Abstract
Objective
This study aimed to evaluate the clinical characteristics, surgical outcomes, and prognostic factors of cervical spondylosis in patients with dysphagia caused by anterior osteophytes.
Methods
This retrospective study included 28 patients who underwent anterior cervical surgery for dysphagia associated with anterior osteophytes between January 2006 and January 2023. Clinical data, including preoperative evaluations, surgical reports, and follow-up visits, were collected from electronic medical records. Cervical spine degeneration was confirmed by radiographic examinations. Dysphagia severity was assessed by the Functional Outcome Swallowing Scale (FOSS) preoperatively and at follow-up. Statistical analyses were conducted to compare pre- and postoperative outcomes.
Results
The average age was 62.4 years, and 78.6% were men. All patients experienced dysphagia, with severity ranging from grades 1-4. After surgery, 89.3% of the patients showed symptomatic improvement, defined as a reduction of at least one grade on the FOSS. Complications were infrequent, primarily consisting of axial pain (10.7%), along with less common occurrences of esophageal fistula, stenosis, nutritional insufficiency, and myelitis (each at 3.6%). Both the modified Japanese Orthopaedic Association (mJOA) score and Neck Disability Index (NDI) showed significant postoperative improvement. The most frequently involved segments were C5/6 and C4/5.
Conclusion
Anterior cervical osteophyte resection, with or without fusion, appears effective in relieving dysphagia symptoms and improving neurological function in well-selected patients. Further prospective studies are needed to validate these findings and refine surgical strategies.
Introduction
Cervical spondylosis is a degenerative disorder of the cervical spine, typically presenting with neurological or mechanical symptoms. However, in rare cases, excessive anterior osteophyte formation may cause esophageal compression, leading to symptoms, such as dysphagia. 1
Excessive anterior osteophyte formation, often caused by chronic mechanical stress and ossification of the anterior longitudinal ligament, can impinge upon the esophagus. 2 Other conditions, such as Diffuse Idiopathic Skeletal Hyperostosis (DISH), may further contribute to the development of prominent anterior osteophyte and exacerbate esophageal compression.3,4 Such bony overgrowth can mechanically narrow the esophageal lumen, leading to clinical manifestations including dysphagia, odynophagia, or a sensation of food sticking during swallowing. Although uncommon, these symptoms can be severe and significantly impair the patients’ quality of life. 5
Cervical spondylosis with dysphagia represents a rare but clinically important condition that remains underrecognized and poorly understood. 6 The current literature is largely limited to case reports and small case series, with little consensus on surgical indications or long-term outcomes. Our institution, among the largest cervical spine surgery centers in China, has relatively extensive experience with this condition. Here, we presented a comprehensive review of 28 surgically treated patients to explore their clinical characteristics, surgical strategies, and postoperative outcomes.
Methods
Study Design and Setting
This retrospective case series included 28 patients who underwent anterior cervical spinal surgery for dysphagia caused by anterior cervical osteophytes at our institution between January 2006 and January 2023. All patients had a minimum postoperative follow-up of 12 months. The study was approved by the Institutional Ethics Committee, and the requirement for informed consent was waived because of the retrospective design of the study.
Inclusion Criteria
(1) Adults (aged ≥18 years) presenting with dysphagia not attributable to pharyngeal, esophageal, gastric, or neurological disorders, (2) radiographic evidence of anterior cervical osteophytes (MRI or CT), and (3) no history of cervical spine surgery within the previous 12 months.
Exclusion Criteria
(1) Cervical deformity, infection, tumor, or other conditions affecting outcomes, (2) previous surgery involving the neck, pharynx, or esophagus, (3) severe osteoporosis contraindicating surgery, (4) dysphagia due to alternative causes, such as neurological diseases, malignancy, or gastroesophageal reflux disease (GERD), and (5) noncompliance with follow-up protocols.
Evaluation and Treatment Procedures
The severity of dysphagia was retrospectively assessed using the Functional Outcome Swallowing Scale (FOSS) based on clinical records and imaging evaluations. Evaluations were performed independently by two experienced spine surgeons, based on preoperative records. In cases of disagreement, a third senior reviewer was consulted to reach a consensus. 7 Special examinations, including barium swallow tests and esophagoscopy, were performed to evaluate esophageal function and structure.
Surgical intervention was indicated if conservative measures failed or patients presented with severe dysphagia that impaired their quality of life. An anterior surgical approach was adopted in all cases, allowing direct decompression and osteophyte resection. Interbody fusion was performed using autografts, allografts, or synthetic cages, depending on the individual pathology and surgeon preference. 8
Data Collection
The variables collected included demographic data, preoperative comorbidities, dysphagia severity (FOSS), imaging findings, surgical approach, intraoperative details, and complications. 9 The primary outcome was improvement in dysphagia, defined as a reduction of at least one grade on the FOSS from baseline to final follow-up.
Several measures were implemented to minimize the potential bias inherent in retrospective chart reviews. (1) All eligible patients who met the inclusion criteria during the study period were enrolled. The excluded patients (n = 12) had baseline characteristics comparable with those of the included cohort. A comparative summary of the age, comorbidities, osteophyte location, and DISH status is provided in Supplemental Table 1. (2) Data were independently extracted by two investigators using a standardized collection form. Any discrepancies were resolved by a third reviewer. Key clinical variables (FOSS and mJOA) were cross-validated with the operative and follow-up notes. (3) Multivariate regression analyses were performed after adjusting for potential confounders, including age, DISH status, and comorbidities. Sensitivity analyses were performed to test the robustness of the primary findings.
The secondary outcomes included perioperative complications, patient-reported quality of life, functional status, overall patient satisfaction, incidence of axial pain, neurological function, neck disability, and general health status. Descriptive statistics were used to summarize the baseline characteristics of the participants.
Data Analysis
Continuous variables are expressed as means ± standard deviation, whereas categorical variables are presented as frequencies and percentages. Wilcoxon signed-rank tests were used to compare the preoperative and postoperative FOSS, mJOA, and NDI scores because the data were not normally distributed. Statistical analyses were performed using SPSS version 25.0. A P-value of <0.05 was considered statistically significant.
Results
Demographics and Preoperative Data of 28 Patients.

Individual Patient Levels, Symptoms, Surgery, Complications, and FOSS Scores.
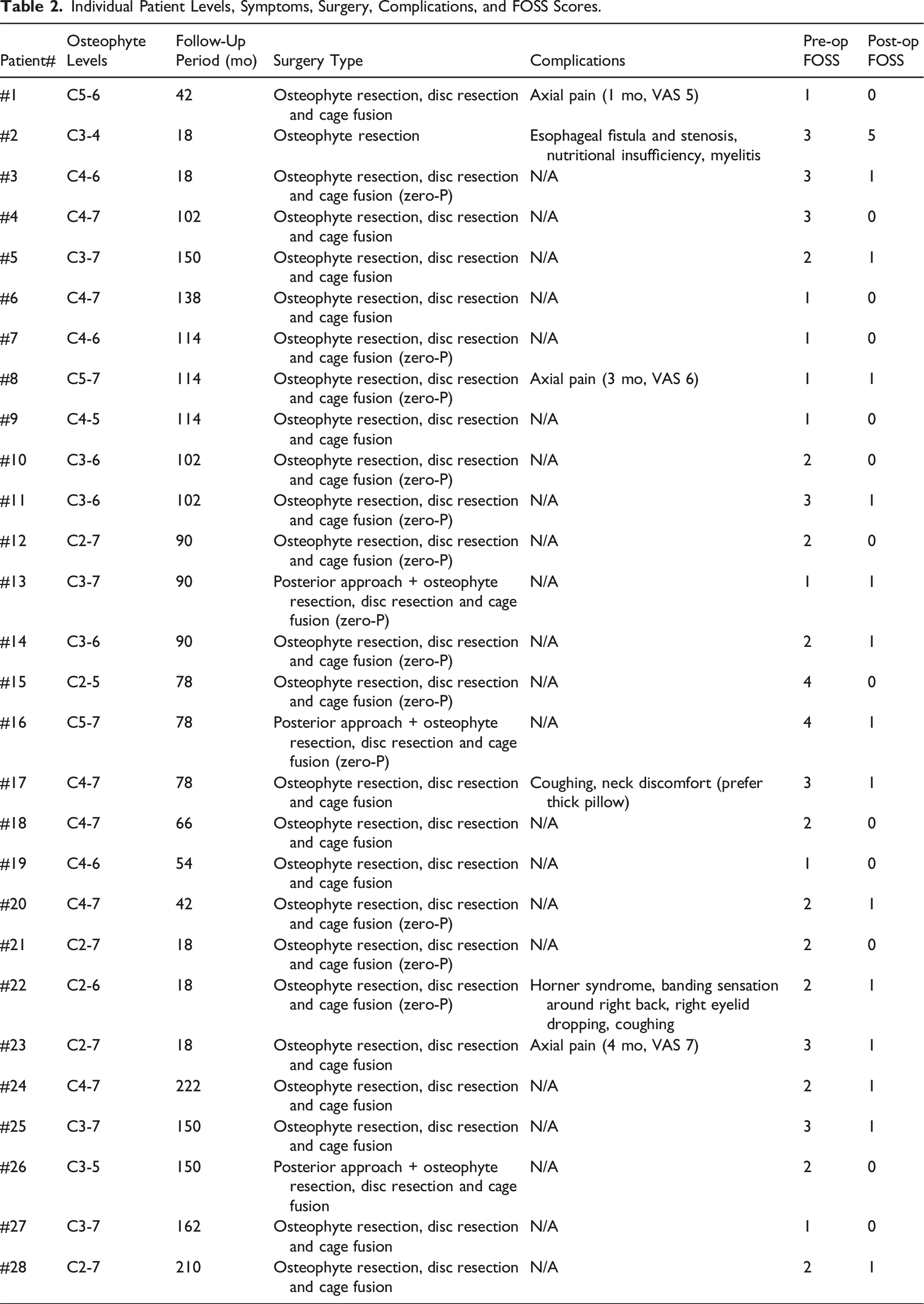
The osteophyte distribution varied across the cervical levels, with the most commonly affected segments being C4/5 and C5/6 (26.7%). The most severely affected osteophytes were concentrated at the C5/6 level (Figure 1). Case #1 Male, 82 years old, progressive dysphagia for 3 years, worsened over the last year. Pre-op FOSS score: 3. Anterior osteophytes at C4-7. Surgery: C4-7 osteophyte resection, C5-7 disc resection and cage fusion. Post-op FOSS score: 0. Recovery rate: 100%. (a) Pre-operative X-ray. (b) Pre Pre-operative CT scan. (c) Pre-operative MRI. (d) Post-operative X-ray.
Surgical resection led to favorable outcomes in all 28 patients. Dysphagia improved in 89.1% of patients, and the mean mJOA score increased to 15.5 ± 2.7. The SF-36 scores showed variable improvements across the domains (Supplemental Table 2).
Perioperative and Postoperative Outcomes for Patients With Cervical Spondylosis and Dysphagia Treated With Anterior Cervical Spine Surgery.

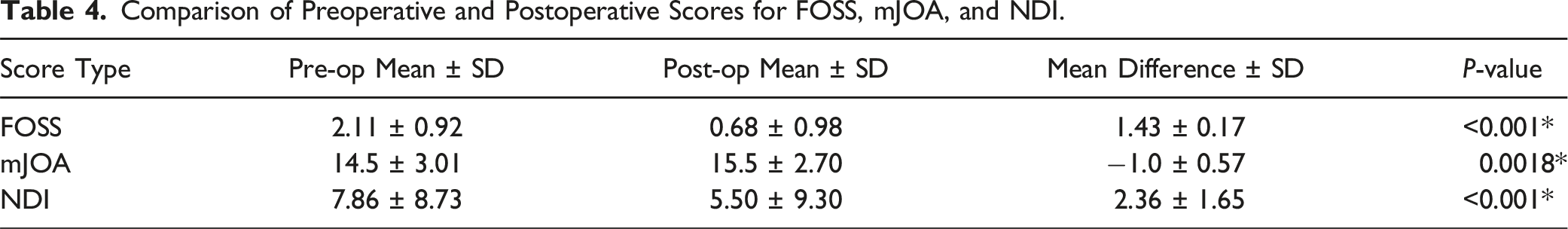
Comparison of Preoperative and Postoperative Scores for FOSS, mJOA, and NDI.
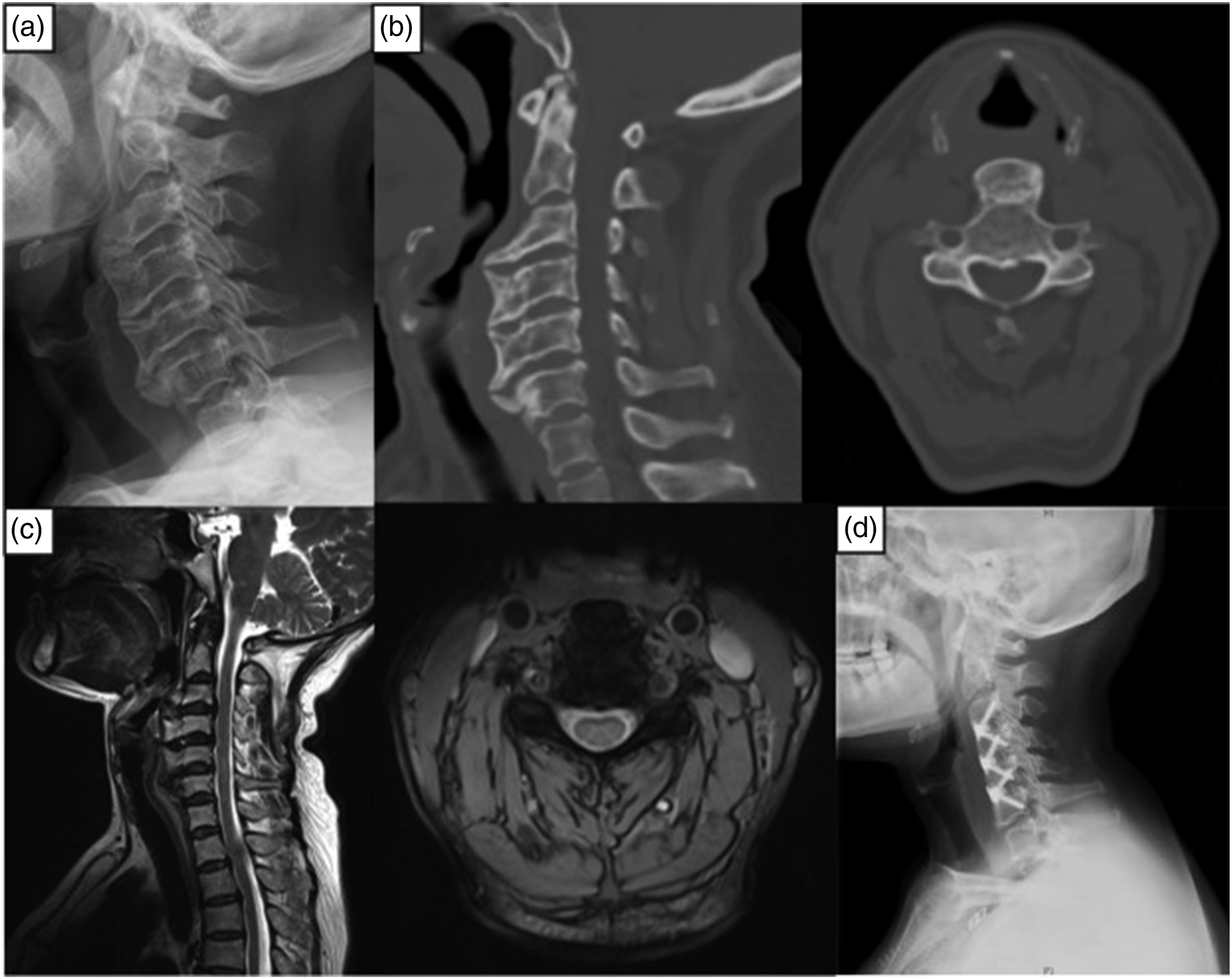
Case #2 Female, 53 years old, dysphagia with upper extremity numbness for 4 years (myelopathy), worsened over the last year. Pre-op FOSS score: 3. Anterior osteophytes at C3-6 (DISH). Surgery: C3-6 osteophyte resection, C3-6 disc resection, and Zero-P cage fusion. Post-op FOSS score: 1. Recovery rate: 90%. (a) Pre-operative X-ray. (b) Pre Pre-operative CT scan. (c) Pre-operative MRI. (d) Post-operative X-ray.
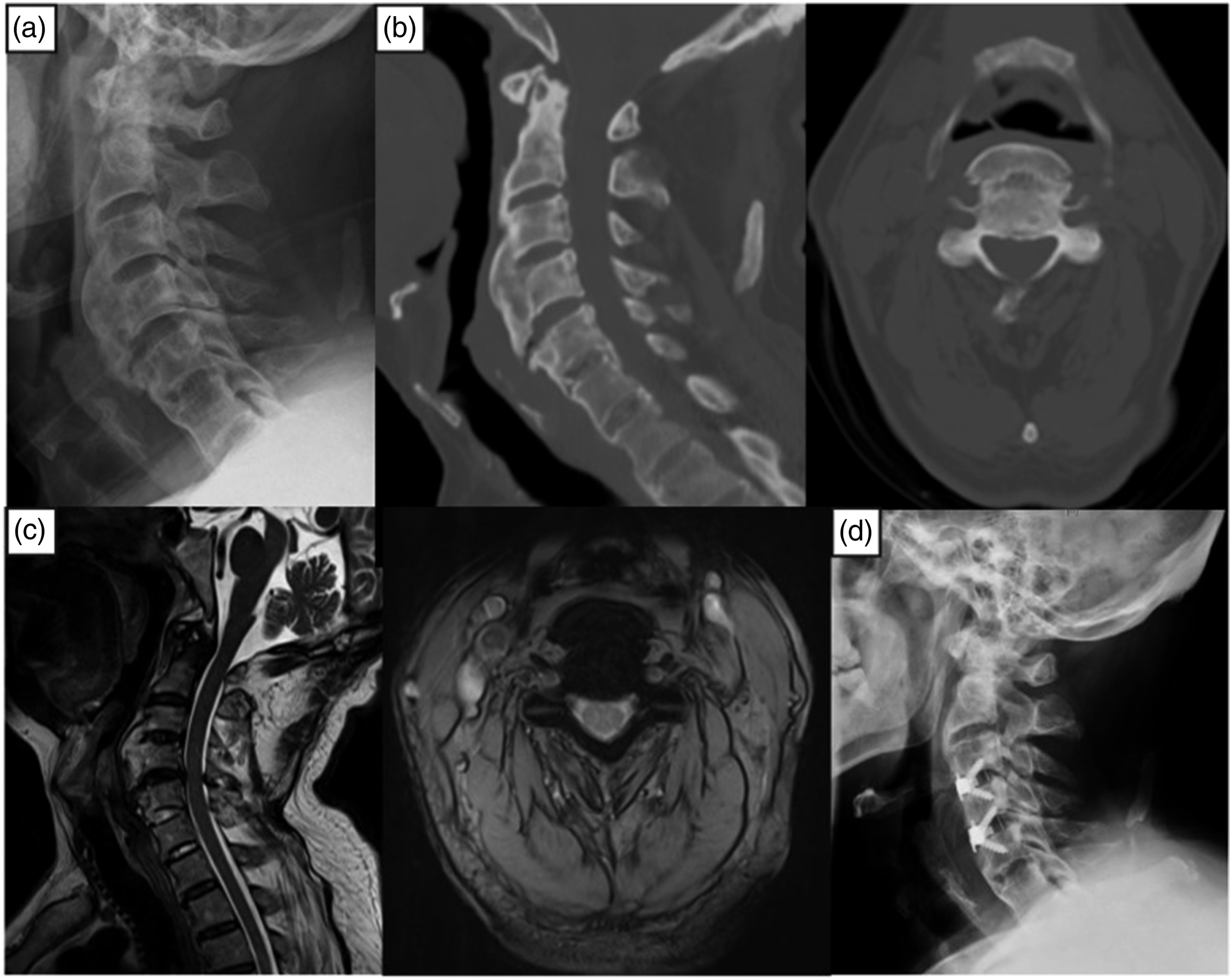
Case #3 Male, 72 years old, dysphagia with choking on water and bilateral shoulder pain for 2 years, gait instability for 1 month (myelopathy). Pre-op FOSS score: 4. Anterior osteophytes at C2-5 (DISH). Surgery: C3-5 osteophyte resection, C3-5 disc resection, and Zero-P cage fusion. Post-op FOSS score: 0. Recovery rate: 100%. (a) Pre-operative X-ray. (b) Pre Pre-operative CT scan. (c) Pre-operative MRI. (d) Post-operative X-ray.
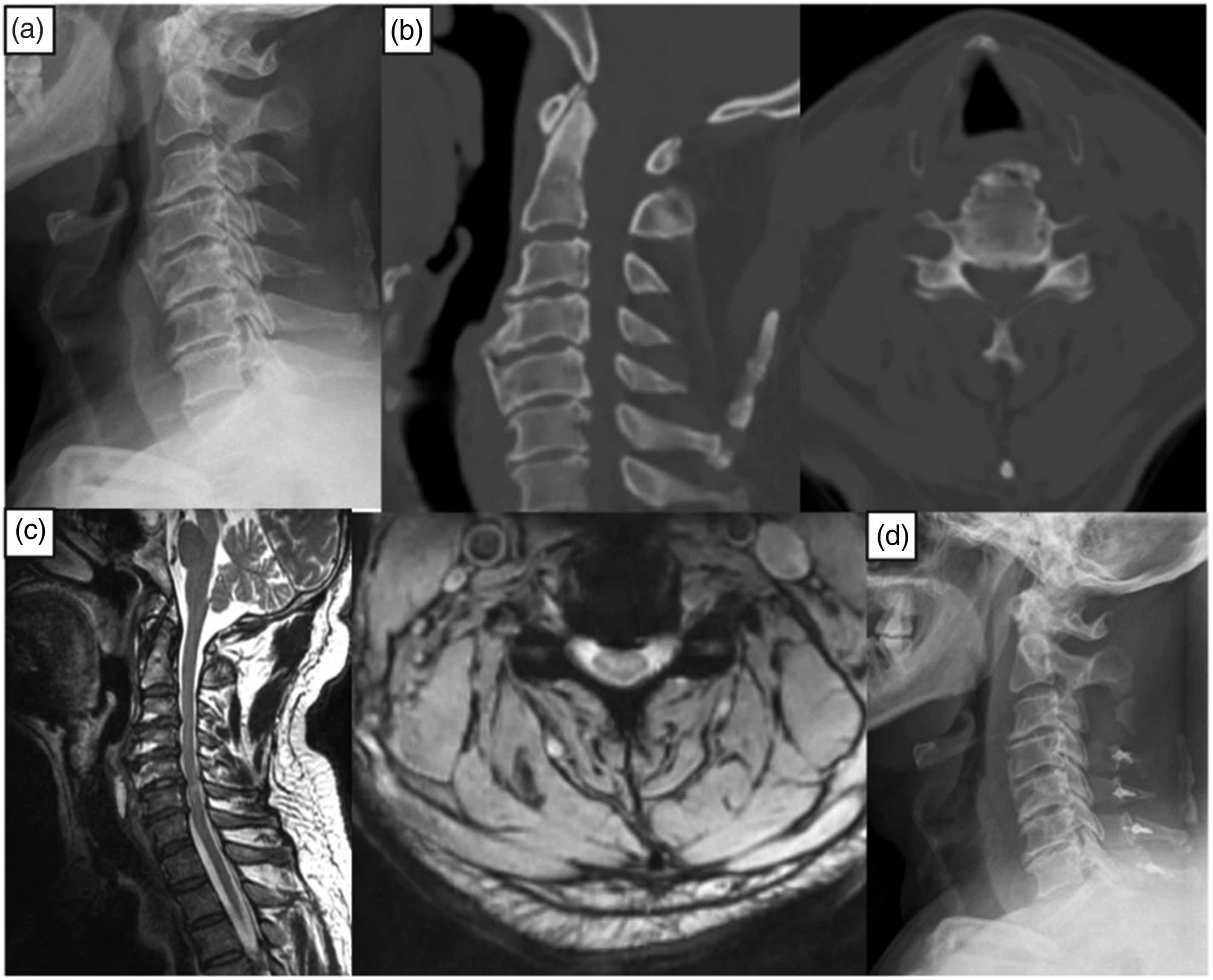
Case #4 Male, 62 years old, intermittent dysphagia with neck and shoulder pain and bilateral upper extremity weakness for 10 years (myelopathy). Pre-op FOSS score: 2. Anterior osteophytes at C3-5. Surgery: C3 laminectomy, C4-7 laminoplasty, C4-5 anterior osteophyte resection. Post-op FOSS score: 0. Recovery rate: 100%. (a) Pre-operative X-ray. (b) Pre Pre-operative CT scan. (c) Pre-operative MRI. (d) Post-operative X-ray.
Discussion
Cervical spondylosis with dysphagia secondary to anterior osteophytes is an uncommon but clinically meaningful condition caused by mechanical esophageal compression from bony overgrowths. 10 Surgical intervention is often required if symptoms are severe or refractory to conservative treatment, with anterior decompression being the preferred approach in most cases. Despite technical advances, the etiology, optimal surgical indications, and long-term outcomes remain poorly defined because of the rarity of the condition and limited systematic evidence. 11
In our study, anterior osteophytes were primarily located at C4/5and C5/6 segments known for their high mobility and susceptibility to degeneration. 12 These osteophytes may form due to chronic disc degeneration and anterior longitudinal ligament ossification, which contribute to esophageal compression through direct mechanical impingement, local inflammation, and adjacent muscle spasms. Notably, osteophytes were mostly situated below the epiglottis, a location that may exacerbate swallowing dysfunction due to proximity to critical pharyngeal structures. Age-related decreases in cervical flexibility may further compromise swallowing mechanics. 13
While half of the patients met diagnostic criteria for diffuse idiopathic skeletal hyperostosis (DISH), dysphagia was not exclusive to those with DISH, highlighting the broader relevance of osteophyte burden in degenerative conditions. 14 Coexisting cervical myelopathy or radiculopathy was observed in one-third (33.3%) of patients, emphasizing the need to evaluate spinal cord and nerve root compression if planning surgery. Conditions, such as DISH, characterized by calcifications and ossifications along multiple vertebral bodies without significant disc disease or bony ankylosis of facet joints, predominantly affect C5/6 and contribute to dysphagia through mechanisms including inflammation and impaired laryngeal function. 15 Other conditions, such as ankylosing spondylitis and cervical osteomyelitis with pre-vertebral abscesses, can lead to dysphagia. 16 Recognizing these distinctions allows for tailored therapeutic strategies aimed at optimizing clinical outcomes and alleviating symptoms early in the disease progression.
Differential diagnosis remains critical. Disorders such as cricopharyngeal dysfunction, pharyngeal neuromuscular disease, and vocal cord paralysis must be ruled out using tools like barium swallow, FEES, and laryngoscopy. In our series, multimodal imaging, especially dynamic studies, confirmed the mechanical etiology of dysphagia and informed surgical planning. The clinical presentation extended beyond dysphagia to include neck pain, stiffness, hoarseness, and in some cases, neurological symptoms—reflecting the multifactorial impact of anterior osteophytes. Dysphagia and hoarseness likely result from esophageal and recurrent laryngeal nerve compression, emphasizing the need for early symptom recognition.17,18
Recent studies on dysphagia have primarily focused on postoperative complications following cervical spine surgery, rather than on dysphagia caused specifically by anterior osteophytes. For example, Mazmudar et al found that preoperative dysphagia significantly prolonged postoperative symptoms in patients who underwent ACDF, whereas Ohba et al identified preoperative dysfunction as a predictor of postoperative dysphagia in patients with cervical spondylotic myelopathy.19,20 These studies, which are valuable for understanding postoperative outcomes, did not address the distinct pathophysiology of osteophyte-induced dysphagia. Contrastingly, our study investigated this specific etiology and its management and showed that osteophyte resection is an effective intervention for relieving dysphagia. We observed a postoperative dysphagia rate of 17%, which is lower than the 40% rate reported in studies on ACDF. This difference may reflect the targeted approach of osteophyte resection in our study, which contrasts with the broader surgical populations examined in previous studies. Our findings align with those of Egerter et al, 21 reinforcing the need to differentiate osteophyte-related dysphagia from general postoperative dysphagia. Future research should aim to compare long-term outcomes across different surgical techniques.
Most patients in our study underwent anterior osteophyte resection combined with discectomy and interbody fusion, with either traditional or zero-profile cages. This approach not only decompresses the esophagus but also restores cervical alignment and stabilizes the spine, reducing the risk of recurrent osteophyte formation and adjacent segment degeneration. Zero-profile implants may offer additional benefits by minimizing hardware prominence, which has been associated with postoperative dysphagia. In more complex or multilevel cases, a posterior approach was selectively used to achieve adequate decompression. 22 Real-time intraoperative navigation and imaging facilitated precise osteophyte removal while minimizing the risk of injury to adjacent structures. 23 Thorough preoperative evaluation, including imaging and swallowing assessments, remains essential for identifying mechanical causes of dysphagia and optimizing surgical planning. Despite the benefits observed, the ideal surgical technique—including implant choice and fusion strategy—remains an area requiring further comparative research.
Two patients in our cohort developed dysphagia following prior anterior cervical surgery, suggesting that postoperative osteophyte formation may occur as a delayed complication. This can result from abnormal mechanical stress at adjacent levels, triggering new bone formation as a compensatory response to altered spinal dynamics. 24 Postoperative scar formation, fibrosis, and changes in cervical alignment may further contribute by tethering soft tissues or altering the esophageal trajectory, ultimately impairing swallowing. These cases underscore the multifactorial nature of dysphagia in the postoperative setting and highlight the importance of long-term surveillance. 25 Early recognition of these mechanisms can inform preventive strategies, such as motion preservation at adjacent levels or careful attention to spinal alignment during index surgery, to mitigate the risk of delayed complications.
However, our retrospective study has several limitations that warrant acknowledgment. The relatively small sample size limits the generalizability of our findings and the retrospective nature of the study precludes causal inferences. Additionally, the lack of a control group hinders the comparison of surgical outcomes between conservative management approaches. Future prospective studies with larger cohorts and longer follow-up periods are required to validate our findings and address these limitations.
Cervical esophageal spondylosis presents complex diagnostic and therapeutic challenges that necessitate a multidisciplinary approach. Our retrospective study highlighted the efficacy and safety of anterior decompressive cervical surgery in the management of this condition. However, further research is needed to elucidate the pathogenesis, refine surgical techniques, and optimize treatment outcomes. By integrating insights from our study with the existing literature, we can advance our understanding of esophageal cervical spondylosis and improve patient care.
Conclusion
Anterior osteophyte resection, combined with disc resection and cage fusion, appears effective in treating cervical spondylosis with dysphagia caused by anterior osteophytes, but it requires careful consideration of its complex etiology and diverse clinical presentations.
Supplemental Material
Supplemental Material - Treatment of Cervical Spondylosis With Dysphagia Caused by Anterior Osteophytes: A Retrospective Case Series Study
Supplemental Material for Treatment of Cervical Spondylosis With Dysphagia Caused by Anterior Osteophytes: A Retrospective Case Series Study by Rui Chen, Ben Wang, Yanbin Zhao, Yinze Diao, Xin Chen, Shengfa Pan, Fengshan Zhang, Yu Sun, Feifei Zhou, Zhongjun Liu in Global Spine Journal.
Footnotes
Author Contributions
Rui Chen and Ben Wang carried out the studies, participated in collecting data, and drafted the manuscript. Rui Chen and Ben Wang performed the statistical analysis and participated in its design. Rui Chen, Ben Wang, Feifei Zhou and Zhongjun Liu participated in acquisition, analysis, or interpretation of data and draft the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81874023) and Peking University Third Hospital (301-2404-01-03).
Ethical Statement
Data Availability Statement
All data generated or analysed during this study are included in this published article. Research data supporting this publication are available from the Peking University Third Hospital, but restrictions apply to the availability of these data, which were used under license for the current study, and so are not publicly.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
