Abstract
Understanding the development of depression can inform etiology and prevention/intervention. Maternal depression and maladaptive patterns of temperament (e.g., low positive emotionality [PE] or high negative emotionality, especially sadness) are known to predict depression. Although it is unclear how these risks cause depression, altered functional connectivity (FC) during negative-emotion processing may play an important role. We investigated whether maternal depression and age-3 emotionality predicted FC during negative mood reactivity in never-depressed preadolescents and whether these relationships were augmented by early-life stress. Maternal depression predicted decreased medial prefrontal cortex (mPFC)–amygdala and mPFC–insula FC but increased mPFC–posterior cingulate cortex (PCC) FC. PE predicted increased dorsolateral prefrontal cortex–amygdala FC, whereas sadness predicted increased PCC-based FC in insula, orbitofrontal cortex, and anterior cingulate cortex (ACC). Sadness was more strongly associated with PCC–insula and PCC–ACC FC as early stress increased. Findings indicate that early depression risks may be mediated by FC underlying negative-emotion processing.
Depression is an etiologically complex mental disorder that develops via an array of vulnerabilities and pathways. Understanding these vulnerabilities is imperative for informing both etiology and prevention/intervention efforts. Although depression most commonly onsets in adolescence and early adulthood, markers of risk are identifiable during the first years of life (Birmaher et al., 1996). Specifically, children with a maternal history of depression and maladaptive patterns of temperament (e.g., low positive emotionality [PE] or high negative emotionality [NE]; Watson et al., 1988) and children who have been exposed to early-life stress are at elevated risk for depression (Klein & Finsaas, 2017). However, the mechanisms by which these early risks lead to depression are not fully understood. Maladaptive patterns of brain connectivity, particularly during negative mood states, may serve as an important pathway to depression given that such patterns mark difficulties in down-regulating or recovering from negative mood (Ho et al., 2014, 2015; Perlman et al., 2012). This possibility would be consistent with the notion that alterations in brain activity during negative-emotion processing index ruminative cognitive styles (Berman et al., 2011; Mandell et al., 2014), a “depressogenic” cognitive style characterized by passive and mostly self-referential reflection on feelings of distress and possible causes and consequences of one’s dysphoria (Nolen-Hoeksema et al., 2008). Building on this literature, in this study, we aim to examine whether children with early depression risks (e.g., maternal depression, maladaptive patterns of temperament, exposure to early-life stress) show distinct patterns of brain connectivity during induced negative-emotion reactivity.
Functional Connectivity in the Context of Depression
Functional connectivity (FC) refers to the temporal correlation, derived from functional MRI (fMRI), between the blood-oxygen-level-dependent (BOLD) time series extracted from two different brain structures (van den Heuvel & Hulshoff Pol, 2010). FC reflects the functional coupling between brain regions during a resting state or a task, and greater absolute values of the correlation (i.e., closer to one) indicate stronger coupling over time. In depressed individuals, the literature has identified altered FC patterns across distributed brain networks during negative-emotion reactivity and regulation; the default mode network (DMN) and fronto-limbic network have emerged as the networks most clearly implicated in these processes (e.g., Carballedo et al., 2011; Ho et al., 2014, 2015; Perlman et al., 2012).
The DMN comprises regions anchored along the cortical midline, spanning from anterior regions, such as the medial prefrontal cortex (mPFC), to posterior regions, such as the posterior cingulate cortex (PCC; Buckner et al., 2008; Mason et al., 2007). The mPFC and PCC are hypothesized to be involved in introspective processes, including the representation, evaluation, and processing of self-relevant stimuli (Northoff et al., 2006). The PCC is also thought to support adaptive switching between internally and externally oriented attention (Brewer et al., 2013). Nondepressed individuals typically show activation across DMN regions during resting or introspective states and deactivation of DMN regions during nonintrospective tasks (Buckner et al., 2008). Depressed youths and adults, however, often show ineffective suppression of DMN activation during nonintrospective affective processing, or heightened FC between DMN regions during rest (for meta-analyses, see Hamilton et al., 2015; Kaiser et al., 2015). Increased activation or connectivity within DMN may reflect excessive self-focus or ruminative cognitive styles in depressed individuals (Hamilton et al., 2015).
The fronto-limbic network is known to mediate emotion reactivity and emotion regulation, processes that go awry in youths at risk for depression (Helm et al., 2018). Among the limbic regions, insula is thought to support salience detection and monitoring, and amygdala subserves immediate responses to emotionally arousing stimuli (Iordan et al., 2013; Leppänen, 2006). Within the prefrontal cortex (PFC), the mPFC is hypothesized to support emotion evaluation and context-appropriate emotion expression by integrating limbic information (Etkin et al., 2011); lateral PFC down-regulates amygdalar reactivity to emotional cues, thereby maintaining goal-directed cognitive processes (Iordan et al., 2013; Wager et al., 2008).
Altered PFC-limbic connectivity has been documented in depressed youths (Kerestes et al., 2014) and adults (Helm et al., 2018) in both resting and task-modulated states, albeit with inconsistencies that may be associated with participant characteristics (e.g., age, severity and duration of depression, medication) and/or methodological differences. These inconsistencies indicate the need for further study of the role of fronto-limbic FC in depressive psychopathology. Note that heightened negative-emotion reactivity and impaired regulation of negative emotions are core features of depression and may play an etiological role in depression (Joormann & Stanton, 2016; Kovacs et al., 2008). Examining FC using paradigms that elicit negative-emotion reactivity may yield important information concerning the neural substrates of a key pathway to depression.
A relevant literature has investigated FC during emotion processing in clinically depressed individuals. Compared with control participants, depressed adults showed decreased PFC-amygdala FC in response to unpleasant pictures (Friedel et al., 2009) and negative faces (Carballedo et al., 2011; Kong et al., 2013) and decreased cortical-limbic FC while viewing emotional pictures (Anand et al., 2005). Likewise, depressed teenagers showed decreased mPFC-amygdala FC when they were viewing negative pictures while attending to their emotional responses (Perlman et al., 2012). During an emotional face-identification task, depressed adolescents demonstrated heightened FC between DMN structures (e.g., mPFC–precuneus, PCC–precuneus), although they did not differ from control participants in resting-state FC (Ho et al., 2015). Altered FC has also been found in depressed adolescents during implicit processing of fearful faces such that they showed increased anterior cingulate cortex (ACC)–amygdala FC but decreased FC of ACC–precuneus and ACC–insula (Ho et al., 2014).
These past studies have focused on adults and youths who were currently (or previously) depressed; hence, findings from these samples cannot speak to whether the observed FC alterations are trait markers of an early predisposition to depression or state markers of depressive disorder. Studies of high-risk youths without diagnosed depression are an important complement to the extant literature. Furthermore, most past work has used tasks that involve processing of emotional pictures (faces or scenes) or words; it is unclear whether these stimuli reliably induce negative emotion, which raises the possibility that such paradigms fail to tap the neural processes associated with negative-emotion reactivity and regulation (Isaacowitz & Stanley, 2011; Parsons, 2015).
Studies of Brain Function in Individuals at High Risk for Depression
Maternal depression
Mothers’ lifetime history of depression increases the risk of depression in offspring from threefold to fivefold (Gotlib & Colich, 2014; Williamson et al., 2004). Nondepressed youths with maternal depression exhibited heightened resting-state FC between DMN structures (e.g., PCC or mPFC) and orbitofrontal cortex (OFC) compared with low-risk control participants (Chai et al., 2016). Youths with parental depression showed heightened neural responses to fearful faces in amygdala and PCC but decreased responses to happy faces in ACC and nucleus accumbens (Chai et al., 2015; Monk et al., 2008). Our group recently found that preadolescents with maternal depression showed heightened activation in the ventrolateral PFC and mPFC when processing positive self-descriptive information (Liu et al., 2020). While watching a sad movie clip, daughters of depressed mothers showed heightened amygdalar reactivity; during subsequent mood regulation, however, they showed decreased activation in regulatory regions including the dorsolateral PFC (dlPFC; Joormann et al., 2012). Note that all of these past studies focused on resting-state FC or activation analysis and that none explored FC modulated by a negative-emotion-reactivity paradigm.
Temperamental emotionality
Temperamental emotionality refers to individual differences in emotional predispositions that are biologically based, early emerging, and relatively stable (Rothbart & Bates, 2006). Lower PE during early childhood prospectively predicts depressive symptoms in late childhood (for reviews, see Klein & Finsaas, 2017; Kujawa & Burkhouse, 2017). Relative to PE, NE is more heterogeneous and has distinct facets of NE that are differentially associated with different forms of psychopathology. For example, temperamental sadness is more strongly associated with depression, whereas fearfulness is more closely related to anxiety (Aktar et al., 2016; Kryski et al., 2018; Rapee & Coplan, 2010).
A small literature has reported associations between temperamental emotionality and children’s neural functioning. Higher PE at age 3 predicts greater electrophysiological activity when children won monetary credits in a rewarding task at age 9 (Kujawa et al., 2015). NE observed at age 3 predicts greater amygdalar and fusiform activation but decreased amygdala–fusiform FC in response to emotional faces around age 11 (Kann et al., 2017). However, none of the past studies explored whether emotionality predicts FC in the context of negative-emotion reactivity. Compared with fearfulness (Pérez-Edgar et al., 2007; Roy et al., 2014), less work has been done on sadness, which is problematic given its relatively important role in the development of depression.
Moderation of risk by early chronic stress
According to diathesis-stress theories, associations between early vulnerabilities and maladaptive outcomes are strengthened by exposure to life stress (Ingram & Luxton, 2005; Monroe & Simons, 1991). For example, whereas emotion-evoked amygdalar reactivity typically decreases with age in adolescents, adolescents with familial depression showed increased emotion-elicited amygdalar reactivity over time; this pattern was accelerated by stressful life events such that youths with both familial depression and exposure to life stress exhibited faster age-related increases in amygdalar reactivity (Swartz et al., 2014). Note that Swartz et al. (2014) examined recent life stress that occurred within the past year; it is unclear how exposure to chronic stress during early childhood predicts brain function in preadolescence. Given the greater neuroplasticity and vulnerability during early development, it is important to explore potential interactions between individual differences in depression vulnerability and early-life stress in predicting later brain functions subserving negative-emotion reactivity.
The Present Study
The high-risk studies reviewed above have largely focused on resting-state FC or regional activation in processing emotional stimuli (typically pictures) as the outcome measure. Many of these studies examined single, rather than multiple, risk factors despite the etiological heterogeneity of depression, and most studies did not investigate the moderating role of early chronic stress in predicting later FC underlying negative-emotion processing. We aimed to fill these gaps in this study by investigating whether maternal history of depression and early temperament (PE and sadness) predict later FC underlying negative-emotion reactivity in preadolescence. We also examined whether associations between these risk factors and later FC were moderated by youths’ exposure to early-life stress. To these aims, we recruited a group of never-depressed youths oversampled with depression risk to enhance our power to detect neurobiological markers of risk that are present before the onset of clinical depression. In tapping negative-emotion reactivity, we used a negative-mood-induction procedure to elicit negative mood and capture individual differences during this process.
Eighty-seven never-depressed youths around age 11 were drawn from an existing longitudinal cohort that had participated in laboratory tasks tapping child temperament at age 3 and interviews assessing maternal psychopathology and early chronic stress in the home. Around age 11, youths completed a negative-mood-induction procedure in the MRI scanner by watching a sad movie clip, a standard paradigm that taps negative-emotion reactivity. They also contributed resting-state fMRI data, which were used as a comparison condition in data analysis. Given the literature, we focused on 13 a priori regions of interest (ROIs) selected from the DMN (mPFC, ACC, PCC) and fronto-limbic system (left and right areas of dlPFC, OFC, amygdala, insula, and hippocampus), all of which are critical for depression risks and emotion processing. For FC analysis, we took an ROI-to-ROI approach by computing correlation coefficients of BOLD time series between a priori ROIs (see details in the Method section), which were then treated as dependent variables in multiple regressions with early risks as predictors. Given previous findings (e.g., Ho et al., 2014, 2015; Perlman et al., 2012), we predicted that early depression risk would be associated with maladaptive alterations of FC during negative mood induction (e.g., heightened FC within DMN; weaker FC between PFC and limbic regions) but not during the resting state. We also expected that exposure to early chronic stress would strengthen associations between maternal risk/temperament and FC.
Method
Participants and procedure
Data were drawn from an ongoing longitudinal study that began at child age 3, when 409 typically developing children (201 boys; mean age = 3.43 years, SD = 0.30) and their primary caregivers (93% mothers) were recruited from the community. Children with major medical or psychological problems were excluded, and normative cognitive development was verified by the Peabody Picture Vocabulary Test (Dunn & Dunn, 2007). Children were mostly White (93.4%), and 50.4% of the families were middle class and had an annual income between $40,001 and $100,000. At age 3, child PE and sadness were assessed by laboratory measures. Early-life stress was assessed at child age 3, age 4 (M = 4.73 years, SD = 0.38), and age 5 (M = 5.93 years, SD = 0.31) via family income, marital satisfaction, and structured interview with the primary caregiver.
Around age 11, 87 children (49 boys; M = 11.09 years, SD = 0.66) were recruited for MRI assessment. On the basis of the mother’s psychiatric history, 29 (17 boys) were identified as high risk, and 58 children (32 boys) were identified as low risk. Mothers were interviewed by trained clinical graduate students for lifetime psychopathology using the Structured Clinical Interview for the DSM-IV-TR Axis I Disorders Non-Patient Edition (First et al., 1997). Inter-interviewer reliability was examined in 10 participants and showed good interrater reliability indexed by high values of Cohen’s κ (κ = 1) for depressive disorders, panic disorder, and agoraphobia. 1 High-risk children’s mothers had at least two major depressive episodes (MDEs; n = 26) or one MDE and a major anxiety disorder (n = 3) 2 ; both mark risks for offspring depression (Barnett et al., 1991; Kendler et al., 1992). Low-risk children’s mothers had no history of depressive or anxious disorder. No child had past or current depressive disorder as indicated by the Kiddie Schedule for Affective Disorders and Schizophrenia, Present and Lifetime version (K-SAD-PL; Kaufman et al., 1997) administered by trained clinical graduate students to both the primary caregiver and the child. Inter-interviewer reliability of child and caregiver K-SAD-PL was examined in 11 children, none of whom met criteria for any diagnoses (including depression), and demonstrated 100% interrater agreement on the absence of any diagnoses. 3
Mothers completed the Child Behavior Checklist (Achenbach, 2007), of which the withdrawn-depressed subscale was used to index maternally reported symptoms (Cronbach’s α = .71); children completed the Children’s Depression Inventory (CDI; Kovacs & Multi-Health Systems Staff, 2003; Cronbach’s α = .84). Of the 87 children, 85 (29 high risk; 48 boys) contributed usable fMRI data during both negative mood induction and resting state (one had braces; one discontinued).
PE and sadness (age 3)
Children’s PE and sadness at age 3 were assessed via 12 standardized tasks of the Laboratory Temperament Assessment Battery (Goldsmith et al., 1995) conducted by a female experimenter. Tasks were designed to elicit a range of behaviors relevant to early temperament and were videotaped for coding. In each task, PE and sadness were coded by trained graduate and undergraduate coders by counting instances of the two affects in facial, vocal, and bodily channels at three intensity levels (low, moderate, and high). Coders had to achieve an intraclass correlation (ICC) of .80 with a master coder on approximately 20 participants before coding independently, after which periodic reliability checks were conducted on 15% to 20% of all participants for each task. Finally, to calculate an aggregate score for PE and sadness, counts of each affect at each intensity level were weighted by multiplying 1 (low), 2 (moderate), or 3 (high) and then averaged across channels and tasks (PE: Cronbach’s α = .88, interrater ICC = .67, n = 16; sadness: Cronbach’s α = .77, interrater ICC = .63, n = 14). Each task is briefly described below in the order of conduction:
Early-life stress (ages 3–5)
Early-life stress was assessed at child ages 3, 4, and 5 via three indicators. At child ages 3 and 5, the primary caregiver reported annual family income on a 5-point scale (1 = < $20,000; 2 = $20,000–$40,000; 3 = $40,001–$70,000; 4 = $70,001–$100,000; 5 = > $100,000) and completed the Dyadic Adjustment Scale (DAS; Spanier, 1976), a 32-item scale measuring quality of intimate relationship (age 3: Cronbach’s α = .92; age 5: Cronbach’s α = .93). At child ages 4 and 5, trained clinical graduate students administered the UCLA Chronic Stress Interview (Hammen, 1991) to the primary caregiver to assess the chronic familial stress during the past approximately 15 months. The UCLA interview covers chronic stress across multiple domains in the home, including quality of intimate relationship, friendship, social life, relations with family members, finances, work, and health of self and family members (age 4: Cohen’s α = .74, interrater ICC = .72, n = 10; age 5: Cohen’s α = .72, interrater ICC = .74, n = 13).
Family income and DAS scores were reversed such that higher scores reflected lower family income and lower relationship quality. We then computed a composite stress score by standardizing and averaging scores of family income, DAS, and UCLA interview for each family across all time points of assessments. A higher composite score indicates children’s exposure to greater chronic life stress during early to middle childhood (Cronbach’s α = .71).
Negative mood induction and MRI data collection (age 11)
MRI data were collected at the UWO Robarts Research Institute on a Siemens Magnetom Prisma fit 3T scanner with a 32-channel head coil. Resting-state fMRI data were first collected for a period of 6 min as children looked at a fixation at the center of the screen while remaining still. Next, a negative-mood-induction procedure was conducted in the scanner by showing children a 169-s-long movie clip from The NeverEnding Story (Petersen, 1984) in which a boy and his horse were passing through a swamp in which the horse subsequently drowned. Children were instructed to watch the clip while staying still and to pretend that what happened in the movie was happening in real life. They rated their mood before and after watching the clip on a 5-point scale (1 = very sad, 5 = very happy). A paired-samples t test comparing preinduction and postinduction mood ratings indicated that children showed significant mood change after watching the movie clip toward the sad end of the scale, which supports the effectiveness of the mood-induction procedure; preintervention: M = 3.72, SD = 0.75; postintervention: M = 2.18, SD = 0.76, t(83) = 15.68, p < .001, 95% confidence interval = [1.34, 1.73]. An independent-samples t test showed that high- and low-risk children did not differ in their response to negative mood induction, p = .88.
During negative mood induction, high-resolution T1-weighted anatomical images were acquired using a magnetization-prepared rapid gradient-echo sequence (repetition time [TR] = 2,300 ms, echo time [TE] = 2.98 ms, inversion time [TI] = 900 ms, flip angle = 9°, number of slices = 192, field of view [FOV] = 256 mm, voxel size = 1 mm3). Functional T2-weighted gradient echo images were acquired with 48 contiguous axial interleaved slices with a 0-mm gap (TR = 1,000 ms, TE = 30 ms, flip angle = 45°, FOV = 210 mm, voxel size = 3 mm3, matrix size = 642).
fMRI data processing
The fMRI data were processed using the CONN toolbox (Version 18.a; Whitfield-Gabrieli & Nieto-Castanon, 2012) in MATLAB (Version 7.14.0; The MathWorks, Natick, MA). The same protocol was used for data collected during both negative mood induction and resting state. First, data were processed by following a standard preprocessing pipeline: Functional images were realigned to the first image for motion correction and were corrected for slice timing, the mean realigned functional image was coregistered to each individual’s T1 image and normalized to Montreal Neurological Institute (MNI) space, and the normalized images were then resampled to 2-mm3 voxels and spatially smoothed with a 6-mm Gaussian kernel.
In addition to the basic preprocessing steps, we implemented further measures in CONN for FC analysis to account for spurious variances of signals such as physiological and motion-related noises. For each individual, white matter and cerebrospinal fluid were estimated and regressed out using the anatomical CompCor method (Behzadi et al., 2007), temporal filtering was implemented with a band pass of 0.008 Hz to 0.09 Hz, scans with frame-wise displacement exceeding 0.9 mm (Siegel et al., 2014) or average global signal intensity exceeding 5 SD from the individual’s mean were labeled as invalid and ignored in subsequent analysis, motion parameters (three rotation, three translation, and six first-order temporal derivatives) and global signals were modeled as nuisance variables, and the effect of condition (e.g., hemodynamic response function-convolved task block) was also modeled as a confound to control for potential coactivation of different regions in response to the presented stimuli. No significant effect of risk group (high risk vs. low risk) or brain state (negative mood induction vs. resting state) was found in average frame-wise displacement (negative mood induction high risk: M = 0.23 mm, SD = 0.07; negative mood induction low-risk: M = 0.21, SD = 0.08; resting state high risk: M = 0.21 mm, SD = 0.09; resting state low risk: M = 0.20 mm, SD = 0.06, p = .31) or the percentage of invalid scans (negative mood induction high risk: M = 0.03, SD = 0.03; negative mood induction low risk: M = 0.03, SD = 0.06, p = .85; resting state high risk: M = 0.02, SD = 0.04; resting state low risk: M = 0.02, SD = 0.02). None of the correlations between average frame-wise displacement and other variables of interest (PE, sadness, early-life stress) were significant (ps > .27).
FC analysis of data during negative mood induction
FC analysis was first conducted on the fMRI data collected during negative mood induction. To increase the sensitivity of analyses, we used an ROI-to-ROI approach and constrained our analyses within 13 a priori anatomical ROIs as defined by the Harvard-Oxford cortical and subcortical structural atlases (Desikan et al., 2006). These consisted of three DMN regions (mPFC, ACC, PCC), four PFC regions (left and right areas of dlPFC and OFC), and six limbic regions (left and right areas of amygdala, insula, and hippocampus). Given our interests in introspective processes and emotion regulation, we focused on seven of the 13 ROIs as the seeds, including three DMN regions (mPFC, PCC, ACC) and four PFC regions (left and right dlPFC, left and right OFC). Each seed was paired with a target ROI (i.e., one of the other a priori ROIs) to generate both cortical-cortical and cortical-subcortical FCs. This resulted in 57 ROI-to-ROI associations in total because we were not testing any directional relationships between ROIs and removed the redundant pairs (see Tables 2 and 3).
In first-level analysis, Pearson’s correlation between the average BOLD time series of each of the 57 pairs was computed for each participant. Correlation coefficients were then converted to Fisher’s z scores and subjected to second-level analysis as the dependent variables. In second-level analysis, a multiple linear regression model was conducted on the Fisher’s z score of each of the 57 ROI-to-ROI pairs; maternal depression, age 3 PE, age 3 sadness, and early-life stress were the regressors. The initial model included three interaction terms (Maternal Risk × Stress, PE × Stress, and Sadness × Stress) to examine the moderating effect of early-life stress. Nonsignificant interactions were dropped to reserve statistical power. Each child’s age, sex, concurrent depressive symptoms (self-reported CDI), and average frame-wise displacement were treated as covariates. Within the CONN toolbox, we applied correction of false discovery rates (FDR; p < .05) for each of the seven seeds to correct for the multiple tests conducted on the FC between each seed and multiple target ROIs. The FDR correction was applied on the seed level but not across all seeds to balance between false positive error and false negative error.
Post hoc analyses controlling for resting-state FC
We were interested in the extent to which any relationships between early risk factors and FC during negative mood induction were specific to brain activity during negative-emotion reactivity compared with during the non-task-based resting-state activity. We therefore based all analyses of the resting-state data on findings from the data of negative mood induction; specifically, we focused on any ROI–ROI pairs that demonstrated significant associations with early risk factors during negative mood induction and extracted the FC indices (i.e., Fisher’s z scores) from data during resting-state and negative mood induction for these same ROI–ROI pairs. 4 To examine whether the observed associations between the risk predictors and FC during negative mood induction would hold after varying out resting-state activity of the same ROI–ROI pairs, we conducted post hoc linear regression analyses on the z scores extracted from the data of negative mood induction; the z scores of resting-state data were controlled as a covariate, and the risk factors (i.e., maternal depression, PE, sadness, stress, and any significant interaction term) were treated as the predictors. Age, sex, concurrent depressive symptoms, and mean frame-wise displacement served as other covariates (IBM SPSS Version 24.0.1). Note that we ran these post hoc analyses for the sole purpose of examining the significance of the effects of predictors above and beyond resting-state FC, not to estimate, or overestimate, the effect sizes of any associations between early risk markers and FC during negative mood induction.
Results
Bivariate correlations between major study variables
Table 1 presents the descriptive statistics and bivariate correlations for primary study variables. Maternal depression was associated with early-life stress and youths’ depressive symptoms at age 11, the latter indexed by both mothers’ and youths’ self-report. Life stress was associated with youths’ self-reported depressive symptoms at age 11. Youths’ symptoms reported by mothers were positively correlated with youths’ self-reported symptoms.
Mean, Standard Deviation, and Bivariate Correlations for Main Study Variables
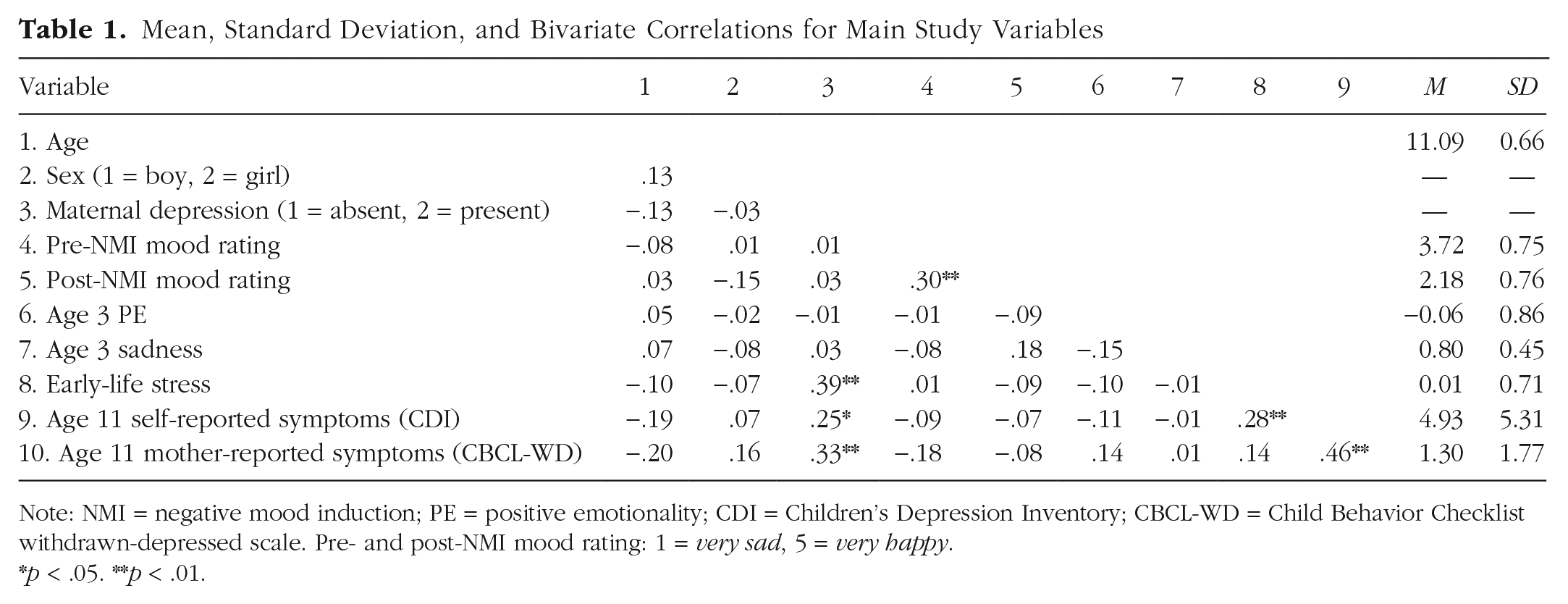
Note: NMI = negative mood induction; PE = positive emotionality; CDI = Children’s Depression Inventory; CBCL-WD = Child Behavior Checklist withdrawn-depressed scale. Pre- and post-NMI mood rating: 1 = very sad, 5 = very happy.
p < .05. **p < .01.
ROI-to-ROI FC during negative mood induction
In the initial regression model, two of the three interaction terms (Maternal Depression × Stress, PE × Stress) did not predict FC between a priori ROIs and were therefore dropped from analyses. In the final regression model (see Tables 2 and 3 and Figs. 1 and 2), maternal depression, age 3 PE, age 3 sadness, early-life stress, and Sadness × Life Stress all predicted FC between certain a priori ROIs with concurrent depressive symptoms being controlled. Compared with low-risk children, the high-risk group showed decreased (less positive or more negative) connectivity between mPFC and bilateral regions of amygdala and insula but increased (i.e., more positive) connectivity between mPFC and PCC (Fig. 1a). Across risk groups, age 3 PE predicted increased (more positive or less negative) connectivity between left dlPFC and left amygdala (Fig. 1b).
Multiple Regression Models in ROI-to-ROI Analyses Within A Priori ROIs: Results of Early Risk Factors
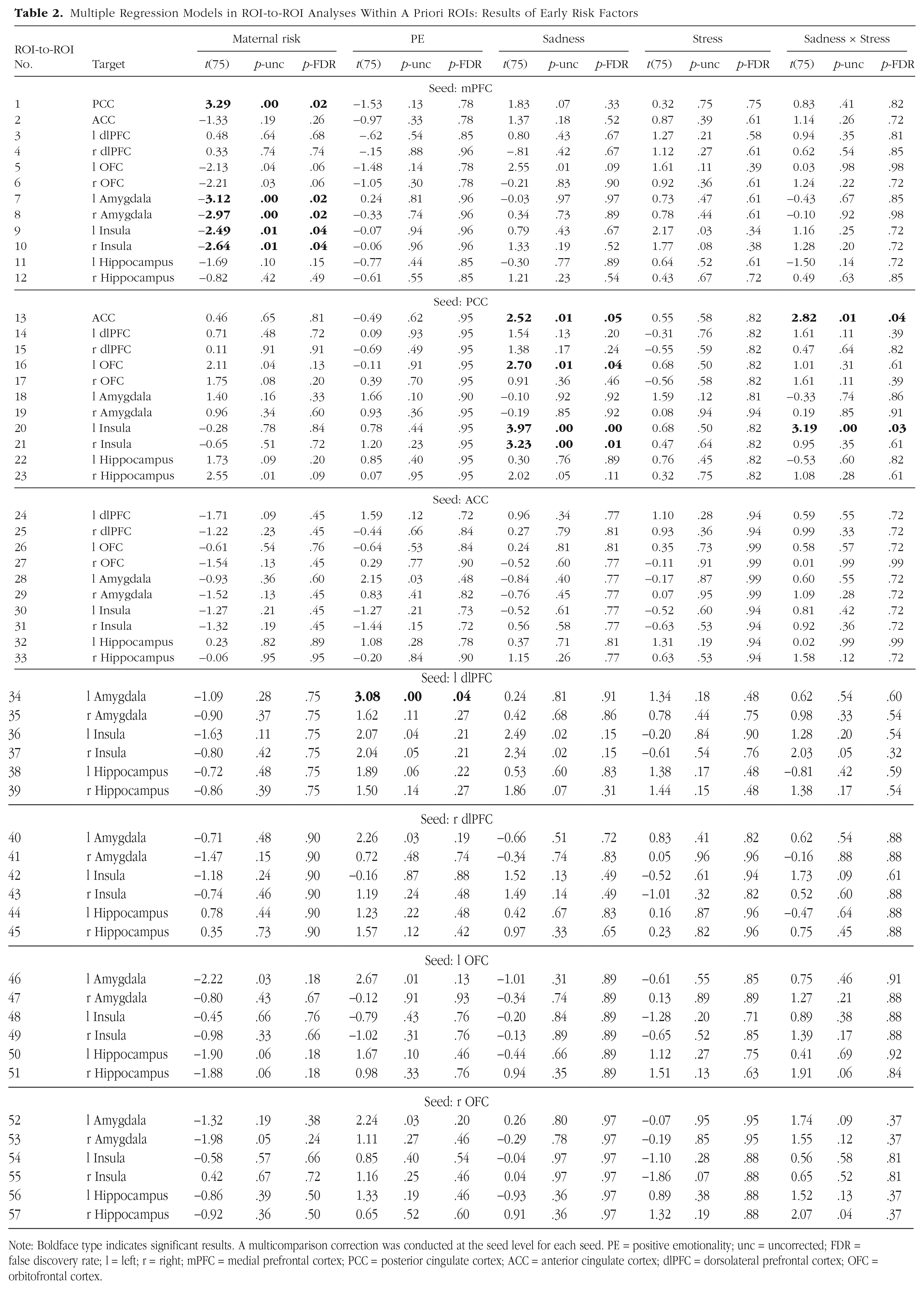
Note: Boldface type indicates significant results. A multicomparison correction was conducted at the seed level for each seed. PE = positive emotionality; unc = uncorrected; FDR = false discovery rate; l = left; r = right; mPFC = medial prefrontal cortex; PCC = posterior cingulate cortex; ACC = anterior cingulate cortex; dlPFC = dorsolateral prefrontal cortex; OFC = orbitofrontal cortex.
Multiple Regression Models in ROI-to-ROI Analyses Within A Priori ROIs: Results of Covariates
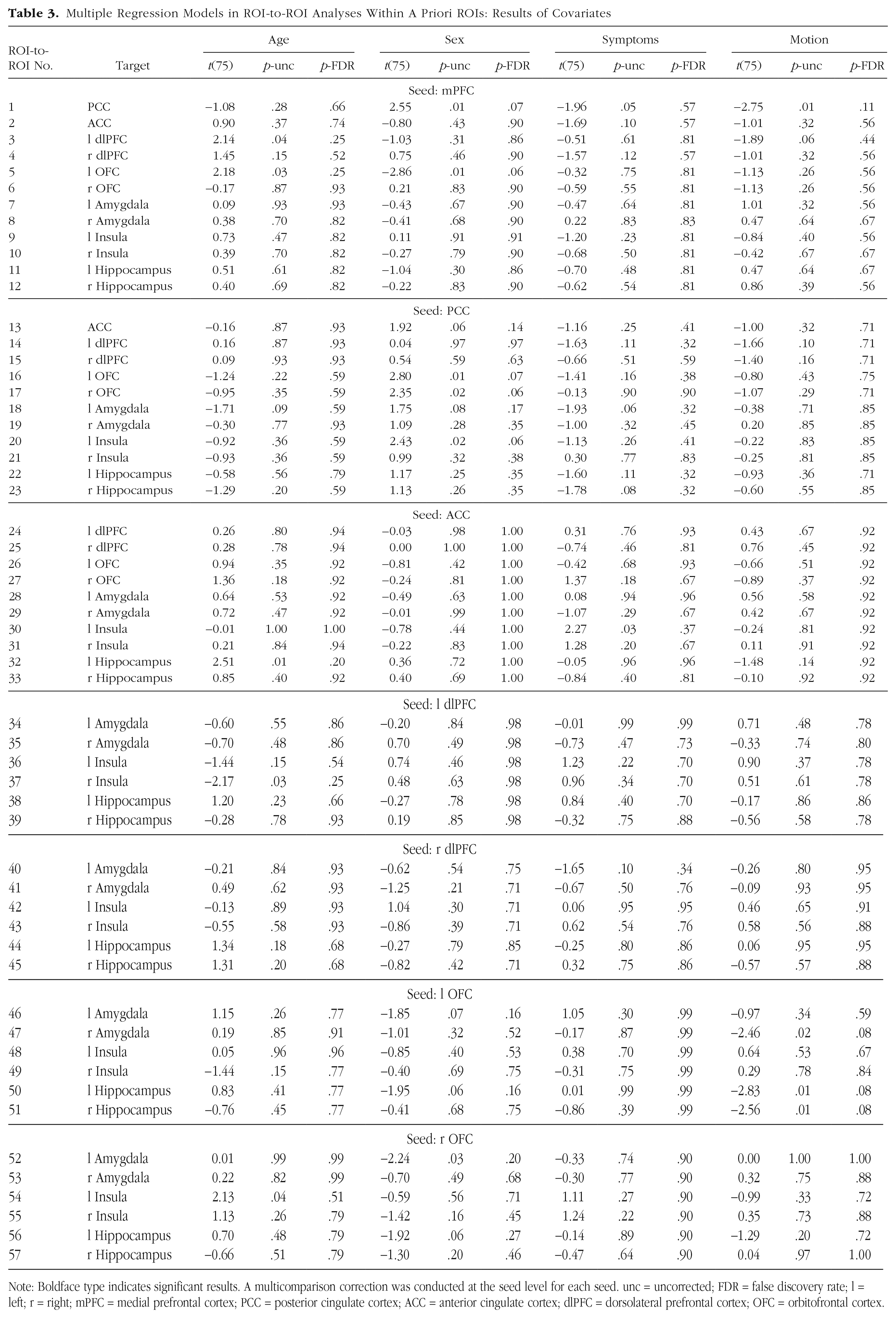
Note: Boldface type indicates significant results. A multicomparison correction was conducted at the seed level for each seed. unc = uncorrected; FDR = false discovery rate; l = left; r = right; mPFC = medial prefrontal cortex; PCC = posterior cingulate cortex; ACC = anterior cingulate cortex; dlPFC = dorsolateral prefrontal cortex; OFC = orbitofrontal cortex.
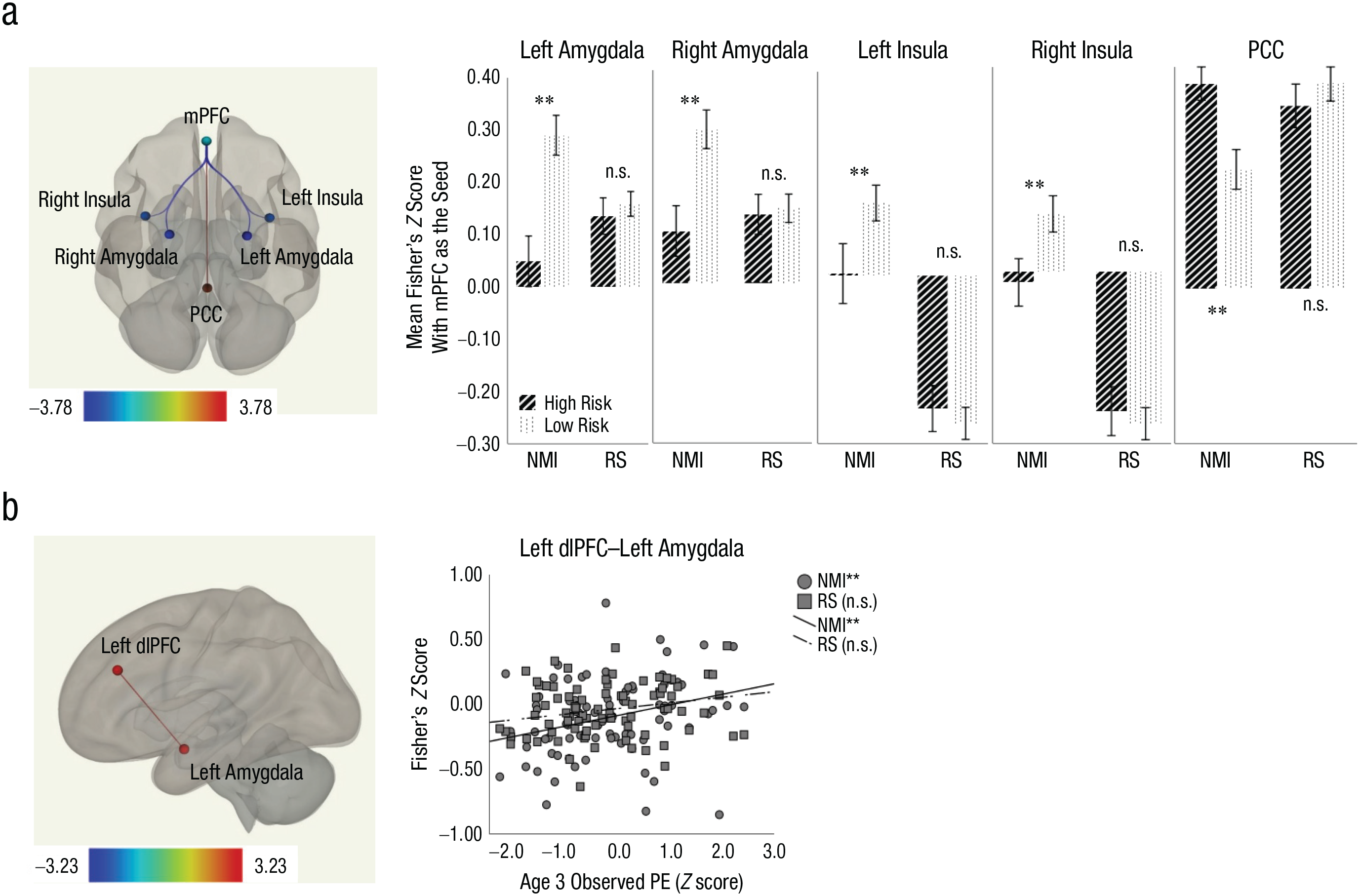
Main effect of maternal depression risk (a) and age 3 positive emotionality (PE; b). The bar graphs show Fisher’s z scores of significant ROI–ROI pairs for high-risk and low-risk youths. Error bars indicate ±1 SE. The scatterplots (with best-fitting regression lines) show the relationship between Fisher’s z scores for the significant ROI–ROI pair and age 3 PE. ROI = region of interest; NMI = negative mood induction; RS = resting state; PE = positive emotionality; mPFC = medial prefrontal cortex; OFC = orbitofrontal cortex; PCC = posterior cingulate cortex; ACC = anterior cingulate cortex; dlPFC = dorsolateral prefrontal cortex; n.s. = nonsignificant.
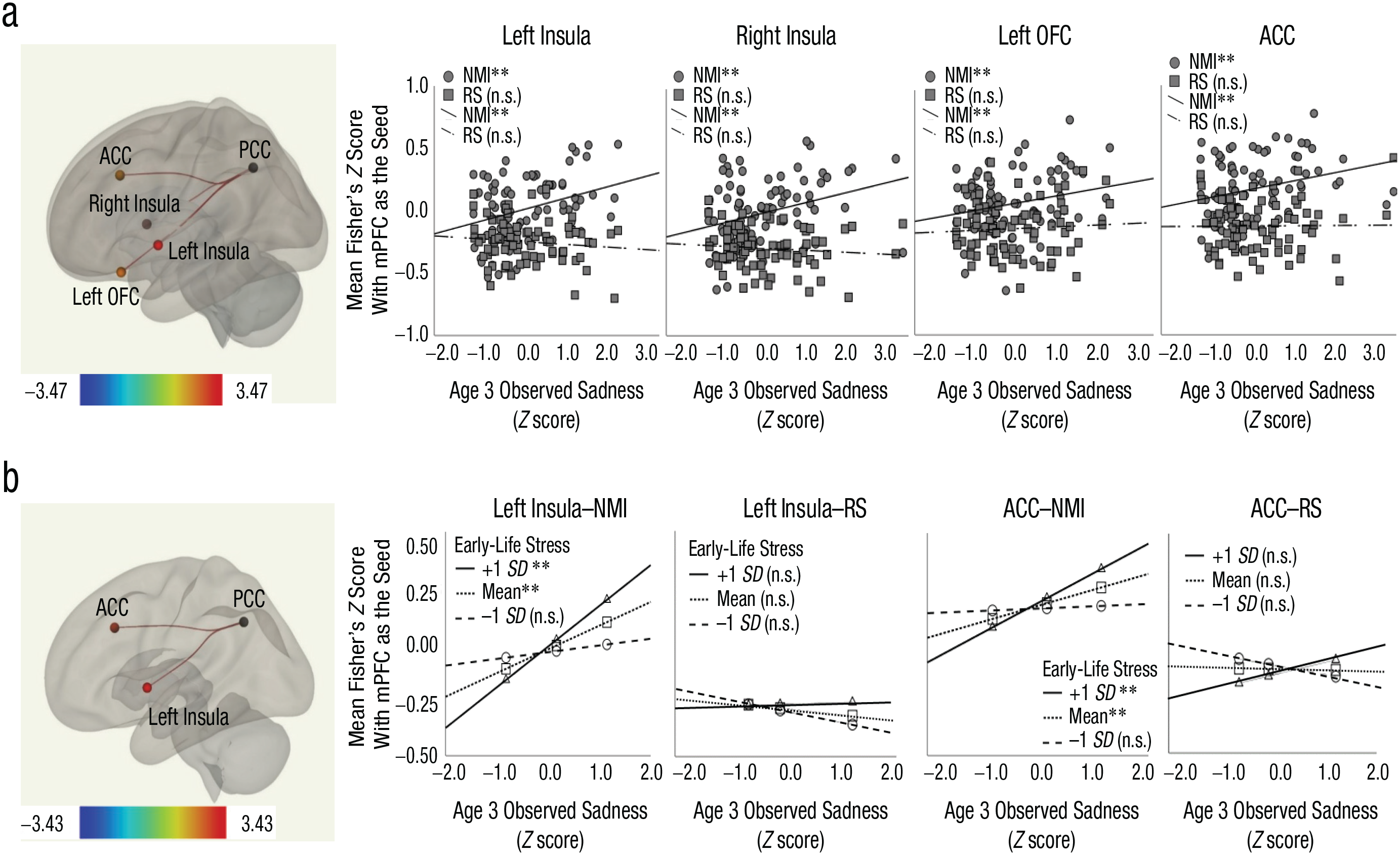
Main effect of age 3 sadness (a) and interaction between age 3 sadness and early-life stress (b). The scatterplots (with best-fitting regression lines) show the relationship between Fisher’s z scores for significant ROI–ROI pairs and age 3 sadness. The bar graphs plot Fisher’s z scores of significant ROI–ROI pairs at three levels of early-life stress against age 3 sadness. Error bars indicate ±1 SE. ROI = region of interest; NMI = negative mood induction; RS = resting state; OFC = orbitofrontal cortex; PCC = posterior cingulate cortex; ACC = anterior cingulate cortex; n.s. = nonsignificant.
Although age 3 sadness predicted increased (more positive or less negative) connectivity between PCC and bilateral insula, left OFC, and ACC (Fig. 2a), this main effect was qualified by a significant interaction with early chronic stress in predicting PCC–left insula and PCC–ACC FC (Fig. 2b). To further probe the interaction patterns, we ran simple-slopes analyses by examining the associations between sadness and the PCC–left insula and PCC–ACC FC at mean and mean ± 1 SD life stress (Hayes, 2018). Results showed that the sadness-FC associations were significant for children experiencing average (M) and above average (+1 SD) life stress, but not lower life stress (–1 SD), indicating that exposure to greater early-life stress strengthened the longitudinal associations between sadness and PCC–left insula and PCC–ACC FC during negative mood induction.
Results of post hoc analyses controlling for resting-state FC
To examine whether the significant associations between the risk predictors and FC during negative mood induction would hold after controlling for resting-state activity, post hoc linear regression analyses were conducted on the z scores extracted from the FC data of negative mood induction with the z scores extracted from resting-state FC controlled as a covariate. Note that effect sizes should not be interpreted using these post hoc results. Results of these analyses indicated that the observed effects of early risk predictors remained significant above and beyond resting-state FC. Specifically, the effect of maternal depression remained significant for all five ROI–ROI pairs; mPFC–left amygdala: b = −0.25, SE = 0.07, p < .001; mPFC–right amygdala: b = −0.23, SE = 0.07, p < .001; mPFC–left insula: b = −0.17, SE = 0.07, p < .05; mPFC–right insula: b = −0.17, SE = 0.07, p < .05; and mPFC–PCC: b = 0.19, SE = 0.06, p < .01. The effect of age 3 PE remained significant on the left dlPFC–left amygdala FC: b = 0.08, SE = 0.03, p < .05. The effect of age 3 sadness also remained significant on all four ROI–ROI pairs; PCC–left insula: b = 0.10, SE = 0.03, p < .001; PCC–right insula: b = 0.09, SE = 0.03, p < .01; PCC–left OFC: b = 0.07, SE = 0.03, p < .05; and PCC–ACC: b = 0.07, SE = 0.03, p < .05. Finally, the Sadness × Stress interaction remained significant on PCC–left insula (b = 0.10, SE = 0.04, p < .05) and PCC–ACC (b = 0.08, SE = 0.04, p = .05). Overall, the associations between early risk factors and FC during negative mood induction remained significant when the variances of resting-state FC were varied out, which suggests that these observed FC patterns during negative mood induction reflected brain connectivity during negative-emotion processing and could not be attributed to resting-state activity.
Discussion
In this study, we examined how maternal depression and age 3 temperamental emotionality (PE and sadness), all established risk factors for depression, were related to never-depressed preadolescents’ FC during a negative mood state; we also examined whether any associations between these early risk markers and FC were moderated by exposure to early chronic stress. We found that youths with a maternal depression history showed decreased (less positive or more negative) mPFC-based FC in bilateral amygdala and bilateral insula but increased (more positive) FC between mPFC and PCC. Children’s PE at age 3 was associated with stronger (more positive) FC between left dlPFC and left amygdala. Although age 3 sadness was associated with stronger (more positive or less negative) PCC-based FC in left and right insula, left OFC, and ACC, this main effect was qualified by an interaction with early chronic stress in predicting PCC-based FC in left insula and ACC. Specifically, the associations between sadness and PCC–left insula FC and between sadness and PCC–ACC FC were significant in the presence of greater early chronic stress. Note that these results were significant despite the absence of a personal depression history in the youths, even after adjusting for concurrent depressive symptoms, which suggests that the observed FC patterns were markers of early depression vulnerabilities rather than state markers of concurrent symptoms. In addition, similar patterns were not observed in the resting-state data of the same sample, which indicates that the observed associations were specific to brain activity during emotion processing/reactivity rather than intrinsic brain activity. By taking a longitudinal, multimethod approach, this study provides novel information concerning the longitudinal associations between early vulnerability and potentially maladaptive patterns of FC during negative-emotion processing.
First, maternal depression and lower PE at age 3 were associated with decreased (less positive) FC between PFC subregions (mPFC and dlPFC) and limbic areas (amygdala and insula) during negative mood induction. This is consistent with findings from clinically depressed individuals: Compared with control participants, depressed adolescents (Perlman et al., 2012) and adults (Carballedo et al., 2011; Friedel et al., 2009; Kong et al., 2013) show decreased PFC-limbic FC in processing negative pictorial stimuli. The PFC subregions and limbic areas are known to mediate emotion regulation and emotion reactivity, respectively (Helm et al., 2018). For adaptive processing of negative stimuli, sufficient PFC-mediated regulatory resources are needed to effectively regulate limbic reactivity, which may manifest as positive PFC-limbic coupling. Indeed, healthy adults show more positive FC between PFC regions (e.g., mPFC, OFC, dlPFC) and amygdala when they are exposed to threat (Gold et al., 2015) or down-regulating feelings elicited by aversive pictures (Banks et al., 2007); greater PFC-amygdala FC is also correlated with better performance on a goal-directed task (Gold et al., 2015) or decreases in negative feelings (Banks et al., 2007). Considered alongside this past work, the decreased PFC-limbic FC during negative mood induction we report here may indicate dysfunction in emotion regulation such that PFC-mediated regulatory processes are insufficient to effectively regulate heightened limbic reactivity to negative stimuli. Consistent with this notion, previous work reported that during a similar negative-mood-induction procedure, school-age children with preschool onset depression showed decreased prefrontal activity compared with control participants; children with more severe symptoms also show increased amygdalar activity (Pagliaccio et al., 2012). These maladaptive patterns of brain activity and connectivity may further portend or enhance risks for later depression.
Maternal depression was also associated with increased PCC–mPFC FC during negative mood induction. This is consistent with previous findings in depressed youths and adults who, compared with control participants, showed heightened FC within the DMN during resting-state or nonintrospective emotion processing (Hamilton et al., 2015; Kaiser et al., 2015). As two key components of DMN, PCC and mPFC are thought to mediate introspective, self-referential processes; heightened PCC–mPFC FC is thought to be a neural marker of ruminative processes or indicate greater involvement of the self during information processing (Hamilton et al., 2015; Whitfield-Gabrieli & Ford, 2012). In this study, however, because we did not instruct youths to self-reflect while watching the movie clip, it is unclear whether some of them might have engaged in self-related processes (e.g., recalling similar events from their own lives while watching the video). Future research that explicitly instructs participants to think about themselves while watching mood-evocative movie clips may more directly tap into introspective processes during emotion processing, which are important for depression risk.
Age 3 sadness was associated with increased, more positive FC between PCC and bilateral insula, left OFC, and ACC. However, this main effect was qualified by an interaction with early chronic stress such that only children raised in moderate to relatively high-stress homes showed significant associations between sadness and the FC of PCC–left insula and PCC–ACC. The observed interaction is consistent with diathesis-stress models of depression, which supports the moderating role of exposure to early stress in predicting later brain connectivity during emotion processing. In a low-risk, community-dwelling sample such as ours, mild early stress may be insufficiently potent to exert a significant main effect on brain functions; rather, mild stress conditionally influences brain development through its interaction with other vulnerabilities. For our participants, the presence of stress may activate or further maintain maladaptive processes such as rumination, thereby strengthening the associations between sadness and later brain activity during induced negative mood.
Among the ROIs that showed associations with sadness, both insula and ACC are part of the salience network, subserving the detection and monitoring of the biological and socioemotional salience of the stimuli (Seeley et al., 2007). OFC, in addition to its role in emotion regulation, is also activated by nonreward, punishment, or unpleasant stimuli (Grabenhorst & Rolls, 2011; Small et al., 2001). PCC is thought to support autobiographical memory search and retrieval (Spreng et al., 2008) as well as attention shifting between the external world and internal world (Brewer et al., 2013). For example, healthy adults show heightened PCC activation before lapses in attention during an attentional control task, which may indicate an attention shift from the external task to internal mentation (Weissman et al., 2006). Consistent with these proposed functions of PCC, a recent neural model of rumination (Hamilton et al., 2015) suggests that although the salience network assesses the significance of stimuli in the external environment, the DMN (including PCC) assigns and elaborates on the valence of self-related, internally represented stimuli (e.g., introspective thoughts, feelings, and memories). However, because the mood-induction paradigm used did not explicitly evoke introspective processing, we cannot establish whether the observed increases in PCC connectivity reflected internally oriented attention shifts or activations of autobiographical memory. Again, future research will benefit from paradigms that directly elicit introspective processes, for example, by instructing participants to think about themselves during mood-induction procedures.
It is unclear why the Sadness × Stress interaction was significant for PCC–left insula and PCC–ACC FC but not for PCC–right insula and PCC–left OFC FC. We know of no past work that speaks to why only PCC-OFC FC in the left hemisphere was related to sadness, a topic for future researchers to consider. However, regarding the insula, previous work has reported lateralization of insular function during emotion processing; for example, a meta-analysis showed greater involvement of left insula compared with right insula when a subjective mood was induced in participants (Duerden et al., 2013). Although our data cannot directly examine the magnitude of activation in left and right insula during negative mood induction, we speculate that this functional laterality of insula may be associated with the observed interaction patterns for the PCC-insula FC. No interaction with early stress was found for maternal depression or PE. Compared with PE, early sadness also shows associations with FC across a greater number of ROIs, which suggests that sadness may be more strongly associated with the neural substrates of negative mood reactivity. This is unsurprising given the shared valence of these two constructs. The relatively stronger association between sadness and FC (compared with PE and FC) may be related to rumination given that rumination has been found to be associated with both early NE (Schweizer et al., 2018) and altered neural patterns during negative-emotion processing (Mandell et al., 2014). Although the paradigm used in this study did not tap into ruminative processes, future studies that directly measure rumination will help illuminate the relationships between early temperament, rumination, and brain function subserving emotion processing.
Our findings also expand the extant literature of high-risk studies that examine how early risk factors, including maternal depression and maladaptive child emotionality, predict brain functions in never-depressed youths. As described earlier, previous work in this literature has largely focused on resting-state FC (e.g., Chai et al., 2016; Roy et al., 2014) or regional activation in response to emotional stimuli (e.g., Joormann et al., 2012; Monk et al., 2008; Pérez-Edgar et al., 2007); in addition, most of these studies examined a single risk factor only and did not test any interactions. To our knowledge, the present study is the first one that explores how multiple early depression vulnerabilities and their interactions with early chronic life stress predict later brain connectivity during negative-emotion reactivity elicited by a negative-mood-induction paradigm. Examining FC across a number of a priori ROIs provides richer information on brain functioning compared with activation analysis in isolated regions. Incorporating multiple risk factors in the same model is also important to more precisely capture the multidetermined nature of the development of depression.
In post hoc analyses, the observed associations between early risk predictors and FC during negative mood induction remained significant after partialing out the variance of resting-state FC, which supports our conclusion that the observed FC patterns reflect the neural bases of negative mood reactivity rather than intrinsic brain connectivity. However, we did not include induction of other mood states as part of our task battery, which is an important limitation of our study because it renders it unclear to what extent the observed FC patterns during negative mood induction were valence dependent. In adults, FC alterations during positive-emotion processing are associated with clinical (Price et al., 2017) and subthreshold (Green et al., 2019) depression. In youths, blunted responses to positive or reward stimuli, including alterations in associated neural activity and connectivity, are known to confer risks for depression across development (Luking et al., 2016). Future research will benefit from exploring the associations between depression vulnerabilities and later FC during positive mood reactivity in never-depressed youths, which will add to the knowledge of early neurobiological markers of depression risks in youths.
Future studies may also consider the effect of task instructions during the mood-induction procedure on different cognitive processes relevant to depression risk. In this study, we instructed participants to pretend that “what happened in the movie was happening in real life” as opposed to other instructions that could have enhanced self-referent thought (e.g., “think of a time something similar happened to you”). Given the importance of self-focused processes in the development of depression, future research may consider using different instructions designed to enhance self-focus.
Of the 29 high-risk children in this study, 12 of their mothers had lifetime depression before the child’s birth, whereas 17 mothers had depression during the child’s life; however, the modest sample sizes of these two groups prevented us from testing the effect of maternal depression timing on children’s negative-emotion reactivity and psychopathology. Furthermore, although fathers’ depression history may also have important implications for offspring psychopathology, only seven of the 87 children had a father with a lifetime history of depression, which precluded analyses of associations between paternal depression and children’s negative-emotion reactivity and depression risk. Finally, the interrater reliability (ICC) of our temperamental measures was modest, albeit still within the acceptable range as suggested by the literature (ICCs > .55; Fleiss, 1986).
This study has a number of strengths. We used a longitudinal, multimethod approach in which we incorporated neuroimaging measures with an objective, observed measure of child temperament; a clinical interview to diagnose maternal depression; and a composite measure for early-life stress combining parent interview and questionnaire. The multimeasurement approach allowed us to eliminate the shared measurement error that may inflate associations between constructs. We used a standard negative-mood-induction procedure to effectively elicit sad mood and tap negative-emotion reactivity. None of our participants had current or lifetime depressive disorder, which ensures that the observed neural patterns were markers of early risks rather than concomitants or consequences of the disorder. Taking a rigorous approach, we found that early depression vulnerabilities were associated with decreased PFC-limbic FC and increased PCC-based FC, which may indicate dysfunctional emotion regulation and tendencies of rumination or excessive self-focus during negative-emotion reactivity. These patterns, in turn, may portend or maintain risks for later depression.
Footnotes
Acknowledgements
We thank the families who participated in our studies and the many individuals who contributed to data collection.
Transparency
Action Editor: Erin B. Tone
Editor: Kenneth J. Sher
Author Contributions
E. P. Hayden conceptualized and designed the study. M. F. Joanisse contributed to study design and data analysis. D. M. Barch contributed to data analysis and data interpretation. M. R. J. Vandemeer, O. Mohamed Ali, and A. R. Daoust contributed to neuroimaging data acquisition. P. Liu performed data analysis and drafted the manuscript. All of the authors provided critical revisions and approved the final manuscript for submission.
