Abstract
Study Design
A multimethod experimental study.
Objectives
Spinal cord injury (SCI) has devastating neurological consequences, mainly through secondary injury mechanisms. Chinese medicine-derived flavonoids, including rutin (RUT) and ochnaflavone (OCE), have shown potential in modulating these processes, although their molecular mechanisms are not understood. This study aims to elucidate the neuroprotective mechanisms of RUB and OCE in SCI, with an emphasis on their regulatory function in microglia and the PI3K/AKT and NF-κB signaling pathways.
Methods
Single-cell RNA sequencing (scRNA-seq) of SCI mouse models was used to identify the inflammatory microglial subtype and define its molecular signature. Network pharmacology predicted that RUB and OCE targets overlap with SCI pathology. Their anti-inflammatory and anti-apoptotic effects were tested by vitro assays using LPS-stimulated BV2 microglia and a microglia-neuron co-culture system. In vivo validation was conducted using a murine SCI model to test protein expression by western blotting, immunofluorescence, and Enzyme-linked immunosorbent assay (ELISA).
Results
RUB and OCE synergistically inhibited LPS-induced microglial activation with significant downregulation of pro-inflammatory cytokines (TNF-α, IL-1β, and IL-6) and neuronal apoptosis markers (Bax and cleaved Caspase-3) and upregulation of anti-apoptotic B. Mechanistically, the combination therapy suppressed the phosphorylation of PI3K, AKT, IKKβ, and NF-κB p65 without affecting their protein levels. These molecular effects were parallel in vivo, significantly reducing microglial hyperactivation and apoptotic signaling.
Conclusion
Our findings suggest that targeting the PI3K/AKT and NF-κB pathways may be an effective strategy for inhibiting secondary damage post-SCI, offering a novel therapeutic approach to reshape the post-injury environment and restore neural homeostasis.
Introduction
Spinal cord injury (SCI) is a devastating condition resulting from traumatic or non-traumatic insults to the central Nervous System (CNS), disrupts structural and functional integrity, and causes subsequent impairment of sensory, motor, and autonomic functions. 1 With an estimated annual incidence of 250 000 to 500 000 cases worldwide, Spinal cord injury (SCI) has profound psychological and socioeconomic burdens.2,3 The condition progresses through two phases: primary injury, referred to as immediate mechanical damage to the spinal cord, and secondary injury, characterized by a cascade of complicated molecular events, including neuroinflammation, oxidative stress, apoptotic cascades, and glial scar formation, which exacerbate the initial disruption.4,5 Despite therapeutic strides in acute-phase interventions (surgical decompression and methylprednisolone protocols) and chronic rehabilitation, functional recovery remains suboptimal. 6 This therapeutic impasse underscores the need to identify immune-mediated secondary injury mechanisms.
Recent advances in high-throughput omics, particularly single-cell RNA sequencing (scRNA-seq), have revolutionized our understanding of post-SCI cellular heterogeneity.7,8 This technology delineates immune cell dynamics within the injury microenvironment, illuminating the spatiotemporal transcriptome shifts underlying synaptic remodeling and regenerative failure. 9
Microglia, the resident mononuclear phagocytes of the CNS, play essential roles in maintaining neural homeostasis by continuously monitoring the microenvironment, clearing cellular debris, and modulating synaptic plasticity.10,11 Following SCI, microglia can polarize in response to environmental stimuli: the pro-inflammatory M1 phenotype (induced by lipopolysaccharide (LPS) or interferon-γ (IFN-γ)) or the anti-inflammatory M2 phenotype, which facilitates inflammation resolution and tissue repair. 12 These polarized microglia mediate SCI outcomes through several signaling pathways, including NF-κB, STAT1/3, AMPK-mTOR, and Nrf2/HO-1, which regulate inflammatory responses, autophagy, and oxidative stress. 13 Although mechanistic studies have elucidated the multiple regulatory roles of microglia in SCI, their clinical translational potential remains unclear.
Ochnaflavone (OCE), a natural biflavonoid isolated from plants of the Ochnaaceae family, exhibits potent anti-inflammatory, antioxidant, and anti-apoptotic properties.14,15 Recent studies have shown that Ochnaflavone exerts broad anti-inflammatory effects by inhibiting key enzymes, such as cyclooxygenase-2 (COX-2) and 5-lipoxygenase (5-LOX), and by suppressing the biosynthesis of leukotrienes. 16 Moreover, this compound modulates multiple signaling pathways, including NF-κB and Nrf2/HO-1, to attenuate oxidative stress and prevent apoptosis. 17 Notably, it inhibits nuclear translocation of the p65 subunit of NF-κB, thereby blocking the transcriptional activation of pro-inflammatory genes such as iNOS and COX-2. 18 Collectively, these mechanisms highlight OCE’s potential as a therapeutic candidate for inflammation.
Rutin (RUB) is a naturally occurring flavonoid found in fruits and vegetables. Numerous studies have demonstrated that Rutin possesses a broad spectrum of pharmacological activities, including anti-inflammatory, analgesic, and antidepressant effects. 19 Mounting research has revealed this flavonoid plays a protective role in various neurodegenerative disorders by modulating multiple signaling pathways, including NF-κB, Nrf2, and PI3K/Akt, thereby exerting cytoprotective and anti-apoptotic effects and suppressing astrogliosis.20,21 This evidence highlights RUB’s potential therapeutic value for inflammation and neurodegenerative diseases.
This study is the first to report that OCE and RUB dual therapy attenuates neuroinflammation and neuronal apoptosis by concurrently targeting the PI3K/AKT and NF-κB signaling pathways. These findings provide novel insights into microglia-driven persistent inflammation in patients post-spinal cord injury (SCI). They may also pave the way for the development of biologics or small-molecule cocktails to improve the quality of life of patients with SCI.
Materials and Methods
Bioinformatic Analysis of Public Datasets
Three SCI-related transcriptomic datasets (GSE125630, GSE129694, GSE166009) were retrieved from GEO.22,23 Raw data were processed using R (version 4.1.0) for normalization, batch correction, and differential expression analysis (criteria: |log2FC| > 1, FDR <0.1). Common DEGs across the datasets were identified using Venn analysis.
Single-Cell RNA Sequencing Analysis
The datasets of Single-cell RNA sequencing (scRNA-seq) which are: (GSE205037, GSE203330, and GSE182803) from mouse SCI models (10X Genomics) were analyzed. The samples of post-injury (n = 47) were selected. Data pre-processing in Seurat included quality control (cells: >1000 UMIs, <20% mitochondrial genes; genes: detected in ≥3 cells), doublet removal, and batch correction. Subsequently, dimensionality reduction and clustering were performed. The cell types were annotated manually using published marker genes. Functional enrichment (GO/KEGG) was performed using clusterProfiler (P < 0.05) and visualized using ggplot2. GSEA was conducted against KEGG gene sets (FDR < 0.25), prioritizing the pathways associated with neuroinflammation and neural repair.
Network Pharmacology
Potential therapeutic targets of RUB and ONCE were predicted using SwissTargetPrediction, SEA, and PharmMapper databases, with species restricted to Homo sapiens. The predicted targets were subsequently standardized using the UniProt database. The related targets of SCI-(relevance score ≥25) were retrieved from GeneCards and OMIM databases, followed by deduplication. Intersection genes between drug and disease targets were identified using Venn tools and were defined as candidate therapeutic targets.
The network of the protein-protein interaction (PPI) was constructed using the STRING database (confidence score ≥0.9) and visualized using Cytoscape. Topological parameters (degree centrality) were calculated using NetworkAnalyzer software. The core hub genes were screened using the CytoHubba plugin (degree algorithm and MCODE clustering).
Functional annotation (GO) and KEGG pathway enrichment analyses were performed using MetaScape (P < 0.05). The signaling pathway of PI3K/AKT was prioritized because of its relevance in neuroprotection and inflammatory regulation.
Cell Culture and Treatment
The microglial cell line BV2 (CL-0493; Wuhan Pricella Biotechnology Co, Ltd, China) and mouse spinal cord neurons (CP-M178; Wuhan Pricella Biotechnology Co, Ltd, China) were used in this study. The cells of BV2 were cultured in complete DMEM (Dulbecco’s Modified Eagle Medium supplemented with 10% fetal bovine serum (FBS) and 1% penicillin-streptomycin) (Thermo Fisher Scientific Inc, USA), and cultured in mouse spinal cord neuron complete culture medium (CM-M178, Wuhan Pricella Biotechnology Co, Ltd, China) at 37°C under 5% CO2. Cell density and potential pathogenic contamination were monitored daily using a microscope. Subculturing was performed when cell confluence reached approximately 90% at a 1:3 to 1:4 ratio, with a passage frequency maintained 2-3 times per week. Cells were resuspended in 1 mL serum-free cell freezing medium and immediately transferred to a −80°C freezer for 24 h, followed by long-term storage in liquid nitrogen for cryopreservation. For cell recovery, the frozen cryovials were rapidly thawed in a 37°C water bath with gentle agitation for 2 min. The thawed cell suspension was transferred to a culture dish, and the medium was replaced within 24 h post-thawing.
A co-culture system was established using Transwell inserts (6.5 mm diameter, 0.4 μm pore size; Corning Inc, USA) to evaluate the interaction between BV2 microglial cells and mouse spinal cord neuronal cells. Briefly, neuronal cells were seeded at a density of 5 × 104 cells/well into the bottom chamber of a 24-well culture plate and allowed to adhere for 24 h until they reached a healthy growth status. The cells of BV2 pretreated with the indicated compounds were seeded at a density of 2 × 105 cells/inserted into the upper Transwell chambers. Once BV2 cells had adhered stably, the Transwell inserts were gently placed into wells containing neurons, establishing an indirect co-culture system.
The pharmacological agents used in this study were lipopolysaccharide (LPS), rutin (RUB), and ochnaflavone (OCE). All compounds were dissolved according to the manufacturer’s recommended protocols to achieve stock concentrations and stored at −20°C until use. BV2 cells were stimulated with 1 μg/mL LPS for 24 h to establish a neuroinflammatory cell model.
In a solvent mixture, the Rutin (RUB) was prepared of 10% DMSO, 30% polyethylene glycol 300, 5% Tween-80, and 45% physiological saline. In experiments investigating RUB’s effects on apoptosis and inflammation, cells were treated with RUB at the concentration gradients specified in the Results section.
In serum- and antibiotic-free DMEM, the Ochnaflavone (OCE) was dissolved to prepare the stock solutions. To evaluate OCE’s regulatory effects on apoptosis and inflammatory responses, cells were exposed to OCE at the concentration gradients indicated in the results section for 24 h before sample collection.
In the combined drug administration experiments, the individual compounds were first diluted to their target concentrations in separate media. These solutions were then simultaneously added to the cell culture system. After 24 h of exposure, cells were harvested for subsequent analyses.
Animal Experiments
Female C57BL/6J mice (8 weeks old, with an initial body weight of approximately 20 g) were purchased from Nanjing Model Animal Research Center (NMARC, China). Animals were housed under specific pathogen-free (SPF) conditions with a 12-hour light/dark cycle and provided ad libitum access to food and water. The mice were randomly allocated to each experimental group.
To establish a spinal cord injury (SCI) model, a modified infinite Horizon (IH) impactor (Precision Systems and Instrumentation, USA) was employed to induce thoracic spinal cord contusion. 24 Briefly, the mice were anesthetized with 2% sodium pentobarbital (50 mg/kg, intraperitoneal injection). The absence of pedal withdrawal reflexes was used as an indicator of surgical anesthesia. After shaving and disinfecting the dorsal thoracic region, a midline incision was made over the T10–T12 vertebrae. Following laminectomy at T10–T11, the spinal cord was exposed and stabilized. A standardized contusion injury (force: 5 kdyn, dwell time: 0.1 s) was delivered using an IH impactor to mimic moderate SCI. The sham-operated mice underwent identical procedures without any mechanical impact. The incisions were closed in layers, and postoperative analgesia (meloxicam, 2 mg/kg, subcutaneous) was administered for 72 h.
Manual bladder expression was performed thrice daily until spontaneous micturition resumed. Mice with persistent urinary retention (>48 h) were euthanized under deep anesthesia to comply with ethical standards.
Ceftiofur sodium (5 mg/kg) was subcutaneously injected daily for 7 days post-surgery.
28 days after surgery, the mice were transcardially perfused with ice-cold PBS under deep anesthesia. Spinal cord segments (T10–T12) were collected for histopathological and molecular analysis.
Protein Extraction and Quantification
Tissue samples or adherent cells were lysed in ice-cold RIPA buffer containing phosphatase/protease inhibitors and PMSF (Cell Signaling Technology). After 30 min of incubation on ice and vortexing, lysates were centrifuged (12 000 rpm, 15 min, 4°C), and supernatants were collected. Protein concentrations were determined via BCA assay (Thermo Fisher) following the manufacturer’s instructions.
Western Blotting
Protein samples were denatured in 5× Laemmli buffer (95°C, 10 min), resolved on SDS-PAGE gels, and transferred to PVDF membranes. Membranes were blocked with 5% BSA/TBST (2 h, room temperature), incubated with primary antibodies (3% BSA/TBST, 4°C overnight): Anti-Bcl-XL (1/1000, ab32370, abcam), Anti-Bax (1/1000, ab32503, abcam), Anti-Caspase-3 (1/1000, ab13585, abcam), Anti-Cleaved Caspase-3 (1/1000, ab214430, abcam), Anti-TNF alpha (1/1000, ab183218, abcam), Anti-IL-1 beta (1/1000, ab283818, abcam), Anti-IL-4 (1/1000, ab84269, abcam), Anti-IL-6 (1/1000, ab290735, abcam), Anti-IL-10 (1/1000, ab133575, abcam), Anti-EC-SOD (1/1000, ab80946, abcam), Anti-Phospho-PI3 Kinase (1/1000, #17366, Cell Signaling TECHNOLOGY), Anti-PI3K-gamma (1/1000, ab302958, abcam), Anti-Phospho-Akt (1/2000, #4060, Cell Signaling TECHNOLOGY), Anti-AKT (1/1000, ab38449, abcam), Anti-Phospho-mTOR (1/2000, #5536, Cell Signaling TECHNOLOGY), Anti-mTOR (1/1000, ab134903, abcam), Anti-Phospho-p70 S6 Kinase (1/2000, #9205, Cell Signaling TECHNOLOGY), Anti-S6K1 (1/1000, ab32359, abcam), Anti-Phospho-IKKα/β (1/2000, #2697, Cell Signaling TECHNOLOGY), Anti-IKK alpha (1/1000, ab32041, abcam), Anti-IKK beta (1/1000, ab124957, abcam), Anti-NF-kB p65 (1/1000, ab32536, abcam), Anti-Histone H3 (1/1000, ab1791, abcam), Anti-GAPDH (1/10000, ab8245, abcam), Anti-β-Actin (1/2000, #4967, Cell Signaling TECHNOLOGY) washed (TBST, 3 × 5 min), and probed with HRP-conjugated secondary antibodies (3% BSA/TBST, 1 h, RT): Anti-rabbit IgG (1/3000, #7074, Cell Signaling TECHNOLOGY). After the final washes, signals were developed using ECL substrate and captured using a Tanon 5200 imaging system. Band intensities were quantified using ImageJ software (NIH) and normalized to GAPDH or β-actin.
Immunofluorescence Staining
Spinal cord tissues were fixed in 4% paraformaldehyde (PFA) for 24 h, cryoprotected in 30% sucrose solution at 4°C until sinking, and embedded in OCT compound (Sakura Finetek). Coronal sections (10 μm thickness) were cut using a cryostat (Leica CM1950) and stored at −80°C. For staining, sections were rewarmed, permeabilized with 0.3% Triton X-100/PBS for 15 min, blocked with 20% goat serum/PBS for 1 h at room temperature, and incubated with primary antibodies (diluted in 5% goat serum/0.1% Triton X-100/PBS): Anti-TNF-α (1/400, #8184, Cell Signaling TECHNOLOGY), Anti-Iba1 (1/400, #17198, Cell Signaling TECHNOLOGY), Anti- IL-6 (1/400, #12912, Cell Signaling TECHNOLOGY), anti-Phospho-Akt (1/400, #4060, Cell Signaling TECHNOLOGY), and anti-IKKα (1/400, #11930, Cell Signaling TECHNOLOGY) at 4°C overnight. After washing with PBS (3 × 5 min), the sections were stained with Alexa Fluor 488/594-conjugated secondary antibodies (1:500, Cat#A-11008, Cat#A-11012, Invitrogen) for 2 h at room temperature. Nuclei were counterstained with DAPI (1 μg/mL, 5 min), and slides were mounted with ProLong Diamond Antifade Mountant (Thermo Fisher). Images were acquired using a laser-scanning confocal microscope (Zeiss LSM 900).
Enzyme-Linked Immunosorbent Assay (ELISA)
Target protein levels in cell culture supernatants were quantified using a 96-well ELISA kit (R&D Systems), following the manufacturer’s protocol. Briefly, plates pre-coated with capture antibodies were incubated with the samples or serially diluted standards (100 μL/well, triplicate) for 2 h at 37°C. After washing with PBST (3 × 5 min), HRP-conjugated detection antibodies (100 μL/well) were added and incubated for 1 h at 37°C. After additional washes, TMB substrate (50 μL each of solutions A and B) was added for 15 min at 37°C. Reactions were terminated with 2 M H2SO 3 (50 μL/well), and absorbance was measured at 450 nm using a SpectraMax M3 microplate reader.
Statistical Analysis
All statistical analyses were performed using SPSS 22.0 (IBM). Continuous variables were compared using the independent samples t-test (normally distributed data) or the Wilcoxon signed-rank test (non-parametric paired data). One-way analysis of variance (ANOVA) followed by Bonferroni post-hoc correction was used for multi-group comparisons. Data are presented as the mean ± standard deviation (SD). Statistical significance was defined as P < 0.05, denoted as follows: P < 0.05 (*), P < 0.01 (**), P < 0.001 (***).
Results
Integrated scRNA-seq and Network Pharmacology Identifies RUB/OCE Target PI3K/AKT and NF-κB Signaling in SCI
To elucidate the therapeutic potential of RUB and OCE in spinal cord injury (SCI), we integrated three single-cell RNA sequencing (scRNA-seq) datasets from murine SCI models. We explored their cellular composition and transcriptional profiles. After quality control and doublet removal, 86972 high-quality cells from 47 mice were retained for further analysis. Unsupervised clustering partitioned these cells into 25 subclusters, which were annotated as 11 major cell types: macrophages, microglia, granulocytes, monocytes, astrocytes, oligodendrocytes, B cells, T cells, epithelial cells, endothelial cells, and erythrocytes (Figure 1A). Integrated scRNA-seq and Network Pharmacology Identifies RUB/OCE Target PI3K/AKT and NF-κB Signaling in SCI (A) t-SNE Plot Annotated Single Cells Into 11 Major Cell Types, including Microglia, Macrophages, Monocytes, Granulocytes, Astrocytes, Oligodendrocytes, B Cells, T Cells, Epithelial Cells, Endothelial Cells, and Erythrocytes. (B) Microglia Were Classified Into Four Subtypes: Inflammatory, IFN-Responsive, Homeostatic, and Proliferating. (C) KEGG Pathway Enrichment Analysis of Inflammatory Microglia B Subtype Marker Genes Revealed Activation of PI3K-AKT, NF-κB, Leukocyte Transendothelial Migration, Chemokine Signaling, and Antigen Processing and Presentation Pathways. (D) Venn Diagram Showing 95 Overlapping Targets Between SCI-Related Genes and Candidate Targets of Rutin (RUB) and Ochnaflavone (OCE). (E) KEGG Enrichment of Shared Targets Revealed Significant Involvement in Neurodegeneration, PI3K-AKT Signaling, and Alzheimer’s Disease Pathways. (F) A Compound–Disease–Gene Pathway Network that Integrates SCI, RUB, and Once With Their Predicted and Shared Targets Highlights Key Biological Processes and Signaling Axes. (G) PPI Network Analysis Identified TNF as a Key Hub Gene and Other Inflammation- and Apoptosis-Related Targets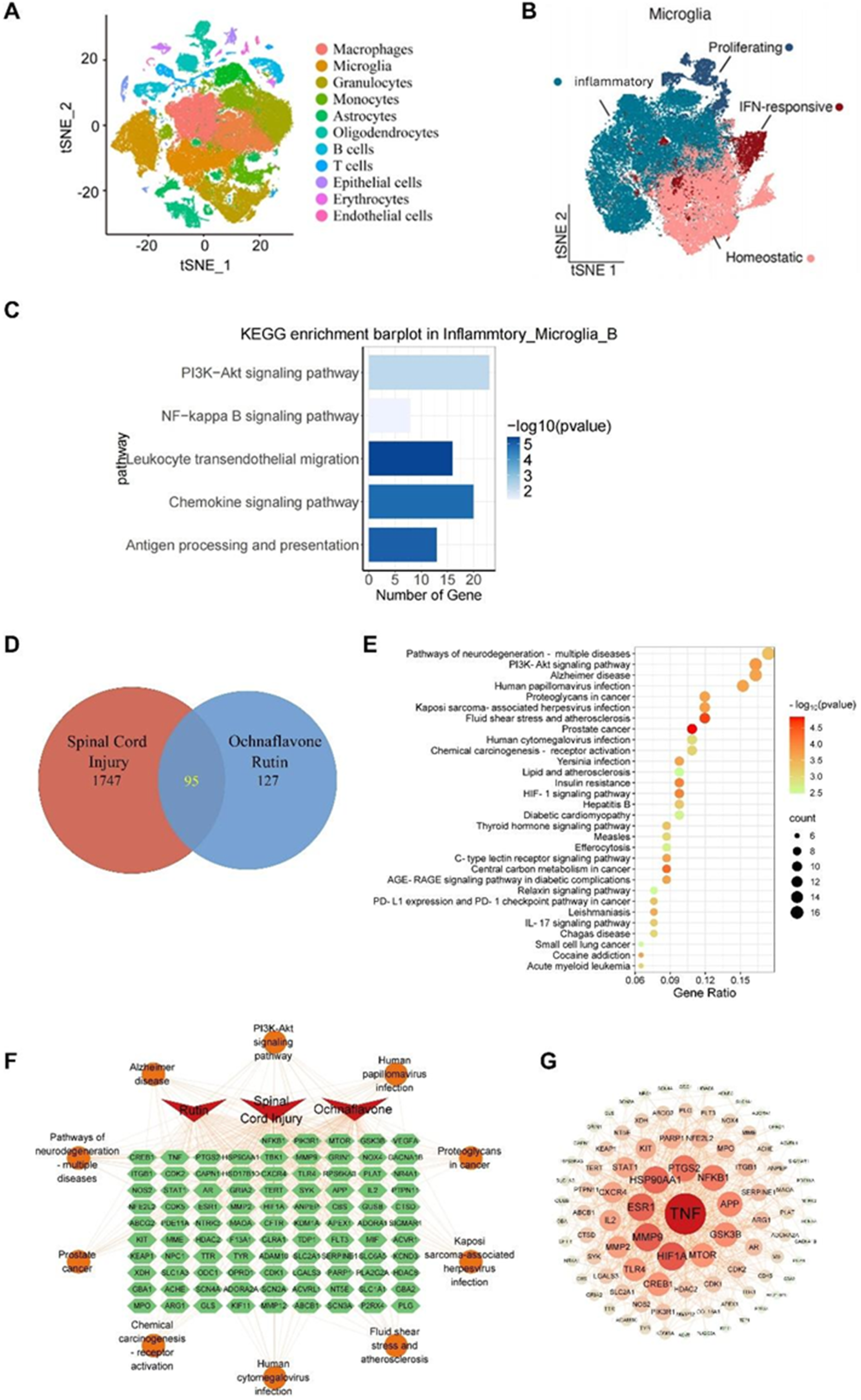
Subsequently, microglia were subclustered into inflammatory A, inflammatory B, homeostatic, and proliferating microglia. SCI mice exhibited significant inflammatory B-type microglial enrichment compared to homeostatic dominance in the sham controls (Figure 1B).
Furthermore, KEGG enrichment analysis demonstrated that the marker genes of inflammatory microglia B were enriched in PI3K/AKT signaling, NF-κB signaling, leukocyte transendothelial migration, chemokine signaling, and antigen processing and presentation (Figure 1C), suggesting their pro-inflammatory roles in SCI progression.
Subsequent network pharmacology analysis revealed 95 overlapping targets between SCI-related genes and candidate RUB/OCE targets (Figure 1D). KEGG analysis demonstrated a significant overlap between RUB/OCE targets and inflammatory microglia B gene signatures, particularly in neurodegeneration, PI3K/AKT, and Alzheimer’s (Figure 1E). Network analysis suggested that these compounds may exert therapeutic effects by interacting with key targets such as NFκB1, PIK3R1, MTOR, and TNF (Figure 1F).
The network analysis of Protein-Protein Interaction (PPI) revealed the core targets of TNF, highlighting its pivotal regulatory role in inflammatory microglial B-mediated SCI (Figure 1G). Other high-degree nodes, including NFKB1, PTGS2, HSP90AA1, MMP9, ESR1, HIF1A, and MTOR, have also been identified as core hubs to be crucial targets that orchestrate neuroinflammation and apoptosis. These findings suggest that RUB and OCE are multi-target agents capable of modulating PI3K/AKT and NF-κB signaling to counteract microglia-driven neuroinflammation and neuronal apoptosis in SCI.
Combined RUB and OCE Treatment Attenuates Pro-inflammatory Cytokine Release and Neuronal Apoptosis in SCI Cellular Models
Previous studies have reported that lipopolysaccharide (LPS) activates BV2 microglia to induce neuroinflammatory responses.
25
To validate the therapeutic effects of RUB and OCE on SCI progression, we established an LPS-induced neuroinflammatory cell model and treated BV2 cells with RUB, OCE, or their combination. Western blotting verified that LPS treatment significantly upregulated pro-apoptotic markers (Bax, cleaved Caspase-3) and downregulated Bcl-x compared to the controls. In contrast, the monotherapy of RUB or RUB + OCE co-treatment markedly reversed these changes (Figure 2A and B). RUB and OCE Attenuate LPS-Induced Neuroinflammation and Apoptosis in BV2 Cells and Spinal Neurons (A) Representative Western Blot Images of Apoptosis-Related Proteins (Bax, Cleaved Caspase-3, Bcl-xL) in LPS-Stimulated BV2 Cells Treated With RUB, OCE, or Their Combination. (B) Quantitative Analysis of (A) (n = 3). LPS Increased Bax and Cleaved Caspase-3 while Reducing Bcl-xL, Effects Reversed by RUB or RUB + OCE (**P < 0.01, ***P < 0.001 vs LPS). (C) Western Blot Images of Inflammatory Cytokines (TNF-α, IL-1β, IL-6, IL-4), Anti-inflammatory IL-10, and SOD in BV2 Cells. (D) Quantification of (C) (n = 3). RUB + OCE Significantly Suppressed Pro-inflammatory Cytokines and Enhanced IL-10/SOD (*P < 0.05, P < 0.01 vs LPS). (E) ELISA of TNF-α and IL-6 in Cell Supernatants (n = 4). RUB + OCE Synergistically Reduced Cytokine Release (*P < 0.001 vs LPS). (F) Immunofluorescence Staining of TNF-α (Red) and IL-6 (Green) in BV2 Cells. Scale bar = 50 μm. (G) TUNEL Assay (Green: Apoptotic Cells) in Mouse Spinal Neurons. Scale bar = 50 μm. Data: Mean ± SD; One-way ANOVA With Post-hoc Test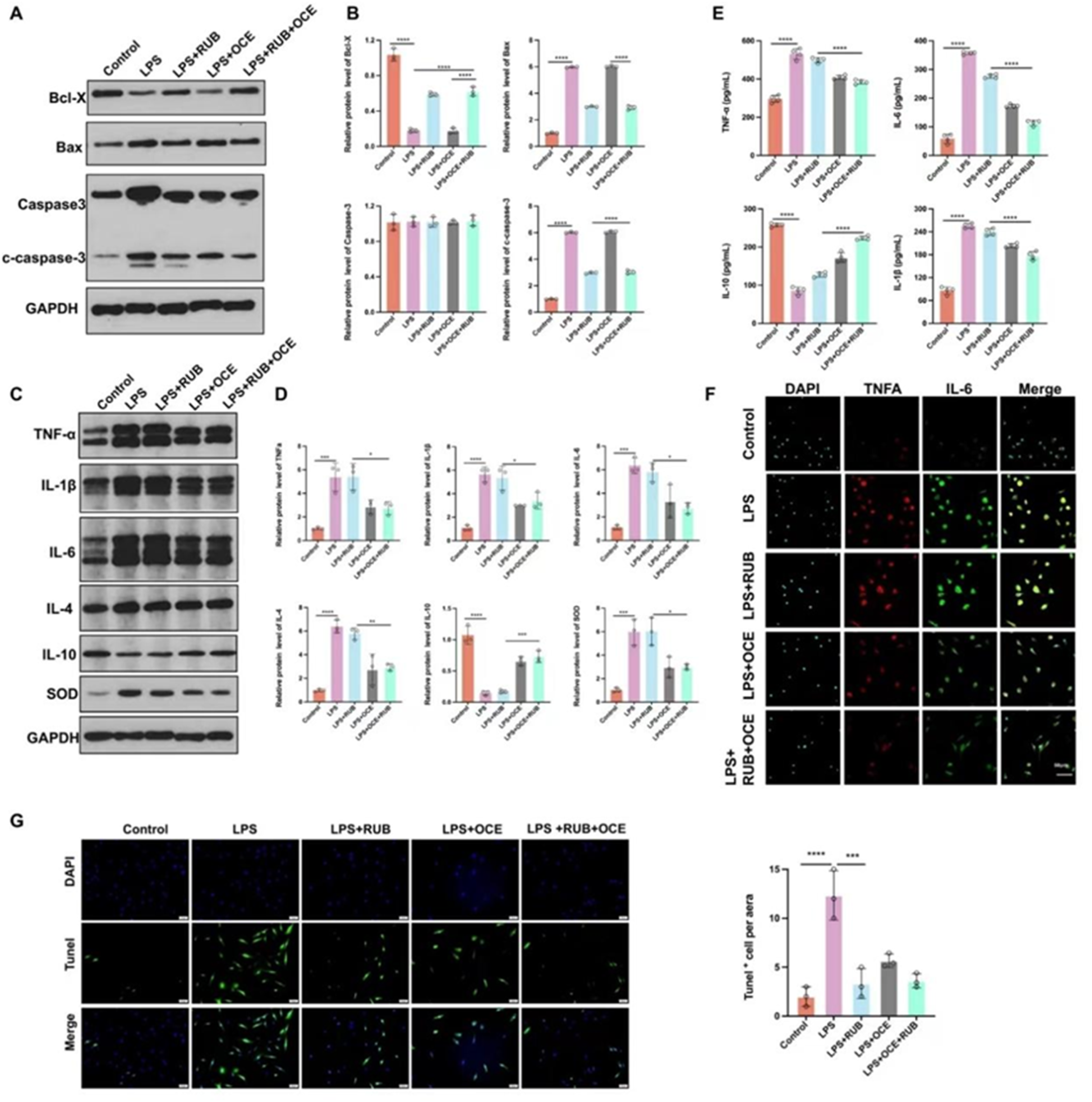
Stimulation of Lipopolysaccharide (LPS) dramatically elevated pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, and IL-4) and SOD while suppressing IL-10. Ochnaflavone (OCE), alone or in combination with RUB, significantly mitigated these inflammatory perturbations (Figure 2C and D). ELISA (Figure 2E) and immunofluorescence (Figure 2F) consistently validated the suppression of TNF-α and IL-1β secretion by the OCE therapy. TUNEL staining further corroborated the anti-apoptotic efficacy of RUB in the mouse spinal neurons (Figure 2G).
These findings suggest that activated microglia-driven neuroinflammation exacerbates neuronal apoptosis, with OCE attenuating the inflammatory cascades and RUB inhibiting apoptosis. Their combined application synergistically targets both the pathological axes, highlighting a dual therapeutic strategy for SCI.
RUB and OCE Synergistically Inhibit Microglia-Driven Neuroinflammation and Neuronal Apoptosis via PI3K/AKT and NF-κB Signaling
To validate network pharmacology predictions implicating the PI3K/AKT and NF-κB pathways in RUB/OCE-mediated neuroprotection, we assessed the phosphorylation dynamics in LPS-stimulated BV2 microglia. Western blot analysis revealed no significant differences in total PI3K, AKT, IKKβ, or IκBα expression between the groups. However, treatment of Lipopolysaccharide (LPS) markedly increased the phosphorylation levels (p-PI3K, p-AKT, p-IKKβ, and p-IκBα) compared to the control group. Notably, the co-treatment of RUB + OCE suppressed LPS-induced phosphorylation (Figure 3A). RUB and OCE Synergistically Inhibit Microglia-Driven Neuroinflammation and Neuronal Apoptosis via PI3K/AKT and NF-κB Signaling (A) PI3K/AKT and NF-κB Signaling in LPS-Stimulated BV2 Cells. Top: Representative Western Blots of PI3K, p-AKT, AKT, NF-κB p65, and IκBα. Bottom: Quantification (n = 3). RUB + OCE Suppressed LPS-Induced PI3K/AKT/NF-κB Activation (**P < 0.01 vs LPS). (B) mTOR/p70S6K Pathway Modulation. Top: Western Blots of mTOR, p-mTOR, p70S6K, and p-p70S6K. Bottom: Quantification (n = 3). RUB + OCE Reversed LPS Effects Similarly to LY294002 (PI3K Inhibitor) (***P < 0.001 vs LPS; ns vs LY294002). (C) NF-κB Nuclear Translocation. Top: Western Blots of Nuclear p65 (P65-Nu), Cytoplasmic p65 (P65-Cy), p-IκBα, and IκBα. Bottom: Quantification (n = 3). RUB + OCE or BAY 11-7082 (NF-κB Inhibitor) Reduced Nuclear p65 and Increased p-IκBα (****P < 0.0001 vs LPS). Data: Mean ± SD; One-way ANOVA With Post-hoc Test. ns, not Significant.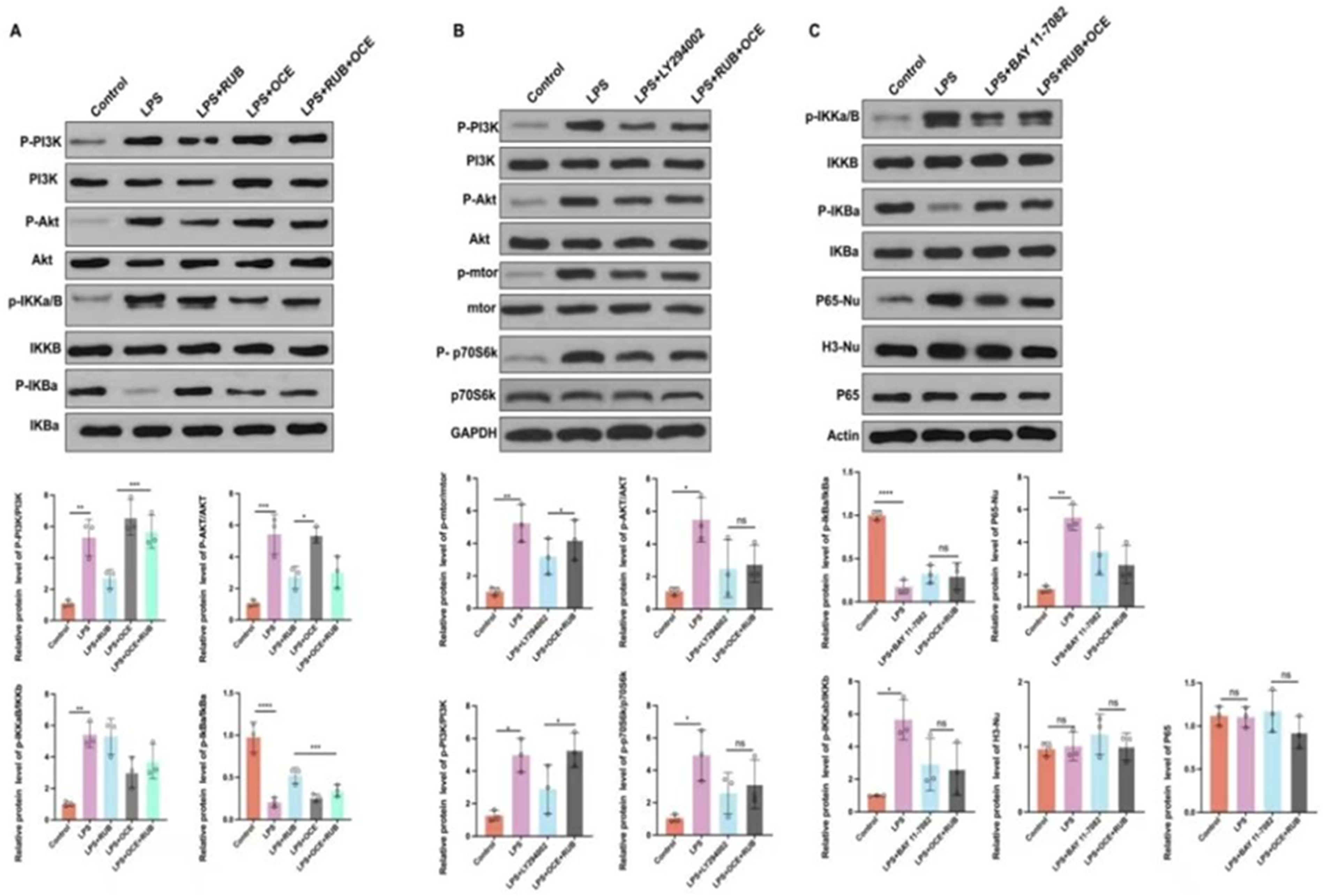
Treatment With RUB + OCE or the NF-κB Inhibitor BAY 11-7082 Significantly Reduced Nuclear p65 (P65-Nu) Levels and Increased IκBα Phosphorylation. Data are Presented as the Mean ± SD; *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001, ns = not Significant (One-way ANOVA)
The signaling pathway of the Mammalian target of rapamycin (mTOR) is a key intracellular regulator vital for modulating the inflammation and proliferation of microglial cells, such as BV2. 26 Its downstream target, p70S6K, is involved in ribosomal protein synthesis and serves as a classic marker for mTOR pathway activation. Western blotting and quantitative analysis results demonstrated that the phosphorylation levels of PI3K, AKT, mTOR, and p70S6K were increased in the LPS treatment group, and RUB + OCE co-treatment suppressed LPS-induced phosphorylation to levels comparable to those of LY294002 (AKT pathway inhibitor) (Figure 3B).
Similarly, Lipopolysaccharide (LPS) triggered robust nuclear translocation of p65 (p65-nu) but reduced the phosphorylation of IκBα without altering the total expression of p65 or IκBα. Both BAY 11-7082 (an NF-κB inhibitor) and RUB + OCE combination therapy significantly reduced nuclear p65 levels and increased IκBα phosphorylation (Figure 3C).
These results demonstrate that RUB and OCE synergistically suppress microglial hyperactivation and neuronal apoptosis by targeting the dual PI3K/AKT and NF-κB signaling cascades, aligning with their predicted multi-pathway regulatory roles.
RUB and OCE Combination Therapy Attenuates Post-SCI Neuroinflammation and Apoptosis via PI3K/AKT/NF-κB Pathways in Vivo
To validate the therapeutic efficacy of RUB and OCE in vivo, we established a murine SCI model and administered RUB, OCE, or a combination of both. Immunofluorescence and quantitative analysis demonstrated that SCI significantly upregulated pro-apoptotic markers (Bax, cleaved Caspase-3) and pro-inflammatory mediators (TNF-α, IL-1β, IL-6, IL-4, and SOD) while downregulating Bcl-x and IL-10. These pathological alterations were robustly reversed by RUB and OCE co-treatment (Figure 4A). RUB and OCE Combination Therapy Attenuates Post-SCI Neuroinflammation and Apoptosis via PI3K/AKT/NF-κB Pathways in Vivo (A) IBA1+ Microglia (Red) Co-localized With TNF-α (Green) in Spinal Cord Sections. Right: Quantification of TNF-α+ Microglia (*P < 0.001 vs SCI; n = 5). Scale bar = 10 μm. (B) IBA1/IL-10 Co-staining. Right: IL-10+ Microglia (*P < 0.001 vs SCI). Scale bar = 10 μm. (C) IBA1/IL-6 Co-staining. Right: IL-6+ Microglia (*P < 0.05 vs SCI). Scale bar = 10 μm. (D) Synaptic Markers (Synaptophysin, SYN, Green;) in the Spinal Cord. Right: Quantified Synaptic Puncta Density (***P < 0.001 vs SCI). Scale bar = 5 μm. (E) p-AKT (Green) Immunofluorescence. Right: Quantified p-AKT Intensity (*P < 0.001 vs SCI; n = 5). Scale bar = 50 μm. (F) p-IKKα/β (red) Immunofluorescence. Right: Quantified Intensity (P < 0.01 vs SCI). Scale bar = 50 μm. (G) p-PI3K (Green) and PI3K (red) immunofluorescence. Right: Quantified Intensity (***P < 0.001 vs SCI). Scale bar = 50 μm. Data: Mean ± SD; One-way ANOVA With Post-hoc Test. SCI, Spinal Cord Injury; Sham, Uninjured Controls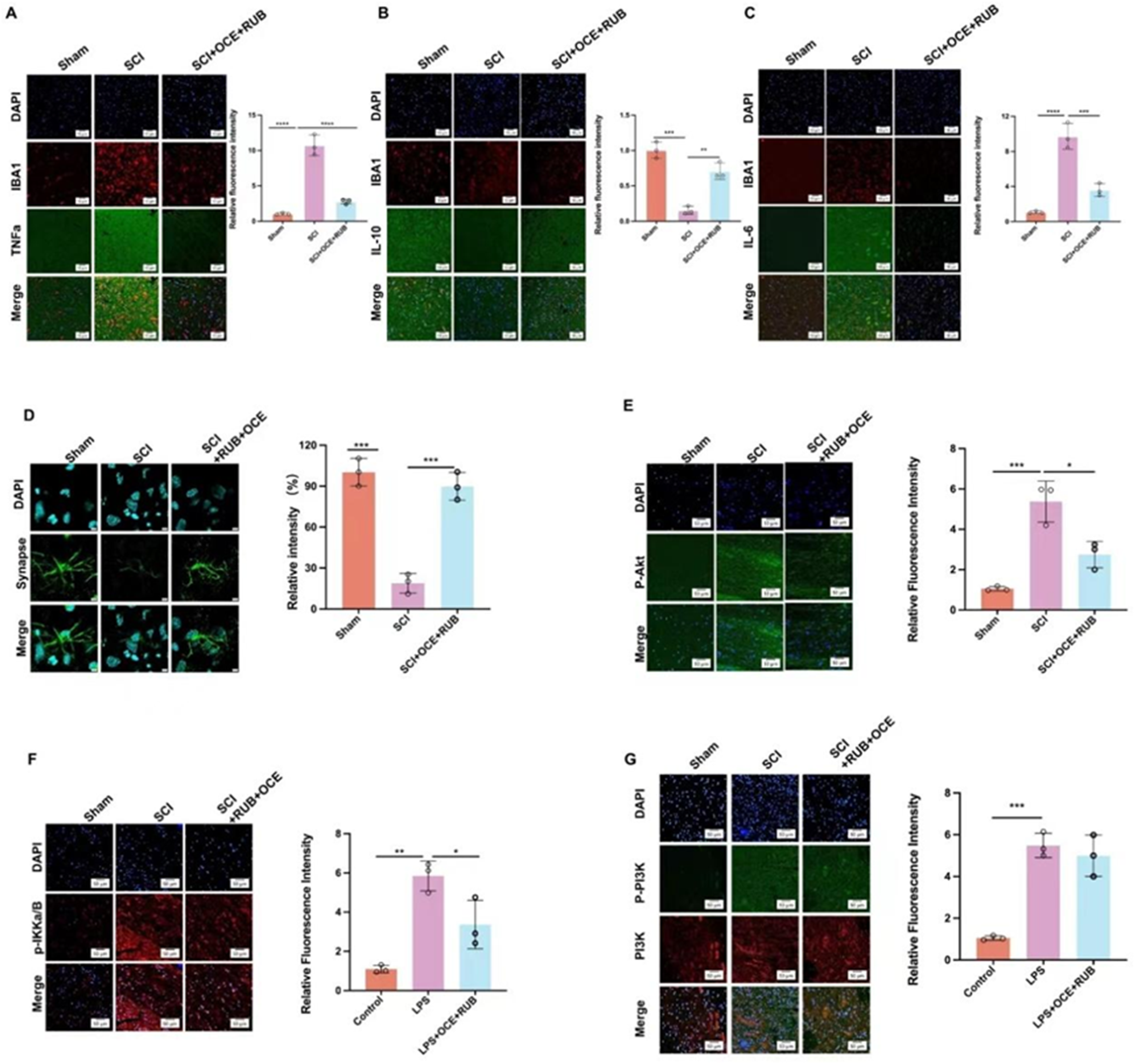
Notably, the total protein levels of PI3K, AKT, and IKKβ remained unchanged across all groups. However, SCI dramatically increased the phosphorylation of these signaling nodes (p-PI3K, p-AKT, and p-IKKβ), in which RUB + OCE was suppressed to near-control levels (Figure 4B). These findings corroborated by Immunofluorescence (Figure 4C), revealing diminished microglial activation and neuronal apoptosis in SCI mice.
Our data indicate that RUB + OCE mitigates SCI-driven neuropathology by dual inhibition of the PI3K/AKT and NF-κB pathways, thereby counteracting microglia-mediated neuroinflammation and secondary neuronal loss.
Discussion
Spinal cord injury (SCI) is a profoundly disabling condition that triggers a cascade of primary and secondary pathophysiological responses that seriously hampers neurological recovery. Although primary mechanical trauma is not reversible, a body of evidence has highlighted the crucial role of secondary events, especially neuroinflammation and apoptosis, in enhancing the deleterious effects of tissue destruction and loss of functionality after brain injury. Some of the cell mediators include microglia, which are central orchestrators of this secondary response and are fast to respond to CNS insults and release a wide range of pro-inflammatory cytokines, chemokines, and reactive oxygen species (ROS), further contributing to neuronal death, axonal demyelination, and synaptic dysfunction The present study prove that combinatorial therapy, including rutin (RUB) and ochnaflavone (OCE), has synergistic neuroprotective effects by modulating crucial intracellular signaling pathways such as PI3K/AKT and NF-κB, which play a significant role in immune responses, inflammation, and cell survival.27,28
The findings of the current study on the dual inhibition of the PI3K/AKT and NF-κB axes are consistent with previous reports highlighting the pervasiveness of these pathways in SCI. For example, activation of the PI3K/AKT signaling system has been found to control neuronal survival, axonal regeneration, and synaptic integrity, and its dysregulation may also lead to gliosis and inhibit reparative processes. 29 In a murine model of SCI, pharmacological manipulation of PI3K/AKT with agents, such as LY294002, alleviated microglial activation. It enhances motor recovery, demonstrating the bidirectional effects of this pathway, which depend on cellular context. 30 Our findings indicate that RUB inhibits PI3K phosphorylation, as well as downstream effectors such as Akt and mTOR, resulting in a reduction in apoptosis markers such as cleaved Caspase-3 and Bax, which corroborates the emerging literature that posits the PI3K/AKT/mTOR axis as a viable option for SCI therapy. 31
Similarly, the signaling pathway of NF-κB is a well-established regulator of the innate immune response and is rapidly activated in microglia after SCI. When activated, NF-κB translocate to the nucleus, promoting the transcription of pro-inflammatory genes, such as TNF-α, IL-1β, and IL-6, which further triggers neuronal damage and glial scar formation.32,33 The present study revealed that OCE blocks the nuclear translocation of the NF-κB p65 subunit and rescues IκBα phosphorylation, successfully quelling inflammatory cytokine release. These observations echo earlier studies that suggest that NF-κB blockade through natural compounds (celastrol or curcumin) provides neuroprotection through glial deactivation and neural protection. 34 Furthermore, no evidence has emerged from RNA-seq-based transcriptome profiling that the signature genes of NF-κB remain upregulated for weeks after injury, thereby sustaining the chronology of neuroinflammation and hampering neurogenesis. 35
The synergistic approach of combining RUB with OCE is a novel aspect of this research, as their interplay appears to produce anti-inflammatory and anti-apoptotic effects more effectively than either monotherapy. Synergistic drug strategies have been increasingly acknowledged for their ability to affect various pathological axes simultaneously, reduce therapeutic doses, and minimize side effects. 36 The additive or synergistic interplay detected in the current study can be explained by the bivalent modulating effects of RUB and OCE on the overlapping but distinct biological targets of the PI3K/AKT and NF-κB cascades. Previous research has demonstrated that bioflavonoids may enhance BBB permeability, pharmacokinetics, and receptor binding affinity. 37 Although RUB preferentially affects apoptotic routes and ROS scavenging, OCE displays a more pronounced immunomodulatory action, providing a complementary clinical profile.
Another noteworthy aspect of this study was the application of single-cell RNA sequencing (scRNA-seq) to determine inflammatory microglial subtypes. This state-of-the-art method has revealed remarkable heterogeneity among CNS immune populations and has mapped the transcriptional trajectories of microglial activation states. 38 Our analysis of scRNA-seq revealed a shift towards an inflammatory microglia subtype B following SCI, characterized by increased gene expression associated with antigen processing, cytokine production, and cell migration. The data reported here are consistent with findings from other groups who previously described disease-associated microglia (DAMs) in models of neurodegeneration and exhibited parallels with our inflammatory B subtype. 39 Such granular-level insights are essential for creating precise therapeutics that selectively impact pathogenic microglial states without destabilizing homeostatic surveillance functions.
Additionally, the reported modulation of the cytokine profile (decrease of TNF-α, IL-1β, and IL-6; the increase of anti-inflammatory cytokine IL-10) corresponds to the notion of polarization of immunity from M1 (pro-inflammatory) to M2 (anti-inflammation), which is essential for promoting axon regeneration, remyelination, and neurovascular remodeling in the injured spinal cord. 40 By modulating upstream signaling mechanisms, RUB and OCE may facilitate a phenotypic shift in microglia, which promotes the establishment of a more regenerative microenvironment. This hypothesis is supported by studies in which other compounds obtained from plants, such as resveratrol and baicalin, promoted M2 polarization and enabled functional recovery in SCI and stroke models.41,42
While this study primarily focused on elucidating the molecular mechanisms underlying the neuroprotective effects of rutin and ochnaflavone, it is essential to consider the clinical implications of our findings. 37 The observed dual inhibition of PI3K/AKT and NF-κB pathways, resulting in significant attenuation of neuroinflammation and neuronal apoptosis, addresses two fundamental pathological processes that directly contribute to functional impairment after SCI. Preclinical evidence consistently demonstrates that interventions reducing neuroinflammation (particularly TNF-α and IL-1β suppression) and preventing neuronal apoptosis correlate with improved locomotor recovery in SCI models. Although functional assessments were beyond the scope of this mechanistic investigation, our demonstration of pathway modulation provides strong rationale for future studies incorporating comprehensive behavioral analyses (eg, BMS scoring, grid walk tests, and electrophysiological assessments) to evaluate functional outcomes directly. The synergistic effects observed at the cellular level suggest this combinatorial approach may offer superior therapeutic potential compared to single-target strategies currently under investigation.
Although the findings are robust, several limitations of this study deserve discussion. First, although the current study revealed molecular and cellular changes after treatment, functional outcomes, including locomotor recovery, sensory thresholds, and electrophysiological measures, were not described and should be included in future experimental studies. Second, the long-term safety profile of RUB and OCE is yet to be fully realized, particularly in the context of chronic administration. Third, although murine models provide informative results, interspecific differences exist in microglial physiology and immunological responses, which must be considered when projecting results to human SCI settings. 43 The pharmacodynamics and pharmacokinetics (including BBB permeability and metabolic stability) of RUB and OCE in humans are also key aspects of future translational research. Furthermore, the study did not cover the possibility of off-target effects or interactions with other CNS cells (eg, astrocytes and oligodendrocytes) that significantly contribute to SCI pathology. For instance, astrocytes can exhibit reactive gliosis and form glial scars that can hinder axon regeneration, and their interaction with microglia can significantly influence inflammatory outcomes. 44 Multi-omics methods, including spatial transcriptomics and metabolomics, may help to better understand the cross-talk between such cell types and the overall effect of RUB/OCE therapy.
Conclusion
The current study provides novel evidence that the combination of Rutin and Ochnaflavone attenuates spinal cord injury-induced neuroinflammation and neuronal apoptosis via dual inhibition of the PI3K/AKT and NF-κB signaling pathways. These findings not only validate the ability of bioactive flavonoids to treat SCI but also promote the efficacy of multi-target approaches as a plausible strategy for overcoming complex neurodegenerative processes. Further studies should be devoted to validating these results in higher-order preclinical models, improving delivery systems (eg, nanoparticles and hydrogels), and initiating clinical feasibility studies to prepare the ground for new adjunct therapies in spinal cord injury treatment.
Footnotes
Acknowledgments
The authors would like to express their gratitude to everyone who assisted in the preparation of this manuscript. We also appreciate the support of the XXX Hospital.
Ethical Considerations
Ethical approval for these procedures has been obtained from the Medical Ethics Committee of The XXX HOSPITAL. All SD rats were housed in the animal facility of XXX HOSPITAL and maintained in a pathogen-free environment. All methods were carried out according to the approved guidelines, and all experimental procedures were conducted under the guidance of the XXX HOSPITAL. All methods are reported in accordance with the ARRIVE guidelines.
Author Contributions
All authors contributed equally.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by 2024 Jilin Provincial Department of Science and Technology, Natural Science Foundation of Jilin Province (Free Exploration Key Project (Stability Support Project)) (YDZJ202401411ZYTS).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The authors declare that all the data supporting the findings of this study are contained within the paper.
Significance Statement
This study demonstrates that Rutin and Ochnaflavone synergistically attenuate spinal cord injury-induced neuroinflammation and neuronal apoptosis by targeting the PI3K/AKT and NF-κB signaling pathways. Applying single-cell RNA sequencing (scRNA-seq) determines inflammatory microglia subtypes. This state-of-the-art method has revealed remarkable heterogeneity among CNS immune populations and mapped the transcriptional trajectories of microglial activation states. It offers novel insights into microglia-driven inflammation and paves the way for innovative, combinatorial treatments to improve functional recovery in patients with spinal cord injury.
