Abstract
Dozens of studies have indicated that individuals more prone to experiencing disgust have stronger symptoms of anxiety disorders—especially contamination sensitivity. However, no work has informed the degree to which this relationship arises from genetic factors versus environmental factors. In the present study, we fill this gap by measuring disgust proneness and contamination sensitivity in a sample of 7,199 twins and siblings of twins, including 1,411 complete twin pairs. Disgust proneness was related to contamination sensitivity, r = .32. Multivariate twin modeling revealed that genetic factors accounted for 34% and 40% of the variance in disgust proneness and contamination sensitivity, respectively, and that the correlation between the two traits reflected overlapping genetic (54%) and unshared environmental (46%)—but not shared environmental—influences. Although consistent with work indicating that disgust proneness relates to contamination sensitivity, results suggest that prevailing parental-modeling hypotheses for explaining this relationship be reevaluated.
Keywords
People vary in the degree to which they experience disgust toward things such as bodily waste, dead animals, and spoiled foods—that is, they vary in disgust proneness. A recent meta-analysis of 83 studies indicated that this trait covaries with symptoms of anxiety disorders (i.e., dimensional assessments of categorical diagnoses) even after controlling for dispositional negative affect (Olatunji, Armstrong, & Elwood, 2017). This relationship is stronger for so-called disgust-related disorders (e.g., obsessive-compulsive disorder, blood-injury-injection phobia, posttraumatic stress) than other anxiety disorders (e.g., social anxiety disorder, agoraphobia, generalized anxiety disorder), and it is especially strong for a specific facet of obsessive-compulsive disorder: contamination sensitivity (i.e., aversions to direct and indirect contact with objects interpreted as dirty or contaminating).
Clinical research on disgust proneness has sparked hypotheses regarding the etiology of such disorders with the goal of tailoring interventions to target characteristics of disgust not shared by general anxiety or fear (e.g., its distinct developmental inputs, mechanistic underpinnings, and resistance to extinction; Amoroso et al., 2020; Knowles, Cox, Armstrong, & Olatunji, 2019; Olatunji et al., 2017). One prominent hypothesis suggests that higher disgust proneness leads to an overestimate of the threat posed by infectious disease, which in turn leads to vigilance toward pathogen cues and, consequently, downstream anxiety (Olatunji et al., 2017; Widen & Olatunji, 2016). Critically, this hypothesis also proposes factors that might lead to the development of both disgust proneness and contamination sensitivity. Although acknowledging genetic influences, it emphasizes potential effects of parents’ behavior. Candidate parental behaviors include frequent expressions of disgust and a controlling parenting style, with the former putatively leading children to model disgust and the latter putatively inhibiting the extinction of children’s disgust-related anxieties that might follow from exposure. These possibilities seemingly resonate with multiple lines of evidence, including findings that children’s disgust proneness and fears of disgust-eliciting animals (e.g., spiders) covary with their parents’ disgust proneness (Davey, Forster, & Mayhew, 1993), that mothers of spider-phobic girls find spiders to be more disgusting than do mothers of nonphobic girls (De Jong et al., 1997), that parents and offspring score similarly on contamination-sensitivity measures (Rozin, Fallon, & Mandell, 1984), that parents’ verbal communication of disgust toward animals influences their children’s reactions to those animals (Muris, Mayer, Borth, & Vos, 2013), and that parents of younger children avoid contact with disgust elicitors more than parents of older children, potentially to model such avoidance especially for younger children (Stevenson, Oaten, Case, Repacholi, & Wagland, 2010).
Caution is warranted in interpreting such observations given that evidence that adults’ disgust proneness and contamination sensitivity stem from such parental dispositions or behaviors. Extensive research has found that similarities among family members are better accounted for by shared genetic factors than by shared environmental ones (Plomin, DeFries, Knopik, & Neiderhiser, 2016). Furthermore, although some laboratory experiments suggest that exposure to parents’ disgust expressions influences offspring behavior, other studies have reported similar effects resulting from exposure to unrelated individuals (Askew, Çakır, Põldsam, & Reynolds, 2014). If disgust proneness, contamination sensitivity, or both are indeed influenced by others’ expressions of disgust, then parental effects might be overwhelmed by the influence of the thousands of other people encountered throughout a child’s development. Indeed, aspects of the shared environment (including, potentially, exposure to the same parents) underlie many types of psychopathology in children, but these effects decline over time, to the point that they are undetectable in adults (Bergen, Gardner, & Kendler, 2007).
The rich literature investigating the relationship between disgust proneness and contamination sensitivity exclusively comprises phenotypic correlations—that is, bivariate correlations between observable characteristics. Such correlations alone cannot evaluate the hypotheses that disgust proneness and contamination sensitivity are shaped by parental modeling or parenting styles, as is their covariance. These limitations result from the fact that phenotypic correlations can arise because the same environmental factors influence both traits or because the same genetic factors influence both traits. Twin studies are uniquely positioned to partition such phenotypic correlations into genetic, shared environmental, and nonshared environmental components because monozygotic (MZ) twins share nearly all of their segregating genes (~100%), whereas dizygotic (DZ) twins and siblings share, on average, 50% of their segregating genes. At the same time, DZ twins share the same family environments (including being exposed to the same parents), just as MZ twins do. Thus, greater similarities between MZ twins relative to DZ twins indicate genetic influences on a trait. Given the similarity of MZ twins relative to DZ twins, we can estimate the degree to which DZ twins should be similar on the basis of genetic influences alone. Any similarity that is not genetic in nature is the result of environmental factors shared by twins. If disgust proneness and contamination sensitivity are jointly influenced by exposure to parents who model disgust or adopt a specific parenting style, we should find that shared environmental components influence both traits and their covariance. The current study tested the genetic and environmental underpinnings of disgust proneness, contamination sensitivity, and their covariance.
Method
Participants
Invitation letters were sent to twins and siblings of twins identified from the Central Population Registry in Finland. Individuals were invited only if (a) their mother tongue was listed as Finnish in the Central Population Registry, (b) they resided in Finland at the time of gathering contact addresses, and (c) they were older than 18. A subset of individuals (n = 7,716) had participated in similar data collections conducted by the same research group before (for an elaboration on this cohort, see Johansson et al., 2013) and indicated a willingness to participate in future studies (in 2006 or 2012–2013). We also obtained addresses for a cohort of twins and siblings of twins who had not previously participated in studies performed by our research group. These twins were born between March 13, 1988, and October 4, 2000 (their siblings were any biological siblings at least 18 years old). In total, addresses of 33,390 individuals were obtained. Of these, 179 had addresses abroad, leaving us with 33,211 eligible addresses. Recipients were invited to respond to an online survey with an individual eight-character code that was used to identify twin pairs and individuals from the same family, and they were offered entry into a raffle that contained 40 gift vouchers to a Finnish network of companies operating in the retail and service (e.g., restaurants and petrol stations) sectors worth €100 each as prizes. The first invitation letters were sent to potential participants in November 2018. Individuals who did not respond were sent a maximum of two reminder letters spaced 2 to 3 weeks apart. We used a postal service that did not include an option to return undeliverable letters; thus, the response rate presented here conservatively estimates that all invitation letters were received. We were informed that 31 individuals were unable to participate (e.g., because of relocation, severe disability, or death). Data collection concluded in the first week of January 2019.
In total, 9,564 individuals (6,965 twins, 2,592 siblings, and seven unknown) responded, resulting in a total response rate of 29%; 9,319 (97%) of these respondents consented to their data being used for scientific purposes. The current analyses include the 7,199 participants who completed both the disgust-proneness and contamination-sensitivity instruments and were either twin singletons, members of a twin pair, or siblings of at least one twin who provided data (66.3% women; age: M = 29.51 years, SD = 7.84). Of these participants, 2,822 were members of complete twin pairs, including 274 MZ male, 832 MZ female, 240 DZ male, 740 DZ female, and 736 DZ opposite-sex twins. An additional 3,502 were twin singletons, and 875 were siblings of participating twins. This sample is ~71 times larger than the median sample size included in a recent meta-analysis on this topic (Olatunji et al., 2017) and, to the best of our knowledge, is the largest sample to have ever completed instruments measuring disgust proneness and contamination sensitivity. The sample affords between 80% and 90% power to detect shared environment components of .20 (depending on additive genetic components ranging between .10 and .50; for a power curve, see Fig. S1 in the Supplemental Material available online). Zygosity was available for some twins on the basis of earlier genotyping, and for other twins, it was estimated using instruments with excellent accuracy when validated using DNA polymorphisms (for further details, see the Supplemental Material).
Ethical review
The Ethics Review Board of Åbo Akademi University in Turku, Finland, reviewed and approved the research plan describing the data-collection procedure. Data collection involved no invasive procedures, and participants were informed that participation was voluntary and that they were free to terminate their participation at any time without giving a reason. All participants provided written, informed consent in accordance with the Declaration of Helsinki before responding to the survey.
Measures
All items were presented in Finnish. English versions of the scales were translated by a native Finnish speaker fluent in English, and they were back-translated to English by another native Finnish speaker also fluent in English and without access to the original English items. A native English speaker then compared the back-translated version with the original English versions and flagged discrepant items. The original translator then reviewed and revised those items as appropriate. Finally, two other native Finnish speakers checked all items for grammar and comprehensibility.
Contamination sensitivity
To measure contamination sensitivity, we administered the contamination subscale of the Padua Inventory–Revised (Burns, Keortge, Formea, & Sternberger, 1996). This 10-item instrument asks participants to respond to statements such as “I wash my hands more often and longer than necessary” on a scale from 0 (not at all) to 4 (very much).
Disgust proneness
To measure disgust proneness, we used the seven-item pathogen domain of the Three Domain Disgust Scale (TDDS; Tybur, Lieberman, & Griskevicius, 2009), which asks participants to report how disgusting they find experiences such as “Stepping in dog poop” and “Seeing some mold on old leftovers in your refrigerator” on a scale from 0 (not at all disgusting) to 6 (extremely disgusting). We chose the TDDS because it has less item overlap with contamination-sensitivity instruments than do other frequently used disgust instruments. For example, the Disgust Scale (Haidt et al., 1994), which was used most widely in the literature meta-analyzed by Olatunji and colleagues (2017), contains multiple items with content that overlaps with Padua Inventory items. Compare, for example, the Disgust Scale item, “I would never let any part of my body touch the toilet seat in a public restroom,” with the Padua Inventory item, “I avoid using public toilets because I am afraid of disease and contamination.” Given such shared item content, previously observed relationships between disgust-proneness instruments and contamination sensitivity might partially reflect method variance. Using the TDDS presumably attenuates this issue because item content does not similarly overlap with the Padua Inventory.
Before twin modeling, we conducted a principal axis factor analysis on the seven TDDS pathogen items and the 10 Padua Inventory contamination items in IBM SPSS (Version 25). 1 A visual inspection of the scree plot indicated the presence of two factors (the five highest eigenvalues were 5.71, 2.54, 1.01, 0.91, 0.76). After oblimin rotation, the pattern matrix revealed that all seven disgust proneness items (α = .79) loaded saliently on one factor (all primary loadings > .46, no cross-loadings .06) and that all 10 contamination-sensitivity items (α = .88) loaded saliently on the other factor (all primary loadings > .56, no cross-loadings > .08). The relationship between disgust proneness and contamination sensitivity (r = .32; .38 when disattenuated for unreliability) was similar to that reported in a recent bias-corrected meta-analysis of 83 published effect sizes estimating the relationship between disgust proneness and anxiety disorder symptoms (r = .36; Olatunji et al., 2017).
Statistical analyses
Genetic analyses were conducted with the OpenMx package (Boker et al., 2011) in the R Software environment (Version 3.5.2; R Core Team, 2018) using the full information maximum likelihood option with responses from twin singletons contributing only to estimates of means and variances. We began with a saturated model, which allows means and standard deviations to differ between the sexes and correlations to differ between both zygosity categories and sexes. Differences between the negative log-likelihoods (LLs) of more as opposed to less constrained models were tested using χ² tests (α < .01). Degrees of freedom reflect differences in parameter estimates across models. If equality constraints did not significantly diminish model fit, the more parsimonious model was retained.
We first tested for sex differences in means and variances by constraining those values to be equal across the sexes. We then imposed equality constraints on the correlations between male-male and female-female twin pairs to test whether genetic and environmental influences varied across the sexes (quantitative sex differences). Then, we imposed equality constraints on the correlations between same-sex and opposite-sex DZ twins to test for sex differences in the genetic and environmental factors underlying variance in men and women (qualitative sex differences). Finally, we constrained sibling correlations to be equivalent to DZ twin correlations to test for twin-specific environmental effects.
Estimating genetic and environmental effects
Structural equation modeling allowed observed phenotypic variance to be partitioned into additive genetic (A), nonadditive genetic (D), shared environmental (C), and nonshared environmental (E) components. Because MZ twins share nearly all of their segregating genes (~100%), whereas DZ twins and siblings share, on average, 50% of their genes, higher MZ compared with DZ and sibling correlations indicate that genetic factors underlie trait variance (with MZ correlations more than double DZ and sibling correlations indicative of nonadditive genetic factors). DZ twin and sibling correlations more than half MZ twin correlations indicate that shared (common) environmental influences underlie trait variance. The remaining variance is attributed to nonshared environmental influences and measurement error. Because C and D have opposing effects on DZ and sibling correlations, they cannot be simultaneously estimated in the classical twin design. In a way similar to that described above, cross-twin–cross-trait correlations were used in a bivariate model to estimate the additive genetic, nonadditive genetic, shared environmental, and unique environmental influences on the covariance between disgust proneness and contamination sensitivity.
Results
Contamination sensitivity did not differ between twins and siblings, χ2(2) = 4.28, p = .12, although disgust proneness did, χ2(2) = 11.74, p = .003, albeit slightly (d = 0.03). Consistent with past work, both disgust proneness and contamination sensitivity varied across the sexes. Women scored higher on both traits (p < .001; for sex-specific means and standard deviations as well as all MZ, DZ, and sibling correlations, see Table S1 in the Supplemental Material). However, twin correlations did not differ between same-sex male and female twin pairs (quantitative sex differences) or between the same-sex DZ and opposite-sex twins (qualitative sex differences; all ps > .16). Sibling correlations did not differ from DZ twin correlations (ps = .77, .85, and .06 for disgust proneness, contamination sensitivity, and their covariance, respectively). Thus, correlations between additive genetic factors were estimated at 1.0 for MZ twins and 0.5 for both siblings and DZ twins (for all test statistics, see Tables S2 and S3 in the Supplemental Material).
MZ correlations for disgust proneness and contamination sensitivity (rs = .42 and .44, respectively) were more than double DZ correlations (rs = .11 and .15, respectively), as was the cross-twin–cross-trait correlation (i.e., the correlation between one twin’s disgust proneness and the other twin’s contamination sensitivity; rs = .17 and .07 for MZ and DZ twins, respectively). Consistent with these patterns, constraining C paths to 0 in an ACE model did not diminish model fit (ps > .999)—that is, we observed no effect of shared environment on disgust proneness, contamination sensitivity, or their covariance. We thus tested for dominance effects by constraining D paths to 0 in an ADE model. None of the three constraints diminished model fit (ps > .01). Constraining the additive genetic effects to 0 did diminish model fit (ps < .001), so we retained an AE model (see Table 1 for model fit statistics).
Standardized Genetic and Environmental Estimates and Model Fit Statistics for the Bivariate Model
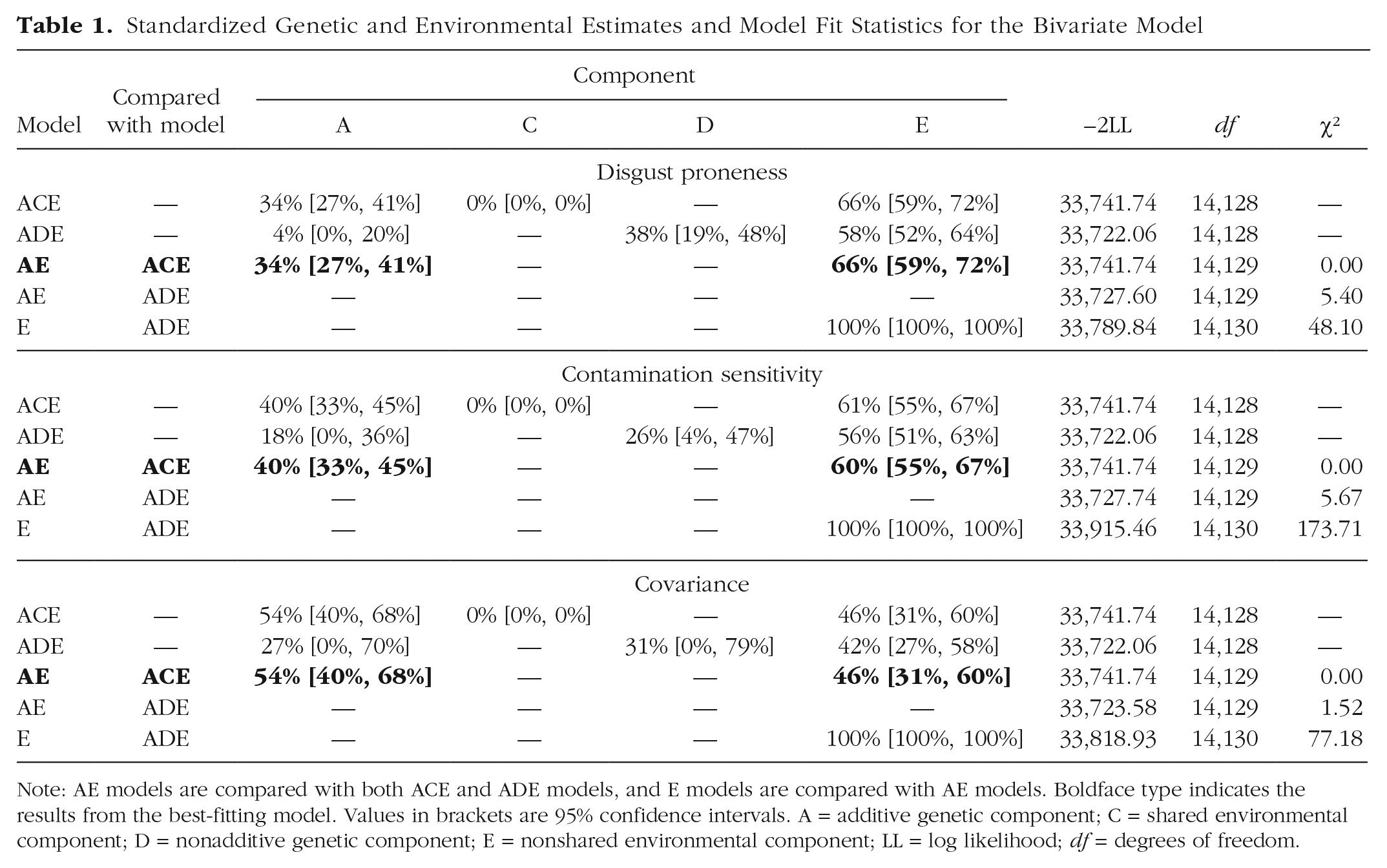
Note: AE models are compared with both ACE and ADE models, and E models are compared with AE models. Boldface type indicates the results from the best-fitting model. Values in brackets are 95% confidence intervals. A = additive genetic component; C = shared environmental component; D = nonadditive genetic component; E = nonshared environmental component; LL = log likelihood; df = degrees of freedom..
Genetic factors accounted for 34% and 40% of the variance in disgust proneness and contamination sensitivity, respectively, and unique environmental factors accounted for the remaining variance (see Table 1 and Fig. 1). The overlap between genetic influences on disgust proneness and contamination sensitivity (i.e., the genetic correlation) was .43, and the overlap between (unshared) environmental influences on the two traits (i.e., the nonshared environmental correlation) was .21. Fifty-four percent of the phenotypic correlation between the traits reflects genetic factors, and 46% reflects unshared environmental factors.
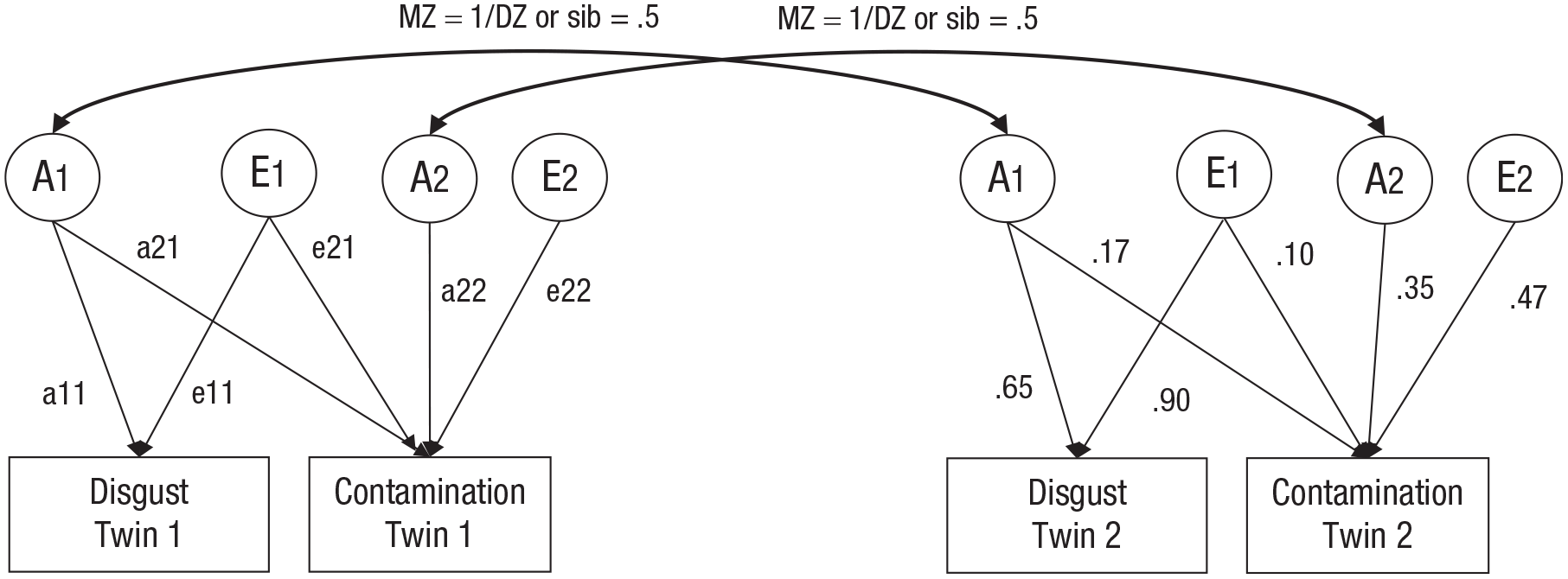
The bivariate genetic model. A1 and A2 are additive genetic factors with factor loadings a11, a21, and a22, and E1 and E2 are unique environmental factors with factor loadings e11, e21, and e22. For monozygotic (MZ) twin pairs, correlation between A in Twins 1 and 2 is 1; for dizygotic (DZ) and sibling pairs, this correlation is .5 (Neale & Cardon, 1992). The nonshared environmental effects (E) in Twin 1 are uncorrelated with those in Twin 2. Standardized path coefficient estimates are displayed on the right half of the figure.
Discussion
More than two decades’ worth of research has uncovered a relationship between disgust proneness and symptoms of anxiety disorders—especially contamination sensitivity. This is the first genetically informed study to examine the relationship between the two traits, and it is thus uniquely situated to inform the hypothesis that disgust proneness arises from social transmission from parents to offspring and that this social transmission leads to higher contamination sensitivity (Olatunji et al., 2017; Widen & Olatunji, 2016). Twin modeling revealed no evidence that environmental factors shared by twins—which presumably include exposure to the same parents’ fastidious cleaning behavior, expressions of disgust, or controlling parenting style—underlie this covariance. Instead, 54% of the covariance was attributable to genetic factors, and the remainder was accounted for by nonshared environmental factors.
At first blush, these results might seem at odds with the observations motivating the parental-modeling hypothesis, including young children’s apparent absence of disgust and sense of contamination (Rottman, 2014), parent–offspring resemblance in contamination sensitivity (Rozin et al., 1984), and greater expressions of disgust from parents of younger children than parents of older children (Stevenson et al., 2010). Further reflection suggests that none of these findings strongly supports the parental-modeling perspective. Many traits not present before age 5 develop independently of parental modeling (e.g., psychological changes during puberty). Even if exposure to others’ expressions of disgust influences the development of disgust proneness, then exposure to thousands of nonparents could overwhelm any parental effects (e.g., Askew et al., 2014). Most critical to this study is that within-family similarities do not necessarily indicate that a phenotype is socially transmitted by parents—indeed, decades of behavioral genetics work has shown that within-family similarities are largely the product of shared genetic rather than shared environmental influences (Plomin et al., 2016).
Naturally, results reported here should not be interpreted as discounting the importance of environmental influences on disgust proneness and contamination sensitivity. Indeed, more than half of the variance in each trait was attributable to environmental factors that twins do not share (and measurement error), and some of these factors influenced both traits. These results suggest that rather than searching for the parental behaviors that lead to high levels of disgust and contamination sensitivity, researchers can instead focus their efforts on generating and testing hypotheses of environmental factors that create differences between siblings. We discuss two that have been generated via adaptationist perspectives (e.g., Tybur, Lieberman, Kurzban, & DeScioli, 2013) but have received only preliminary testing.
First, a history of infectious disease might promote stronger motivations to avoid pathogen cues, either via disgust responses or via discomfort with indirect social contact. Some work has supported this hypothesis (Stevenson, Case, & Oaten, 2009); other work has not (De Barra, Islam, & Curtis, 2014; for a review, see Tybur, Çınar, Karinen, & Perone, 2018). Second, having a child might either increase disgust proneness (given a protective effect on offspring) or decrease it (given the need to contact offspring bodily wastes). One study reporting that mothers are less disgust prone than childless women (Prokop & Fancˇovicˇová, 2016) provides preliminary support for the latter hypothesis. Similar thinking might inform the genetic correlation between disgust proneness and contamination sensitivity, which could reflect a pleiotropic effect in which the same genes affect both traits. Across taxa, animals engage in multiple strategies to neutralize pathogens, including resistance (e.g., via inflammatory responses) and avoidance (e.g., via motivational states such as disgust or via behavioral avoidance). If individuals compensate for a deficit in the former by increasing the latter, then those genes underlying some aspects of immune function could simultaneously influence both the motivational states specialized for pathogen avoidance (i.e., disgust) and ruminations regarding routes of pathogen transmission. Alternatively, if greater disgust proneness leads to greater contamination sensitivity (Widen & Olatunji, 2016), then those genes underlying disgust proneness will partially also underlie contamination sensitivity. And, of course, the cross-sectional design employed here cannot rule out the possibility that contamination sensitivity—and the genes underlying it—causally influences disgust proneness. Longitudinal designs would be required to distinguish among these accounts.
The current study also offers some insights into sex differences in the heritability of disgust. Of the two recent twin studies on disgust, one sampled only women (Sherlock, Zietsch, Tybur, & Jern, 2016), and the other sampled only 180 pairs of twins of both sexes (Olatunji, Taylor, & Zald, 2019). The former could not test for sex differences, and the latter concluded that trait disgust is heritable for women but not for men. Note that these conclusions were limited by a modest number of male twins (41 MZ and 19 DZ male twin pairs), which afforded low power to detect moderate genetic effects (Verhulst, 2017), and an absence of formal tests of quantitative sex differences. With a much larger sample, we find no evidence for sex-specific heritabilities in disgust proneness, contamination sensitivity, or their covariance (see Table S2 in the Supplemental Material). Hence, disgust-proneness and contamination-sensitivity phenotypes differ across the sexes; however, the degree to which genes underlie these traits does not.
We note a few caveats to our conclusions. First, the methods employed here cannot speak to the possibility that within-family differences in disgust proneness stem from differences in how often children within the same household observe specific parental behaviors or how they react to those behaviors (Turkheimer & Waldron, 2000). Second, our decision to interpret AE over ADE models was based on a conservative criterion for differences in model fit (p < .01), although it was further justified by the complexities of interpreting nonadditive genetic effects from classical twin designs (e.g., Keller & Coventry, 2005). Third, we did not measure parents’ disgust proneness or parenting style. Future work using more complex designs that also assess parental phenotypes and can simultaneously estimate dominant and additive genetic components and shared environment components as well as correlations and interactions among those (e.g., nuclear twin family designs or children-of-twins design) could address these shortcomings. Finally, like most studies investigating the relationship between disgust proneness and contamination sensitivity, we sampled adults only. Thus, we cannot rule out the possibility that aspects of the shared environment influence children’s disgust proneness and/or contamination sensitivity but that these influences dissipate in adulthood, just as shared environment effects for so many other characteristics do (Bergen et al., 2007). Genetically informed studies of children would be necessary to compare shared environment effects in childhood as opposed to adulthood.
According to a recent meta-analysis (Olatunji et al., 2017), no study investigated the relationship between disgust proneness and anxiety disorder symptoms before 1996. In just over 20 years since then, at least 83 published studies have done so, and they have shown that disgust-prone individuals have higher anxiety-related symptoms, most notably contamination sensitivity. Generating and testing hypotheses to explain why these traits covary is a necessary step toward leveraging the psychology of disgust to better understand psychopathology (Amaroso et al., 2019). Although the current study suggests that aspects of the shared environment do not underlie disgust proneness, contamination sensitivity, or their covariance, it offers directions for multiple other tests of the environmental and genetic underpinnings of disgust and its related disorders.
Supplemental Material
Tybur_Open_Practices_Disclosure – Supplemental material for Genetic and Environmental Influences on Disgust Proneness, Contamination Sensitivity, and Their Covariance
Supplemental material, Tybur_Open_Practices_Disclosure for Genetic and Environmental Influences on Disgust Proneness, Contamination Sensitivity, and Their Covariance by Joshua M. Tybur, Laura W. Wesseldijk and Patrick Jern in Clinical Psychological Science
Supplemental Material
Tybur_Supplemental_Material – Supplemental material for Genetic and Environmental Influences on Disgust Proneness, Contamination Sensitivity, and Their Covariance
Supplemental material, Tybur_Supplemental_Material for Genetic and Environmental Influences on Disgust Proneness, Contamination Sensitivity, and Their Covariance by Joshua M. Tybur, Laura W. Wesseldijk and Patrick Jern in Clinical Psychological Science
Footnotes
Acknowledgements
We are grateful to Annika Karinen and Kimmo Karinen for assistance in translating research instruments.
Transparency
Action Editor: Michael F. Pogue-Geile
Editor: Scott O. Lilienfeld
Author Contributions
J. M. Tybur developed the study concept. P. Jern coordinated all data collection efforts. L. W. Wesseldijk conducted analyses, and J. M. Tybur and P. Jern provided feedback on these analyses. J. M. Tybur drafted the manuscript, and L. W. Wesseldijk and P. Jern contributed to revisions. All of the authors approved the final manuscript for submission.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
