Abstract
The regulatory framework that governs research involving human subjects in the United States was developed based on the ethical principles described in the Belmont Report. The principle of respect for persons demands that research consider an individual’s right to determine whether or not to participate in research. In the regulations, respect for persons is manifested in the informed consent requirements. However, the regulations do not provide clear direction for institutional review boards (IRBs) when presented with research that involves adult subjects who lack the capacity to give their informed consent. The regulations merely require that researchers obtain the legally effective informed consent of the subject or the subject’s legally authorized representative. To assist IRBs with their assessment of such research, we propose a framework for assessing the appropriateness of inclusion of such subjects through the application of a framework that takes into account the level of risk presented by the proposed research as well as the expected benefits that individual subjects are likely to accrue through their participation in the research. This framework takes into account the principle of respect for persons as well as the principles of beneficence and justice.
One of the most difficult decisions in research protocol design and ethics review is determining when it is appropriate to allow adults who lack the capacity to provide legally effective informed consent for themselves (hereafter referred to as “adults lacking capacity”) to participate in research. This article discusses the application of the Belmont Report and the US Food and Drug Administration (FDA) human subject protection regulations to this issue, provides examples of research where the inclusion of adults lacking capacity is appropriate or not, and proposes categories of research that allow institutional review boards (IRBs) to make appropriate decisions on this issue.
Some research will require the participation of adults lacking the capacity to provide legally effective informed consent. The need to include such subjects can be derived from the need to obtain knowledge about conditions that affect their population, such as research on Alzheimer’s disease or stroke, or when there are direct benefits to individual subjects that can only be obtained through participation in research. However, there is also a spectrum of research activities that neither automatically excludes these individuals nor compels their inclusion.
The Belmont Report provides the moral framework for the human subjects regulations that IRBs must apply to proposed research. 1 Determining whether to include adults lacking capacity in a protocol involves the 3 principles of research ethics that are described in the Belmont Report: beneficence, justice, and respect for persons. Respect for persons recognizes the autonomy of the individual and requires informed consent as a vehicle for exercising autonomy. When there are vulnerable subjects who are unable to fully exercise their autonomy, respect for persons requires consideration of additional protections. The principle of beneficence requires that there be an appropriate balance of risk to the subjects against the benefit to the individual subjects and the knowledge that results from the research. Justice requires that there be equitable selection of subjects in order to avoid exploitation of vulnerable populations. When considering vulnerable groups such as adults lacking capacity, there must be an appropriate balancing of the demands of the ethical principles. The protection of such subjects may be achieved through exclusion from the research, or augmented consent processes that ensure that permission is sought from an individual speaking on behalf of the subject coupled with an attempt to obtain the subjects’ assent, when possible.
From a regulatory perspective, the issues involve minimization of risk and appropriate risk-benefit relationship under 21 CFR 56.111(a)(1) and (a)(2), equitable selection of subjects under 21 CFR 56.111(a)(3), protection of vulnerable subjects under 21 CFR 56.111(b), and informed consent under 21 CFR 50.52. However, the regulations do not offer a definition of vulnerability, and instead provide examples that include “children, prisoners, pregnant women, handicapped, or mentally disabled persons, or economically or educationally disadvantaged persons.” Within the FDA’s regulatory framework, the only category of vulnerable subjects that are provided with explicit additional regulatory protections, known as the Subpart D regulations, are children. 2 The Subpart D regulations impose additional requirements on IRBs that restrict their inclusion in research following a framework that is tiered based on the degree of risk and possibility of benefit. Unlike research involving children, there are no additional regulatory protections in place to protect adults lacking capacity to consent to research. IRBs must therefore determine on their own when participation of this population is acceptable.
For certain categories of research, there is agreement that it is clearly appropriate to enroll adults lacking capacity. One such category is minimal risk research. In human subjects research regulations, research is considered to be minimal when “the probability and magnitude of harm or discomfort anticipated in the research are not greater in and of themselves than those ordinarily encountered in daily life or during the performance of routine physical or psychological examinations or tests.” 3 Provided there are adequate provisions for consent and assent, it is appropriate to enroll adults lacking capacity into research involving minimal risk because the regulatory criteria above are satisfied when research is minimal risk. Minimal risk stands alone from the other types of research described below that involve more than minimal risk, based solely on the level of risk.
For research that involves more than minimal risk, there is a spectrum of types of research studies, from studies where it is clearly inappropriate to enroll adults lacking capacity to studies where it is clearly appropriate (Figure 1). The decision is straightforward at each end of the spectrum. At one end of the spectrum, there is research that has more than minimal risks to participants, but from which the population with impaired decision-making capacity will receive neither direct nor indirect benefits. For example, there should be agreement that it is inappropriate to include women with impaired decision-making capacity in research on in vitro fertilization. In vitro fertilization is not clinically indicated for women lacking capacity. A little further down the spectrum, there is widespread agreement that it is usually not necessary or appropriate to include adults lacking capacity in research involving chronic diseases with large, capable populations and a relatively low incidence of subjects without capacity. Examples include obesity, diabetes, arthritis, blindness, and psoriasis. In these circumstances, it is not necessary or appropriate to include adults lacking capacity because it violates the principle of justice by burdening a vulnerable population when their exclusion does not deprive the vulnerable population of the societal benefit of the research.
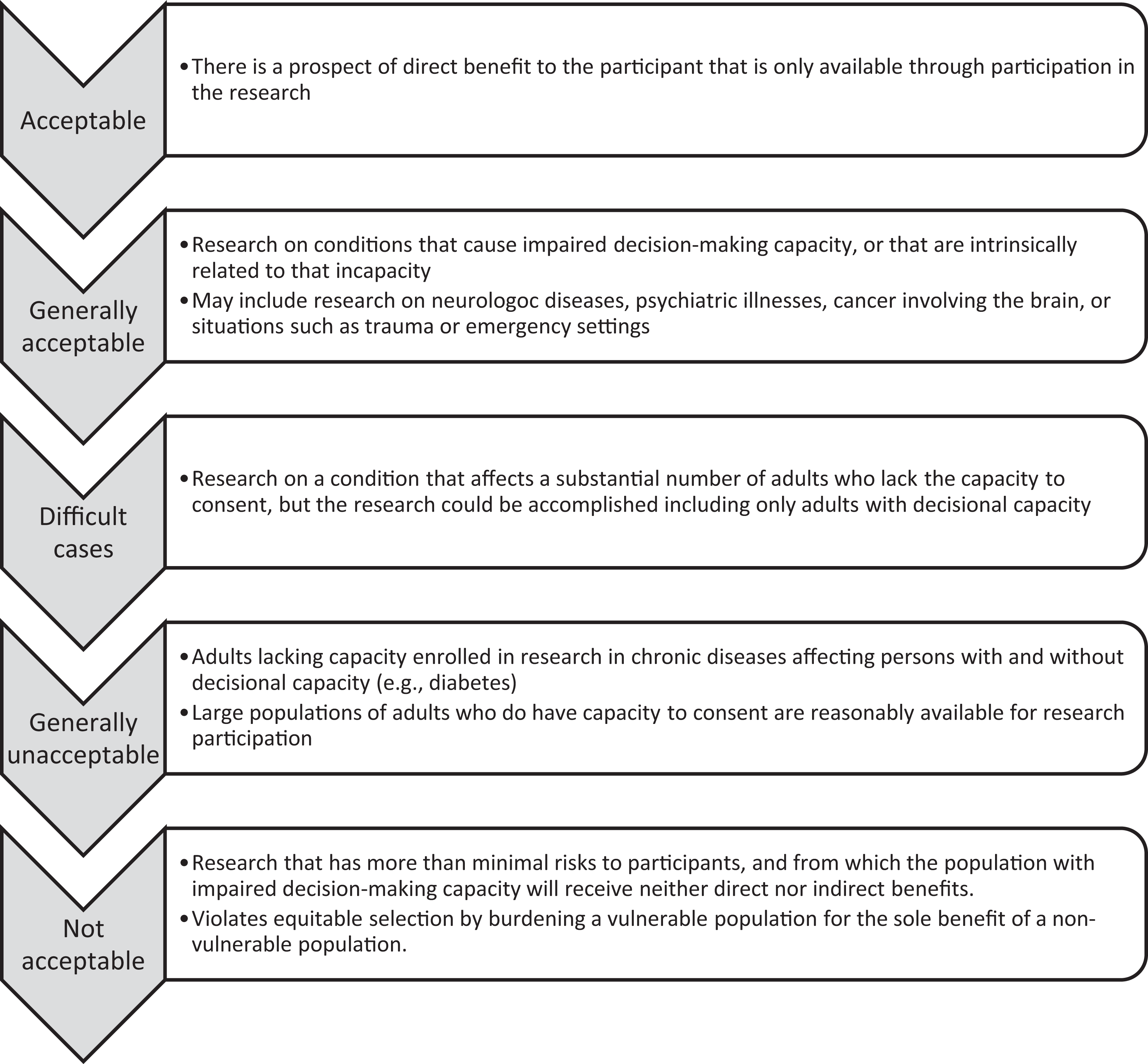
Decisional Impairment Research Spectrum.
At the other end of the spectrum, there is agreement that it is appropriate to include adults lacking capacity when there is a prospect of direct benefit that is available only in the research based on the direct clinical benefit to the subject. Likewise, there is agreement that it is appropriate and necessary to include adults lacking capacity in research on diseases and conditions that cause such incapacity or are intrinsically related to incapacity. Examples include Alzheimer’s disease, stroke, schizophrenia, and research in emergency situations such as massive trauma.
However, at the center of this spectrum, the decision is most difficult. The goal of this article is to provide a framework of criteria for evaluating whether the inclusion of adults lacking capacity in research is appropriate. We believe that this framework could be applied to the majority of diseases and conditions that are experienced by individuals lacking capacity, including cancer, Alzheimer’s and other neurologic diseases, and genetic diseases.
We propose 4 categories of research that could involve adults with impaired decision-making capacity (Figure 2). Our proposed categories follow the framework similar to that employed by the FDA for determining when research involving children is permissible. This includes measures for obtaining the assent of the potential subject, when in the judgment of the IRB the subject is capable of providing assent, taking into account the maturity, and psychological state and legal status of the individual.

Four Categories of Research Involving Adults with Impaired Decision-making Capacity.
Category 1 is research involving no more than minimal risk to subjects.
Category 2 is research with prospect of direct benefit to the individual subjects, and the subject population includes both adults with capacity and adults lacking capacity to consent.
Category 3 is research with a prospect of direct benefit to the individual subject where the subject population by scientific necessity requires the inclusion of adults lacking capacity to consent.
Category 4 is research that holds out a prospect of direct benefit not available outside the research context.
The criteria for each of these categories of research are as follows. For the purposes of brevity, the assent criteria will only be provided in full for category 1, with a place holder noting their applicability in categories 2, 3, and 4.
The goal of this framework is to better parse out the categories of research so that the research at the ends of the spectrum, in category 1 and categories 3-4, is easy to categorize. The difficult cases will tend to fall into category 2. Of course, as appropriate, an IRB can apply component analysis and assign different categories for different arms of the research in order to clearly think through the justice and risk-benefit issues. As a result, different categories might be applied to the same protocol, as is often done when assessing research with children.
There are 4 criteria in category 2: research with prospect of direct benefit to the individual subjects, and the subject population includes both adults with capacity and adults lacking capacity to consent. The research holds out the prospect of direct benefit for the individual subject. The relation of the anticipated benefit to the risk is at least as favorable to the subjects as that presented by available alternative approaches. The inclusion of adults unable to consent is equitable, taking into account the prevalence of adults unable to consent in the research population and the importance of the knowledge to adults with cognitive impairment. There are adequate provisions for soliciting the permission of an LAR and assent of subjects.
The first 2 criteria are required to meet the regulatory criteria relating to risk and benefit (21 CFR 56.111(a) (1) and (a)(2)) and the fourth criterion is required to meet the criterion related to consent (21 CFR 50.52). The challenging criterion, and the one that will cause debate and controversy, is criterion 3. When the regulatory criteria relating to risk and benefit and consent are met, inclusion of adults lacking capacity can be appropriate given the setting (adults without capacity are a meaningful part of the subject population) and the purpose of the research (important to adults without capacity as a distinct population). An example is the inclusion of adults lacking capacity in a study of a new antibiotic intended for use in an intensive care unit (ICU) setting. These judgments are subjective, but we believe that they can provide useful criteria for distinguishing which studies should include subjects unable to consent.
There is one criterion that was not included in category 3, the criterion that the objectives cannot be met with research involving subjects who can give consent personally. The reason was that it became the rule that consumed the other criteria. It needs to be weighed against the other criteria, but not serve as an absolute threshold. One way to provide a contrast regarding this criterion is to compare it to the inclusion of children in research. Sometimes research studies include children and adults contemporaneously because the disease or condition affects both populations equally. When reviewing such research, IRBs use the categories in Subpart D to consider whether the inclusion of children is appropriate. One result of using the criterion that the objectives cannot be met with research involving subjects who can give consent personally is that the research can be approved for the inclusion of children but not for the inclusion of incapable adults. It is arguably inappropriate to be more conservative regarding the inclusion of adults lacking capacity than regarding the inclusion of children.
A final factor is that there are many causes of impaired decision-making capacity in adults. One population is adults who have never had capacity and now are suffering a disease, including groups such as Down syndrome. However, in contrast, numerically the bulk of adults lacking capacity are individuals who had capacity as adults and subsequently lost that capacity for medical reasons, and this is a diverse group. Representative populations include individuals in an ICU who are expected to regain capacity in a few weeks if their disease resolves, patients with psychiatric illnesses, patients with Alzheimer’s, those who have had strokes, and those in a persistent vegetative state. Unfortunately, at the time of IRB review it is usually not known which type of subject without capacity might end up under the care of an investigator doing research. For diseases where there is often incapacity and the incapacity might resolve if the underlying disease is cured, like brain cancer, it will often be appropriate to include adults without capacity.
Conclusion
The Belmont Report and the FDA regulations require that IRBs carefully consider if the inclusion of individuals lacking capacity to give consent in research is appropriate and if so what additional protections may be needed to offset the limitations on autonomy. At the same time, the regulations do not offer an explicit framework that IRBs can apply when making such determinations. In the absence of clear regulatory requirements, IRBs may consider developing a framework for working through these questions.
Footnotes
Declaration of Conflicting Interests
No potential conflicts were declared.
Funding
No financial support of the research, authorship, and/or publication of this article was declared.
