C). *P < 0.05, ***P < 0.001, ****P < 0.0001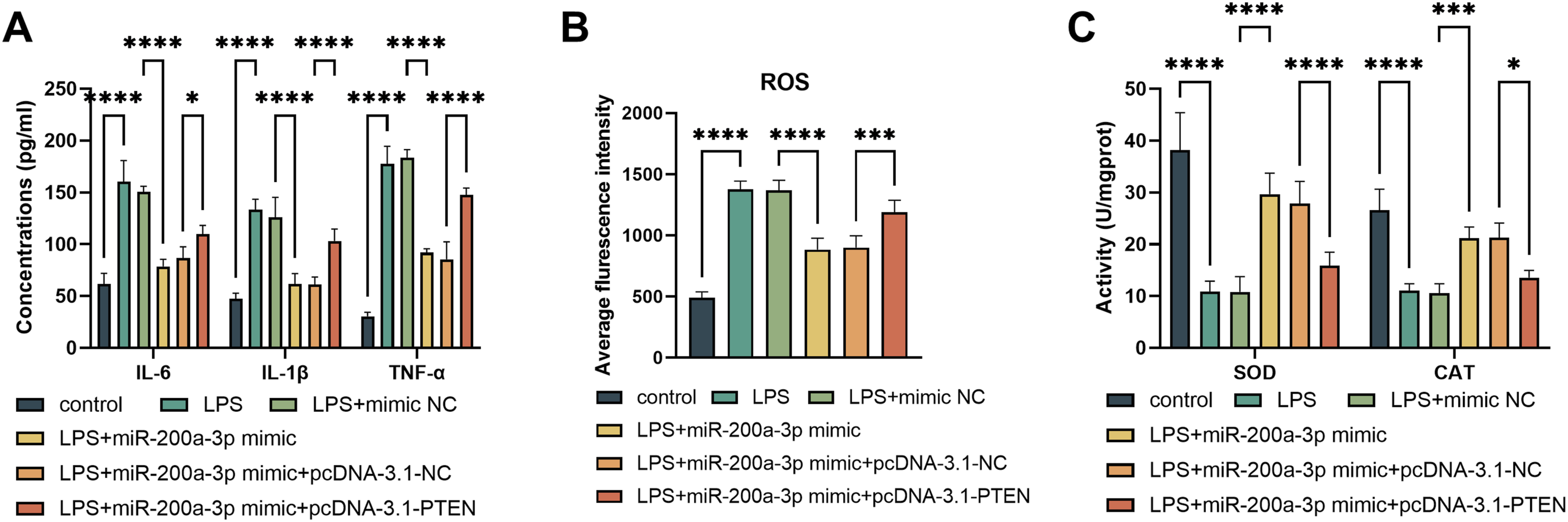
Discussion
SCI remains a destructive disease with a high incidence and long-term disability rate worldwide. 23 Although some drugs have demonstrated excellent improvements in neurological symptoms, such as erythropoietin (EPO) and 4-aminopyridine,24,25 their exact efficacy remains controversial, and none can be considered standard treatment options. 26 Currently, imaging techniques such as MRI and CT scans are the most commonly used methods for diagnosing SCI, 27 but they often fail to accurately reflect functional and molecular changes that occur after injury. 28 These methods are limited in their ability to provide real-time, dynamic insights into ongoing pathological mechanisms such as inflammation, oxidative stress, and neuronal cell death, making early diagnosis and prognosis evaluation particularly difficult. In this context, there is an urgent need for reliable, non-invasive biomarkers that can accurately reflect the underlying molecular alterations in SCI, facilitate early detection, and guide clinical decision-making.
In clinical diagnostics, the detection of miRNA levels in blood, cerebrospinal fluid, or other biological fluids has emerged as an innovative research direction. 29 miRNAs are stably present in human plasma and may serve as potential biomarkers for disease activity, pathogenesis, and prognosis. 30 Recent research has uncovered several miRNAs, like miR-142-5p and miR-223-5p, that hold promise as biomarkers for diagnosing and predicting outcomes in SCI.31,32 miR-200a-3p is also believed to have a close connection to SCI; studies have shown that miRNA levels decrease in zebrafish models of spinal cord injury and that these molecules are also involved in other neurological diseases like multiple sclerosis and AD.33,34 In this study, a cohort of SCI patients was recruited, and serum analysis revealed that miR-200a-3p levels were notably decreased in SCI patients. These results align with earlier studies on different neurological disorders. Importantly, ROC analysis demonstrated that miR-200a-3p could distinguish SCI patients from individuals with normal neurological function and differentiate between complete and incomplete SCI, indicating its potential clinical utility.
In musculoskeletal diseases such as osteoarthritis, tendon injuries, and spinal cord injuries, the expression of non-coding RNAs such as siRNA,35-37 circRNA, 38 lncRNA and miRNA is often abnormal, affecting inflammatory responses, cell apoptosis, and tissue repair processes. In-depth investigation of the mechanisms of these non-coding RNAs provides a solid foundation for the development of new diagnostic tools and therapeutic strategies. miRNAs influence gene expression by altering the translation efficiency or stability of target mRNAs. 39 By analyzing the regulation patterns between miRNAs and mRNAs, researchers can gain a deeper understanding of the underlying biological mechanisms. To this end, databases such as TarBase and TargetScan, along with dual-luciferase reporter assays and RNA pull-down experiments, were used to verify the targeting relationship between miR-200a-3p and PTEN. Further analysis showed that PTEN was upregulated in SCI patients, consistent with Guan et al 40 's findings of increased PTEN expression in rat models of SCI. The ROC curve also indicated that PTEN could distinguish SCI patients effectively. Previous research has shown that PTEN not only plays a crucial role in regulating neuronal differentiation 41 but also participates in various central nervous system disorders, with inhibition of PTEN being a promising approach to promote functional recovery after SCI.42,43 Although changes in peripheral blood following spinal cord injury reflect systemic damage, to investigate the mechanisms of miR-200a-3p and PTEN in neurons, we selected PC12 cells to establish an in vitro SCI model to explore the molecular mechanisms of the miR-200a-3p/PTEN axis in SCI. Using LPS-induced PC12 cells to mimic inflammation and oxidative stress associated with SCI, we observed that overexpression of miR-200a-3p significantly improved cell viability, reduced apoptosis, suppressed pro-inflammatory cytokines (IL-6, IL-1β, TNF-α), decreased ROS production, and enhanced antioxidant enzymes (SOD and CAT). These results are similar to Yang et al 44 's findings that miR-200a-3p can partially counteract the neuronal damage caused by LINC00319 overexpression following oxygen-glucose deprivation. Conversely, PTEN overexpression reversed these protective effects, indicating that miR-200a-3p exerts neuroprotective, anti-inflammatory, and antioxidant effects via inhibition of PTEN. This mechanism aligns with previous research on PTEN’s role in neuroprotection 45 and provides a systematic demonstration of how miR-200a-3p regulates oxidative stress and inflammatory responses through PTEN.
In this study, we first explored the expression changes, functions, and clinical potential of the miR-200a-3p/PTEN axis in SCI, but several limitations remain. First, this study is cross-sectional in nature, with all clinical samples collected at a single time point during the acute phase. It lacks longitudinal dynamic monitoring data across different post-injury periods (eg, subacute phase, rehabilitation phase), thus preventing a comprehensive assessment of the temporal trajectory of miR-200a-3p expression levels and its correlation with neurological recovery (eg, improvement in ASIA scores). Second, in vivo functional gain or loss experiments in animal models have not been conducted to further confirm the neuroprotective effects of this axis. Additionally, recent studies suggest other miRNAs (eg, miR-142-5p, miR-223-5p) hold diagnostic and prognostic value in SCI; this study did not perform parallel comparisons with these molecules to clarify the unique advantages of miR-200a-3p. Looking ahead, we will undertake the following initiatives: First, establish a longitudinal follow-up cohort of SCI patients to systematically analyze the relationship between miR-200a-3p expression dynamics and long-term clinical outcomes, advancing its role as a prognostic biomarker. Second, utilize in vivo gene editing and inhibitor techniques in animal models to clarify the neuro-reparative effects of targeted intervention on the miR-200a-3p/PTEN axis. Third, integrate multi-omics data with machine learning methods to construct a multi-molecular biomarker predictive model, enhancing diagnostic and prognostic accuracy. These studies will advance the translational journey of miR-200a-3p from fundamental mechanisms to clinical applications.
Conclusions
The results of this study suggest that serum levels of miR-200a-3p and its target PTEN are differentially expressed in SCI patients, with ROC analysis suggesting their potential diagnostic value. Additionally, miR-200a-3p effectively alleviates inflammation and oxidative stress by targeting PTEN, thereby protecting neurons. These findings provide new insights into the molecular mechanisms underlying SCI and offer promising biomarkers and therapeutic targets for early diagnosis and intervention.
Footnotes
Ethical Considerations
This study was approved by the Ethics Committee of Zhucheng People’s Hospital.
Consent to Participate
All participants signed informed consent forms.
Author Contributions
Conceptualization, G.X.L. and R.Q.; Data curation, G.G.W., H.T.X. and J.X.Z.; Investigation, G.X.L., R.Q., G.G.W. and H.T.X.; Methodology, G.G.W. and H.T.X.; Roles/Writing - original draft, G.X.L. and R.Q.; Writing - review & editing, H.T.X. and J.X.Z..
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
