Abstract
Study Design:
Retrospective cohort study.
Objectives:
Lateral interbody fixation is being increasingly used for the correction of segmental sagittal parameters. One factor that affects postoperative correction is the resistance afforded by posterior hypertrophic facet joints in the degenerative lumbar spine. In this article, we describe a novel preoperative motion segment classification system to predict postoperative correction of segmental sagittal alignment after lateral lumbar interbody fusion.
Methods:
Preoperative computed tomography scans were analyzed for segmental facet osseous anatomy for all patients undergoing lateral lumbar interbody fusion at 3 institutions. Each facet was assigned a facet grade (min = 0, max = 2), and the sum of the bilateral facet grades was the final motion segment grade (MSG; min = 0, max = 4). Preoperative and postoperative segmental lordosis was measured on standing lateral radiographs. Postoperative segmental lordosis was also conveyed as a percentage of the implanted graft lordosis (%GL). Simple linear regression was conducted to predict the postoperative segmental %GL according to MSG.
Results:
A total of 36 patients with 59 operated levels were identified. There were 19 levels with MSG 0, 14 levels with MSG 1, 13 levels with MSG 2, 8 levels with MSG 3, and 5 levels with MSG 4. Mean %GL was 115%, 90%, 77%, 43%, and 5% for MSG 0 to 4, respectively. MSG significantly predicted postoperative %GL (P < .01). Each increase in MSG was associated with a 28% decrease in %GL.
Conclusions:
We propose a novel facet-based motion segment classification system that significantly predicted postoperative segmental lordosis after lateral lumbar interbody fusion.
Introduction
The surgical correction of spinal deformity is meant to restore spinal balance, relieve pain, obtain solid bony fusion, and, most important, improve patients’ quality of life. 1 –3 While a variety of techniques and technologies have been used to achieve these goals, lumbar interbody grafts are particularly useful tools to improve rates of arthrodesis, restore anterior column height, and assist in deformity correction. 1 Lumbar interbody fixation is most often placed via an anterior, posterior, or, more recently, via a minimally invasive lateral retroperitoneal approach. 4 The application of interbody grafts through the latter technique gives surgeons the ability to utilize the advantages of interbody fixation in the treatment of spinal deformity while reducing the morbidities typically associated with conventional anterior or posterior approaches. 5,6
Commonly referred to as extreme lateral interbody fusion (XLIF; NuVasive, Inc, San Diego, CA) or direct lateral interbody fusion (DLIF; Medtronic Sofamor Danek, Memphis, TN), use of this lateral transpsoas technique for lumbar interbody fusion has increased dramatically since it was first described in 2006, 7 and its application has been demonstrated for both adult degenerative disc disease as well as degenerative scoliosis. 1,8 –14 There is less tissue trauma, less blood loss, less postoperative pain, shorter hospital stays, and faster return to daily activities. 15 Because of these advantages, lateral interbody techniques are given great consideration when feasible for the correction of deformity parameters in degenerative lumbar scoliosis.
It has been shown that DLIF significantly increases the segmental sagittal Cobb angle at the level of instrumentation. 8 Correction of sagittal vertical axis yields clinical improvement across Oswestry Disability Index scores, Scoliosis Research Society assessments, and the Short Form 12 questionnaire, with the best outcomes for those patients with the greatest correction. 4,16 –18 Restoration of sagittal balance has been more closely associated with good outcomes than coronal plane correction; therefore, the importance of minimally invasive lateral interbody fusion to achieve sagittal plane correction is a critical goal to achieve good clinical effect for patient outcome. 8,17
The clinical use of either DLIF or XLIF as a surgical tool for the correction of spinal imbalance depends on the ability to plan, predict, and achieve postoperative correction in this plane. One factor that may affect the expected versus actual correction achieved by lateral interbody techniques is the resistance afforded by the posterior hypertrophic facets of the degenerative lumbar spine. The purpose of this study is to describe a novel preoperative motion segment classification system of these facet joints to assist in the prediction and planning of expected postoperative correction of segmental sagittal alignment at the level of lateral interbody correction.
Materials and Methods
All patients who had undergone minimally invasive lateral lumbar interbody fixation (LLIF) were retrospectively reviewed with institutional review board approval. Those patients with preoperative computed tomography (CT) scans were further reviewed for their segmental facet osseous anatomy. Any patient who had undergone posterior facetectomy or osteotomy was excluded from the study. Similarly, any patient who had undergone an anterior longitudinal ligament release during the lateral portion of the operative procedure was excluded as well.
Each level’s facets were assigned facet grades based on the preoperative CT scan (Figure 1). Grade 0 was given if there was no extension of the superior facet beyond a line drawn parallel to the posterior vertebral body wall (posterior vertebral body wall line [PVBWL]) at the posterior border of the inferior facet (posterior facet line [PFL]); Grade 1 was given if there was extension of the superior facet beyond the PFL but not beyond a line drawn perpendicular to the PVBWL at the midpoint of the inferior facet (mid-facet line [MFL]); and Grade 2 was given if there was extension of the superior facet beyond the MFL, or if there was facet ankylosis (Table 1). The final motion segment grade (MSG) was the sum of the bilateral facet grades (min = 0, max = 4).
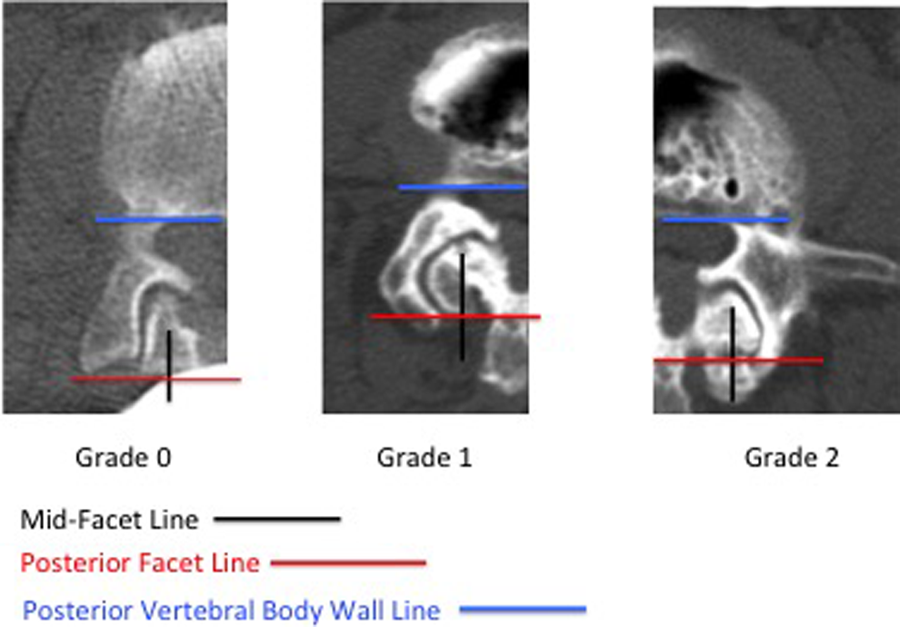
Individual facet grading system. The final motion segment grade (MSG) is the sum of the bilateral facet grades (min = 0, max = 4).
Individual Facet Grading System.

Abbreviations: PFL, posterior facet line; MFL, mid-facet line.
Preoperative and postoperative segmental lordosis was measured on standing lateral radiographs. Because implanted graft cages have different lordotic profiles as determined by surgeon choice and manufacturer, postoperative segmental lordosis was also conveyed as a percentage of the implanted graft lordosis (%GL). Simple linear regression was conducted to predict the postoperative segmental %GL according to MSG, and a post hoc Tukey’s test was used to compare differences in %GL associated with each grade. Wilcoxon signed-rank test was used to determine significant differences between preoperative and postoperative lordosis values, and a post hoc Dunn’s test compared pairs of groups for significance. All statistical analyses were conducted using STATA 13 (StataCorp, College Station, TX), and statistical significance was established if P < .05.
Results
A total of 36 patients with 59 operated levels (average = 1.6 levels per patient) were identified who had undergone minimally invasive LLIF at 3 institutions (Keck Medical Center of University of Southern California, Cedars-Sinai Medical Center, and Ronald Reagan University of California Los Angeles Medical Center). All patients underwent minimally invasive LLIF surgery by DLIF (Medtronic Sofamor Danek, Memphis, TN) or XLIF (NuVasive, Inc, San Diego, CA). Patients underwent treatment for spondylolisthesis, spondylosis, or degenerative scoliosis. All patients had either 10° or 12° lordotic grafts placed in the anterior half of the disc space.
Table 2 demonstrates the characteristics as separated by each operated disc interspace level. There were 4 operated levels at L1-2 (mean MSG 1.0), 20 operated levels at L2-3 (mean MSG 1.1), 25 operated levels at L3-4 (mean MSG 1.7), and 10 operated levels at L4-5 (mean MSG 1.5). Lordosis measurements were also separated by interspace levels at L1-2 (preoperative 2.8°, postoperative 11.5°), L2-3 (preoperative 2.9°, postoperative 10.4°), L3-4 (preoperative 2.6°, postoperative 11.7°), and L4-5 (preoperative 3.6°, postoperative 9.5°). The number of operated levels separated by MSG and disc interspace level is graphically represented in Figure 2. Mean %GL was 101%, 91%, 88%, and 86% for L1-2, L2-3, L3-4, and L4-5, respectively.
Motion Segment Grade by Disc Interspace Level.
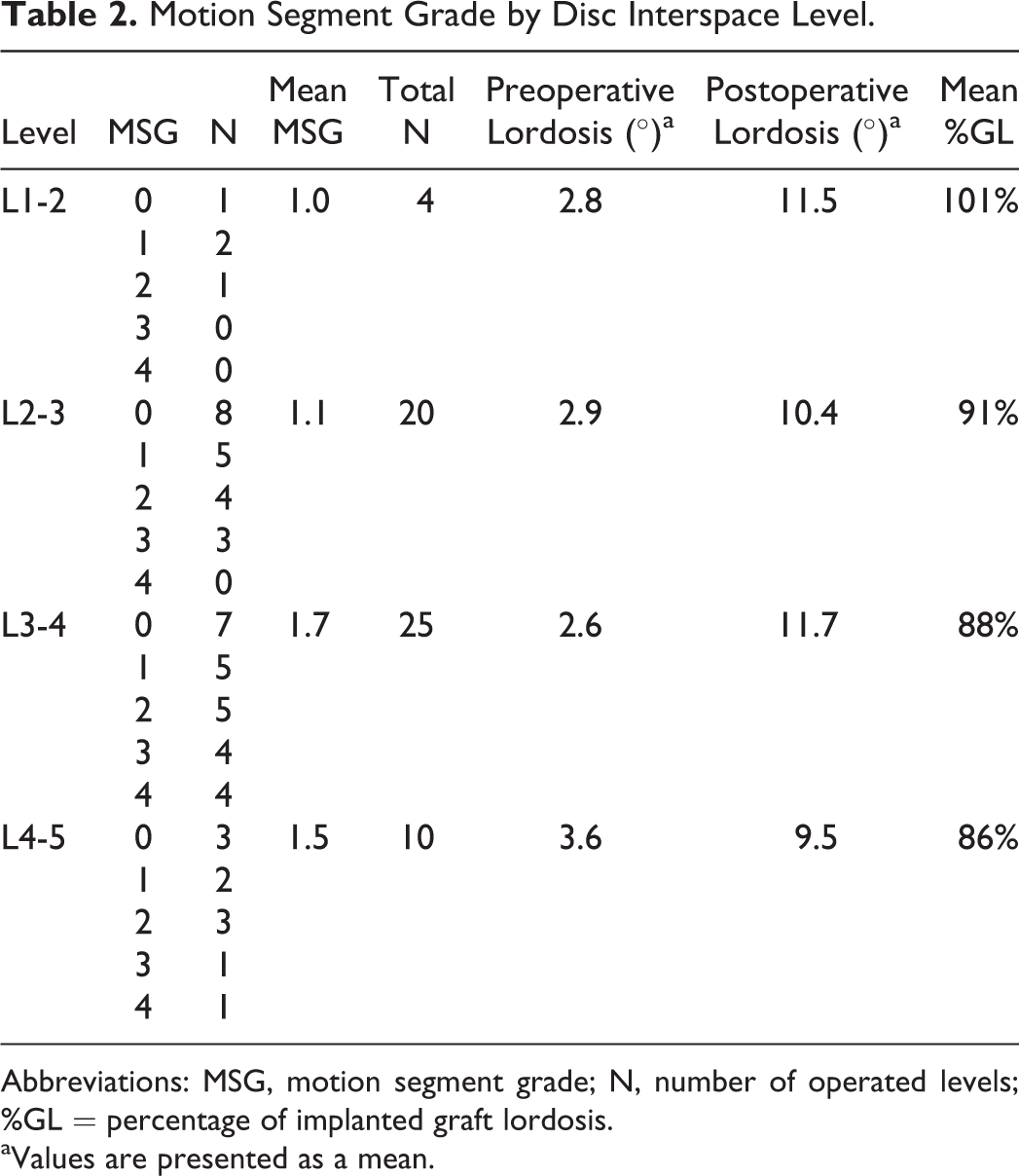
Abbreviations: MSG, motion segment grade; N, number of operated levels; %GL = percentage of implanted graft lordosis.
aValues are presented as a mean.
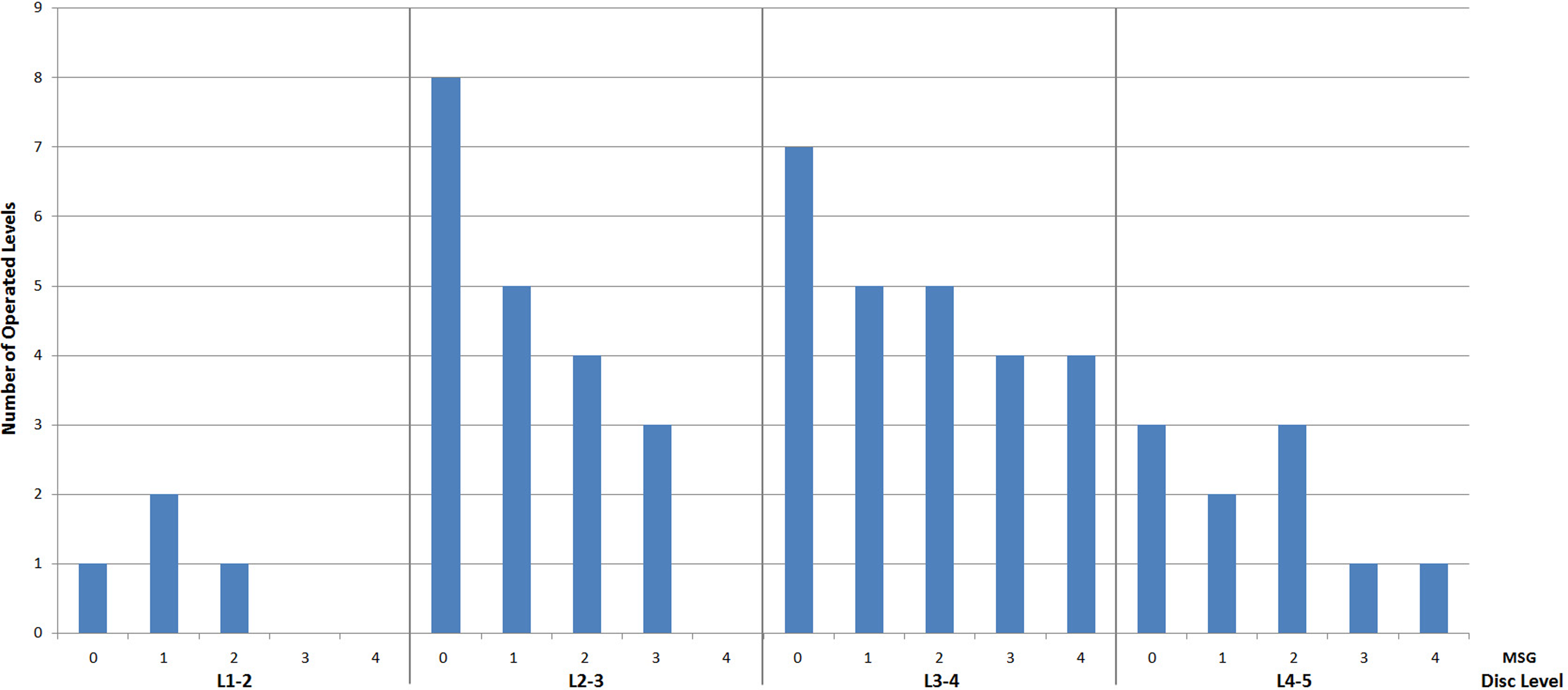
Number of operated levels separated by motion segment grade (MSG) and disc interspace level.
When separated out specifically for MSG, there were 19 levels with MSG 0, 14 levels with MSG 1, 13 levels with MSG 2, 8 levels with MSG 3, and 5 levels with MSG 4 (Table 3). Mean preoperative lordosis for all segments was 2.6° (range = 0° to 8°), and mean postoperative lordosis for all segments was 9.4° (range = 0° to 15°). The mean %GL for all segments was 82% (range = 0% to 130%). When separated out by MSG, mean preoperative lordosis was 3.3°, 2.6°, 3.1°, 2.0°, and 0.2° for MSG 0 to 4, respectively. Mean postoperative lordosis was 12.9°, 10.5°, 8.9°, 5.3°, and 0.6° for MSG 0 to4, respectively. Mean %GL was 115%, 90%, 77%, 43%, and 5% for MSG 0 to 4, respectively. Post hoc evaluation confirmed statistically significant differences between all MSG groups with regard to %GL (P < .01) with the exception of MSG 1 and 2 (P = .18).
Radiographic Data by Motion Segment Grade.
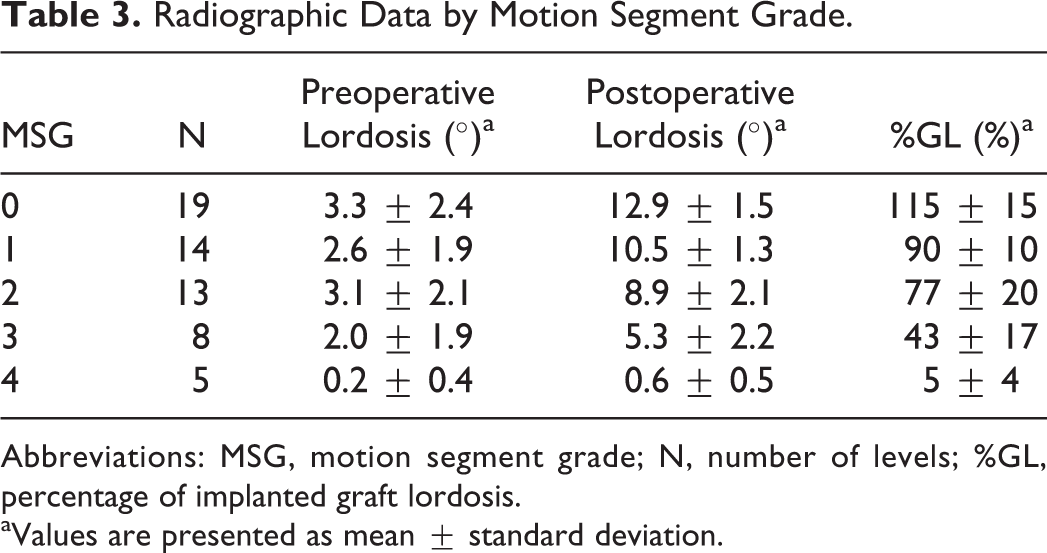
Abbreviations: MSG, motion segment grade; N, number of levels; %GL, percentage of implanted graft lordosis.
aValues are presented as mean ± standard deviation.
MSG significantly predicted postoperative %GL and explained 87.4% of the total variance in lordotic angulation, a significant proportion of variance (adjusted R 2 = 0.874, root mean square error = 15.761, F[1, 41] = 291.65, P < .01). Each increase in MSG was associated with a 28% decrease in %GL (Figure 3). Differences in preoperative lordosis values were not statistically significant except for MSG 4 when compared with all other groups (P < .01). Differences in postoperative lordosis was statistically significant between all MSG groups (P < .01) with the exception of MSG 1 and 2 (P = .19).
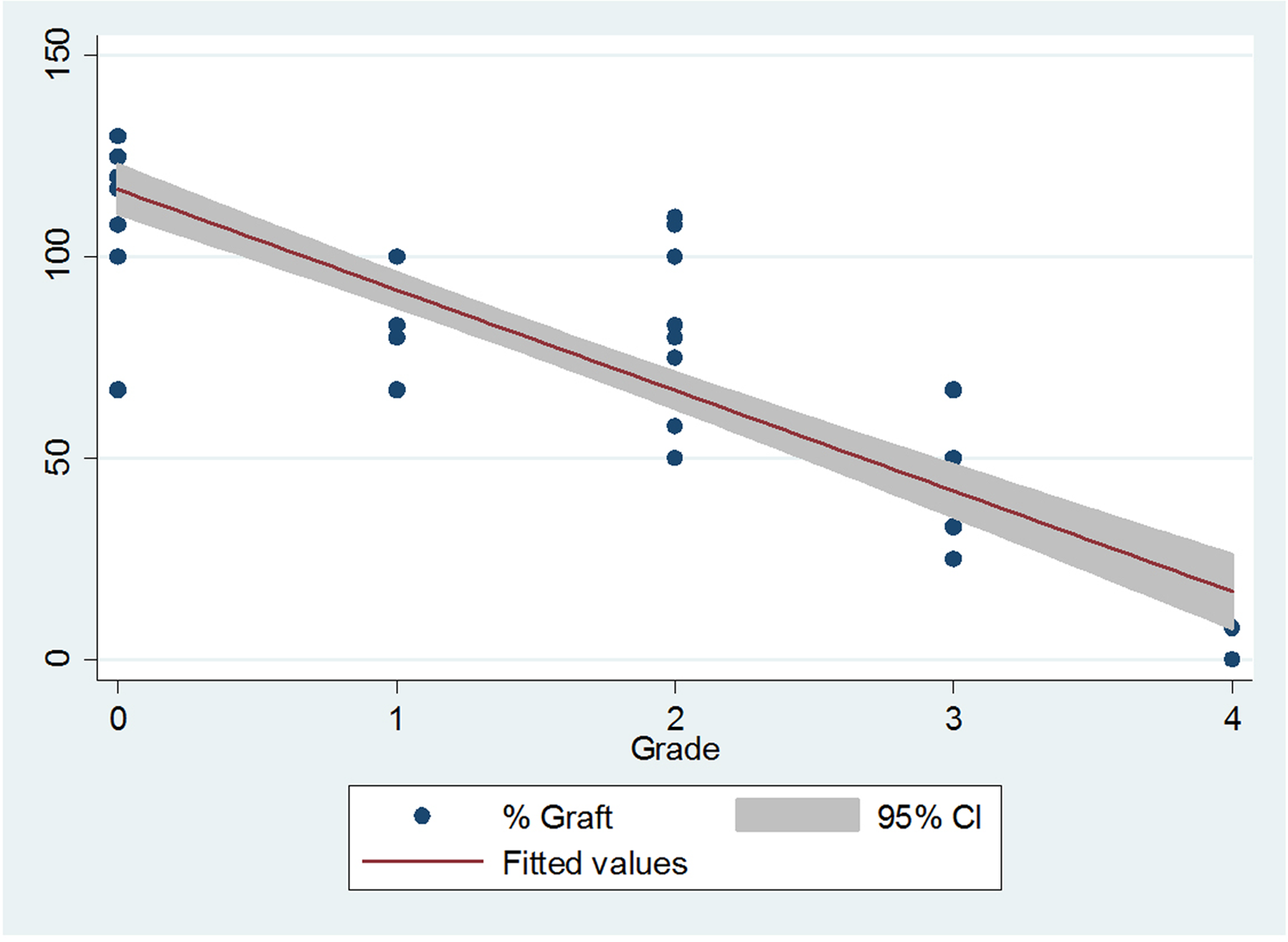
Linear regression model demonstrating how each increase in MSG is associated with a decrease in %GL.
Discussion
Lumbar degenerative disc disease and adult degenerative scoliosis can result in loss of sagittal and coronal balance, which are strong predictors of disability and quality-of-life parameters. 19 –21 Minimally invasive LLIF is an alternative treatment option to traditional open anterior or posterior procedures that allows relatively easier access to multiple levels from T11-L5. Injury to the abdominal viscera and retroperitoneal structures is largely avoided as compared with anterior lumbar interbody fusion (ALIF), and the risks of durotomy and nerve root injury are also reduced as compared with transforaminal and posterior lumbar interbody fusion (TLIF/PLIF). 22 –25 The lateral retroperitoneal surgical corridor of LLIF, however, does carry with it the risks consistent with transpsoas surgery that can result in groin pain, psoas mechanical deficits, or a lumbar plexus motor deficit. 26,27
Several studies have shown a positive postoperative correction in segmental lordosis after LLIF in the treatment of lumbar degenerative disease. Yson et al 28 and Kotwal et al 29 both found significant restoration of segmental lumbar lordosis after LLIF in 56 patients (88 levels) and 118 patients (237 levels), respectively. Other studies, however, have found significant changes in segmental lordosis after LLIF, but no significant change in regional lumbar lordosis or global sagittal alignment. Kepler et al found increases in segmental lordosis at instrumented levels, but no significant changes in overall lordosis in their review of 29 patients (67 levels). 30 Similarly, Johnson et al found postoperative correction in coronal alignment and segmental lumbar lordosis, but not regional lumbar lordosis or other spinopelvic indices (pelvic incidence, pelvic tilt, and sacral slope). 5 The senior author in this current study reviewed 36 patients who had undergone LLIF and also found improvements in segmental, regional, and global coronal alignment along with segmental lumbar lordosis, but LLIF had not improved regional lumbar lordosis or global sagittal alignment. 8 In contrast, Baghdadi et al reviewed their degenerative scoliosis patients who underwent LLIF ± ALIF and found that these patients had statistically significant improvement in lumbar lordosis, with a trend toward improved sagittal alignment that did not reach statistically significance. 4 These sagittal plane improvements were attributable to the LLIF levels and not the ALIF levels. Two recent systematic reviews of the literature on LLIF found significant changes in postoperative sagittal segmental lordosis but limited corrections of regional lumbar lordosis and sagittal alignment. 19,31 Both commented that LLIF appears to be effective if regional lumbar lordosis and sagittal balance correction goals are less than 10° and 5 cm, respectively.
Multiple technical factors have been investigated for the purposes of maximizing the amount of postoperative lordosis after graft placement. Kepler et al found that anterior cage placement resulted in the largest segmental lordosis gain (+7.4°) as compared to posterior cage placement which was prokyphotic (−1.2°). 30 Cage obliquity and height were not significant associated with lordosis change. Marchi et al pointed to the importance of protecting endplate integrity and reported in the cases of its violation that only 23% of the cage slope was reported on segmental lordosis measurements. 32
Because the instantaneous axis of rotation of the lumbar vertebra lies in the vicinity of the upper endplate of the next lower vertebra, changes in sagittal alignment will be influenced anatomically by both anterior and posterior structures. 33 Release of the anterior longitudinal ligament has been shown to provide more lumbar lordosis than lordotic cage placement alone. 34 We hypothesized that different characteristics of the posterior osseous facet anatomy also contributed a limiting factor that could be measured and classified.
The MSG quantifies the amount of degeneration and hypertrophy seen in the bilateral facets at the level of interest as a measurement of osteoarthritis. Our patient population was fairly homogenous with regard to their preoperative lordosis levels with the exception of MSG 4 patients who had statistically significant lower preoperative lordosis values. This is not surprising as MSG 4 patients likely had severe degenerative disc disease that would be consistent with advanced lumbar spondylosis, which would include our high grade facets. Similarly, it was noted that the lower disc space levels of L3-4 and L4-5 had higher mean MSG (1.7 and 1.5, respectively) as compared to L1-2 and L2-3 (1.0 and 1.1, respectively). This also likely represents the fact that the lower lumbar segments bear more axial load than the upper segments, manifesting as higher mean MSG. The only pairwise comparison that did not reveal any significant differences was MSG 1 and 2, which indicates that those grades may functionally respond similarly after LLIF in graft conformation. Using simple linear regression, we show here that the MSG significantly predicted segmental sagittal changes after LLIF as well as the extent to which the postoperative segmental lordosis matched the lordotic angle of the implanted graft. For every increase in MSG, there was an associated 28% decrease in %GL. This suggests that as facets become more hypertrophied and reach a higher grade on the MSG scale, they will increasingly prevent the disc space from opening enough to accommodate and conform to a lordotic LLIF graft. Thus, while magnetic resonance imaging and standing scoliosis radiographs are often sufficient to determine the indication and type of spinal reconstructive surgery necessary, this CT-based classification scheme of lumbar motion segments may assist surgeons in preoperative planning such that higher grade motion segments may require additional facetectomies or posterior column osteotomies rather than LLIF alone to enhance lordosis correction.
There are several limitations to this study. This analysis was not designed to investigate other variable factors that would affect postoperative lumbar lordosis. As such, it is possible that there may be other dependent variables that could have affected the surgical outcome in these patients. Future studies will need to examine MSG in relation to other potential dependent or independent factors. Patients in this review underwent preoperative CT scans at various time points prior to their index LLIF surgery. Depending on the length of time between CT scan and surgery, it is possible that facets may have further degenerated, affecting their true MSG at the time of intervention. Because several patients in this study presented with degenerative scoliosis, measurement of segmental lordosis on lateral radiographs may have been difficult due to the rotational curvature and lateral listhesis. This is also a fairly small review with a limited number of patients and operated levels. Additionally, lumbar lordosis should always be tailored to a patient’s particular pelvic morphology. However, as this study was intended only to analyze the effect of MSG on segmental lordosis after LLIF, we did not investigate the effect on regional lordosis nor did we analyze the final regional lordosis as it relates to pelvic morphology or global sagittal alignment. Nevertheless, these are important parameters to be taken into consideration during surgical planning. Last, this study is open to many of the biases inherent with all retrospective cohort studies.
Conclusion
LLIF remains a promising alternative for the minimally invasive treatment of lumbar degenerative disease including the treatment of degenerative scoliosis. We propose a novel facet-based motion segment classification system that significantly predicted postoperative segmental lordosis. Further studies will be needed to fully delineate the role of LLIF in the management of regional lumbar lordosis and global sagittal plane alignment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Acosta reports personal fees from NuVasive, outside the submitted work. Dr Hsieh reports personal fees from Depuy Synthes Spine, personal fees from Medtronic, and personal fees from Siemens, outside the submitted work. The remaining authors have no conflicts of interest to disclose.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
