Abstract
Study Design
Retrospective Cohort Study.
Introduction
The 11-item modified Frailty index (mFI-11) by the ACS-NSQIP database was used to predict which patients are high risk for complications and inpatient mortality. ACS-NSQIP now has switched to the 5-item MFI. However, there are no studies on how these frailty indices fare against each other and their prognostic value of functional independence in patients with spinal cord injury (SCI).
Objective
To compare the mFI-5 and mFI-11 in order to standardize frailty assessment in the SCI population.
Methods
Retrospective analysis of 272,174 patients with SCI from 2010 to 2020 from the Pennsylvania Trauma Systems Foundation (PTSF) registry. Multivariable logistic regression was used to determine the predictive value of mFI for functional independence as determined by locomotion and transfer mobility.
Results
A total of 1907 patients were included with a mean age of 46.9 ± 15.1 years. The 3 most common MFI factors were hypertension (32.2%), diabetes mellitus (13.7%) and chronic obstructive pulmonary disease (8.5%). Multivariable logistic regression analyses using MFI-5 and MFI-11 showed that a higher frailty score in MFI-5 (OR 1.375, P < .001) and in MFI-11 (OR 1.366, P < .001) were each predictive of poor functional status at discharge. ROC curves for the MFI-5 (AUC = .818, P < .001) and MFI-11 (AUC = .819, P < .001) demonstrated excellent diagnostic accuracy.
Conclusion
The new MFI-5 is equivalent to its predecessor, the MFI-11, and predictive of functional outcomes in patients with SCI. MFI-5 can serve as the preferred frailty index at the point of care and in research contexts.
Introduction
Each year, there are nearly 17,000 new spinal cord injury (SCI) cases in the United States and as many as 450,000 people living with SCI today. 1 SCI is a devastating neurological event that can leave patients with life-long changes in their physical, psychological, and social well-being. Depending on the severity of SCI, a patient may face a decline or complete loss of independent functional status following injury, 2 which predisposes this patient population to lengthy hospital stay, a higher risk of in-hospital mortality and long course of rehabilitation if they survive the hospitalization.3,4 Given these patients’ acute presentation, it is challenging for physicians to give patients and families accurate guidance on potential morbidity and mortality of surgical care. Using the patient’s comorbidities to assess frailty and predict their ability to tolerate surgery and rehabilitation can be a valuable tool for decision-making.
In addition to SCI severity, hemodynamic instability, 5 and assessing other concurrent injuries, determining a patient’s frailty can help identify patients that are high risk for poor health outcomes, worsening disability despite optimal treatment, and inpatient mortality. 6 Presently, there is limited literature on the use of frailty measures in patients with SCI, however, this limited data has encouraging results regarding its prognostic value.7-10 Most of this literature uses the 11-item modified frailty index (mF-11), while one study used the mFI-5. 11 The five-factor modified frailty index (mFI-5) was developed once the eleven-factor modified frailty index (mFI-11) was no longer reported. MFI-5 includes hypertension requiring medication, diabetes mellitus, chronic obstructive pulmonary disease (COPD) or active pneumonia, partially dependent or fully dependent functional status at time of surgery, and congestive heart failure within 30 days prior to surgery. 12 In addition to these 5 items, the mFI-11 also includes myocardial infarction, previous percutaneous procedure or angina, peripheral vascular disease, impaired sensorium, transient ischemic event or cerebrovascular accident, and neurologic deficit after previous cerebrovascular accident.13,14
There is a paucity of literature comparing the 2 frailty indices as predictors of functional independence after suffering SCI8,11 Given that calculating the mFI-11 is no longer feasible, the present study aims to compare the mFI-5 to mFI-11 and demonstrate its ability to predict functional independence based on a statewide traumatic SCI registry in an effort to standardize frailty assessment in this patient population.
Methods
Patient Selection and Variables
The study protocol was approved by the University Institutional Review Board (#20D.1092). We conducted a retrospective analysis of 272,174 patients admitted to a hospital in Pennsylvania from January 2010 to December 2020 after sustaining a traumatic injury from the Pennsylvania Trauma Systems Foundation (PTSF) patient registry. The registry was queried using the ICD-9806 and 952 codes and ICD-10 S14, S24, and S34 codes and identified 3060 patients admitted with SCI. Patient age, gender, race, ethnicity, cause of SCI, insurance, GCS at the scene, Injury Severity Score (ISS), level of SCI, vital signs at the scene and on admission, patient comorbidities, surgery status, functional status at discharge (FSD) were provided for these patients. FSD is scored based on a scale that is comprised of locomotion, transfer mobility, feeding, expression, and social interaction. Only locomotion and transfer mobility are included in this analysis given the focus on SCI. Of the 3060 patients with SCI, 1153 had incomplete functional outcome data and were excluded from this analysis, resulting in 1907 patients studied (Figure 1). The severity of injuries on admission is provided using the injury severity scale that ranges from 0 (no injury) to 75 (unsurvivable injury).
15
Using the patient characteristics, the modified frailty indices mFI-11 and mFI-5 were calculated.
12
According to established methodology, frailty indices were risk stratified by 3 levels: no frailty (mFI = 0) mild frailty (mFI = 1), and moderate-severe frailty (mFI ≥ 2).
16
Patient selection.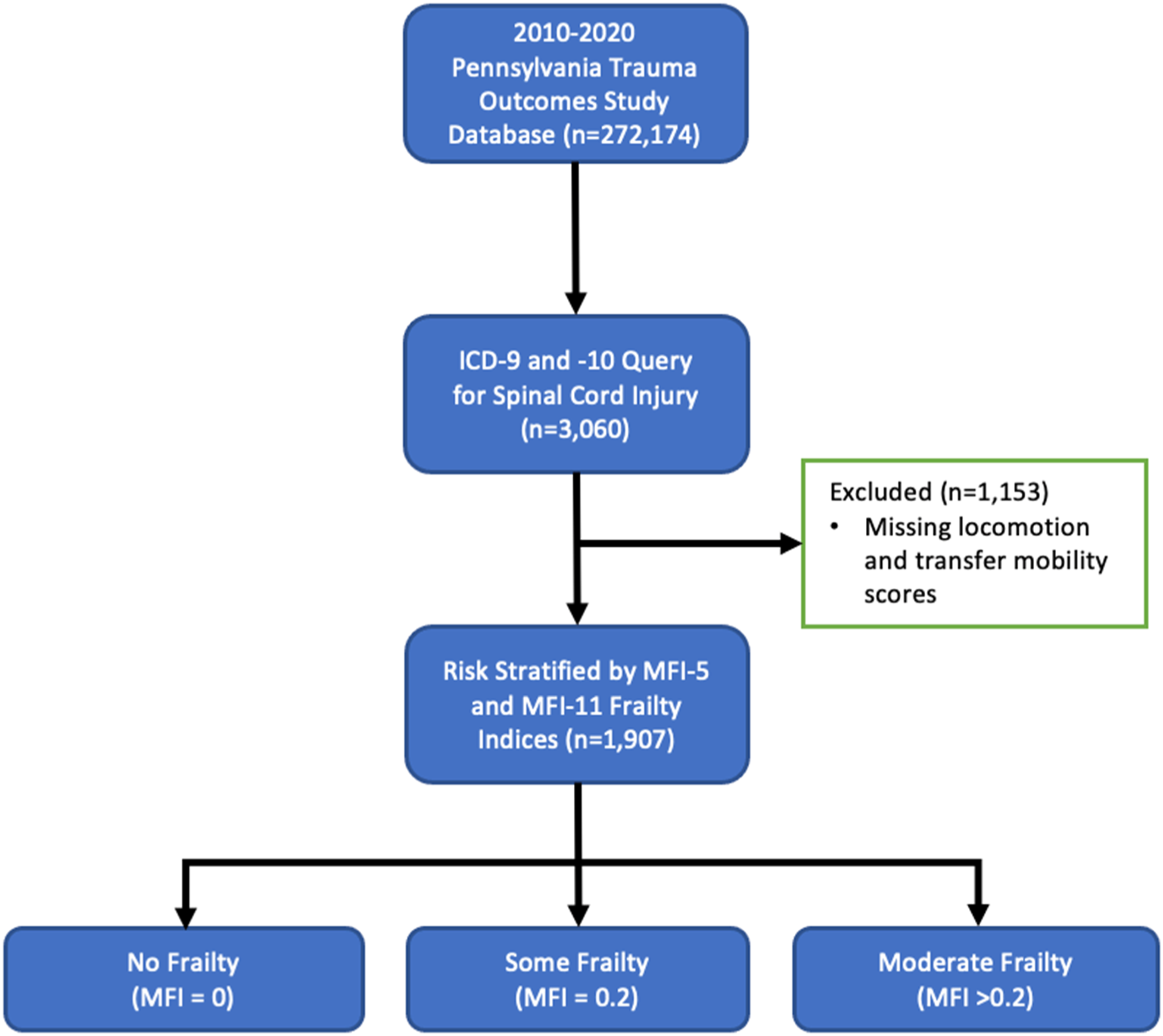
The primary endpoint was functional independence as determined by locomotion and transfer mobility. Each of these 2 functional status variables is scored from 1 to 4 (1 = complete dependence, 2 = modified dependence, 3 = independent with device, 4 = complete independence). 17 For locomotion, complete independence requires walking a minimum of 150 feet without assistive devices or a wheelchair; independent with a device requires walking a minimum of 150 feet, but with a brace (orthosis) or prosthesis on leg, adaptive shoes, cane, crutches, or walkerette. If not walking, operating a manual or electric wheelchair independently for a minimum of 150 feet, turning around, maneuvering the chair to a table, bed or toilet, maneuvering on rugs and over door sills. For transfer mobility, complete independence requires walking, approaching, sitting, and getting up to a standing position from a chair or bed. If in a wheelchair, approaching a bed or chair, locking brakes, lifting foot rests, and performing either a standing pivot or sliding transfer and returning. Independence with a device requires using adaptive or assistive device such as a sliding board, a lift, grab bars, special seat, brace, or crutch.
Functional independence included scores 3 and 4 from both the locomotion and transfer mobility variables, following existing literature using this database in SCI patients.18,19 Patients scoring less than 3 for either locomotion or transfer mobility were therefore designated as functionally dependent. Patients who were missing locomotion or transfer mobility scores were excluded. Data collection was conducted securely in a HIPAA-compliant database without any identifiable personal health information. Individual patient consent was not obtained given the retrospective, noninterventional design of the study that was based on query of an existing database.
Statistical Analysis
Data is presented as mean and standard deviation (SD) for continuous variables that are normally distributed and median and interquartile range (IQR) for non-normally distributed variables. Normality was assessed using the Shapiro-Wilks test. Categorical variables are presented with count and frequency. Associations were determined by χ2, χ2-trend, Fisher’s Exact, Pearson’s correlation coefficient, Spearman’s ρ, as appropriate. Univariable analysis was carried out using unpaired t-test, Chi-square, Fisher’s exact tests, Mann-Whitney U, and Spearman Rank as appropriate. If identified as predictive (defined herein as P < .20) of the endpoint on univariate analysis, the variable was included in a multivariable logistic regression model run in a backward conditional fashion. Redundant variables and those with greater than 5% missing data values were excluded. Results from multivariable logistic regression are reported as odds ratios (ORs), 95% confidence intervals (CI), and P-value. Statistical significance was achieved when P-value was <.05. Statistical analysis was carried out with IBM SPSS (Version 26.0. Armonk, NY: IBM Corp.).
To assess diagnostic strength for functional status at discharge, receiver operator characteristic curves (ROC) of the multivariable risk models were constructed, with predictive performance assessed by area under the curve and asymptotic P value. To assess relative model fit, the Aikaike Information Criterion (AIC) were also generated. 20
Results
Patient Characteristics by MFI-5 Frailty Group.
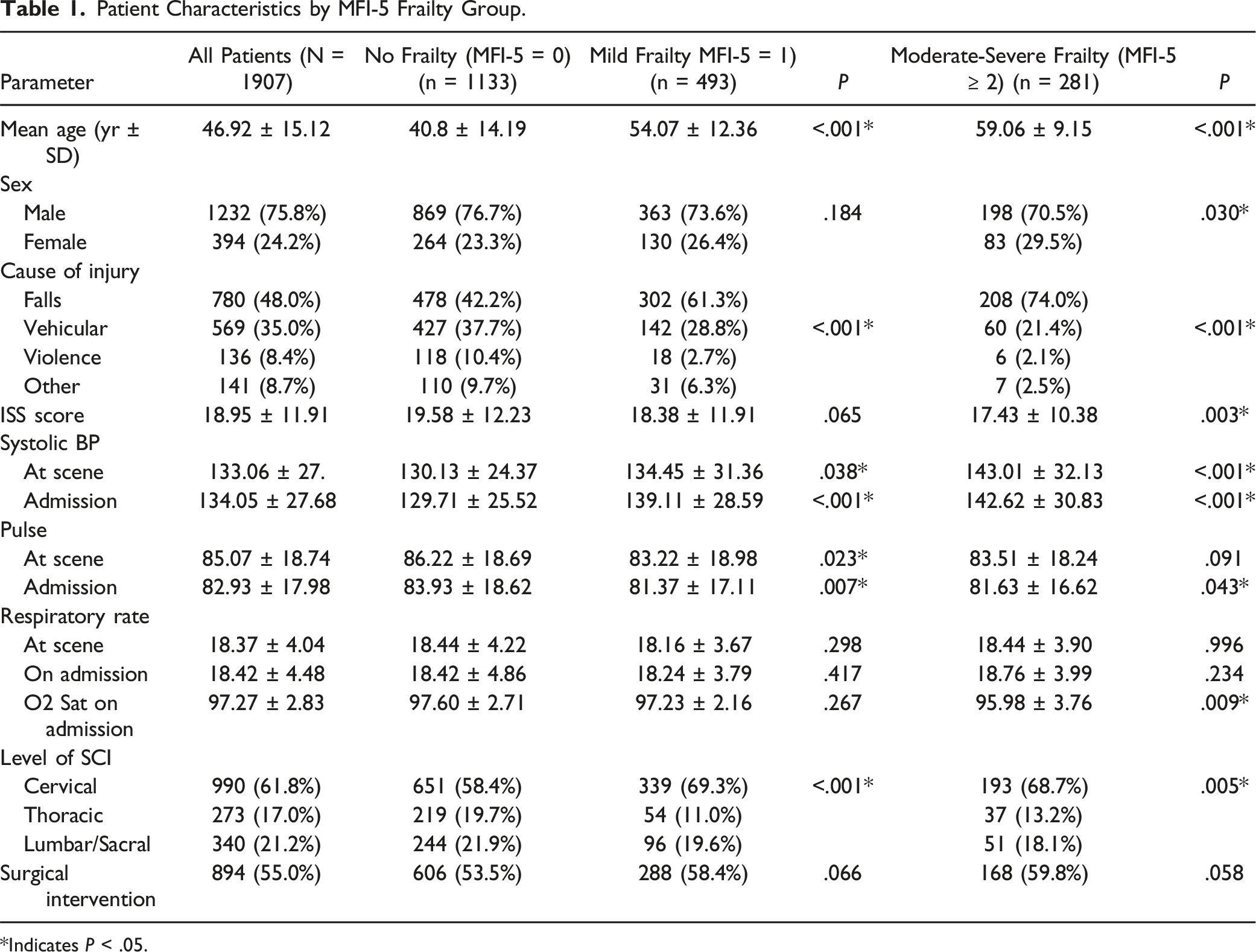
*Indicates P < .05.
Patient Characteristics by MFI-11 Frailty Group.
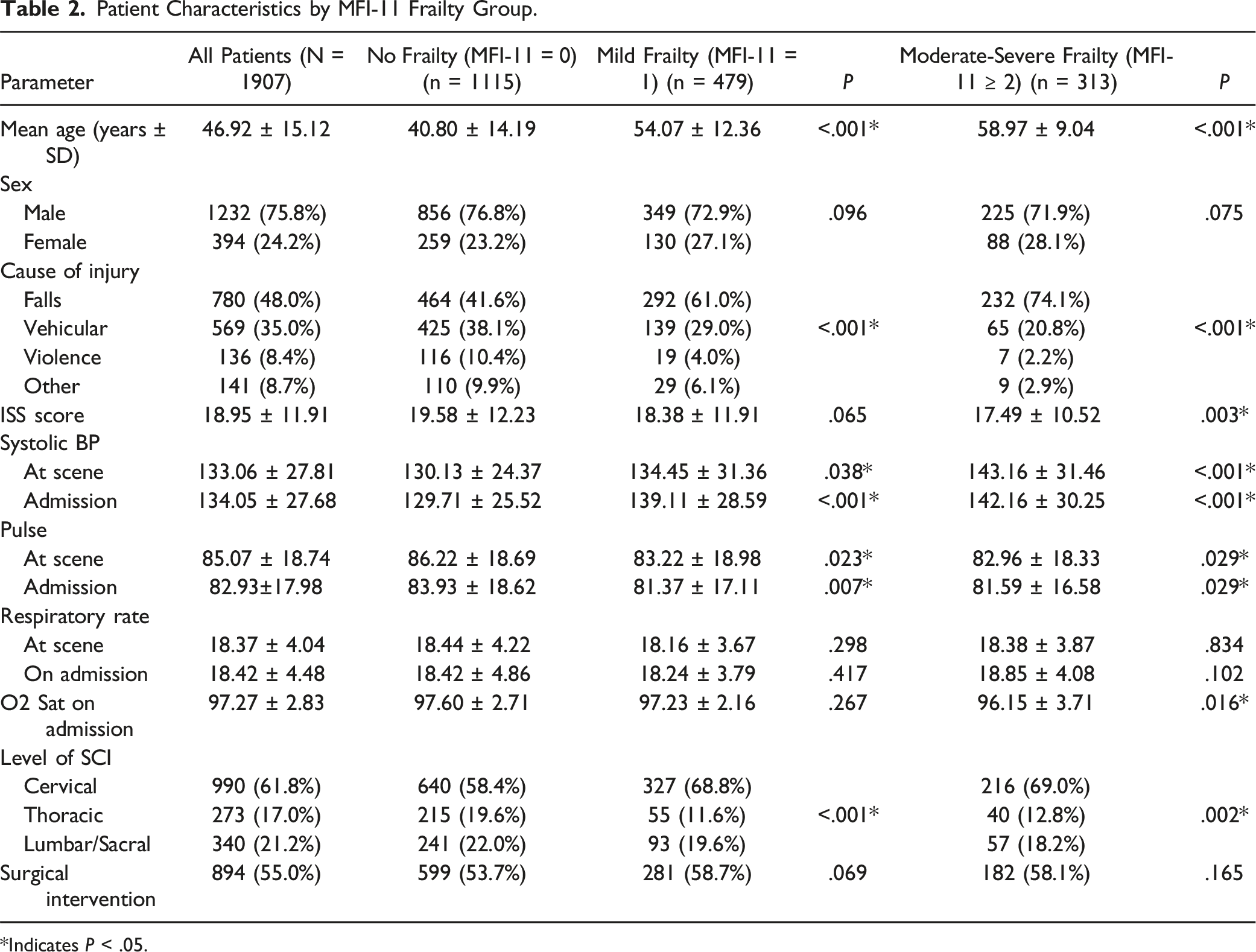
*Indicates P < .05.
Patient Characteristics by Functional Outcome.
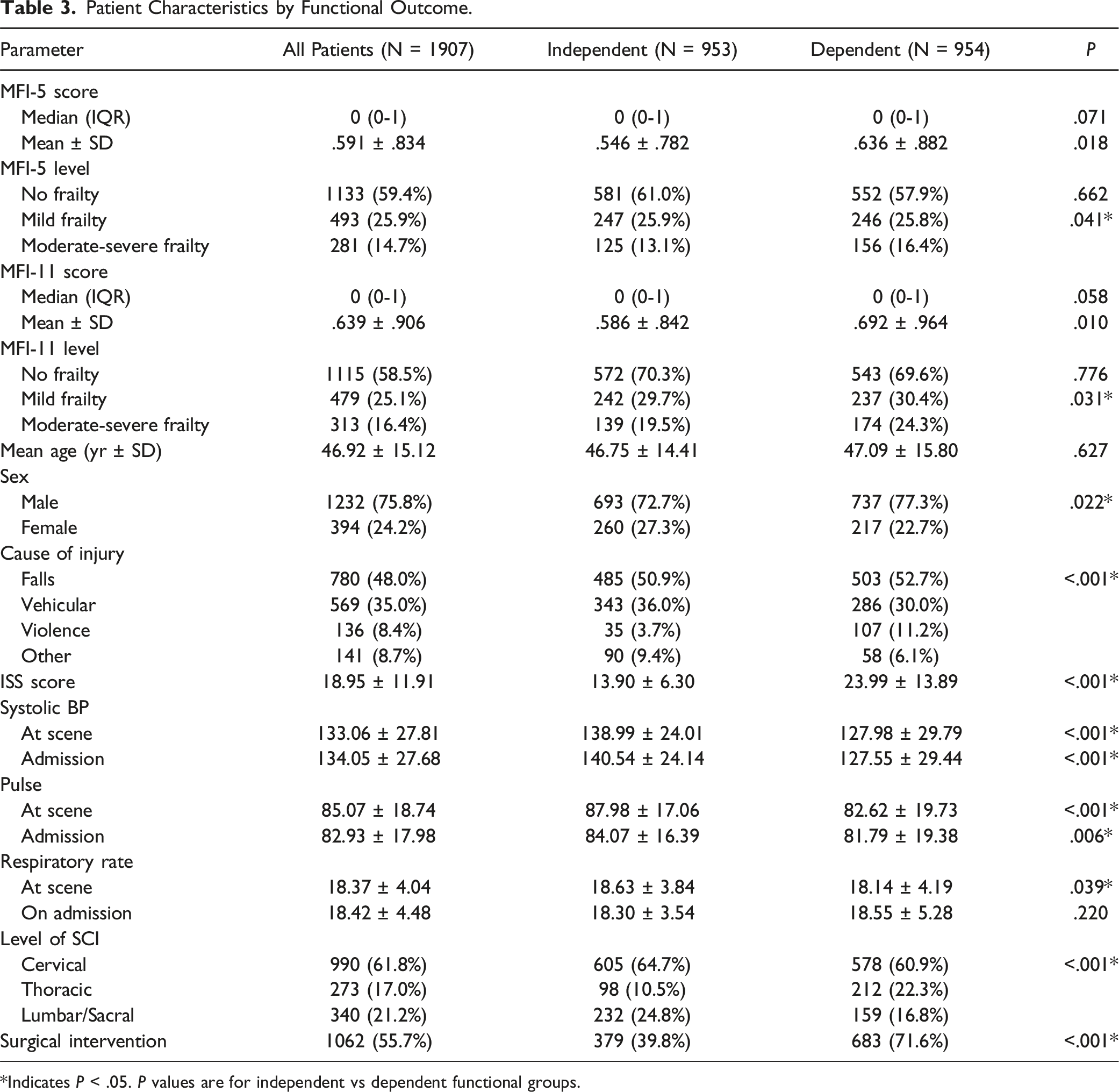
*Indicates P < .05. P values are for independent vs dependent functional groups.
Logistic Regression Analysis of MFI-5 for Poor Functional Status at Discharge.
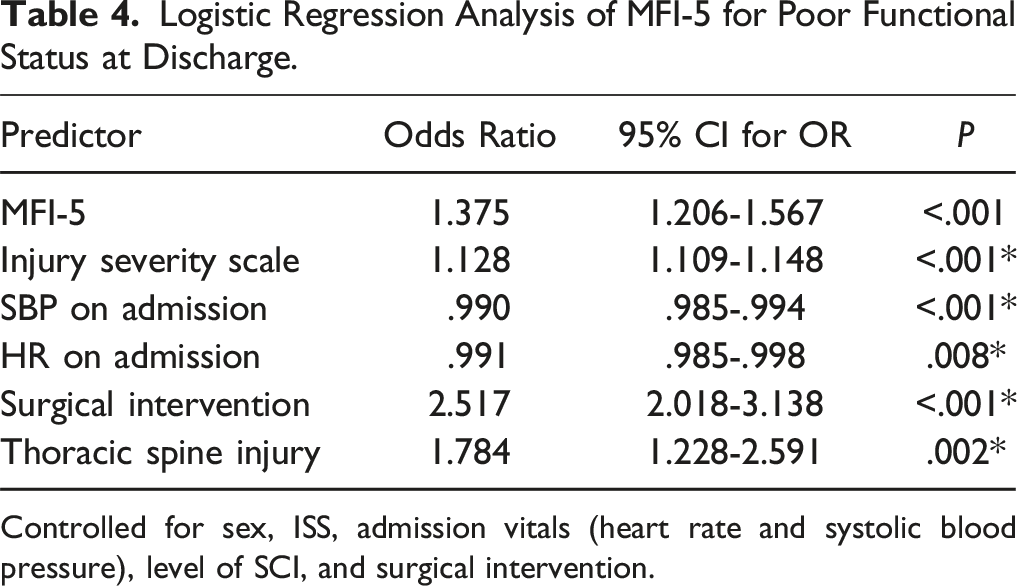
Controlled for sex, ISS, admission vitals (heart rate and systolic blood pressure), level of SCI, and surgical intervention.
Logistic Regression Analysis of MFI-11 for Poor Functional Status at Discharge.
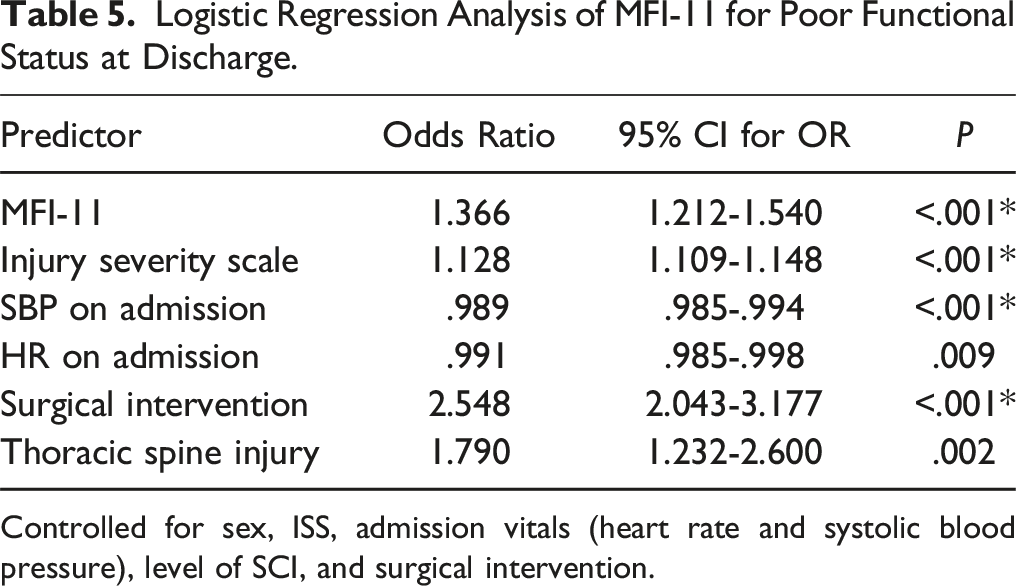
Controlled for sex, ISS, admission vitals (heart rate and systolic blood pressure), level of SCI, and surgical intervention.
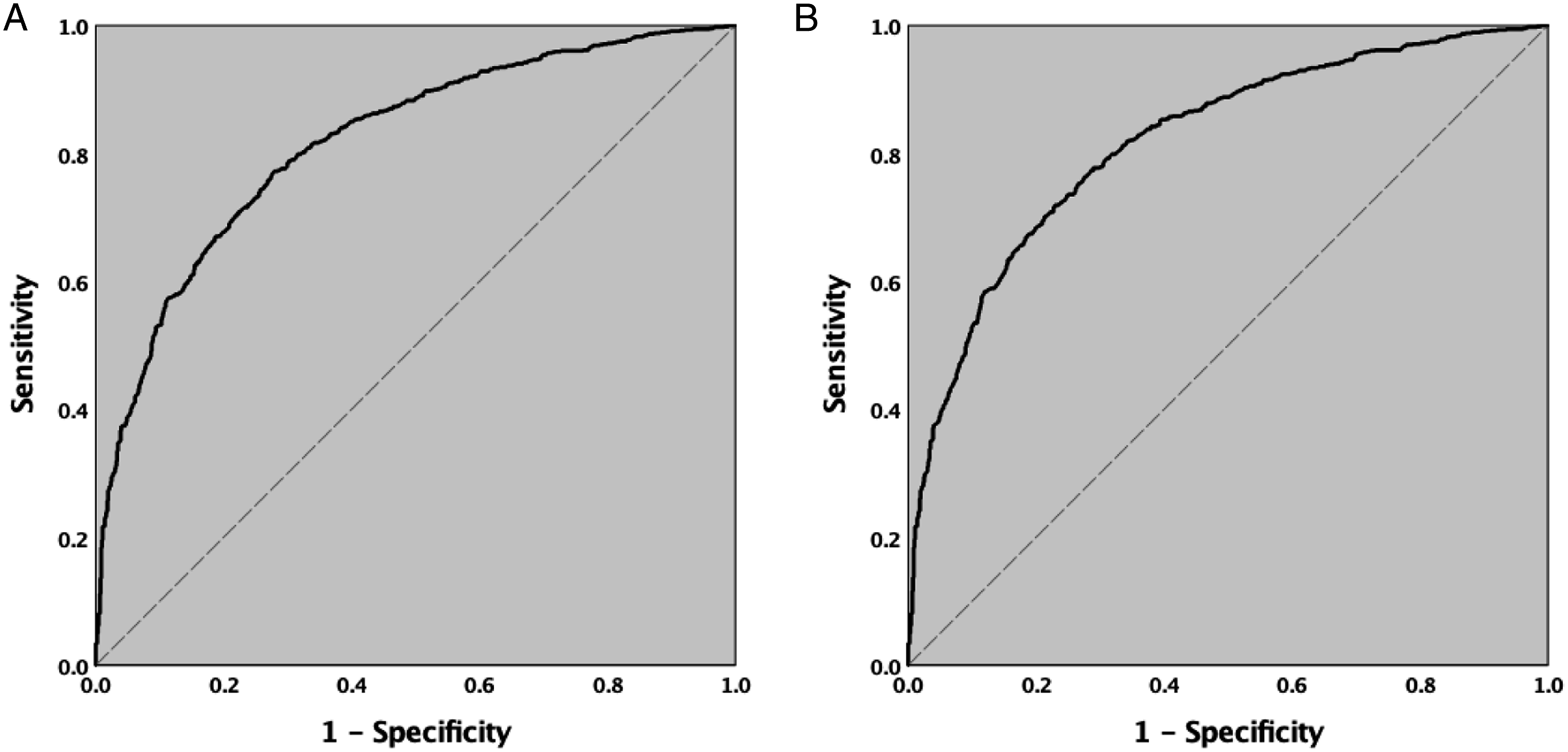
(A). ROC curve for Multivariable Logistic Regression Model of MFI-5 to predict poor functional outcome at discharge (AUC = .818 95% CI .799-837, P < .001). (B). ROC curve for Multivariable Logistic Regression Model of MFI-11 to predict poor functional outcome at discharge (AUC = .819 95% CI .800-.838, P < .001).
Discussion
The initial frailty indices originated in the early 2000s based on data from Canadian population surveys. A frailty index consisting of 38 variables that correlated with mortality was published in 2002 based on the Canadian National Population Health Survey.
21
A second frailty index consisting of 70 variables was developed in 2005 based on the Canadian Study of Health and Aging (CSHA)
There is a limited number of studies that have compared the MFI-5 and MFI-11 indices. These studies are based on the National Surgical Quality Improvement Program (NSQIP) 12 and the Trauma Quality Improvement Program (TQIP) database 16 and found the MFI-5 to be equally predictive for mortality, length of stay and adverse perioperative events. However, these studies focused on a wide range of patients from a variety of surgical specialties. Further, a few studies8,11 investigated these frailty indices in the SCI population and successfully demonstrated a correlation between increasing frailty score and longer length of stay, higher risk of complications and mortality. However, there is yet to be a study of frailty as a prognosticator for functional independence after traumatic SCI. To our knowledge, this is the first study in the literature to bridge this knowledge gap.
Our study utilizes the PTOS registry of patients with SCI from the mature trauma state of Pennsylvania and our findings demonstrate that the MFI-5 and MFI-11 are equally predictive risk stratification tools. Due to its ease of use, the MFI-5 may therefore be the preferred tool for functional prognostication after traumatic SCI. Interestingly, while age correlated with higher levels of frailty, it was not a significant risk factor for poor functional outcome. This fits with the existing literature which demonstrates that while age may be one component of physiologic frailty, it is less significant in predicting outcomes than the sum total of factors that can offset physiologic reserve.16,26,27 Indeed, this finding reinforces that frailty is a distinct risk factor from chronologic age, and is consistent with Banaszek et al.’s subgroup analysis showing that among patients younger than 75 years old, age was not a significant predictor of mortality, length of stay and adverse events. 8
Several important limitations must be taken into consideration when reviewing our findings. As a retrospective study based on a large registry, the data used here is based on coding for medical billing and quality of care review. Further, the study endpoint was functional independence at discharge and therefore outpatient functional status at follow-up is not available through this registry. The indication for surgery was also not available in the database, nor was there a specific characterization of the SCI beyond the level of injury (e.g. compressive lesion vs unstable fracture alone). Lastly, history of stroke without a neurological deficit is the only element of the MFI-11 that was not available in the PTOS registry. However, history of stroke with a neurological deficit is provided for each patient and is more clinically useful.
Despite these limitations, this study achieved our goal of comparing the MFI-5 and MFI-11 in traumatic SCI patients and identifies the MFI-5 as a sufficient predictor of poor functional outcome after traumatic SCI. Future studies should elucidate the role of frailty in long-term outcomes after traumatic SCI and aim to demonstrate its usefulness in clinical decision making.
Conclusion
Our study shows that MFI-5 and MFI-11 have equal prognostic value in terms of functional independence in patients with traumatic SCI. Given its ease of use, we posit that the MFI-5 can serve as the preferred frailty index at the point of care, as well as in research contexts. Future studies will need to demonstrate the usefulness of the MFI-5 in clinical decision making and in long-term prognostication after traumatic SCI.
Footnotes
Acknowledgments
These data were provided by the Pennsylvania Trauma Systems Foundation, Mechanicsburg, PA. The Foundation Specifically Disclaims Responsibility for any analyses, interpretations, or conclusions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author Contributions
NM, TL, and JJ designed the study. NM, TL were involved with data collection and analysis. NM, TL, CY, KL, DF, KH, KL were involved with data collection, manuscript preparation, critical revision, and data interpretation. AS, SP, JH, JSH, JJ were responsible for manuscript preparation, and critical revision.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
