Abstract
Study Design:
Retrospective cohort study.
Objectives:
We aimed to evaluate the effectiveness of terminal ventriculostomy in treating tethered cord syndrome (TCS) combined with terminal syringomyelia (TS) and describe “V”-type ostomy as an effective surgical method to avoid relapsing syringomyelia based on terminal ventriculostomy.
Methods:
We retrospectively analyzed the clinical and radiological data of 28 patients admitted to the Department of Neurosurgery, PLA General Hospital who had been diagnosed with TCS combined with TS and underwent terminal ventriculostomy-associated “V”-type ostomy between January 2011 and January 2016. We classified patients’ clinical outcomes into 4 levels according to the Spinal Bifida Neurological Scale: markedly improved, improved, stable, and deteriorated. The size of the syrinx cavity was quantified using the syrinx index, and there was a difference in syrinx cavity size between pre-operation and post-operation.
Results:
Twenty-eight patients were followed up for 36 months. We found that each syrinx cavity shrunk by at least 50%. More than 90% of patients had achieved “markedly improved” and “improved” outcomes during the follow-up visit. Moreover, no patient relapsed for up to 36 months post-surgery.
Conclusion:
Terminal ventriculostomy has a beneficial effect on TS, particularly on the syrinx cavity extending to the filum terminale. For this special cavity, we advocate the use of terminal ventriculostomy-associated “V”-type ostomy to avoid potential relapse. As a safe, convenient, and persistently effective approach, terminal ventriculostomy-associated “V”-type ostomy can be considered a promising alternative method for treating TCS combined with TS in clinical practice.
Keywords
Introduction
Tethered cord syndrome (TCS), a type of neural tube defect, is caused by fixation and traction of the spinal cord because of various factors, either congenital or acquired. This leads to ischemia of the conus medullaris and damages the cauda equina. Syringomyelia develops as a TCS-accompanying disease. A clinical study reported morbidity of 22.5–53% for TCS associated with syringomyelia. 1 Terminal syringomyelia (TS), in which the lesion involves the terminal spinal cord, is a special type of syringomyelia. Patients with TS have motor and sensory deficits in the lower extremities and experience disturbance in urination and defecation.
Syringomyelia can be treated by syringosubarachnoid shunt placement, syringoperitoneal shunt placement, or myelotomy. 2 In 1977, Gardner proposed a groundbreaking method—terminal ventriculostomy—for treating syringomyelia. 3 To validate its effectiveness, many clinicians have replicated his surgery, but the outcomes vary considerably. Williams et al. considered this novel surgical method to merely manage the condition. 4 Previous studies have suggested that terminal ventriculostomy causes TCS relapse, which may be due to reclosure of the fistula and preclude further drainage.4-6 Conversely, Beaumont et al. confirmed the advantages of this approach. 7 Thus, the effectiveness of terminal ventriculostomy remains controversial.
We considered whether terminal ventriculostomy could benefit patients with TS. This study aimed to evaluate terminal ventriculostomy for patients with TCS combined with TS and optimize a surgical approach based on terminal ventriculostomy to avoid potential reclosing of the fistula.
Materials and Methods
We retrospectively analyzed 28 patients admitted to the Department of Neurosurgery, General PLA Hospital who were diagnosed with TCS combined with TS between January 2011 and January 2016. The patients’ characteristics are shown in Table 1. The preoperative clinical manifestations and radiological data are shown in Tables 2 and 3, respectively. We excluded patients associated with Arnold–Chiari malformation, as this malformation could develop into syringomyelia spontaneously. This study was approved by the Institutional Review Board of PLA General Hospital and followed the tenets of the Declaration of Helsinki. All patients provided written informed consent.
Patients’ Background Characteristics.

Abbreviation: SD, standard deviation.
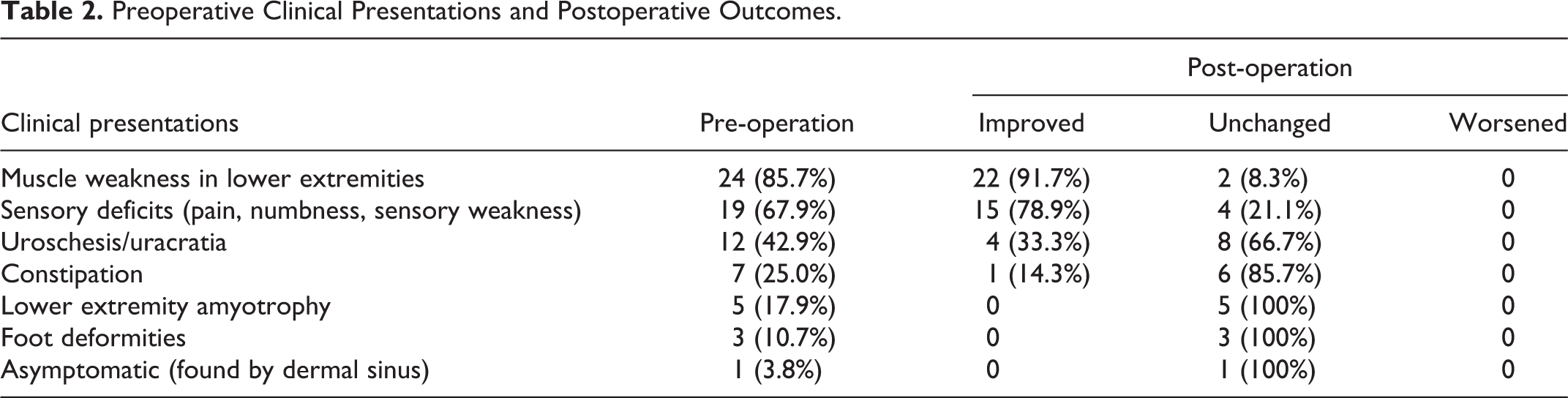
Preoperative Clinical Presentations and Postoperative Outcomes.
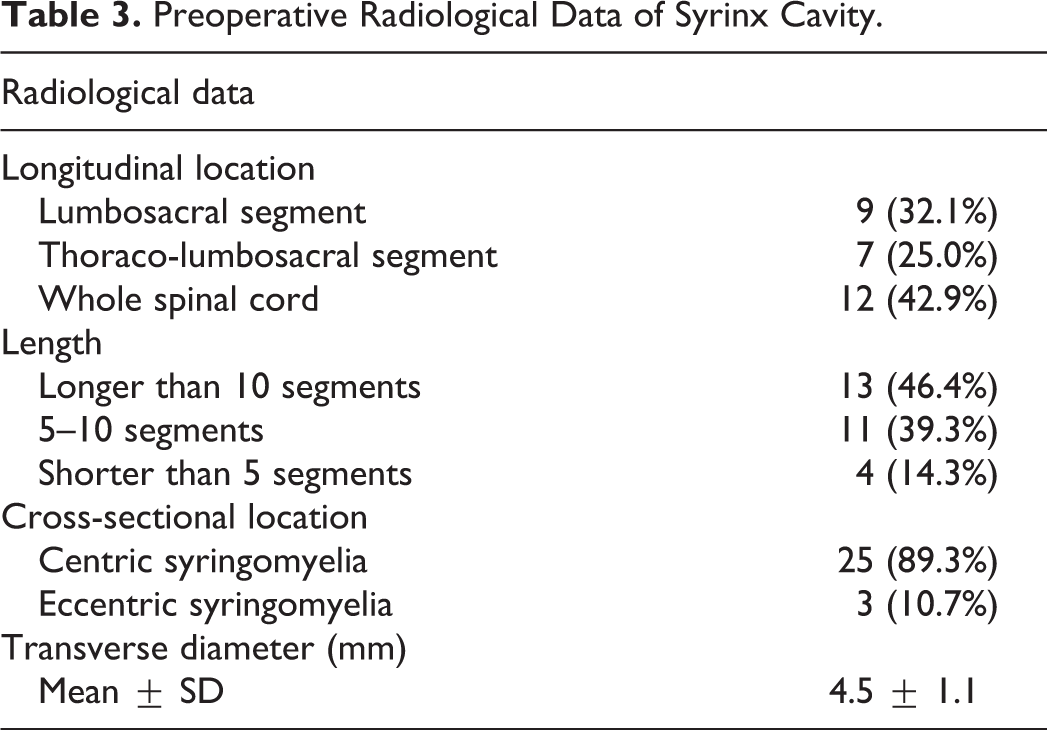
Preoperative Radiological Data of Syrinx Cavity.
Surgical Management
Patients underwent general anesthesia with tracheal intubation in the prone position. At the level of sacral vertebrae 1–2, we made a posteromedian longitudinal incision of 1.5–3 cm. We separated subcutaneous tissue and muscle, creating a bony window of approximately 1.5 cm × 1 cm and made an approximately 1-cm incision along the posterior midline of the dorsal dura mater, revealing the thickened filum terminale. Under electrophysiological monitoring, we then identified the last pair of sacral nerves. Bipolar coagulator was used to burn the vessels on filum terminale until they became transparent. Then, we cut off the filum terminale in the burned range. A “V”-type fistula was made at the proximal end of the severed filum terminale to prevent the opening from reclosing. Finally, we sutured the dura mater and closed the incision layer-by-layer. The patient needed to maintain the prone position for 3–7 days after surgery to avoid local effusion and cerebrospinal fluid (CSF) leakage. The major procedure of terminal ventriculostomy-associated “V”-type ostomy is shown in Figure 1 and Supplemental Video 1.
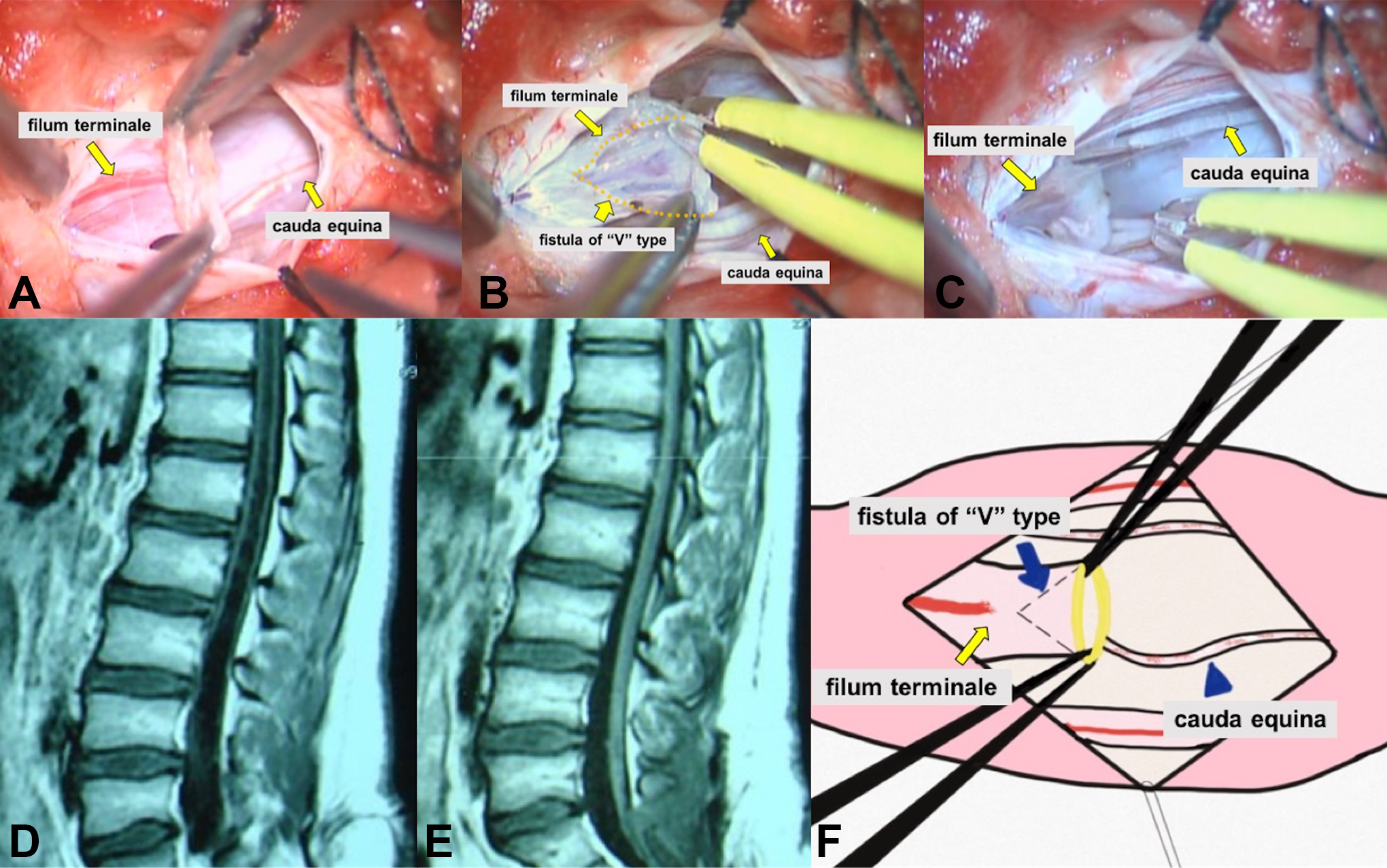
A 14-year-old male patient diagnosed with tethered cord syndrome combined with terminal syringomyelia. Preoperative lumbosacral sagittal T1-weighted magnetic resonance imaging (MRI) sequences (D) reveal cerebrospinal fluid-like hypointense signal in the spinal cord without an identified filum terminale. We operated on her terminal ventriculostomy-associated “V”-type ostomy (A, B, C). Postoperative lumbosacral sagittal T1-weighted MRI (E) reveals the syrinx cavity to be eliminated and that the conus medullaris recovered 12 months after surgery. (F) Diagram of “V”-type ostomy.
Patients who had associated split cord malformation (SCM) or dermal sinus additionally underwent relative lesion-repairing treatment during the same surgery.
Electrophysiological Monitoring
We used intraoperative electrophysiological monitoring to assess neurological function. We measured the somatosensory-evoked potentials (SSEPs) and transcranial electric motor-evoked potentials (MEPs) while searching for the last pair of sacral nerves and cutting off the filum terminale below the final nerve roots.
Evaluation Systems
Clinical evaluation
We used the Spinal Bifida Neurological Scale (SBNS) to evaluate preoperative and postoperative neurological function. 8 The scoring was based on motor (6 points), reflex (4 points), and bladder and bowel function (5 points), for a total of 15 points. Neurological function was graded as follows: Grade I: No neurological deficits of the spinal cord (15 points); Grade II: Autonomous movement, but with uroclepsia or encopresis (10–14 points); Grade III: Autonomous movement, with slight paralysis (5–9 points); Grade Ⅳ: Incapacity to take care of self (4 points); Grade V: Completely bedridden status (3 points).
To accurately describe changes following surgery, we divided post-surgical outcomes into 4 categories according to the SBNS: markedly improved, improved, stable, and deteriorated, which were defined as follows: (i) markedly improved: obvious clinical symptoms disappeared, and neurological function achieved SBNS Grade I; (ii) improved: slight neurological symptoms remained, and the SBNS grade advanced by at least one grade; (iii) stable: the SBNS grade had not advanced and clinical manifestations remained unchanged; (iv) deteriorated: the SBNS grade had reduced by at least one grade.
Radiological evaluation
Using axial magnetic resonance imaging (MRI), we calculated syrinx index, i.e. the ratio of the maximal diameter of the syrinx cavity to that of the spinal cord. We compared the preoperative and postoperative indexes to evaluate changes in the size of the syrinx cavity.
Follow-up
We required all patients to undergo follow-up examinations at 3 months, 12 months, 24 months and 36 months post-operation. We collected data of the fourth examination (36 months following surgery) to analyze the outcomes.
Statistical Analysis
Data is presented as mean ± SD (standard deviation) or as n (%) for statistical analysis.
Results
All patients recovered well without surgery-related complications and were discharged 5–8 days following surgery.
Clinical Results
We found that muscle weakness in the lower extremities was relieved in 91.7% of patients, and sensory deficits were relieved in 78.9% of patients. However, paruria (uroschesis or uracratia) was improved in approximately 30% of patients with these preoperative symptoms, and only one improved case had constipation. Limb impediment (lower extremity amyotrophy and foot deformities) remained unchanged. This data, as shown in Table 2, confirmed that this surgery could benefit muscle weakness and sensory deficits most but did not lead to full recovery of dysporia and paruria and exerted no effect on limb impediment.
Based on our evaluation system, 14 (50.0%) patients achieved “markedly improved”; 13 (46.4%) patients reached “improved”; 1 (3.6%) patient achieved “stable”; and none of the patients deteriorated (Table 2). The data demonstrated that, for each patient in our study, surgery proceeded smoothly, and a satisfactory clinical outcome was achieved. Moreover, none of our patients demonstrated syringomyelia relapse 36 months post-operation.
Radiological Results
The syrinx cavities in 7 (25.0%) cases shrunk remarkably (cavity index reduced by more than 50% but less than 90%); in another 21 (75.0%) cases, the syrinx cavities virtually disappeared (cavity index reduced by more than 90%), including 9 (32.1%) cases in which the cavities completely disappeared and the spinal cords normalized (Table 4). This indicated that syringomyelia in all patients was treated effectively through this surgery. The 36-month-postoperative MRI images of a case with a preoperative whole-spinal syrinx cavity are shown in Figure 2.
Syrinx Cavity Data of Patients.
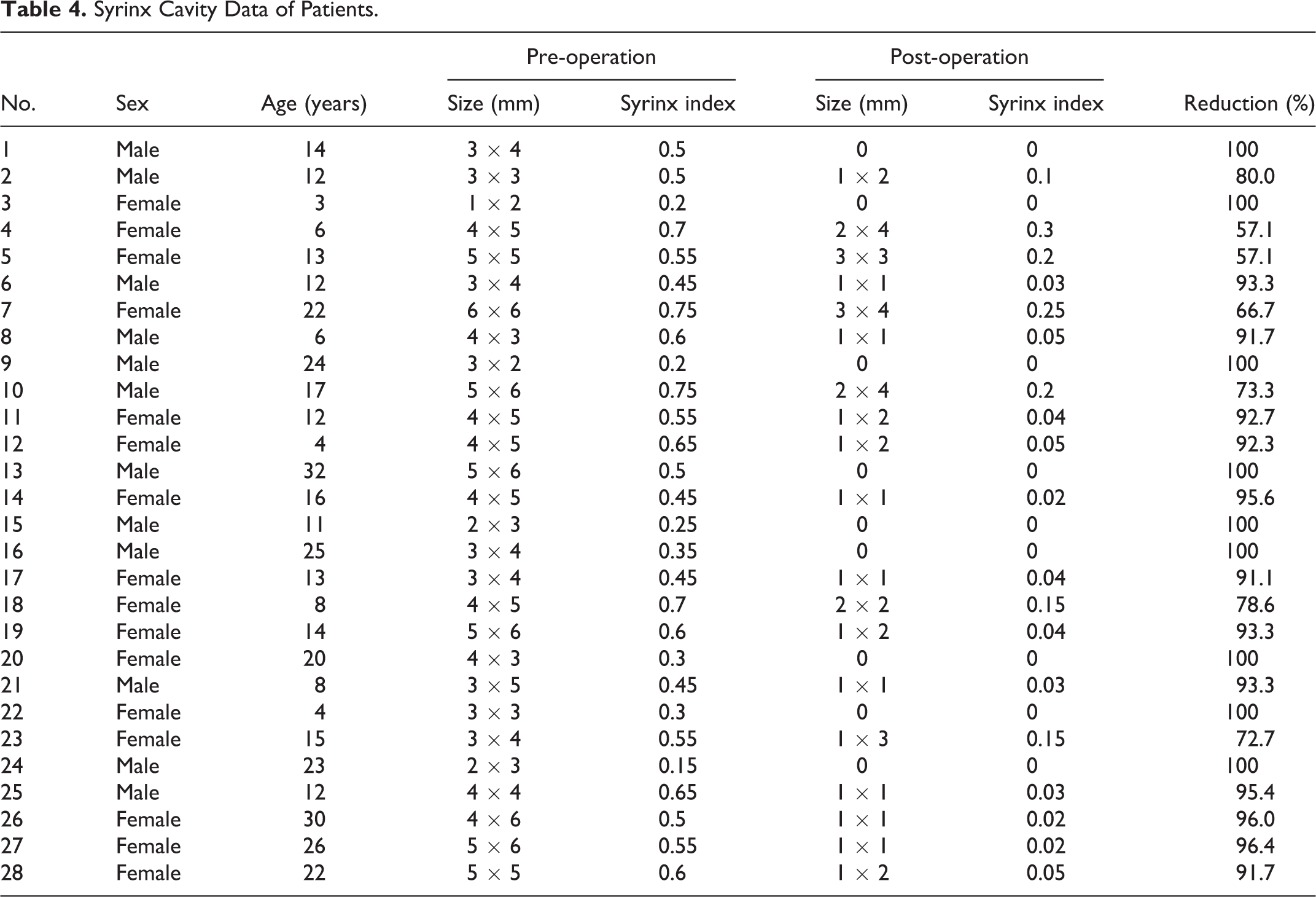
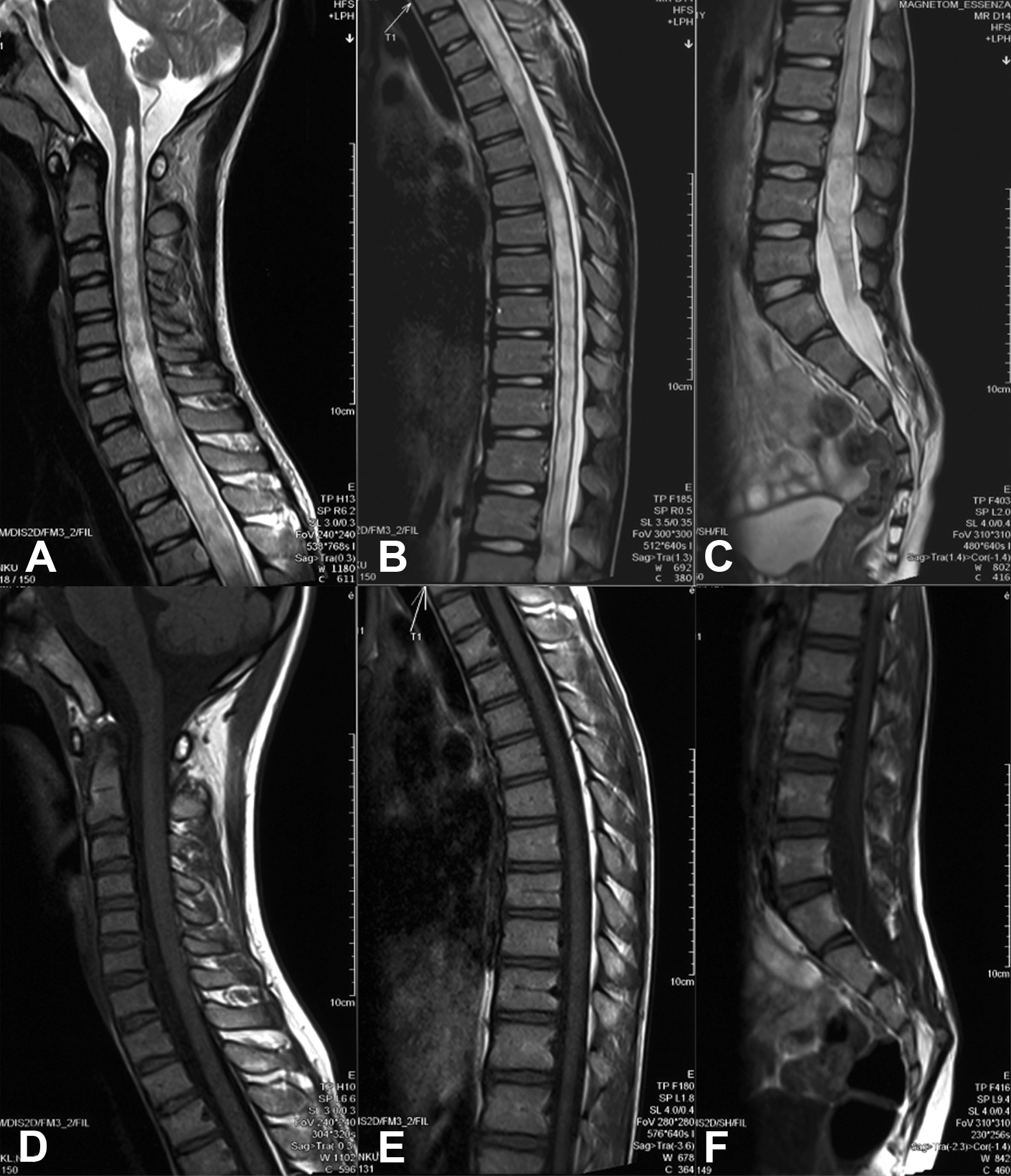
An 8-year-old male patient diagnosed with tethered cord syndrome combined with syringomyelia. Preoperative whole-spinal sagittal T2-weighted magnetic resonance imaging (MRI) (A, B, C) suggests syrinx cavities spanning the whole spinal cord and extending to the filum terminale. The 36-month-postoperative whole-spinal sagittal T1-weighted MRI (C, D, E) suggests a complete disappearance of cavities and a normalized spinal cord.
Discussion
In our study, we found that all patients obtained satisfactory clinical outcomes, with improved symptoms and a minimized or even disappeared syrinx cavity. Thus, terminal ventriculostomy not only detethered the spinal cord but also resulted in decompression of the syrinx cavity. Moreover, we made a short incision, of approximately 1.5–3 cm on the skin, which accelerated wound healing and resulted in little esthetic damage. Notably, to ensure persistent drainage, we produced an additional “V”-type fistula at the proximal end of the severed filum terminale. Our follow-up data also demonstrated no syringomyelia relapse. To the best of our knowledge, this is the first study to adopt “V”-type ostomy to ensure persistent drainage when operating terminal ventriculostomy.
TS most often occurs with TCS caused by a tight filum terminale. 9 The combined pathogenesis of these 2 deformities remains unclear. Pang inferred that both TCS and TS derived from defective retrogressive differentiation. 10 However, this “embryological theory” could not explain the relapse of syringomyelia after radical detethering. Ng et al. proposed that TS might develop secondary to TCS; in other words, a causal relationship underlies the combined pathogenesis. 9 During surgical procedures performed in the present study, we noted that all cases had a thickened filum terminale and that CSF effused from the fistula as soon as we cut off the filum terminale. Therefore, we propose that a thickened filum terminale might be involved in the pathogenesis, by stunting CSF flow in the central canal. Morbid CSF flow could increase the fluid pressure in the spinal subarachnoid and change the pressure gradient between the interior and the exterior of the spinal cord. With confined cycling, CSF would then accumulate in the spinal cord, and consequently, a syrinx cavity would develop and expand.
In 1977, Gardner et al. developed the novel technique of “terminal ventriculostomy” as a suitable therapy for syringomyelia. 3 However, its postoperative effectiveness remains controversial. Hsu et al. performed terminal ventriculostomy on a 3-year-old boy with TCS and thoracic syringomyelia and achieved cavity reduction postoperatively, but observed relapsing syringomyelia in a follow-up visit. 6 Emmez et al. reported a case in which a small terminal syrinx cavity at a lumbar segment extended to the entire spinal cord after terminal ventriculostomy. 5 Beaumont et al., however, indicated that terminal ventriculostomy could sufficiently improve TS without any additional bypass. 7 In 1983, Williams and Fahy presented a 31-case clinical study, in which patients with syringomyelia underwent terminal ventriculostomy. They found that terminal ventriculostomy may be useful for some patients whose syrinx cavities extended to the filum terminale. 4 Wilson et al. also proposed that this particular cavity could be the best indication for terminal ventriculostomy. 11 However, to date, no studies have validated this extrapolation.
All 28 patients undergoing terminal ventriculostomy in the present study had syrinx cavities extending to the filum terminale. The satisfactory outcomes—each syrinx cavity shrunk by at least 50% and more than 90% of the patients achieved “markedly improved” and “improved” status according to our evaluation system by the follow-up visit—validated the efficacy of terminal ventriculostomy. Even when syrinx invaded the whole spinal cord, it ended up in a complete resolution (Figure 3). Preoperative MRI and surgical course both revealed high tension and inner pressure in the syrinx cavities. We postulate that high inner pressure drives the hydrops to release into the spinal subarachnoid space through the fistula, which allows the terminal ventriculostomy to treat the cavity.
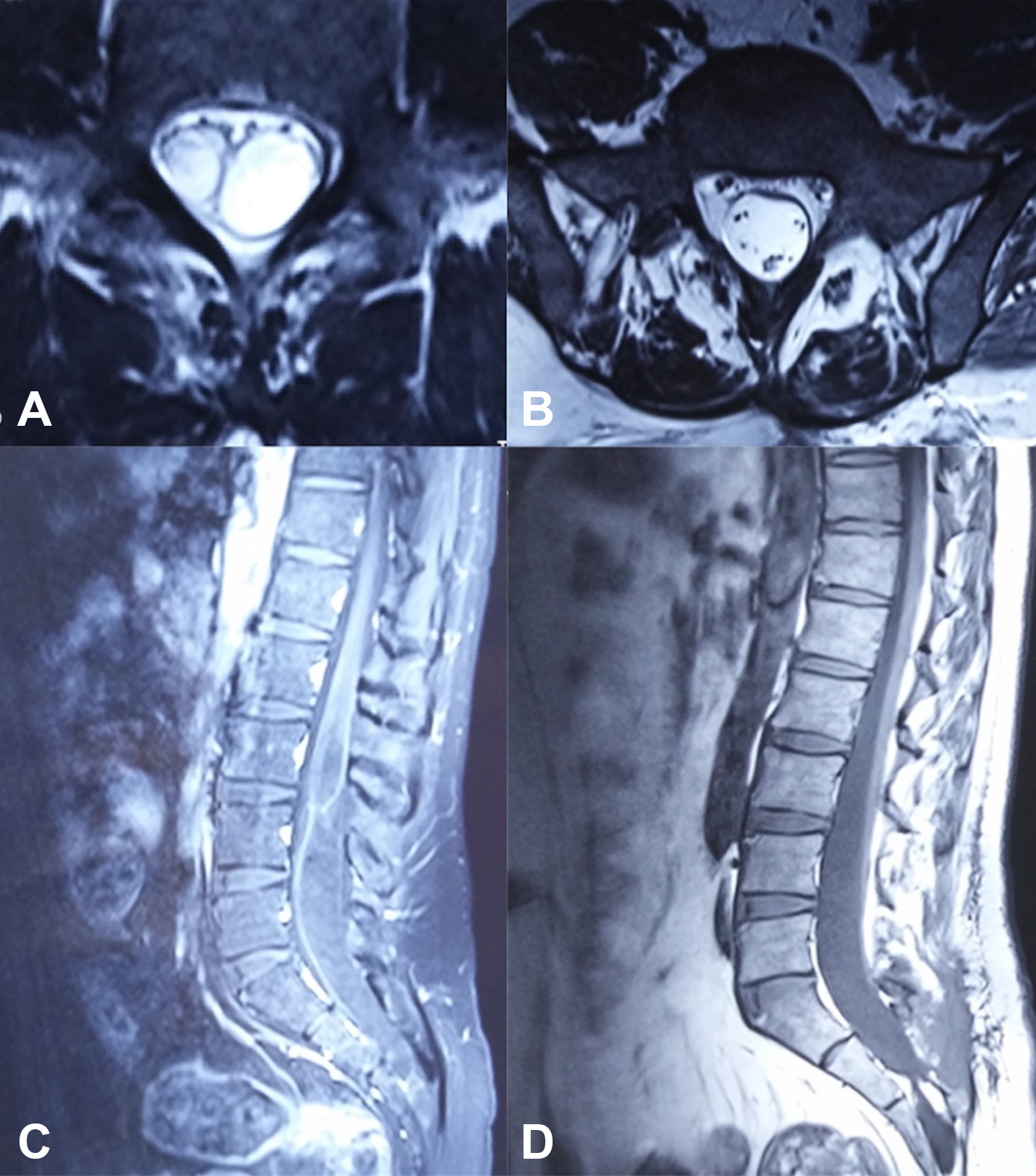
A 32-year-old male patient diagnosed with terminal syringomyelia associated with split cord malformation type I. Preoperative lumbosacral sagittal T1-weighted magnetic resonance imaging (MRI) (C) shows a terminal syrinx cavity extending to the filum terminale and a bony septum intersecting the lumbar spinal cord. Axial T2-weighted MRI (A) shows dual terminal syrinx cavities in the filum terminale. Twelve months after surgery, postoperative lumbosacral sagittal T1-weighted MRI (D) shows elimination of the bony septum and complete disappearance of the terminal syrinx cavity, while axial T2-weighted MRI (B) shows cord detethering and a normal cauda equina.
Still, many authors question the usefulness of terminal ventriculostomy, primarily because of the potential for relapsing syringomyelia. Williams et al. doubted that the possible reclosing fistula, as the result of terminal ventriculostomy, could promise further drainage of the syrinx cavity. 4 Hsu et al. and Emmez et al. also addressed this potential shortfall and attributed the relapse to it.5,6 We concede that this concern is reasonable. Due to the elasticity of tissues, fistula may reclose when the surrounding tissues contract after being detethered. To solve this potential problem, we optimized the method by creating a “V”-type ostomy. We designed this “V”-type ostomy to leave the fistula fragmentary tissue. Due to constant CSF flow, the 2 ends of the “V”-type fistula are not in contact. In this way, we reduced the risk of fistula reclosing and improved efficacy, as shown by our follow-up data, and no patients developed relapsing syringomyelia up to 36 months after surgery. Moreover, given the anatomical hallmarks of the filum terminale—no neurological function and no nerve origins—creating a “V”-type ostomy within the filum terminale is safe and benign. To avoid accidental neuronal damage (especially S1 and S2 nerves, which control the bladder and bowel function), we pinpointed the last pair of sacral nerves by performing intraoperative SSEP and MEP monitoring to detect neurological function, and then cut off the filum terminale below the final nerve roots. Taking both feasibility and safety into account, a “V”-type ostomy has a satisfactory long-term outcome based on terminal ventriculostomy.
Moreover, traditional surgery (syringosubarachnoid shunt placement, syringoperitoneal shunt placement, and myelotomy) requires laminotomy, with a long incision. Because we only worked on the filum terminale, a smaller operative field was sufficient. We made a short incision of 1.5–3 cm on the skin, which allowed rapid wound healing and resulted in less damage to appearance, which is useful for children’s and teenagers’ esthetic concerns.
We included a special case in our study: The patient, 32-year-old male, was diagnosed with TCS, TS, and SCM type I. He had muscle weakness in the lower extremities, sensory deficits, and foot deformities. Preoperative MRI demonstrated a bony septum intersecting the lumbar spinal cord and particularly, dual terminal syrinx cavities extending to the filum terminale (Figure 3). The bony septum divided the vicinal spinal cord, and each half of the spinal cord included a single cavity. Two individual cavities converged below the lower end of the bony septum and extended to the filum terminale. We performed SCM repair along with terminal ventriculostomy; subsequently, he was discharged 7 days later without any surgery-related complications. Three years later, the terminal syrinx cavity disappeared; his spinal cord returned to be normal; and his preoperative symptoms were resolved, except the foot deformities. Radiological and clinical evidence substantiated his remarkable improvements. We consider that opening of the central canal of the filum terminale has allowed drainage of the hydrops and decompression of the syrinx cavity. To date, there have been no reports of TS associated with SCM type I, which is rare but interesting.
This study had some limitations. We had a small sample size of 28 patients that might weaken the credibility and rationality of the present study. Since the cases of TCS combined with TS are rather rare in the clinical practice,12,13 more cases should be included in future studies. Additionally, we did not compare this approach with any conventional surgery on TS in which the syrinx cavity extends to the filum terminale, and we did not explore further surgical indications, such as whether our strategy is applicable in cases where the cavity does not invade the filum terminale. This warrants a more extensive and well-defined study in the future.
In conclusion, the effectiveness of terminal ventriculostomy is determined by 2 factors: communication between the syrinx cavity and the central canal in the filum terminale, and the persistence of drainage, which ensures a good outcome. Terminal ventriculostomy exerts a beneficial effect on TS, particularly when the syrinx cavity extends to the filum terminale. We advocate the use of a terminal ventriculostomy-associated “V”-type ostomy for this particular type of cavity to avoid potential relapse. This approach is safe, convenient, and persistently effective. Thus, terminal ventriculostomy-associated “V”-type ostomy can be considered as a promising alternative method for treating TCS with TS in clinical practice.
Footnotes
Authors’ Note
All data and materials supporting the findings of this study will be available from the corresponding author upon a reasonable request. All patients provided written informed consent. All patients provided written informed consent. This study was approved by the Institutional Review Board of PLA General Hospital (No. S2018-280-01) and followed the tenets of the Declaration of Helsinki.
Authors’ Contributions
Conceptualization: Mengchun Sun.
Methodology: Mengchun Sun, Benzhang Tao.
Formal analysis and investigation: Mengchun Sun, Benzhang Tao.
Writing—original draft preparation: Mengchun Sun.
Writing—review and editing: Mengchun Sun, Gan Gao.
Resources: Gan Gao, Shiqiang Li.
Supervision: Shiqiang Li, Teng Li.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Key Research and Development Program of China under Grant (No. 2018YFC1002500), the Science Foundation of Military Medical Research and Clinical Research Foundation of PLA General Hospital in China (No. 17KMM11); Supporting Project of Clinical Research of PLA General Hospital in China (No. 2017FC-TSYS-2026); and Application of Clinical Features of Capital City of Science and Technology Commission in China (No. Z171100001017140).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
