Abstract
Study Design
Retrospective cohort study.
Objective
Compare the supine vs standing radiographs of patients with adult spinal deformity against ideals defined by healthy standing alignment.
Methods
56 patients with primary sagittal ASD (SRS-Schwab Type N) and 119 asymptomatic volunteers were included. Standing alignment of asymptomatic volunteers was used to calculate PI-based formulas for normative age-adjusted standing PI–LL, L4–S1, and L1–L4. These formulas were applied to the supine and standing alignment of ASD cohort. Analyses were repeated on a cohort of 25 patients with at least 5 degrees of lumbar flexibility (difference between supine and standing lordosis).
Results
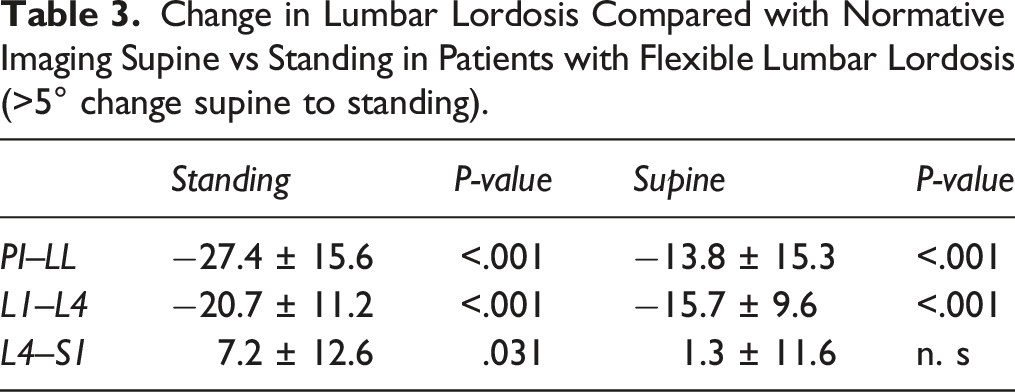
The asymptomatic cohort yielded the following PI-based formulas: PI–LL = −38.3 + .41*PI + .21*Age, L4–S1 = 45.3–.18*Age, L1–L4 = −3 + .48*PI). PI–LL improved with supine positioning (mean 8.9 ± 18.7°, P < .001), though not enough to correct to age-matched norms (mean offset 12.2 ± 16.9°). Compared with mean normative alignment at L1–L4 (22.1 ± 6.2°), L1–L4 was flatter on standing (7.2 ± 17.0°, P < .001) and supine imaging (8.5 ± 15.0°, P < .001). L4-S1 lordosis of subjects with L1-S1 flexibility >5° corrected on supine imaging (33.9 ± 11.1°, P = 1.000), but L1–L4 did not (23.0 ± 6.2° norm vs 2.2 ± 14.4° standing, P < .001; vs 7.3 ± 12.9° supine, P < .001).
Conclusions
When the effects of gravity are removed, the distal portion of the lumbar spine (i.e., below the apex of lordosis) corrects, suggesting that structural lumbar deformity is primarily proximal.
Introduction
Restoration of sagittal alignment during adult spinal deformity (ASD) correction is key to improving patient quality of life1-3 and avoiding the high cost and morbidity of revision surgery.4,5 While poor outcomes following ASD correction are multifactorial in nature, restoration of sagittal vertical axis, and pelvic incidence–lumbar lordosis (PI–LL) mismatch are critical to an optimal outcome. 6 Considerations to this end include age-dependent correction of PI–LL mismatch,7,8 judicious use of osteotomies in “stiff spines,” 9 and accounting for the patient’s intrinsic ability to use thoracic kyphosis to compensate for residual malalignment above the upper instrumented vertebra (UIV).10,11
Standing radiographs are representative of the multifactorial character of sagittal malalignment, which includes impaired muscle strength,12,13 axial loading, and compensatory mechanisms within the axial and appendicular anatomy.14,15 Barrey et al. 16 proposed a multistep process for identifying and accounting for compensatory mechanisms in patients with ASD, which include changes in lumbar lordosis/thoracic kyphosis, the presence of translational pathology such as retrolisthesis, and compensation through the appendicular anatomy such as knee flexion and ankle extension.17,18 However, surgical planning relies more heavily on structural deformity and flexibility. To this end, we have found that supine imaging correlates with intraoperative decision-making as it is more reflective of prone positioning 19 and is superior to standing imaging in the prediction of long-term post-operative lumbar lordosis and thoracic kyphosis. 20 We hypothesized that understanding regional lumbar flexibility by comparing standing and supine radiographs can help identify regions of the lumbar spine that most significantly contribute to PI–LL mismatch. Such knowledge can dictate the need for, extended proximal fusion constructs in order to avoid proximal residual deformity and adjacent segment disease (ASD).21,22
The present work sought to 1) establish a normative algorithm for lumbar lordosis and its proximal and distal components using healthy volunteers without spinal pathology; and 2) use this algorithm as an ideal baseline for assessing the lumbar lordosis of patients with ASD. Findings of this work can highlight where in the lumbar spine ASD patients lose the most lordosis and assess the structural vs non-structural nature of this loss.
Methods
Study approval was obtained through the Hospital for Special Surgery institutional review board (IRB approval number 2015-717). Informed consent was obtained from all healthy participants. Informed consent for ASD patients was not required as this was a retrospective evaluation of radiographs and there was a waiver of consent. Volunteers in the healthy cohort were compensated with a $25 gift card and free parking for their time. This retrospective analysis included patients who underwent primary surgeries for ASD with sagittal malalignment and minimal coronal deformity (coronal deformity SRS-Schwab type N 23 ) treated at a single academic tertiary referral center by 1 of 2 surgeons from 2014 to 2019, and a historical cohort composed of freestanding X-rays of healthy asymptomatic volunteers without a history of back/neck pain from an outside institution in the United States. 24
The primary outcome was lumbar lordosis by segment, defined as proximal lumbar lordosis (superior endplate of L1 to superior endplate of L4) and distal lumbar lordosis (superior endplate of L4 to superior endplate of S1, Figure 1). These definitions were based on prior anatomic studies that identified the L4 vertebral body as the most common apex of lumbar lordosis
25
and that of Presenti et al.,
26
who reported that L1-L4 lordosis varies with PI while L4-S1 remains relatively constant. Additional collected information were patient demographics and sagittal alignment parameters including pelvic incidence (PI), pelvic tilt (PT), lumbar lordosis (LL) between L1 and S1, pelvic incidence–lumbar lordosis mismatch (PI–LL), thoracic kyphosis (TKA), T1 pelvic angle (TPA),
27
and sagittal vertical axis (SVA). Measurements of proximal and distal lumbar lordosis.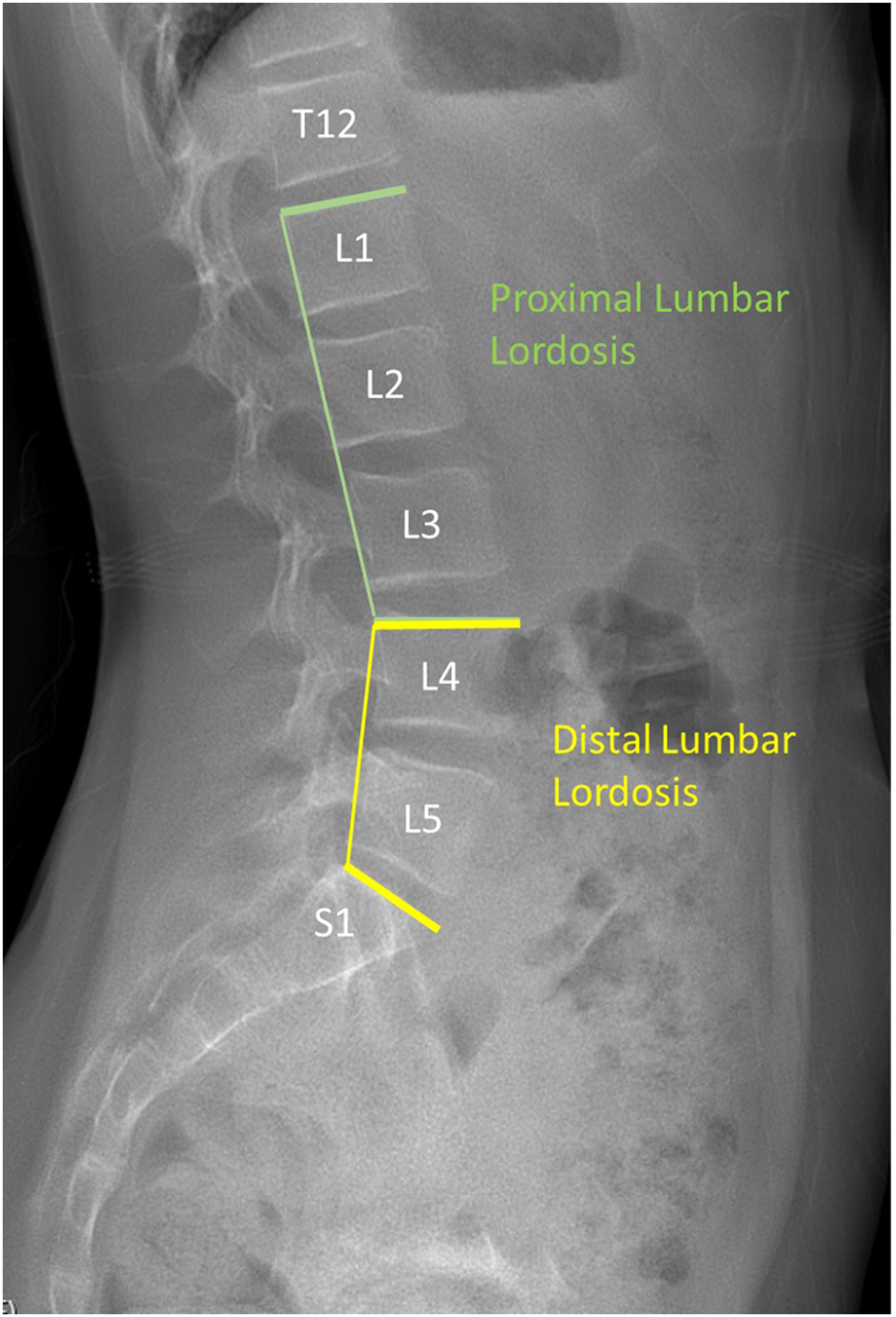
Statistical analysis was performed using IBM SPSS 26.0 (IBM, Armonk, NY, USA). Samples were ones of convenience, derived from patient records from procedures performed during the study period and asymptomatic volunteers recruited during a historical study period. Stepwise linear regression of asymptomatic segmental measurements was used to define formulas describing the normative segmental alignment of the spine (proximal and distal lumbar lordosis and PI–LL). These formulas were then applied to the cohort of ASD patients to identify regional loss of lordosis compared with expected alignment. The absolute values of regional measurements between standing asymptomatic controls, standing ASD and supine ASD patients were compared using a repeated measure analysis of variance (ANOVA) with a Bonferroni correction for between-group comparisons. Finally, given the potential for confounding by ASD patients with stiff lumbar spines, analyses were repeated in a cohort of 25 patients with at least 5° of flexibility (defined as the difference between supine and standing lumbar lordosis) and 5° of offset from regional alignment norms. All two-group comparisons used Student’s t-text. Significance was defined as P < .05 in all cases.
Results
Cohort Sagittal Alignment Characteristics.
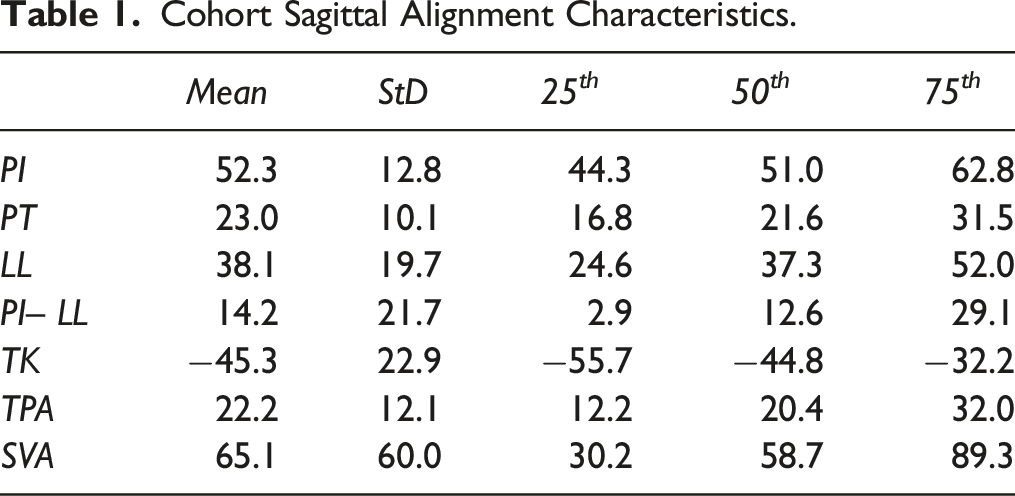
Asymptomatic volunteers had a mean age of 50.8 ± 17.0 years, were 68.1% female and had a mean BMI of 28.0 ± 6.0. Formulas for normative regional alignment in this cohort as measured with stepwise linear regression were as follows: PI–LL = −38.3 + .41*PI + .21*Age; L4–S1 = 45.3–.18*Age; L1–L4 = −3 + .48*PI.
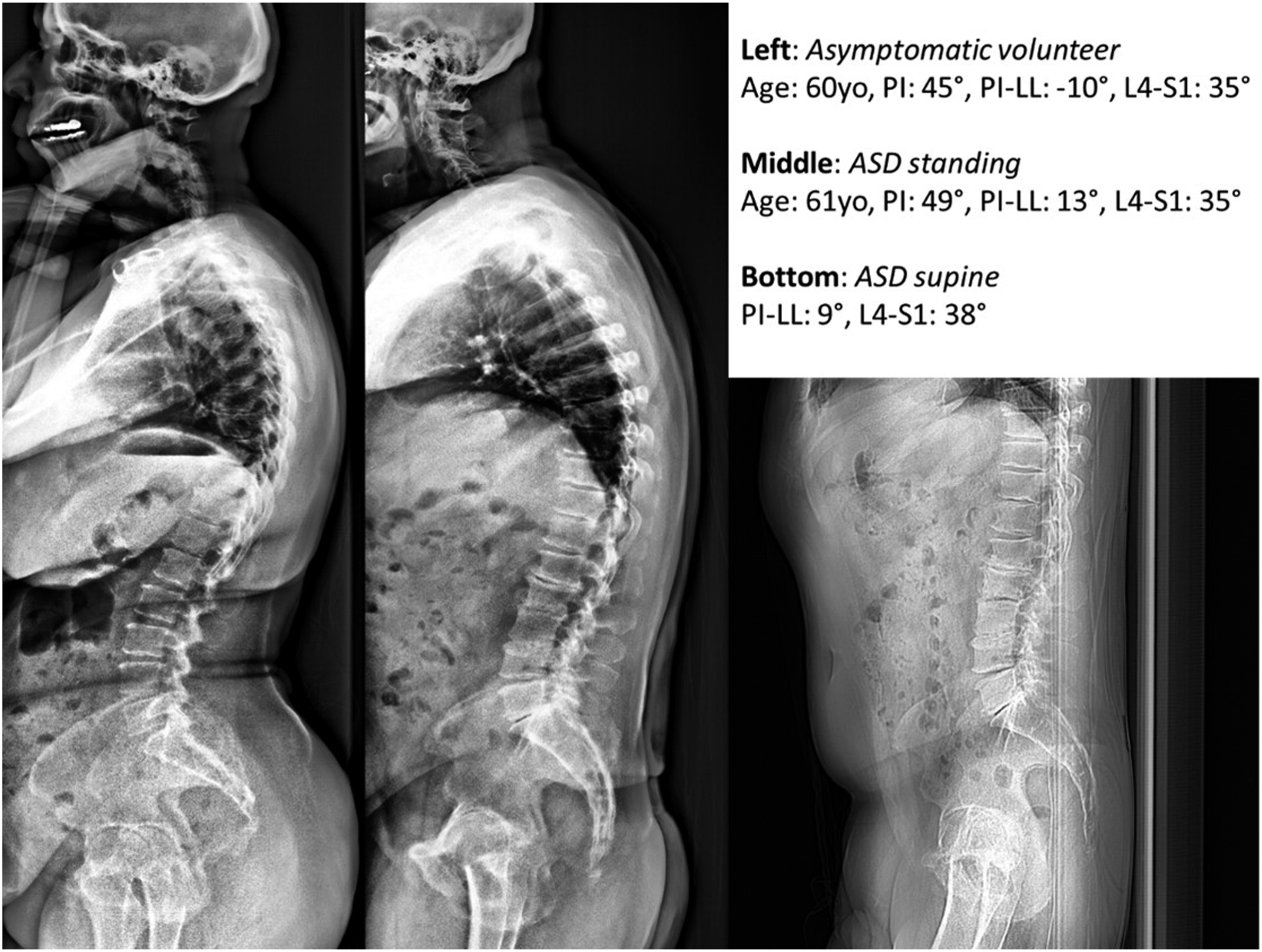
Regional measurements of asymptomatic and ASD patients are shown in Figure 2. PI–LL was significantly increased in ASD patients in both the standing (14.2°) and supine (8.9°) positions vs asymptomatic controls (−3.2°, P < .05 both comparisons). Proximal lumbar lordosis was also significantly lower in ASD patients vs asymptomatic controls (22.1°) in both the standing (7.2°) and supine (8.5°, P < .05 both comparisons) positions. Finally, distal lumbar lordosis was equivalent to that of asymptomatic controls (33.6°) for ASD patients in the supine vs standing positions (resp. 34.5° vs 30.9). Comparison of the regional alignment of patients with adult spinal deformity in the supine and standing positions to age-matched asymptomatic patients. * = P < .05 vs asymptomatic; † = P < .05 vs standing. Case example of an ASD patient with a flexible lumbar spine, with PI–LL reducing from 27 to 10° from standing (middle) to supine (right). Compared with his age and PI normative match (left image), the patient’s loss of lumbar lordosis occurred within the proximal lumbar spine. Case example of an ASD patient with a rigid lumbar spine characterized by an PI–LL between the standing (middle) and supine (right) positions. Compared with his age and PI match (left image), the patient’s loss of lumbar lordosis occurred within the proximal lumbar spine.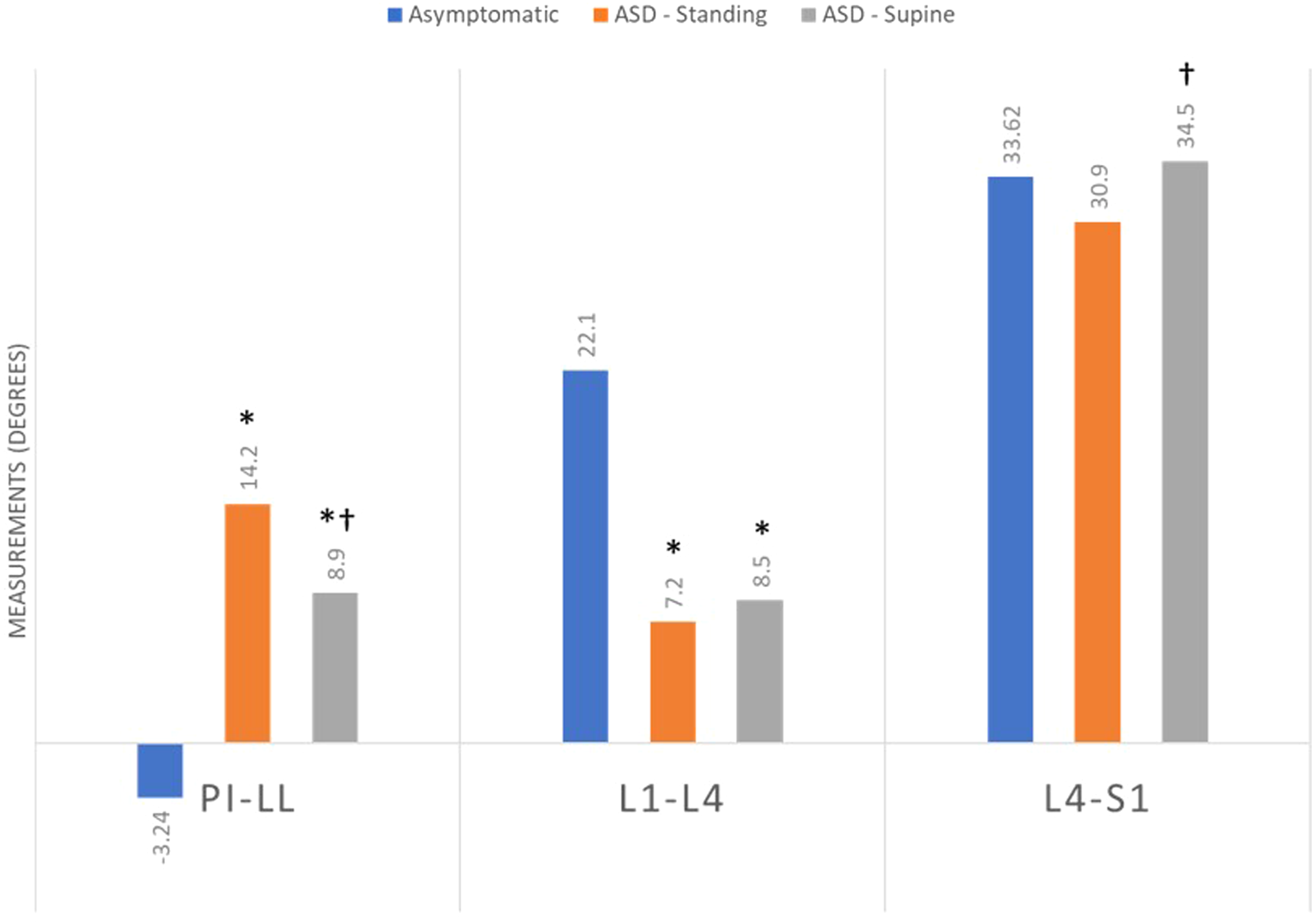
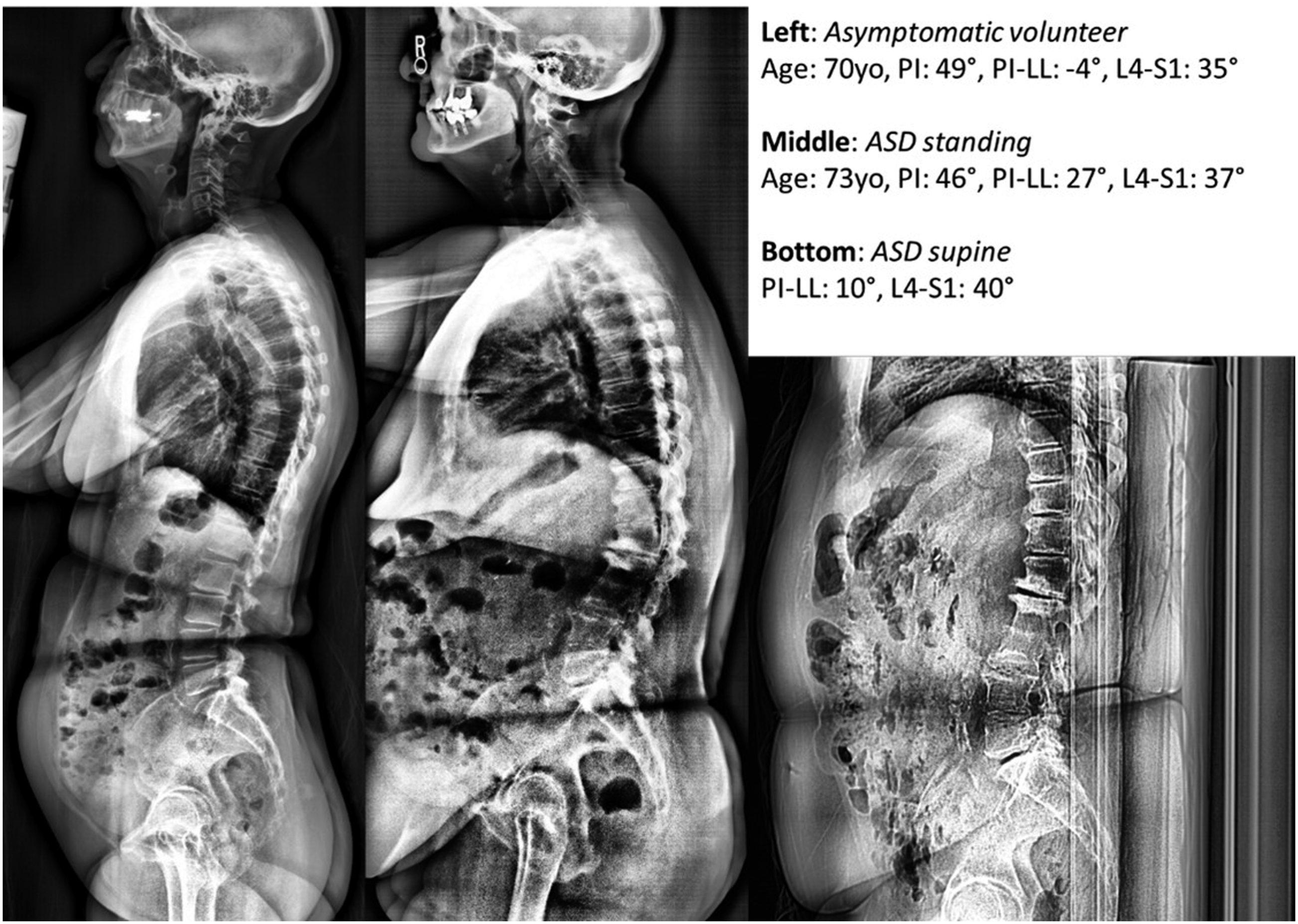
Change in Lumbar Lordosis Compared with Normative Imaging Supine vs Standing.

Change in Lumbar Lordosis Compared with Normative Imaging Supine vs Standing in Patients with Flexible Lumbar Lordosis (>5° change supine to standing).
Discussion
Restoration of normal lumbar sagittal alignment is key to achieving improved patient outcomes and minimizing the need for revision surgery. Using a normative algorithm of healthy spinal parameters as a baseline the present work found that: 1) PI–LL mismatch in the supine position was driven by a loss of proximal (L1–L4) lumbar lordosis; and 2), distal lumbar lordosis (L4–S1) remained largely unchanged vs asymptomatic controls regardless of position. While distal lumbar lordosis did increase in patients with more flexible lumbar spines (PI–LL difference >5° between supine and standing), this corrected with supine positioning while proximal lumbar lordosis did not. This work reinforces the key importance of supine imaging as part of surgical planning during ASD correction. It also suggests that supine proximal lumbar lordosis should be considered when deciding on the upper instrumented vertebra, particularly in patients with a high pelvic incidence.
The importance of supine imaging to ASD correction planning has been previously established. Chevillotte et al. 28 reported that the PI and LL of healthy patients are most strongly correlated when measured in the supine position, which reflects the purely structural relationship between the pelvis and lumbar spine. 29 Karikari et al. 9 note that “stiff lumbar spines,” or those that have a <33% change in LL between the supine and standing position, are more likely to require a three-column osteotomy to achieve satisfactory deformity correction. Elysee et al. 20 in their retrospective analysis of 99 patients with severe sagittal deformity determined that supine LL and pelvic incidence were the only significant predictors of post-operative LL. The authors also found that standing pre-operative lumbar lordosis poorly correlated with post-operative result. While supine imaging may produce more optimal lumbar spine parameters, the reverse may be the case in the thoracic spine. Lovecchio et al., 10 using supine imaging, caution that proximal fusion constructs incorporating a flexible thoracic spine risk the creation of a “flattened” thoracic spine and truncated thoracic kyphosis, which has been shown to contribute to PJK.21,30,31
Our stepwise linear regression of the normative alignment measurements of healthy volunteers found that while distal lumbar alignment was negatively impacted by advancing age, proximal lumbar lordosis was related to pelvic incidence. The relationship between PI and proximal lumbar lordosis has been previously demonstrated by Pesenti et al.,
26
who attributed this finding to the increased need to recruit the proximal lumbar spine into lordosis to compensate for a high pelvic incidence. The authors hypothesized that this also represents the proximal migration of the apex of lordosis in accordance with the classification set forth by Roussouly et al.
25
Recreation of an anatomic lumbar curve in the setting of a high pelvic incidence may necessitate evaluation and correction of discrepant lordosis in the proximal lumbar spine. This understanding can guide osteotomy selection and the restoration of the proportionate balance between the PI–LL and distal/proximal LL.32,33 While lumbar lordosis has been shown to decrease with age, there is disagreement about where that decrease occurs. Non-invasive biomechanical measurements of healthy volunteers performed by Dreischarf et al.
34
demonstrated an age-related decrease in LL, but through the mid-lumbar spine proximal to the apex of lordosis. This finding was similarly supported by a linear regression of radiographic measurements on healthy volunteers by Gelb et al.
35
The impact of age on lumbar lordosis in this and prior works reinforces what we know, which is that older patients have a reduced compensatory reserve that makes them prone to sagittal malalignment as they age. This is particularly prominent in patients with a high pelvic incidence who already required a good deal of proximal lumbar lordosis. Distal lumbar lordosis remains important to achieving overall sagittal balance. However, the present work suggests that patients with a higher pelvic incidence may require more aggressive correction at proximal levels in addition to that already required in the distal lumbar spine in order to recreate their anatomic curve.
The present work observed that proximal lumbar lordosis (L1–L4) accounts for the majority of the lumbar lordosis deficit compared with expected norms following supine positioning. Further, an unfused distal lumbar lordosis in patients with ASD corrects to healthy norms. This does not undervalue the importance of the distal lumbar spine to achieving optimal sagittal balance.
32
However, this report provides an added caveat that short fusions with suboptimal correction of the proximal lumbar spine risks undercorrection, predisposing to PJK and poor quality of life outcomes.
22
It should be emphasized that not every patient requires an extensive fusion construct to achieve optimal lumbar sagittal alignment. It is also unclear how to balance advancing into the thoracic spine to more aggressively correct proximal lumbar lordosis at the cost of thoracic kyphosis. Oren et al.
36
expressed a similar concern in their retrospective analysis of the prone radiographs of 80 patients who underwent >4 level ASD corrections with constructs that extended into to either the lower or upper thoracic spine. The authors reported that fusing into the lower thoracic spine led to distal thoracic hypokyphosis and reciprocal proximal thoracic hyperkyphosis, predisposing to PJK.
37
The authors recommended intraoperative measurement of segmental thoracic kyphosis based on the UIV, which has been shown to predict post-operative standing alignment.
38
There are several limitations to the present work beyond those that are intrinsic to retrospective analyses. First, it only evaluated changes in the regional sagittal alignment of ASD patients with primarily sagittal deformity. The influence of coronal deformity on standing vs supine segmental measurements was not considered. It is likely that the location of the apex of the coronal curve and its relative flexibility will yield different degrees of sagittal correction and deformity. However, this work was a pilot study that relied on a relatively homogeneous sample size to isolate the influence of specific regional anatomy on overall balance. Including coronal differences might have distorted its conclusions. Second, while this work appears to be appropriately powered to achieve its primary outcome, it utilized a small sample size. External validation of the normative measurements and ASD differentials measured here is necessary. This is particularly important with respect to the relationship between age and distal lumbar lordosis, which disagrees with the existing literature. Third, as with all studies that evaluate supine vs standing imaging of the spine, supine measurements should not be considered absolutely equivalent to those obtained in the intraoperative prone position, which is influenced by factors such as patient habitus, table type and pad positioning.39,40 That said, Harimaya et al.
19
noted that pre-operative supine imaging was predictive of the amount of lordosis that could be obtained during intraoperative prone positioning. Future work will compare measurements obtained from intraoperative prone laterals with both supine pre-operative imaging and the normative algorithm to re-establish this equivalence. Fourth, this work uses standardized measurements for lumbar lordosis that assume that the junction between lumbar lordosis and thoracic kyphosis is at L1. This is reflective of previous anatomic studies by Roussouly et al.,
25
who in their cohort of healthy patients reported that the transition occurs at on average at the center of the L1 vertebral body. However, the authors note that this transition occurred as proximally as T10, and as distally as L4. We believe that our findings remain accurate given their agreement with prior works that utilized a patient-dependent apex.
26
However, future studies that include multiplanar imbalance or that consider construct selection and clinical outcomes may choose to employ more patient-specific definitions of lumbar lordosis. Such works should question if the “redefinition” of a transition impacts the quality of life improvement or adjacent segment disease. Finally, while this work suggests that sagittal malalignment originates more proximally in the lumbar spine. It is unable to present clinical recommendations regarding the situational need to extend a fusion construct. It is also unable to associate proximal fusion constructs with a reduced risk of reoperation and improved patient outcomes. It may be that restoration of normative lumbar lordosis via deformity correction is not the ideal “answer” for preventing junctional deformity. However, such a hypothesis requires extensive prospective validation.
In conclusion, a normative algorithm derived from healthy volunteers found that while proximal lumbar lordosis is driven by pelvic incidence, distal lumbar lordosis is age-dependent. When patients with ASD were compared with normative measurements, supine positioning corrected deficits in the distal lumbar lordosis of patients with ASD. This suggests that residual PI–LL mismatch in the supine position is due to persistent deficits in the proximal lumbar spine. These findings prompt questions about the regional flexibility of the lumbar spine and the clinical implications thereof. They may also suggest that the pathogenesis of ASD is driven by proximal changes, although this remains to be assessed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
