Abstract
Study Design
Retrospective Cohort Study.
Objectives
The current recommended treatment for Giant Cell Tumour (GCT) of the spine is en bloc excision. Denosumab is a monoclonal antibody reducing osteoclast activity that shows promising results when used as a neo – adjuvant treatment. However, the current literature remains limited. The purpose of this study was to assess the effect of denosumab on tumour characteristics and symptom relief in the acute phase of treatment of spinal GCT.
Methods
We performed a retrospective review of 16 patients treated with denosumab as neo-adjuvant and stand - alone treatment. MRI and PET tumour characteristics were taken before and after treatment and patients were interviewed for subjective pain responses.
Results
Following treatment, all patients showed improvement of pain, of which 68.7% of patients were pain free with 43.75% noting improvement within 48 hours. Mean relative volumetric reduction in tumour volume was 37.3% (P < .001). Eight patients showed high grade of Bilsky classification (Epidural spinal cord compression scale - ESCC) with seven of them showing significant improvement to low grade of ESCC (P = .016). Median baseline PET Standardised Uptake Value (SUV)max was 14.57 and post treatment was 4.8 (P < .001).
Conclusions
This study provides necessary insight to the limited literature on the use of denosumab for spinal GCT in the acute phase. The clinical and radiographic responses observed demonstrate the critical role that neo-adjuvant denosumab has by reducing the tumour burden around critical adjacent neurovascular structures before eventual resection, significant pain improvement even with presence of fractured vertebra.
Introduction
Giant Cell Tumours (GCTs) are osteolytic lesions that occur most frequently in the long bones. In the axial spine they most commonly involve the sacrum, and in the mobile spine the vertebral bodies, but can also involve the posterior elements, discs and paraspinal soft tissue. 1 They are locally aggressive and can be large at presentation. Typical presentation is with pain which often precedes diagnosis. Up to 72% of patients also experience neurological symptoms such as radiculopathy or symptoms of cord or cauda equina compression. 2 They commonly occur in patients aged 20 - 40 years, with a female–to–male ratio of 1:1. 3
Despite being classified as a benign lesion, GCT can display malignant behaviour. Typical metastasis is via haematogenous spread to the lungs and occurs in up to 5% of cases. 4 A small number may undergo sarcomatous change, most often to osteosarcoma, which can occur as a primary malignant GCT or more commonly as secondary malignancy after radiotherapy. 2
Treatment options include therapeutic embolisation, radiotherapy alone, intralesional surgery +/− radiotherapy and en bloc surgical resection. In cases involving the axial skeleton, en bloc resection is considered the Enneking appropriate treatment.5-7 Surgical resection margins remain the best predictor of local recurrence (LR). Either therapeutic embolization or radiotherapy alone are associated with unacceptably high LR rates.8,9 Similarly, although intralesional resection may spare neural, bony and visceral structures, it does so at the cost of high rates of local recurrence, with rates up to 46% reported. 8 En bloc resection gives the lowest chance of LR and improves survival, but risks increased inter – operative and functional morbidity and may be unachievable due to anatomical constraints.5,6
With the goal of facilitating en bloc resection, interest has grown in the role of denosumab in the neo–adjuvant setting. Denosumab is a human monoclonal antibody that reduces osteoclast activity by binding receptor of activator nuclear factor kappa – B ligand (RANKL). It has previously been shown to be effective in suppressing bone destruction in patients with multiple myeloma and bony metastases in solid tumours such as breast and prostate. 10 In 2010, Thomas et al conducted an open label phase 2 study investigating the effect of denosumab on tumour cell survival and growth on 37 patients with recurrent or unresectable bone GCTs. Eighty–six percent of patients had a tumour response defined as either elimination of at least 90% of giant cells, or no radiological progression of the target lesion up to week 25. 11 In a subsequent study of the histologic analyses of the samples taken, Branstetter et al (2012), concluded that in addition to significantly reducing or eliminating RANK positive tumour giant cells, denosumab, also reduced the content of tumour stromal cells and replaced them with non-proliferative, differentiated, densely woven new bone. 10 Multiple studies to date have demonstrated its efficacy in controlling surgically unresectable GCTs, and approval for its use was granted by the FDA in 2013.2,12 In 2015, Goldschlager et al reported on a series of 5 patients treated pre–operatively with denosumab for GCT of the spine, and demonstrated a 100% radiological response to treatment, although one patient failed to demonstrate a histological response. 13 In contrast to en bloc axial GCT resection there has been growing concern regarding poor LR rates in patients undergoing intralesional resection of extremity GCTs with neo-adjuvant denosumab (up to 60% LR) vs without (as low as 16%). 14
Surprisingly, there remains a paucity of evidence on the effect of this therapy on spinal GCTs. The goal of this study was, therefore, to report the early effect of denosumab on clinical and radiological outcomes in larger series of patients presenting with spinal GCT.
Methods
We performed a retrospective study on 16 patients treated for GCT of the spine with either neo-adjuvant denosumab followed by en bloc resection (D-resection, 11 patients) or denosumab alone in non-resectable tumours (D-only, 5 patients). Treatment regimen with denosumab was uniform for all patients and comprised weekly administration on days 1, 8 and 15, and then monthly thereafter. 15 Mean duration of treatment was 8.2 months. No adverse side effects of denosumab treatment were observed.
Patient Population
Inclusion criteria where patients were 18 years or older at the time of treatment, had a primary diagnosis of biopsy – confirmed GCT of the mobile spine or sacrum, had received denosumab as a primary or neo–adjuvant treatment, and had a minimum follow – up of 6 months after initiation of denosumab treatment. Cases of recurrent GCT were excluded from the study.
Patient and Clinical Variables Collected
Patient demographic data was collected such as age, sex, and ethnicity. Clinical presentation was collected including pain severity and neurological status, duration of symptoms and previous treatment. The patients were contacted retrospectively, and a record was taken of the reported time taken for significant pain improvement (<48 h, 1 week, 6 weeks, >6 weeks). To minimise error, the pain severity was graded as mild (Visual Analogue Scale - VAS 1-3), moderate (VAS 4-7) or severe (VAS 8-10). 16
Radiological Outcomes Collected
Tumour measurements based on imaging were collected at 3-month time intervals following denosumab initiation and continued until cessation of treatment. Volumetric analysis was performed based on these measurements. The Bilsky classification (Epidural spinal cord compression scale - ESCC) for epidural disease 17 as well as the Weinstein-Boriani-Biagini (WBB) layers and sectors 18 and also Enneking staging 19 were recorded. The Boriani classification for denosumab calcification response 7 was also noted. PET SUV (Positron emission tomography Standardised Uptake Value) capitation was measured to assess treatment response meaning a decrease of 25% or more in the sum of maximum SUV compared with screening according to modified European Organization for Research and Treatment of Cancer (EORTC) PET criteria. 20
Ethical Consideration
Approval was obtained from the institutional review board at Oxford University Hospitals NHS Foundation Trust, reference number: OUH9618.
Results
Patient Demographics and Clinical Variables
Patient Demographics and Clinical Variables.
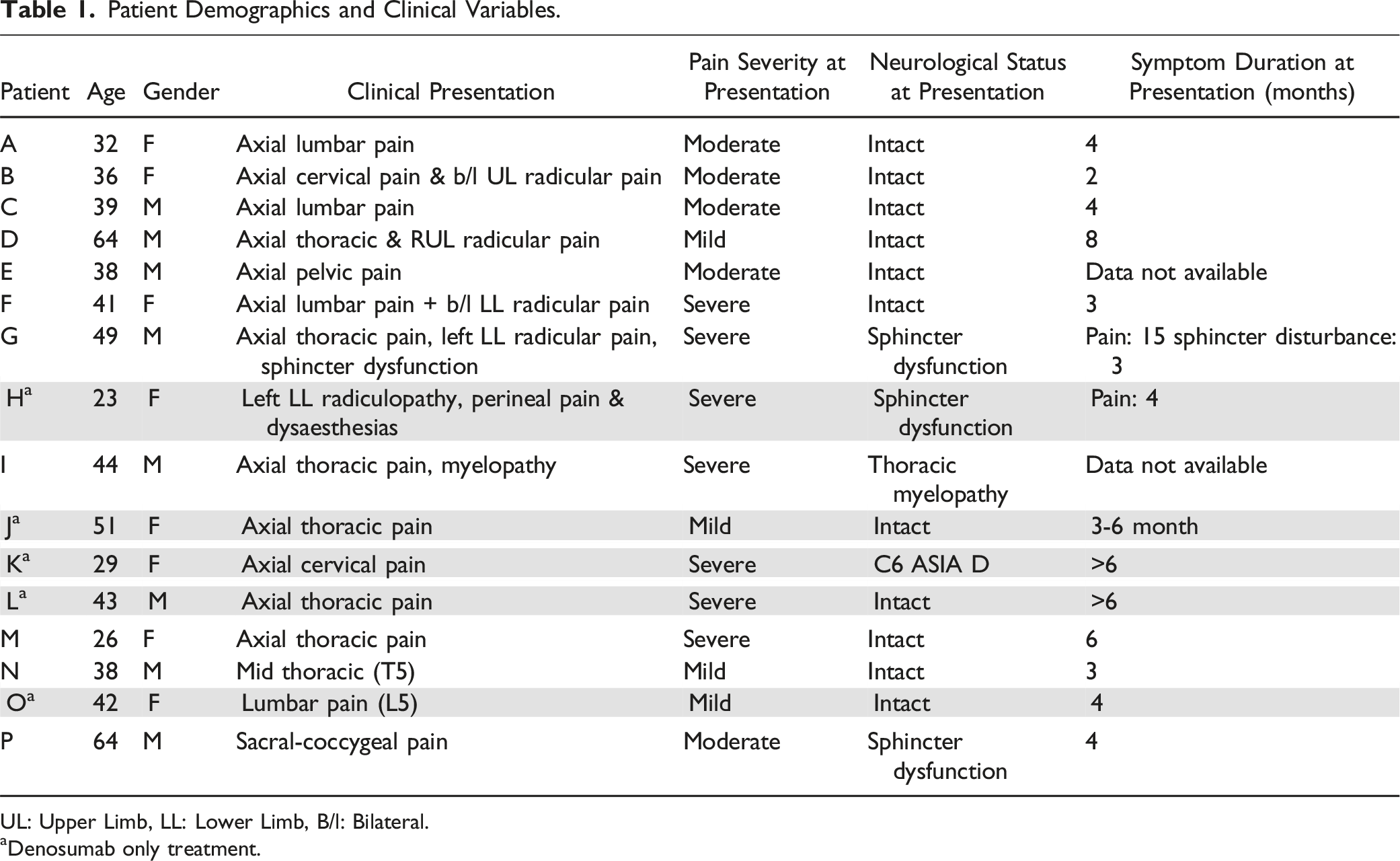
UL: Upper Limb, LL: Lower Limb, B/l: Bilateral.
aDenosumab only treatment.
Pain Outcomes
Pain Outcomes.
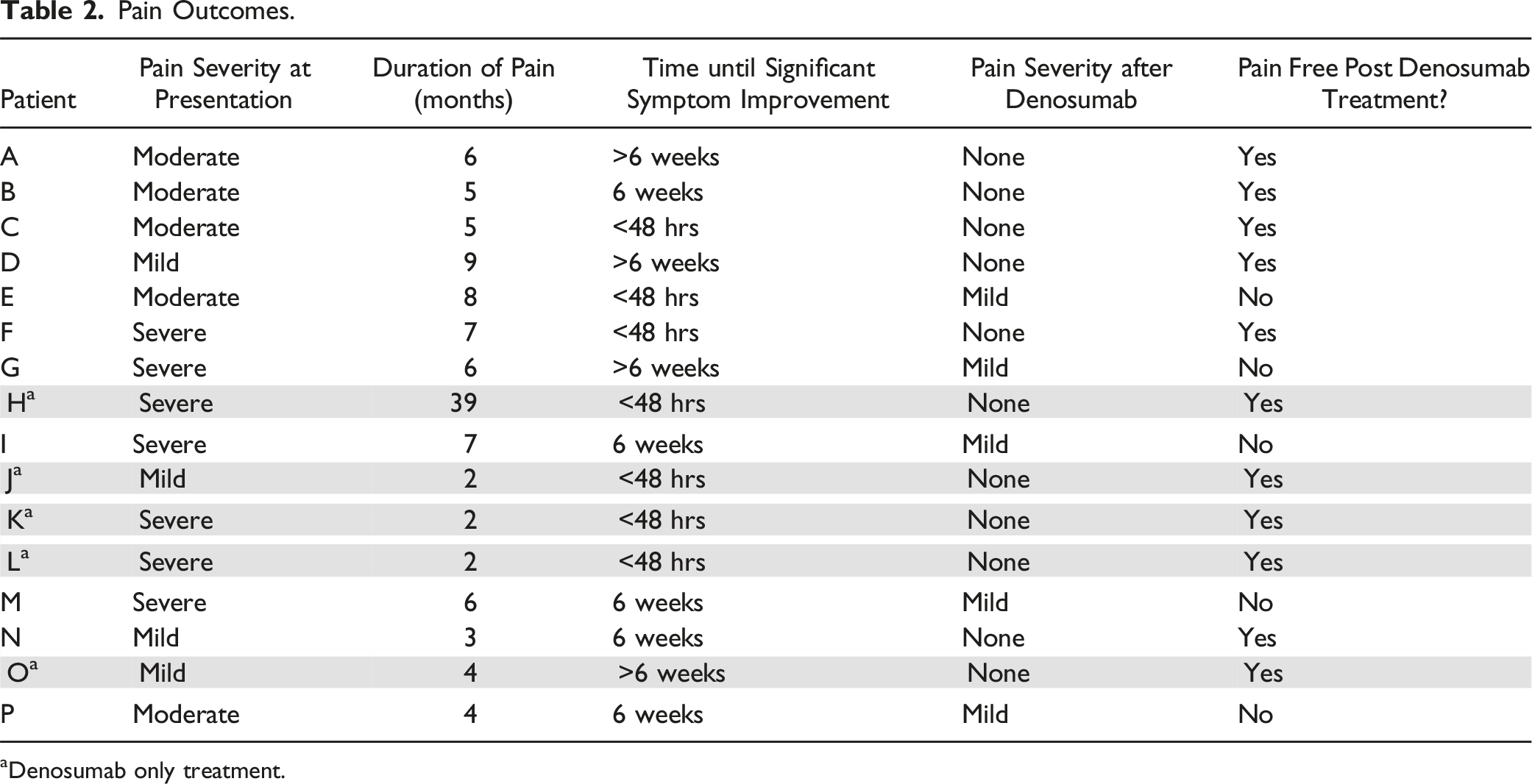
aDenosumab only treatment.
Radiographic Outcomes
Radiographic Outcomes: Tumour Volume.
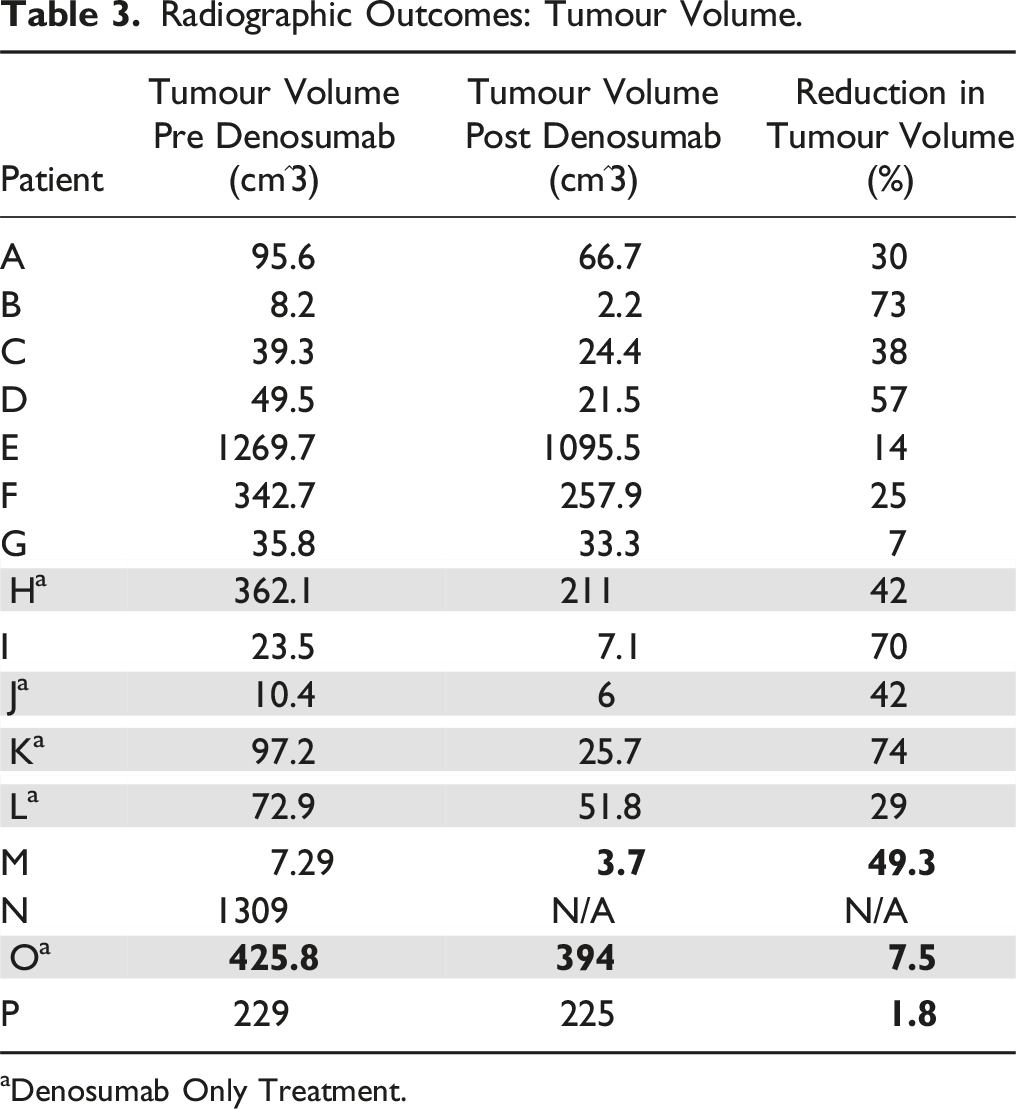
aDenosumab Only Treatment.
Radiographic Outcomes: Epidural Disease, WBB Sectors / Layers, PET Standardised Uptake Values, Calcification Response.
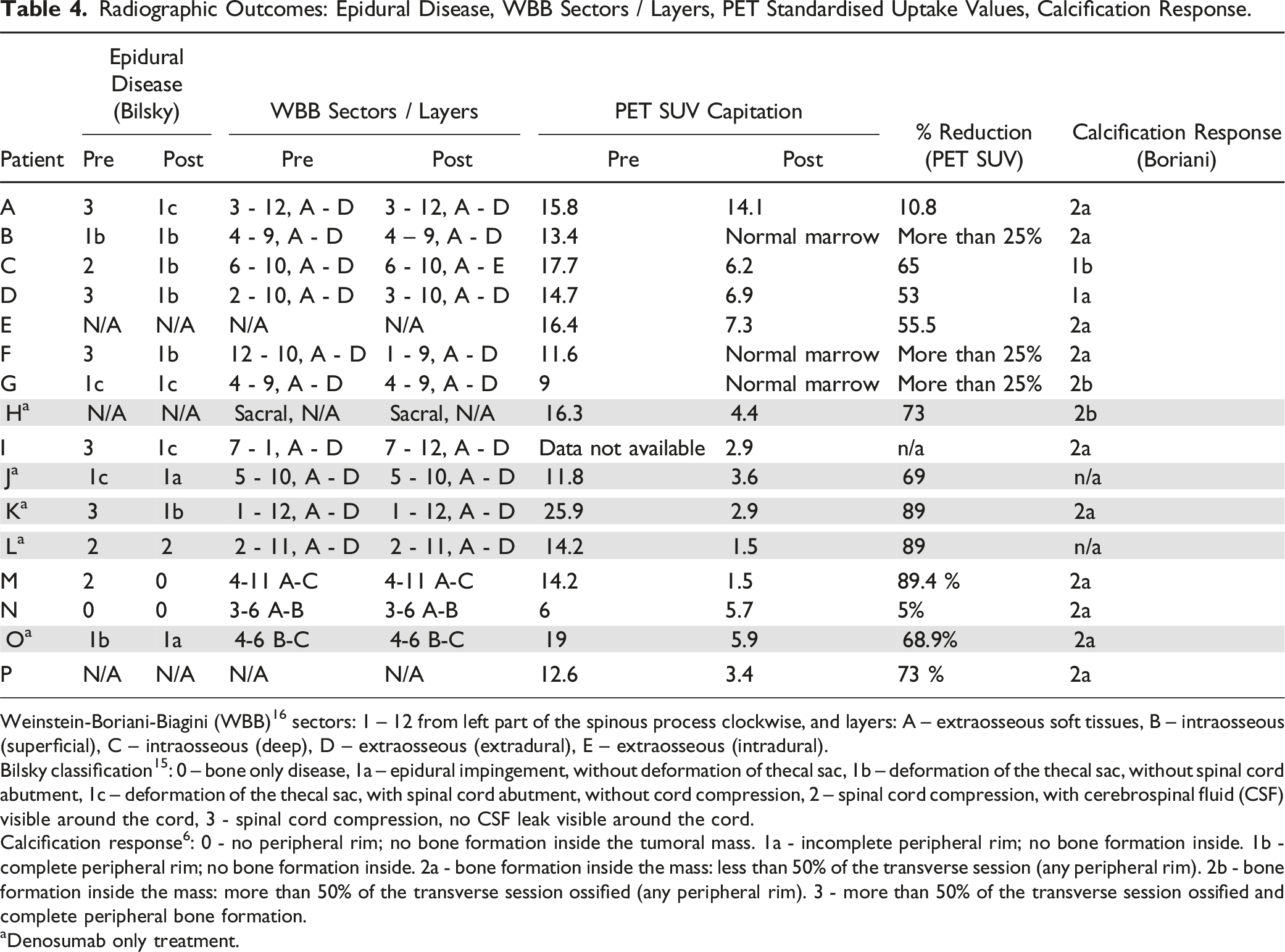
Weinstein-Boriani-Biagini (WBB) 16 sectors: 1 – 12 from left part of the spinous process clockwise, and layers: A – extraosseous soft tissues, B – intraosseous (superficial), C – intraosseous (deep), D – extraosseous (extradural), E – extraosseous (intradural).
Bilsky classification 15 : 0 – bone only disease, 1a – epidural impingement, without deformation of thecal sac, 1b – deformation of the thecal sac, without spinal cord abutment, 1c – deformation of the thecal sac, with spinal cord abutment, without cord compression, 2 – spinal cord compression, with cerebrospinal fluid (CSF) visible around the cord, 3 - spinal cord compression, no CSF leak visible around the cord.
Calcification response 6 : 0 - no peripheral rim; no bone formation inside the tumoral mass. 1a - incomplete peripheral rim; no bone formation inside. 1b - complete peripheral rim; no bone formation inside. 2a - bone formation inside the mass: less than 50% of the transverse session (any peripheral rim). 2b - bone formation inside the mass: more than 50% of the transverse session ossified (any peripheral rim). 3 - more than 50% of the transverse session ossified and complete peripheral bone formation.
aDenosumab only treatment.
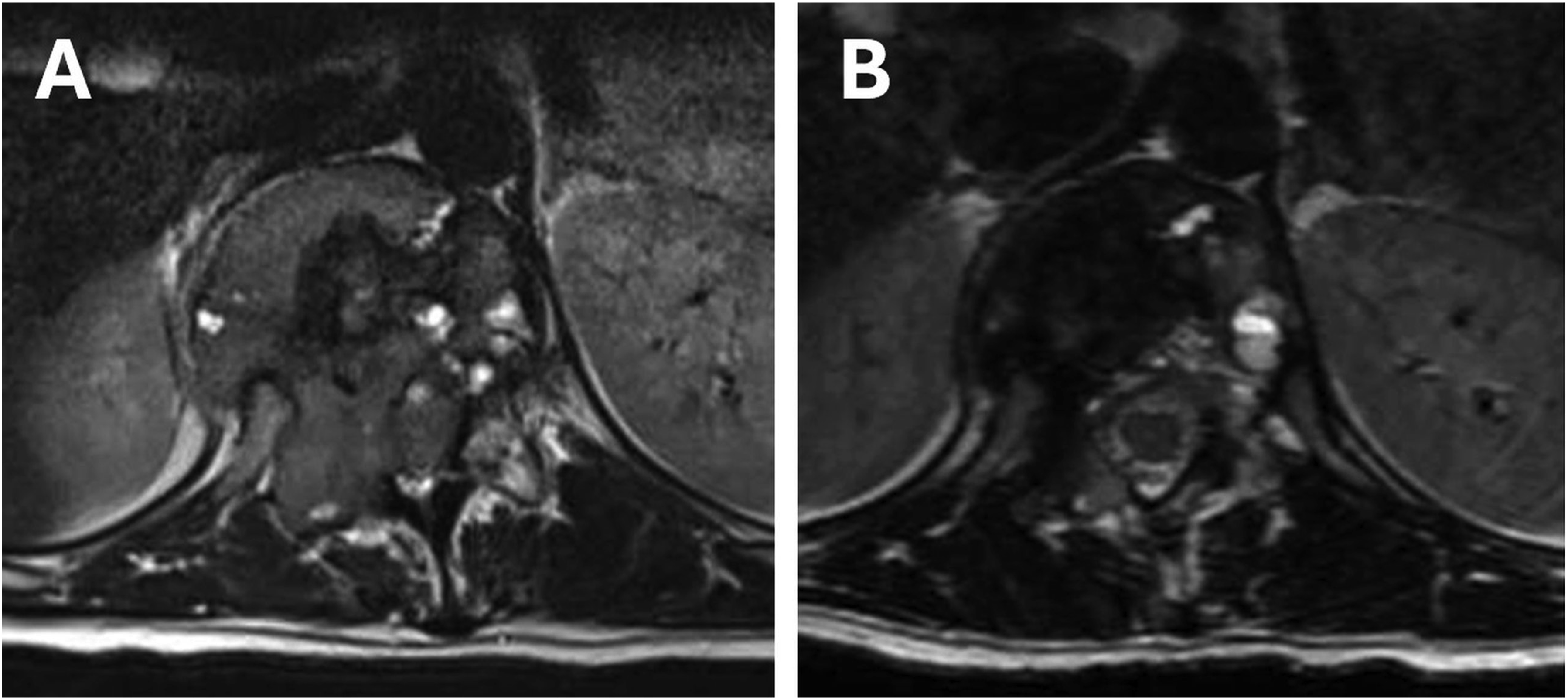
T12 Giant Cell Tumour. A: pre – treatment b: post – treatment (1 cycle), demonstrating sclerosis and reduction in epidural disease.
Fractures of the Affected Vertebrae and Local Recurrence After the Treatment.
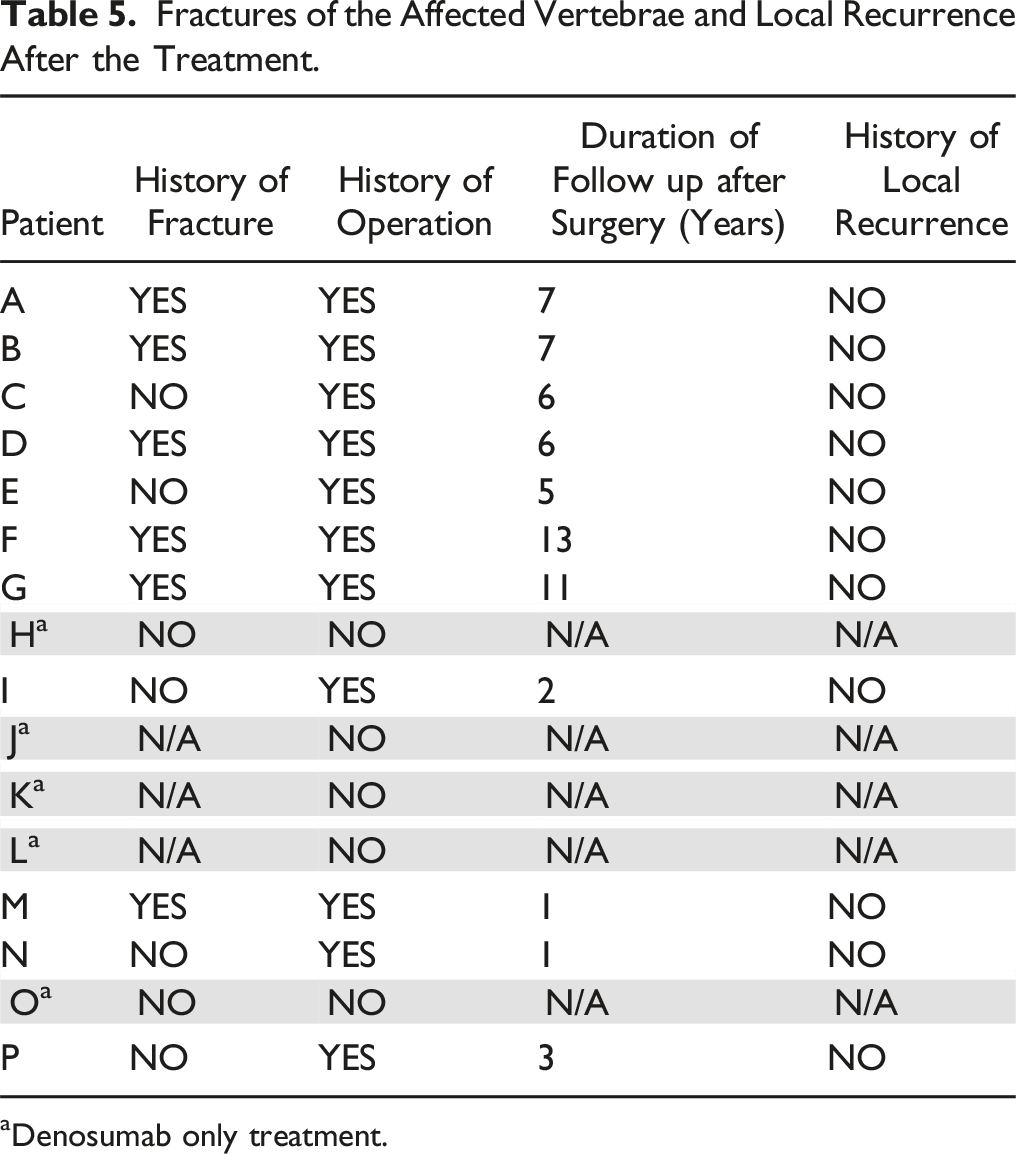
aDenosumab only treatment.
Discussion
This study provides a crucial portrait of spinal GCT treated with denosumab in terms of clinical and radiological outcomes. Mean percentage reduction in tumour volume was 37.3% (P < .001). A correlation was demonstrated between initial tumour volume and percentage reduction in tumour size, with larger tumours undergoing a smaller volumetric reduction. This is perhaps unsurprising given the uniform dosing regimen regardless of initial tumour size. However, a previous study which characterised population pharmacokinetics of denosumab in patients with advanced solid tumours and bone metastases noted that following 120 mg dosing every 4 weeks, as was the case in our study, RANKL occupancy exceeded 97% during the entire dosing interval and concluded dosage adjustments based on body weight, age, race and tumour type is not necessary in these patients. 15 Nonetheless denosumab clearly effected a significant reduction in tumour volume in these cases with no adverse events.
PET standardised uptake values confirmed efficacy of denosumab treatment in reducing metabolic activity in 27% (3/16) cases to normal marrow values. Median baseline SUVmax was 14.57, median SUVmax post treatment was 4.8 (P < .001). The treatment response according to EORTC PET criteria 20 (decrease in SUVmax > 25%) was achieved in 13/16 patients with available data (81.3%). This result is commensurate with the available published literature and further confirms that reduced PET avidity appears to be an early sign of response to denosumab treatment and a useful tool in its assessment.21,22
Our results clearly demonstrate that denosumab effects a significant improvement in pain. Pre–treatment, all but one patient was experiencing moderate to severe pain which had been present on average for 7 months. Following treatment with denosumab, 68.7% of patients reported being pain free. Perhaps most striking, however, is the rapidity with which denosumab took effect in selected cases. In 7/16 (43.75%) of cases, improvement in symptoms was noted within 48 hours. Interestingly, this effect is not uniform, and the remainder of patients took 6 weeks or more to effect improvement. There are several studies in GCT of mixed location (long bones, spine, pelvis) which report an improvement in pain symptoms following treatment and to this end corroborate the findings of our study. The study by Traub et al (2016) reported significant pain relief in all patients after 1 month and complete pain relief in 80% of patients at 6 months. 23 A prospective phase II study of the effects of denosumab treatment on pain for GCT of the bone across all sites reported denosumab to be effective in reducing pain in both resectable and unresectable GCT with rapid and clinically relevant improvement in symptoms in both groups. 24 However, data on symptoms relief in spinal GCTs is very limited. Al Farii et al found in their systematic review from 2023 that neurologic status remained stable or improved in all GCT cases with epidural compression treated with neo-adjuvant denosumab, irrespective of the degree of presenting neurologic deficit. However, results are based on only 22 patients from 12 studies. 25 Only two studies reported the effect of denosumab on pain. Yang et al 26 showed a significant decrease of pain in 6 patients with sacral GCTs. 26 Chen et al. described significant improvement in pain in 80% of their 11 patients treated with neo-adjuvant denosumab, however without mentioning the timeline of pain relief. 27 Therefore, our study provides invaluable benchmark numbers in terms of pain relief.
Improvement in ESCC grade of epidural compression was observed in 7 out of 8 patients (87.5%) who had high ESCC grade and changed to low grade after denosumab (Figure 1). In the rest of the cases, the ESCC grade remained the same. These findings appear to indicate denosumab may protect against spinal cord compression and indeed in some of our cases expediently reverse them. Other studies found similar results that denosumab alone can lead to neurological recovery.7,28 Denosumab better defines the soft tissue margin of the tumour by creating a calcific rim. Our results confirm that treatment either halted disease progression or effected an improvement in all cases. This element almost certainly contributes to the absence of local recurrence in this series. Denosumab’s role in decreasing the local recurrence of GCT has been studied on GCT in extremities. A retrospective study revealed a local recurrence of 60% (15/25) among the group treated with denosumab and curettage (intralesional) where the patients who were treated with curettage had a recurrence rate of 16% (36 of 222). 29 Another systematic review study reported a local recurrence of 50-100% among the group treated with neoadjuvant denosumab and surgery while the patients who were treated with curettage alone had a lower risk of up to 50%. 30 On the other hand, our results confirms the Boriani et al.’s data showing that patients treated with neoadjuvant denosumab and en bloc resection (extralesional) have zero recurrences rate. 7
WBB layers/sectors improved in 3/16 cases and these improvements correlated with the most profound changes in ESCC grade and each had large percentage volumetric reduction. Similarly, this less profound effect on the WBB layers is likely representative of the larger overall tumour volume represented by this system. The cases in which the WBB layers/sectors remained the same nonetheless showed a good calcification response (2a or 2b) demonstrating that denosumab did still have a beneficial effect in these cases.
Six patients had history of vertebral fracture; three of them were suffering from severe pain, 2 patients had a moderate pain, and one patient had mild pain initially. After denosumab treatment, all these patients felt significant improvement - four of them were pain free, other two had only mild pain. This may be partially explained by the early calcification of the lesion and subsequent fracture healing but it remains surprising that such rapid pain relief is achieved, even in this cohort, this may warrant further investigation.23,31
Calcification response in our patient cohort demonstrated good response to denosumab therapy, with 67% patients demonstrating subsequent intra lesional bone formation. One question that our study does not answer is how long denosumab treatment should last as stand-alone therapy, as the majority (11/16 of patients) 68.8% underwent en bloc resection following initial treatment. In a preliminary report of 12 cases and literature review in 2020, Boriani et al noted a radiological withdrawal effect in one case with disappearance of the sclerotic rim and core ossification following 15 months of treatment when treatment was stopped. 7 Previous studies have also lacked long term follow up, or reported on patients still undergoing denosumab treatment11,32 and therefore the results from these studies must be interpreted cautiously. 33 In a prospective non randomised study of neo-adjuvant denosumab treatment for GCT in multiple sites including long bones, Traub et al also noted that the change in the tumour characteristics, from soft tumour to fibro-osseous matrix following denosumab made accurately finding the junction between treated tumour and normal bone difficult, and furthermore speculated that tumour cells may become trapped within the thickened cortical and subchondral bone produced by denosumab and contribute to local recurrence. 23 Clearly this possibility needs to be carefully considered when planning en bloc resection.
Limitations
While this study provides useful insight, its findings should be interpreted in the context of the study design. This single centre study from a supraregional referral hospital provides important data on a rare disease with a complex subspecialized treatment. This explains the small sample size with limited statistical significance of the results as well as the lack of control group. However, such descriptive results are necessary to help guide future efforts in generating multi-centre trials for an adequately powered study. Future multicentre studies should explore patient reported outcomes such as subjective pain with validated tools. Longer follow-up could aid in gaining insight in disease-free survival.
Conclusion
This study highlights key components regarding the investigation and treatment of solitary lesions of the spine confirming the vital aspects of local and systemic staging in prevention of inadvertent tumour breach and optimising oncological outcomes. It demonstrates that patients with aggressive disease, severe pain, mild neurological dysfunction and high grade epidural spinal cord compression can be safely and successfully managed without early intralesional surgery. Even in the presence of fracture, denosumab delivers rapid and large responses in pain relief and excellent reduction of SUVmax and epidural compression whilst protecting against neurological deterioration. The subsequent absence of LR in our surgical series thus managed confirms the clinical rationale.
Footnotes
Author Contribution - CrediT Author Statement
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
