Abstract
This study examined the effect of zinc supplementation on the sleep quality (SQ) of intensive care unit (ICU) nurses. In a double-blind, randomized controlled trial, 54 ICU nurses were randomly assigned to the intervention group (n = 27) that received 220 mg zinc sulfate capsules every 72 hours for 1 month and the control group (n = 26) that received placebo. Sleep quality of ICU nurses was measured by calculating the total quality sleep score using the Pittsburgh Sleep Quality Index (PSQI). The serum zinc level was a secondary outcome. All measurements were calculated at both baseline and 1 month after intervention. The total PSQI, subjective SQ, and sleep latency scores for the intervention group were significantly lower than the control group, which means that the SQ of ICU nurses in the intervention group improved from baseline. The serum zinc level of the intervention group at the end of the study was significantly higher than the control group, which means that zinc supplementation improved participants’ serum zinc level. Based on these findings, the researcher suggested that zinc supplementation could be associated with better SQ, subjective SQ, and sleep latency in this group of workers.
Nurses, the largest group of health care providers, are responsible for delivery of high quality patient care (Gordon, Lorilla, & Lehman, 2012; Nejati, Shepley, & Rodiek, 2016). Nevertheless, round-the-clock nursing care via a variety of rotational shifts could seriously disturb nurses’ sleep quality (SQ; K. Han, Trinkoff, & Geiger-Brown, 2014; Harris, Sims, Parr, & Davies, 2015; Muecke, 2005; Niu et al., 2013). It has been shown that nurses working other than daylight hours experience circadian rhythm misalignment (Yumang-Ross & Burns, 2014). Circadian rhythm misalignment is linked with less alertness and poorer performance (Ji, Grandner, & Liu, 2016). Sleep deprivation impairs workers’ ability to plan, make decisions, and integrate information (Stimpfel, Brewer, & Kovner, 2015). According to reports, approximately 50% of nurses suffer from sleep disorders, 83% reported high levels of work stress (Fang & Li, 2015), and 23% take sleep medications for insomnia (Kageyama, Nishikido, Kobayashi, Oga, & Kawashima, 2001).
Nursing is a stressful profession, and stress is related to impaired sleep (Hasson & Gustavsson, 2010). Intensive care units (ICUs) may be more stressful for nurses because nursing care in ICUs involve activities with various levels of difficulty and require knowledge and skills necessary to care for critically ill patients (Galletta, Portoghese, Coppola, Finco, & Campagna, 2016; Gholipour Baradari et al., 2016). Evidence demonstrates that the prevalence of sleep deprivation and poor SQ among ICU nurses is higher than for nurses working on other hospital services (Akbarzadeh et al., 2014; Galletta et al., 2016; Y. Han, Yuan, Zhang, & Fu, 2016; Kageyama et al., 2001; Karagozoglu & Bingol, 2008; Zverev & Misiri, 2009). Y. Han et al. (2016) found that the total rate of sleep disorders among clinical nurses, particularly female nurses working in ICUs, is high and these sleep disorders are linked to occupational stress. Given nurses’ role in patient care, factors affecting their psychological, physical, and spiritual well-being should be studied (Happell, Gaskin, Reid-Searl, & Dwyer, 2014).
Evidence suggests that sleep disorders such as insomnia and sleep loss may be associated with changes in diet. In addition, some specific nutrients and micronutrients have been found to be associated with sleep disorders (Grandner, Jackson, Gerstner, & Knutson, 2014; Huang, Wahlqvist, & Lee, 2013). As a scarce micronutrient, zinc (Zn) possesses antioxidant and anti-inflammatory properties, which are crucial for physical and psychological well-being (Prasad, 2008). Inadequate intake of Zn or malabsorption of this micronutrient can decrease serum Zn concentrations and cause Zn deficiency (Roohani, Hurrell, Kelishadi, & Schulin, 2013).
Although several measures have been introduced to define Zn deficiency, deficiency prevalence ranges from 17.3% to 58% in the general population (Barnett et al., 2016; Dabbaghmanesh, Taheri Boshrooyeh, Kalantarhormozi, & Ranjbar Omrani, 2011; Wessells & Brown, 2012). Strong evidence has demonstrated that Zn deficiency contributes to sleep, metabolic, hematologic, emotional, psychological, immune, gastrointestinal, and neurological disorders (Kordas et al., 2009; Markiewicz-Żukowska, Gutowska, & Borawska, 2015; Prasad, 2008; Sawada & Yokoi, 2010). In contrast, several studies have shown that Zn supplementation significantly increases serum Zn. For example, Barnett et al. (2016) showed that Zn supplementation of 30 mg per day over 3 months could increase serum Zn concentrations in nursing home residents with low serum Zn levels. On the contrary, some studies have suggested that sleep duration is associated with serum Zn level. Luojus, Lehto, Tolmunen, Elomaa, and Kauhanen (2015) reported that the level of serum Zn was low in men who sleep less than 6 hours per night. In another study, Song, Kim, and Jung (2012) found a significant association between serum Zn level and sleep duration in adult women. In addition, a positive association has been reported between serum Zn level and neuropsychological disorders (Cope & Levenson, 2010; Gholipour Baradari et al., 2013; Sawada & Yokoi, 2010). Several studies revealed that Zn supplementation has beneficial effects on sleep disorders in children (de Moura et al., 2013; Kordas et al., 2009), asthma (Pouramjad, Egtesadi, Moosavi, Nour Mohammadi, & Yazdani, 2009), childhood hyperactivity and attention disorder (Akhondzadeh, Mohammadi, & Khademi, 2004), immunity (Prasad, 2008), and mental conditions such as depression (Sawada & Yokoi, 2010).
Although previous studies have shown a high prevalence of sleep disorders among ICU nurses, no empirical studies have examined the effect of Zn supplementation on SQ of nurses in general, and ICU nurses in particular. Therefore, based on the findings of previous studies, which confirmed that micronutrients such as Zn could improve SQ, the authors hypothesized that Zn supplementation of 220 mg every 72 hours over a period of 1 month would significantly increase serum Zn concentration and improve the SQ of ICU nurses. Study findings could lead to future studies on simple low-cost Zn supplementation interventions to improve SQ.
Method
A multi-center, double-blind, randomized, two parallel group, placebo-controlled trial was conducted to evaluate the effect of Zn supplementation on the SQ of ICU nurses.
Sample and Setting
The study population included staff nurses (N = 176) working in ICUs of two academic hospitals affiliated with Mazandaran University of Medical Sciences, Sari, Iran. Sample enrollment was conducted between February and March 2012. Using a Student t test for two independent samples and a significance level of 5% (two-sided), the authors determined the sample size of at least 27 participants in each group (total of 54) using G*Power (ver. 3.1) calculations with a medium effect size of 0.5, and power (β) of 0.80.
At baseline, potential participants were assessed for serum Zn levels (i.e., ≤79.9 µg/L) and SQ (i.e., total SQ score greater than 5) using the Pittsburgh Sleep Quality Index (PSQI) and other inclusion criteria (i.e., nurses who had worked in the ICU more than 1 year; worked at least five night shifts a month; and held a bachelors or master’s degree from one of two hospitals). By evaluating 112 of the total 176 nurses at two ICUs, the predetermined sample size of 54 study participants (27 nurses from each ICU) was reached. Nurses were excluded if they reported specific chronic illnesses (i.e., diabetes, psychiatric disorders, autoimmune disease, or rheumatic disorders), took any micronutrients (vitamin or mineral supplements) within the last 3 months, smoked cigarettes during the study period, suffered from diarrhea or vomiting, or were pregnant or breastfeeding.
The study was conducted in accordance with the Declaration of Helsinki. The Institutional Review Boards and Ethics Committees on Research involving human subjects at each of the participating sites approved the study protocol (approval number 90177). All study nurses were required to provide both written informed consent and verbal information prior to study enrollment. The signed form was reviewed and approved by the Ethics Review Committee of the Mazandaran University of Medical Sciences. The authors also requested information regarding daily dosage of Zn, possible Zn side effects, time of taking Zn capsule, commitment to completing study, and agreement to refrain from all other supplements.
Randomization and Blinding
At the time of enrollment, each participant was assigned an identification number using a computer-generated random number table. Fifty-four nurses were randomly assigned at a 1:1 ratio to either the Zn supplement group (Zn, n = 27) or placebo group (P, n = 27) by an external individual who was blinded to each group allocation and had no other role in the study. Nurses were asked to take one soft gelatin capsule every 72 hours before sleep (between 21:00 and 23:00) for 1 month. Capsules, which were similar in shape, color, and size, contained the dietary Zn supplement (Zn group) or starch (P group). Nurses assured the researchers that they would take the capsules as prescribed. The intake of 220 mg one soft gelatin Zn sulfate capsule every 72 hours before sleep for 1 month was considered safe (Tomat, Costa Mde, & Arranz, 2011). Investigators were blinded to the randomization table, code assignments, and procedure. This study design and report were followed by the Consolidated Standards of Reporting Trials guideline, which includes numerous initiatives developed by the CONSORT Group to alleviate the problems of inadequate reporting of randomized controlled trials.
Intervention
Nurses in the Zn group were prescribed one capsule containing 220 mg of Zn sulfate every 72 hours for 1 month and nurses in the control group were prescribed one capsule containing placebo similarly. According to Boy et al. (2009), the most effective dosage, frequency, and duration of Zn supplementation to treat sleep disorders have not been determined. Therefore, the researchers used this dosage (220 mg) and time interval based on pharmacist advice and the Tomat and colleagues study. According to Tomat et al. (2011), the intake of one 220 mg soft gelatin Zn sulfate capsule every 72 hours is considered safe. Also, Fosmire (1990) reported that potential side effects of Zn occur with high daily doses (e.g., 225 mg-450 mg). The placebo employed in this study was provided by pharmacy faculty, using starch and adequate coating, similar to the Zn capsules. After 1 month, total SQ, the seven SQ components, and Zn and copper serum levels were re-examined.
Study Outcome
The primary outcome was the SQ of ICU nurses, which was obtained by calculating the total quality sleep score from the PSQI. In addition, the seven major SQ components (from C1 to C7) were evaluated. The serum Zn level was the secondary outcome.
Measurements
Nurses completed the demographic form, which included questions about age, sex, marital status, number of children, education, weight and height, work experience, work hours per week, shifts, number of shifts per day, distance between home and hospital, type of transportation to work, and the previous 24-hour diet.
The PSQI is a well validated, self-report, multiple-indicator sleep instrument, which measures the quality and pattern of sleep on the majority of days or nights over the past month. The instrument consists of nine items, which differentiate “poor” from “good” SQ by measuring seven components (subscales), each of which has a range of 0 to 3. These components include subjective SQ, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleeping medication, and daytime dysfunction. The total score of PSQI is scaled from 0 to 21. As discussed by Buysse, Reynolds, Monk, Berman, and Kupfer (1989), the score of 5 or greater shows problems in at least two components and is indicative of poor SQ with a sensitivity of 89.6% and a specificity of 86.5% in three groups of “good” sleepers (healthy individuals, n = 52) and “poor” sleepers (depressed patients,n = 54; sleep-disorder patients, n = 62). The PSQI has been validated in several studies worldwide (Akbarzadeh et al., 2014; Dietch et al., 2016; Emami Zeydi et al., 2014).
Participants’ serum Zn and Cu levels were measured before and after intervention to control for the confounding effects of Cu because the interaction between Zn and Cu is well documented. It has been shown that high levels of Zn inhibit the intestinal absorption of Cu and induce clinical and biochemical signs of Cu deficiency (Bremner & Beattie, 2007). Before any intervention, a blood sample of 5 mL was taken from an antecubital vein of nurses to determine the Zn and Cu serum levels. Serum samples were collected using trace metal–free tubes. The blood sample was centrifuged at 3,000 rounds per minute (rpm) for 5 minutes to separate the plasma. Then serum was diluted in normal sulfuric acid (0.1). The serum levels of Zn and Cu were measured using the Auto-Analyzer Unit (Cobas Integra, Germany). For this study, Zn serum concentration less than 70 µg/dL was considered to be a severe deficit, between 70 µg/dL to 79.9 µg/dL a slight deficit, 80 µg/dL to 119.9 µg/dL near normal, and more than 120 µg/dL normal (Folin, Contiero, & Vaselli, 1994). The serum level of total copper between 63.7 µg/dL and 140.12 µg/dL was considered normal (Murray, Jacob, & Varghese, 2011). Samples were measured at both baseline and after 30 days of treatment.
Statistical Analysis
Baseline characteristics of nurses with and without intervention were compared using chi-square or Fisher’s exact test for categorical data and t test or Mann–Whitney U test for continuous data. Normality distribution of variables was assessed with the Shapiro–Wilk test. Wilcoxon signed rank test was used to compare median PSQI prior to intervention and postintervention. McNemar’s test was used to compare poor sleep proportion prior to intervention and postintervention. The authors used unpaired tests to compare participants at the same point in time. For analyses that used all of the time points in the data set, the authors used random effects and time-series models using the method of generalized estimating equations (GEE). These models estimated main effects of the drug (Zn vs. placebo), time (before and after study), and their interactions. The interaction effect tests the main hypothesis of the study, that Zn supplementation of 220 mg per 72 hours over a period of 1 month would significantly increase serum Zn concentrations and improve the SQ of ICU nurses as indicated by lower PSQI total score. Statistical analyses were carried out using SPSS (ver. 19). All statistical tests were two-sided with Type I error rate set at 0.05.
Results
Of the 54 study participants, one from the control group was lost during the study period as illustrated in Figure 1. In total, 53 nurses completed the 1-month study; data from all of these individuals were analyzed. No adverse effects of Zn supplementation were reported during the trial period. Figure 1 shows the flow of participants.
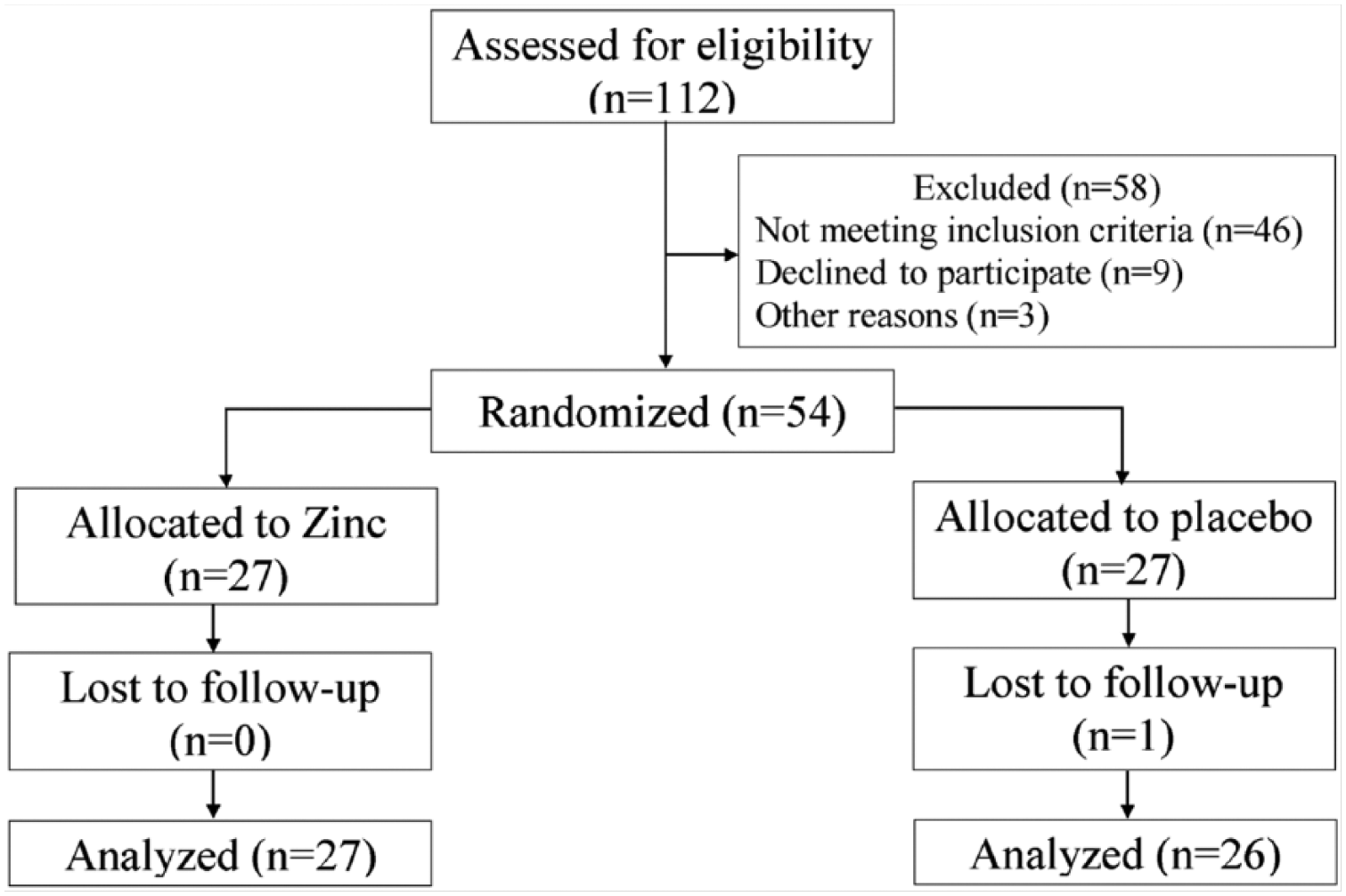
Flowchart for participant selection.
The mean age of all participants was 31.20 ± 5.42 years, more than 90% of the nurses were female, most of the nurses had bachelor’s degrees (96.2%), and more than half of them (60%) were married. More than 98% of the study participants completed the study. Table 1 summarizes the baseline demographic and clinical characteristics of the nurses. Differences between the two groups were not statistically significant in terms of body mass index (BMI), age, sex ratio, academic degree, or shift work (p > .05).
Basic Demographic and Clinical Characteristics of Participants
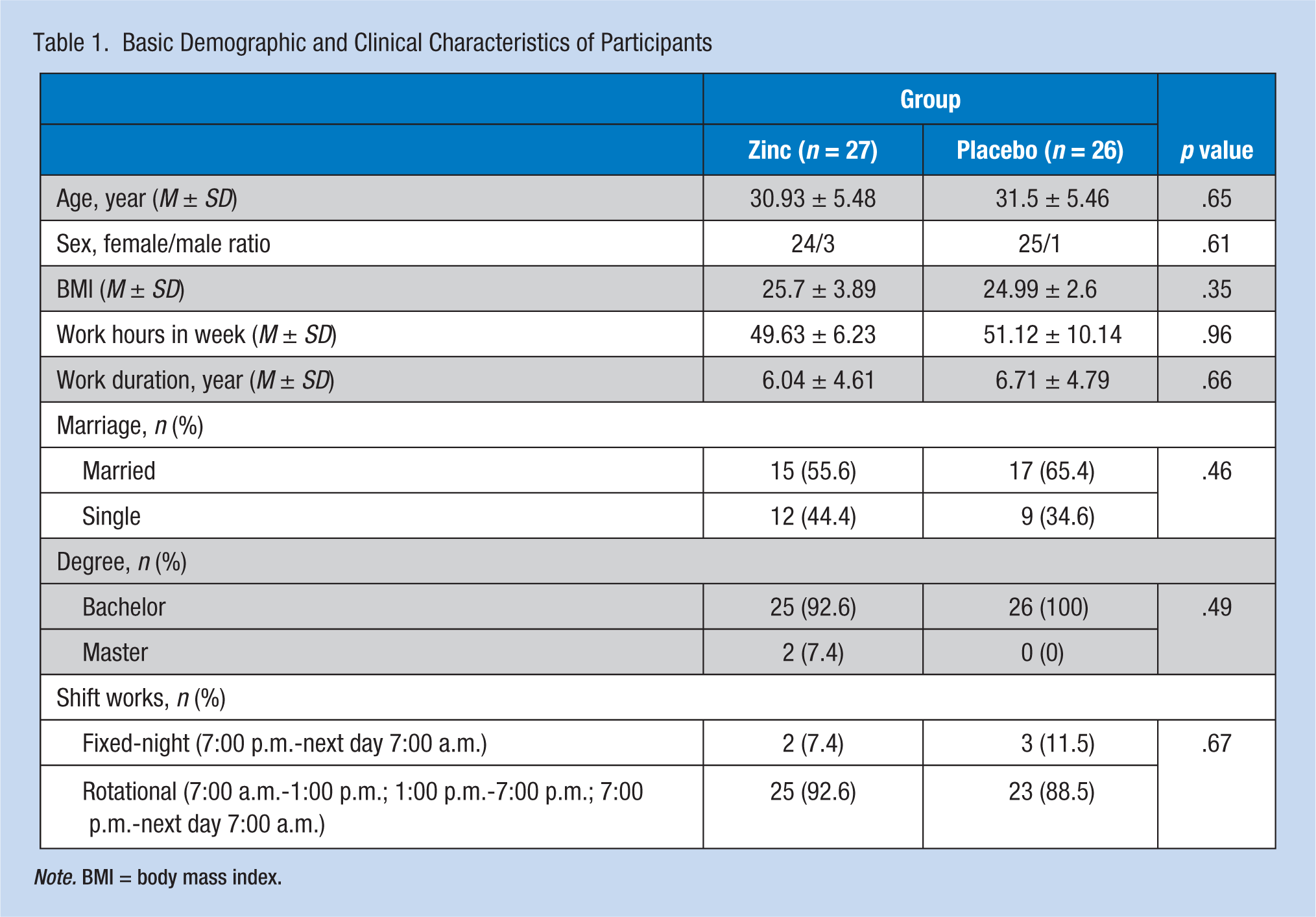
Note. BMI = body mass index.
Zinc and Copper Serum Level for All Nurses
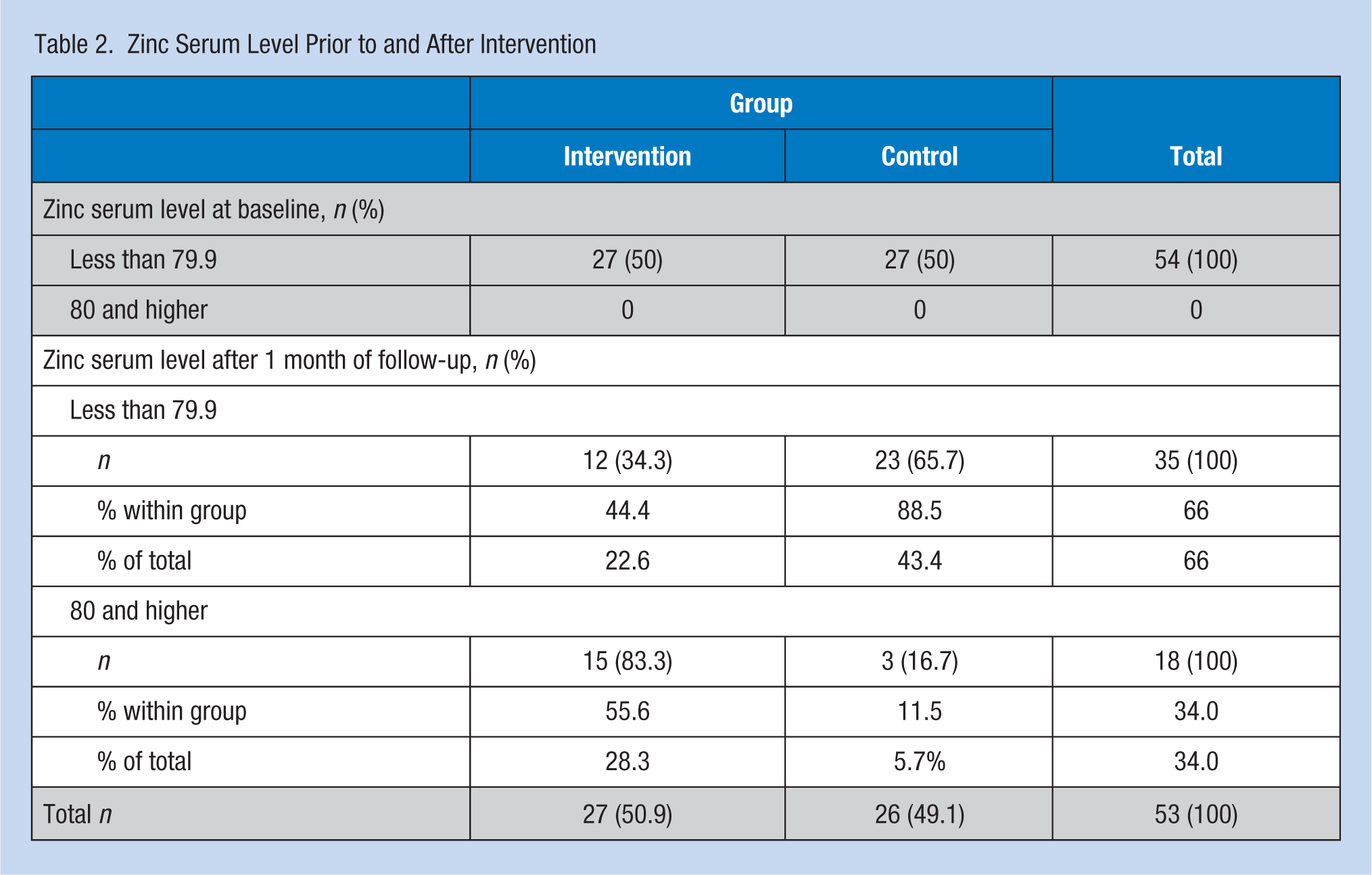
At baseline, more than 48% (n = 54) of screened participants (n = 112) had zinc levels less than 79.9 μg/dL indicating a zinc deficiency. The researchers included those 54 nurses with Zn serum levels less than 79.9 μg/dL in the trial. After only 1 month of intervention, this proportion declined to less than 66% (n = 35) of the total sample. One month of Zn supplementation decreased the percentage of nurses with Zn deficiency from 100% to 22.6% (n = 12) in the supplement group; zinc deficiency remained unchanged in the placebo group (Table 2). A significant difference was observed between Zn and placebo groups after 1 month of intervention (p < .001). In addition, an increase of 22.25 μg/dL in Zn serum levels was also observed after 1 month of treatment (from 60.19 to 82.44 μg/dL). The Cu serum level changes were not statistically significant between the two groups after the intervention month. Table 3 compares Zn and Cu serum levels, total sleep quality score (TSQS), and seven components of PSQI for the two groups.
Zinc Serum Level Prior to and After Intervention
The Seven PSQI Components, and the Serum Level of Zinc and Copper
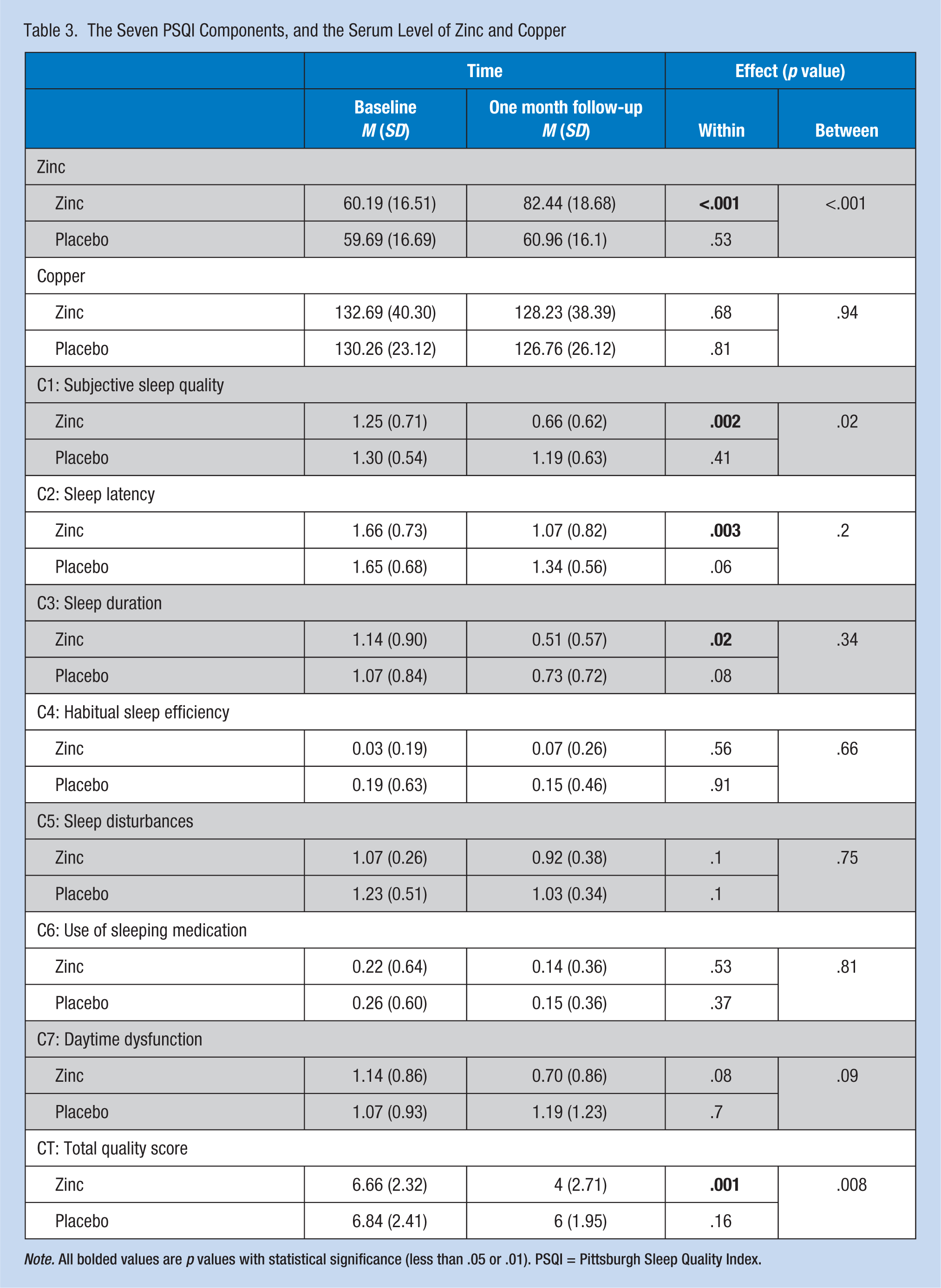
Note. All bolded values are p values with statistical significance (less than .05 or .01). PSQI = Pittsburgh Sleep Quality Index.
Subjective Sleep Data
No differences in PSQI scores between the two groups were found at baseline. As specified in Table 3, the total quality sleep baseline score (abbreviated as CT) was 6.66 ± 2.32 for the Zn group and 6.84 ± 2.41 for the placebo group. The CT score was 4 ± 2.7 for the Zn group and 6 ± 1.95 for the placebo group following 1 month of intervention. The Zn supplementation significantly improved the total SQ score more than 2.5 points for the Zn group compared with the placebo group (p = .008). One month of Zn supplementation improved the total score and the scores of almost all components of the PSQI, except C4. The within-group differences were statistically significant for the Zn group on C1 (subjective SQ), C2 (sleep latency), and total score. The between-group differences at 1 month after intervention remained statistically significant for the subjective SQ component and total score (p < .05).
Poor Sleep Proportion
The proportion of nurses who reported poor sleep prior to intervention and postintervention were assessed. As shown in Table 4 and illustrated in Figure 2, only four nurses in the Zn group reported good SQ before the intervention; however, after 1 month of Zn supplementation, 17 nurses reported good SQ. Of 23 nurses with poor SQ before intervention, only 10 nurses reported poor SQ after 1 month of Zn supplementation. In the Zn group, 13 nurses who reported with poor SQ before the intervention reported good SQ after the intervention (p < .001). Almost 50% of all nurses (n = 27) reported poor SQ. The decreasing poor sleep proportion was greater in the Zn group than the placebo group, but this finding was not statistically significant (p = .13). Zinc serum levels were associated with poor sleep. The GEE model showed that for each unit increase in the serum Zn level, nurses had a 3% decrease in the odds of poor sleep (odds ratio [OR] = 0.97; 95% confidence interval [CI]: [0.94, 0.99]; p = .016).
Poor Sleep Proportion Prior to and After Intervention
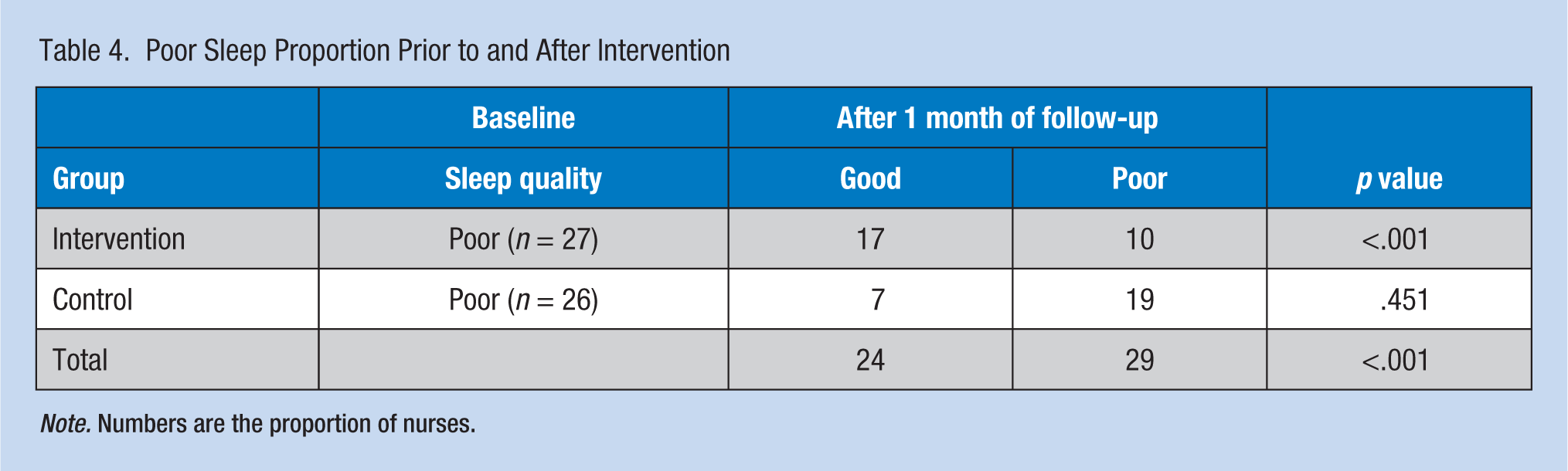
Note. Numbers are the proportion of nurses.
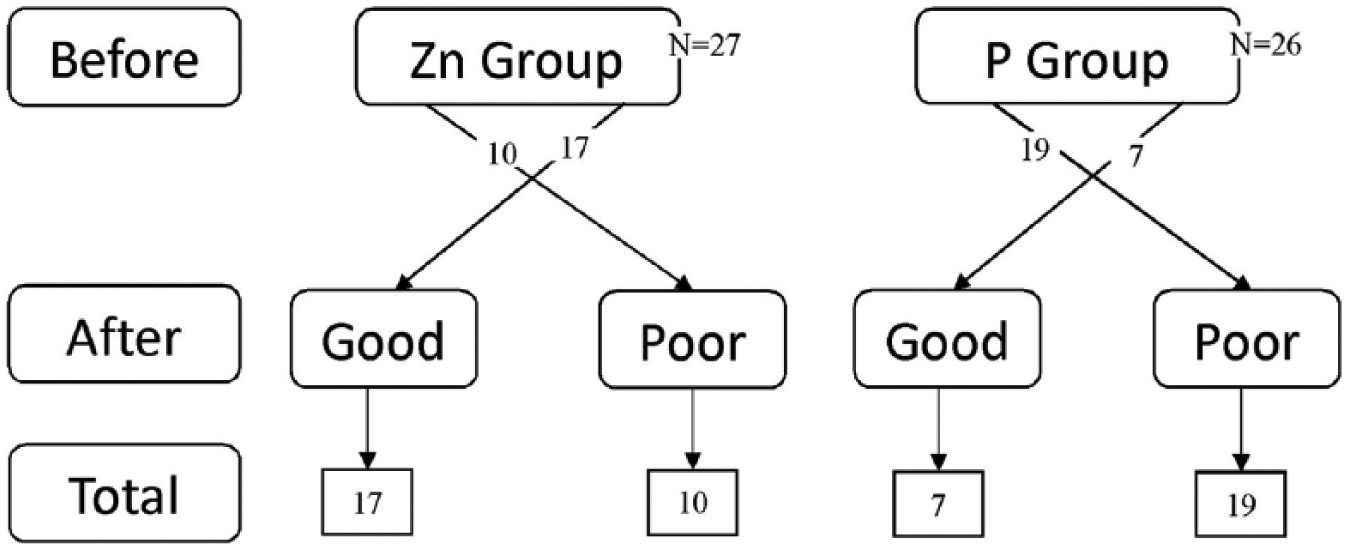
Poor sleep proportion before and after intervention.
Discussion
This is the first study to investigate the effect of Zn supplementation on SQ in ICU nurses. The main finding of this study was that Zn supplementation significantly improved total SQ, subjective SQ, and sleep latency as measured by lower total PSQI score of the Zn group than the placebo group. The present study showed that after 1 month of intervention, both SQ and serum Zn levels improved. At baseline, of 112 screened nurses, more than 48% had Zn deficiency (serum Zn < 79.9 μg/dL). However, after 1 month of Zn supplementation, Zn levels increased to 82.44 μg/dL in the Zn group. On the contrary, 1 month of Zn supplementation decreased the percentage of Zn deficiency more than 55% in the Zn group; in the placebo group, Zn levels were unchanged. Therefore, the authors suggest that improvement in the SQ of ICU nurses could be associated with increased serum Zn levels. This finding is consistent with results from the Barnett et al. (2016) study that revealed supplementation with a sufficient amount of Zn could increase serum Zn levels.
The researchers found that approximately half of ICU nurses reported low SQ. This finding is consistent with findings of previous studies, which estimated that poor SQ was reported by 37% to 58% of nurses (Ghaljaei, Naderifar, & Ghaljeh, 2011; Karagozoglu & Bingol, 2008; Luzzi & Spencer, 2011; Singh et al., 1991). Poor SQ in ICU nurses could be related to several physical and physiological stressors, including high stress environment, working with extremely ill patients, continuous light, noise (e.g., alarm devices), daily confrontation with death, mourning relatives, and rotational work shifts (Akbarzadeh et al., 2014; Cavalheiro, Moura Junior, & Lopes, 2008; Shao, Chou, Yeh, & Tzeng, 2010).
The present study found significant changes in subjective SQ and sleep latency; however, in other aspects of SQ, the intervention was not effective. This finding may be related to the low dosage of Zn supplementation, short duration of the study, specific conditions of the ICU, and stressors specific to ICUs, which merit further studies. Previously, Markiewicz-Żukowska et al. (2015) demonstrated that cognitive dysfunction is associated with low serum Zn levels of nursing home residents (e.g., residents with higher serum Zn concentrations had better mental activity).
It should be noted that the effect of Zn supplementation on SQ of ICU nurses is understudied. Most studies of Zn supplementation have only focused on SQ in the general population. Rondanelli et al. (2011) reported that the simultaneous prescription of melatonin, magnesium, and Zn at night improved the SQ and quality of life in long-term care facility residents with primary insomnia; however, their study did not examine the effect of these supplements separately. These researchers only reported that probably Zn supplementation could improve quality of sleep, which is consistent with study results. Ji and Liu (2015) demonstrated a connection between Zn concentrations and SQ. They found that higher blood Zn concentrations were associated with normal SQ. They also found that lower serum Zn increased the likelihood of poor SQ in preschoolers.
Several studies have examined the side effects of Zn deficiency among children and adults (Hashemi, Jalilvand, Hakimi, & Nourmohammadi, 2006). By contrast, it was shown that Zn supplementation could improve mental fitness (Sawada & Yokoi, 2010), enhance the effect of anti-depressant drugs, reduce mental-social stresses, and improve depression (Mousavi, Habibbollahi, & Mahmoudian, 2006). One potential mechanism of Zn supplementation is the reduction of oxidative stress (Guo & Wang, 2013).
Although Zn has health benefits, very few studies have examined the effect of this valuable micronutrient on overall SQ in nurses, particularly in high stress work settings such as the ICU. As reported by several researchers, longer use of this supplement along with other micronutrients such as melatonin and magnesium could improve all aspects of SQ (Rondanelli et al., 2011). Therefore, prescription of this supplement for nurses who worked in the ICU, specifically nurses with sleep disorders and poor SQ, is recommended. In addition, the prevention and treatment of ICU nurses’ Zn deficiency with micronutrient supplementation may offer an opportunity to enhance the overall quality of patient care and clinical outcomes. Based on the dearth of evidence regarding the prevalence of Zn deficiency among nurses, the researchers believe Zn deficiency continues to be unrecognized around the world.
This study had some strengths and limitations. First, the data for this study were gathered from two hospitals, which slightly increases the generalizability of the results. Second, this study is the first to evaluate the effect of Zn supplementation on the SQ of ICU nurses. As a result, the authors could not compare these study results with other studies using the same population. Third, the authors could not supervise nurse participants to ensure they took their capsules as prescribed. Fourth, this study examined the effect of Zn supplementation on improving SQ for only 1 month. Fifth, the authors could not examine the efficacy of Zn supplementation due to lack of precise control over unknown confounding variables, the small sample size, and the short duration of the study. Sixth, the researchers could not control eating habits and diets of participants; however, participants were instructed to maintain their normal eating habits and diets. Considering these limitations, further studies are warranted to confirm study findings.
Implications for Practice
Poor SQ among ICU nurses may predispose these nurses to health care error. These randomized controlled trial data showed that among ICU nurses reporting poor SQ and with low blood Zn, those who took Zn supplements reported improved SQ compared with those nurses who did not take Zn. Therefore, Zn supplementation may improve the SQ of ICU nurses. These preliminary data warrant further well-designed trials with objective sleep measures to confirm these preliminary findings and determine if Zn supplements might improve SQ for this occupational group. Of course, further pharmacological studies will also be needed to consider the optimum dosage and duration of Zn supplements for sleep disorders.
Applying Research to Practice
Poor sleep quality among intensive care unit (ICU) nurses may predispose them to patient care error. This randomized clinical trial provides evidence that an intervention program for intensive care unit nurses can improve their sleep quality (SQ). The intervention program is a one-month zinc (Zn) supplementation with 220 mg zinc sulfate per 72 hours. Findings showed that among ICU nurses reporting poor SQ and with low blood Zn, those who took Zn supplements reported improved SQ compared with those nurses who did not take Zn. Therefore, Zn supplementation may improve the SQ of ICU nurses. Further pharmacological studies are needed to consider the optimum dosage and duration of Zn supplements for sleep disorders.
Footnotes
Acknowledgements
The authors appreciate the sincere cooperation of intensive care unit nursing staffs in Imam Khomeini and Fatemeh Zahra hospital in Sari. In publishing this article, we would like to thank the Department of Clinical Research Development at Imam Khomeini Hospital in Sari for providing full support during this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this trial was provided by the Mazandaran University of Medical Sciences.
Author Contributions
Dr. Afshin Gholipour Baradari and Mr. Hassan Sharifi were the principal investigators, had access to the data in the study, and took responsibility for the integrity of the data and the accuracy of the data analysis. Afshin Gholipour Baradari, Seyed Mahmood Nouraei, Ali Mahdavi, and Hassan Sharifi were involved in study concept and design. An external individual was involved in the acquisition of data. Abbas Alipour was involved in the analysis and interpretation of data. Hassan Sharifi and Amir Emami Zeydi were involved in the drafting and critical revision of the manuscript. Abbas Alipour was involved in the statistical analysis.
Authors’ Note
Mazandaran University of Medical Sciences had no role in the design and conduct of the study; in the collection, analysis, and interpretation of the data; or in the preparation, review, or approval of the manuscript.
Author Biographies
Afshin Gholipour Baradari, is a professor of anesthesiology in the Department of Anesthesiology, Faculty of Medicine, Mazandaran University of Medical Sciences, Sari, Iran.
Abbas Alipour is an associate professor of epistemology in the Department of Epidemiology, Faculty of Medicine, Mazandaran University of Medical Sciences, Sari, Iran.
Ali Mahdavi is a critical care nurse at Behshahr Imam Khomeini Hospital, Mazandaran University of Medical Sciences, Sari, Iran.
Hassan Sharifi is a medical-surgical nurse and faculty member of the Nursing and Midwifery School, Iranshahr University of Medical Sciences, Iranshahr, Iran and PhD candidate in nursing, Mashhad University of Medical Sciences, Mashhad, Iran.
Seyed Mahmood Nouraei is an associate professor, cardiothoracic surgeon, Department of Cardiac Surgery, Faculty of Medicine, Mazandaran University of Medical Sciences, Sari, Iran.
Amir Emami Zeydi is a critical care nurse, Department of Medical-Surgical Nursing, Faculty of Nursing and Midwifery, Mazandaran University of Medical Sciences, Sari, Iran and PhD candidate in nursing, School of Nursing and Midwifery, Mashhad University of Medical Sciences, Mashhad, Iran.
