Abstract
Background
Cancer treatments, such as chemotherapy, may adversely affect gastrointestinal (GI), physical and mental health in survivors of cancer.
Objective
This study investigated associations between GI, mental and physical health outcomes, and cancer treatment-related variables, such as chemotherapy, in adult cancer survivors.
Methods
A one-time cross-sectional survey with patient-reported outcomes was used. Cancer survivors (N = 317) aged ≥18 years, living in Canada, who completed cancer treatments were included. Descriptive statistics, correlation, and linear regression analyses are reported.
Results
Mean age at diagnosis was 40.90 ± 15.40 years. Most survivors received chemotherapy (86.1%). Persistent GI symptoms include constipation (53.6%), diarrhea (50.5%), and bloating/pain (54.9%). Mean GI symptom duration was 30.53 ± 33.42 months. Severity of GI symptom interference was moderate to extreme for 51.9% of survivors. Compared to normative values of 50 in healthy people, survivors scored poorer for mental health (M = 42.72 ± 8.16) and physical health (M = 45.55 ± 7.93), and reported more belly pain (M = 56.10 ± 8.58), constipation (M = 54.38 ± 6.81), diarrhea (M = 55.69 ± 6.77), and gas/bloating (M = 56.08 ± 8.12). Greater GI symptom severity was associated with poorer mental and physical health (P < .01). Chemotherapy was associated with increased belly pain (B = 4.83, SE = 1.65, P < .01) and gas/bloating (B = 3.06, SE = 1.45, P = .04).
Conclusion
We provide novel evidence that many cancer survivors experience chronic, moderate to severe GI symptoms lasting for years after cancer treatment, which are associated with worse mental and physical health. Chemotherapy is associated with specific GI symptoms. Integrative therapies are needed to address GI symptoms in cancer survivors.
Introduction
Gastrointestinal and Psychosocial Symptoms in Cancer Survivors
Globally, in 2020 an estimated 19.3 million new cases of cancer and nearly 10.0 million cancer deaths occurred. 1 Survival rates have, however, improved considerably, with rates greater than 80% for several specific cancer types (e.g. prostate, breast, Hodgkin lymphoma) 2 and depending on geographical region. This means that many people are surviving for years or decades after a cancer diagnosis. While advances in early detection and medical treatments have improved survival rates considerably, many survivors of cancer live with chronic health conditions as a result of cancer and/or anti-cancer therapies. This has implications for survivors’ quality of life (QoL), financial toxicity for the survivor and their family, and health care and economic system burden.2-5
Survivors of cancer report a plethora of acute and chronic long-term and late-effects of cancer and its’ treatments. These may include physical and psychosocial symptoms, which can compromise overall quality of life.3,4,6-8 GI issues are one of the most common and yet under-reported issues that occur during treatment.8-10 During chemotherapy and/or radiation therapies, common GI issues experienced by patients include nausea and/or vomiting, diarrhea, constipation, pain, and mucositis, the painful inflammation and ulceration of the intestinal epithelium, among others.8-11 The prevalence of GI issues during cancer treatments varies depending on cancer type, treatment regimens, and overall health. Indeed, upwards of 70% of cancer patients (i.e. individuals on active cancer treatments) receiving chemotherapy experience mucositis, which has been associated with adverse changes in the gut microbiota.11-13 McQuade et al. found that during treatment the incidence of chemotherapy-induced diarrhea varied from 40% in patients receiving a standard dose to 100% in patients on high dose chemotherapy, while chemotherapy-induced constipation is estimated to occur in up to 80% of patients on active treatment. 9 Problematic GI issues during cancer treatments can result in treatment delays, dose reductions, impaired QoL, and sometimes the termination of treatment, compromising patients’ survival.8-10
Significant, chronic GI symptoms following the end of cancer treatments have also been reported. A comprehensive review by Andreyev et al. concluded that chronic GI issues are underestimated in the literature and in clinical trials, underreported by patients, and that commonly used GI questionnaires generally fail to investigate key issues that are distressing to patients, such as severe flatulence. 8 The prevalence, severity and type of chronic GI issues vary depending on cancer type and treatment, but are estimated to occur in 15 – 66% of survivors (i.e. people who have completed their anti-cancer therapies), 8 although the duration of time for which symptoms persist is generally unknown. However, given that patients may underreport their GI symptoms and the challenges with measurement and reporting of symptoms, the type, severity and duration of post-cancer treatment GI symptoms remains unclear.
It is well established that many cancer survivors experience psychosocial challenges following a cancer diagnosis and treatments. In a sample of adolescent and young adult (AYA) cancer survivors, Prasad et al. found higher rates of depression, anxiety, and cognitive issues related to task efficiency, emotional regulation and memory compared to healthy siblings. 4 This is consistent with other studies reporting poorer health-related QoL in young cancer survivors versus healthy siblings. 14 Mature adults similarly experience higher rates of psychosocial challenges compared to healthy peers. In a cohort of long-term (i.e. 5 years or more), older adult cancer survivors’ concerns about cancer recurrence, a secondary cancer, and cancer-related symptoms were found in one-third of the survivors and were associated with higher levels of anxiety and depression. 7 This is consistent with other studies reporting higher levels of anxiety, depression, and cognitive issues among adult cancer survivors.3,6 Notably, some of these symptoms are also experienced by patients with irritable bowel syndrome (IBS). For instance, patients with IBS frequently present with comorbid GI and psychosocial symptoms, such as abdominal pain, bloating, constipation and/or diarrhea, anxiety and depression.15-17 Although different chronic health conditions, parallels in symptoms between patients with IBS and cancer survivors may suggest similar underlying physiological mechanisms, such as gut microbiota dysbiosis and dysregulation of the neuroimmune system.12,16
Cancer treatments, such as chemotherapy, are shown to adversely affect the intestinal tract and gut microbiota.18,19 Cancer patients experience various treatment-related toxicities, side-effects, and late-effects; changes in the body as a result of treatment that may last for months or years after a cancer experience, such as cognition and mood.6,20 In both human and animal studies, chemotherapy has been shown to induce a dysbiotic state which may contribute to treatment-related toxicities.19,21–24 The current literature mainly focuses on gut dysbiosis and physical treatment-related symptoms. However, as adverse psychological and cognitive side-effects of cancer treatments are also frequently reported by people with cancer, research must shift to include the investigation of potential mechanisms driving these symptoms, such as dysbiosis of the gut microbiota or neurotoxic factors. However, it is important to first explore the chronicity and symptomology of GI and psychosocial symptoms in diverse samples of cancer survivors. This knowledge can then inform future research investigating potential mechanisms and strategies for intervention.
Present Study
Research studies have focused primarily on immediate GI symptoms related to cancer treatment, while very little attention has been paid to chronic GI symptoms that may arise during or after treatment and persist for an unknown length of time. Moreover, few studies have examined the consequences of persistent GI symptoms on cancer survivors’ psychosocial health, and potential associations between GI and psychosocial symptoms. The current literature further suggests that chronic GI issues after anti-cancer therapies are under-reported in the literature and clinical trials. 8
Recent data from our Chemo-Gut Pilot study 19 and patient engagement with the cancer online community indicate that the duration of time after chemotherapy during which survivors experience GI issues that may co-occur with psychosocial challenges, may extend from months to years after anti-cancer therapies have ended. Given the paucity of research on longer-term GI and psychosocial health outcomes in cancer survivors, the present study investigated post-anti-cancer therapy GI symptoms, and physical and mental health outcomes in cancer survivors in Canada. This study aimed to: (i) describe GI, physical and mental health outcomes in cancer survivors; (ii) determine whether GI symptoms are associated with mental health and physical health; (iii) investigate relationships between type of cancer treatment and GI, physical and mental health outcomes after cancer treatments, with the hypothesis that chemotherapy in particular would be associated with worse GI symptoms.
Methods
Participants
The self-report survey was administered via an online link to cancer survivors in Canada, recruited via social media, and through digital ads and pamphlets at the Tom Baker Cancer Center in Calgary, Alberta, Canada. The survey was anonymous, and participants were asked to complete the survey only if they met the following inclusion criteria: (i) diagnosed with cancer and currently aged 18 years or older; (ii) received anti-cancer therapies (ie chemotherapy, radiation, surgery, hormone therapy, and/or immunotherapy); (iii) had completed their primary anti-cancer therapies (not including hormone maintenance therapy); (iv) live in Canada. Participants did not receive any compensation for participation in this study.
Power Analysis
A target sample size of N = 138 cancer survivors was calculated based on sample size and power analyses using G*Power 3.1, 25 in consideration of previous research conducted with this type of population, 26 and the expected availability of survivors on relevant social media groups. We conducted a power analysis with a proposed sample of N = 138 to determine the ability to detect associations between type of cancer treatment, GI, physical and mental health outcomes for our primary hypothesis that type of cancer treatment, specifically chemotherapy, would be significantly associated with worse GI outcomes. Using G*Power 3.1, we determined that for a multiple regression with a sample size of N = 138, a medium effect size of Cohen’s f2 = .15, at a significance level of P < .05, with a power of .95 could be detected.
Demographic, Clinical and Health Measures
Clinical and treatment-related variables (Sample N = 317).

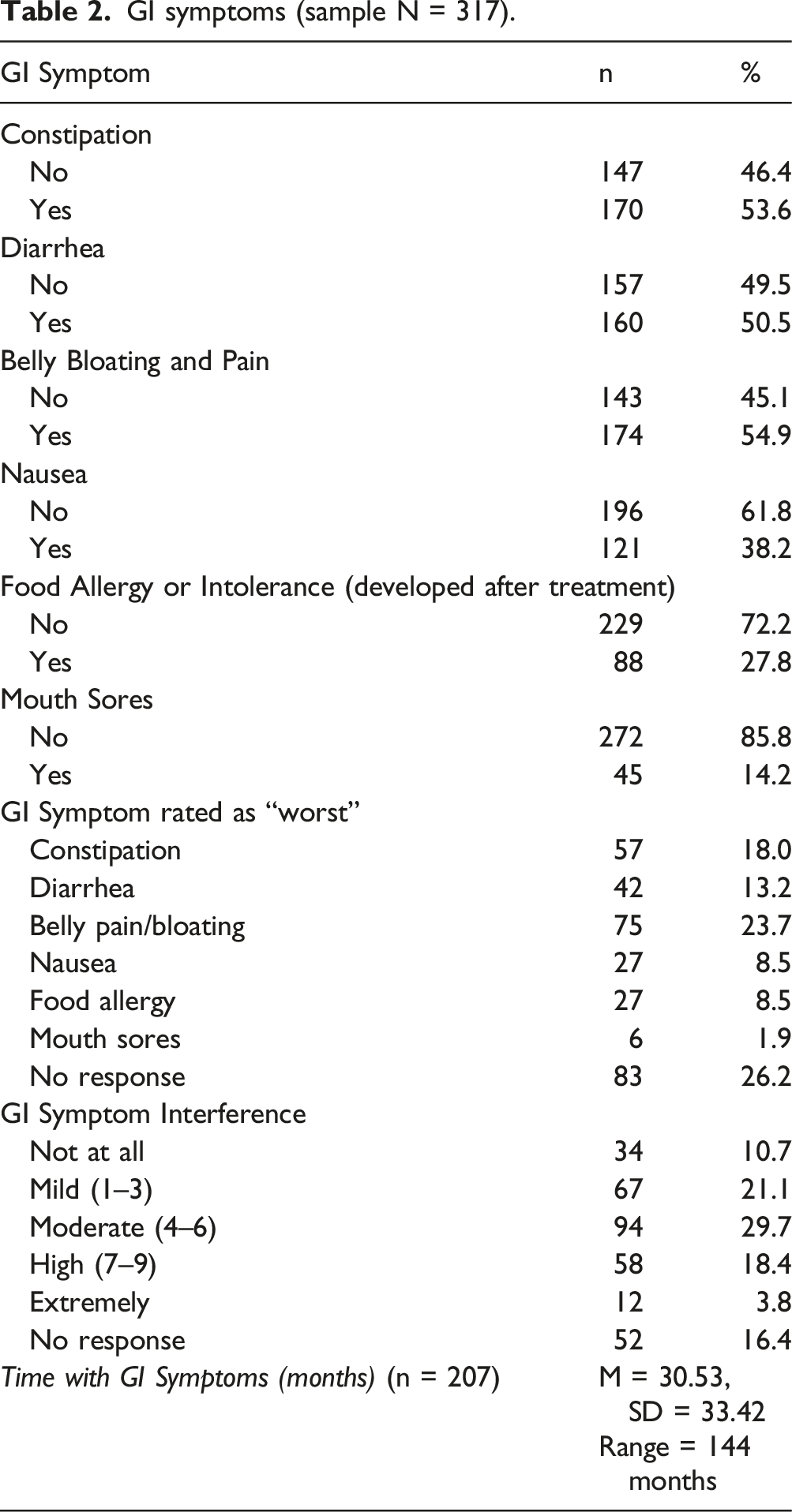
GI symptoms (sample N = 317).
PROMIS Measures
The PROMIS Global Health (v1.2) is a validated scale used to evaluate survivors’ self-reported mental and physical health. 27 The Global Health scale includes 10 statements about physical and mental health, which are rated on a 5-point Likert scale, with the exception of pain severity which is rated on a scale from 0 (no pain) to 10 (worst pain imaginable).
PROMIS Gastrointestinal scales for belly pain (v1.0, 5a), gas and bloating (v1.1, 13a), constipation (v1.0, 9a), and diarrhea (v1.0, 6a) were used. The PROMIS GI scales are reliable and validated measures that have been used in patients with chronic GI diseases, such as irritable bowel syndrome and inflammatory bowel disease, including Crohn’s disease.28,29 These measures assess the frequency, severity, and degree of interference for each GI symptom, which are rated on a 5-point Likert scale.
Procedure
The survey was made available online and shared on relevant cancer social media platforms and via cancer community organizations. The survey took approximately 30 minutes to complete, and participants completed it anonymously through an online link. The data was securely stored in Remote Electronic Data Capture (REDCap), which is a free, secure, browser-based application designed to support Electronic Data Capture (EDC) for research studies provided through the Clinical Research Unit (CRU) in the University of Calgary Cumming School of Medicine. The survey remained open from June 2020 to February 2021. This study was approved by the Health Research Ethics Board of Alberta (HREBA) Cancer Committee (ID: HREBA.CC-19-0018).
Data Analysis
For aim 1, descriptive statistics and frequency analyses were used to describe demographic and clinical characteristics of participants, and GI, physical and mental health outcomes in cancer survivors. Participants who did not respond or indicated no symptoms for a particular PROMIS outcome measure were excluded from subsequent analysis for that given outcome. Treatment types were dummy coded as “No” (0) or “Yes” (1) for each treatment type option (i.e. chemotherapy, surgery, radiation, hormone therapy, and immunotherapy), and a variable for “treatment group” was subsequently created to categorize patients who received chemotherapy only (1), chemotherapy plus other treatments (2), or no chemotherapy (3). GI symptoms were dummy coded as “No” (0) or “Yes” (1) for each GI symptom. For aim 2, Pearson product-moment correlations were used to examine whether GI symptoms were associated with mental and physical health. To address aim 3, 5 linear regression models were used to examine whether treatment type predicted each GI symptom. All treatment types were entered into the models to examine whether chemotherapy was associated with GI symptoms while holding constant receipt of other treatment types. Beta values reported are unstandardized. All statistical analyses were completed using IBM SPSS version 28, with alpha set at p = .05.
Results
Participants
Demographic characteristics of the study participants are shown in supplementary data (Table S1). A total of 317 survivors participated in the study. Mean current age was 49.63 years old (SD = 14.77). Most participants were female (84.2%), white (92.4%), and had post-secondary education (83%). Nearly half (48.3%) had an annual income between $50,000 to less that $120,000, about two-thirds of participants were partnered (63.1%), either married or common-law, and most were from metropolitan living centers (73.2%). While there was participation from survivors in most Canadian provinces, the majority were from Alberta (35.6%) and Ontario (35.3%).
Table 1 summarizes the clinical cancer and treatment-related variables of participants (N = 317). Mean age at diagnosis was 40.90 years old (SD = 15.40). Cancer types ranged, although nearly half of participants were breast cancer survivors (42.6%), followed by hematological cancers (21.2%) and colorectal cancer (6.9%). Most survivors were diagnosed with stage II (31.2%) or III (27.4%) cancers, which were non-metastatic (77%). Cancer treatment related variables for participants are also shown in Table 1. Most survivors received chemotherapy (86.1%), about three-quarters received surgery (79.2%), and two-thirds received radiation therapy (63.4%). Fewer survivors received hormone (29%) or immunotherapy (9.8%). Cumulatively, 76.7% of survivors received chemotherapy plus other treatments, 9.5% received chemotherapy only, and 13.6% received no chemotherapy. Time off treatment ranged from less than 3 months to more than 10 years, with about half (52%) of survivors being up to 3 years post-treatment, and about one-quarter (26.2%) being 5 or more years post treatment.
Aim 1: Describing GI, physical, and mental health outcomes
Table 2 summarizes the type, severity and duration of GI symptoms reported by cancer survivors. Participants reported multiple symptoms, but the most common were constipation (53.6%), diarrhea (50.5%), and belly pain and bloating (54.9%). A little more than one-third reported nausea (38.2%) and just over one-quarter of survivors reported developing a food allergy or intolerance since treatment (27.8%). When asked to rate just one GI symptom as the worst, 23.7% of survivors indicated belly pain and bloating as the worst, followed by constipation (18%). When asked to rate the degree of interference from GI symptoms, most survivors rated theirs as moderate (29.7%), although nearly one-quarter (22.2%) indicated high to extreme GI interference severity. The mean duration of time for GI symptoms experienced since the end of cancer treatment was 30.53 months (SD = 33.42), and ranged from 0 (i.e. finished less than 1 month ago) to 144 months.
PROMIS Gastrointestinal, Global Mental and Physical Health Outcomes.

Aim 2: Association between GI symptoms and mental and physical health
Associations between GI symptoms, and physical and mental health.

Note. Significant associations in
Aim 3: Associations between cancer treatment type and GI symptoms
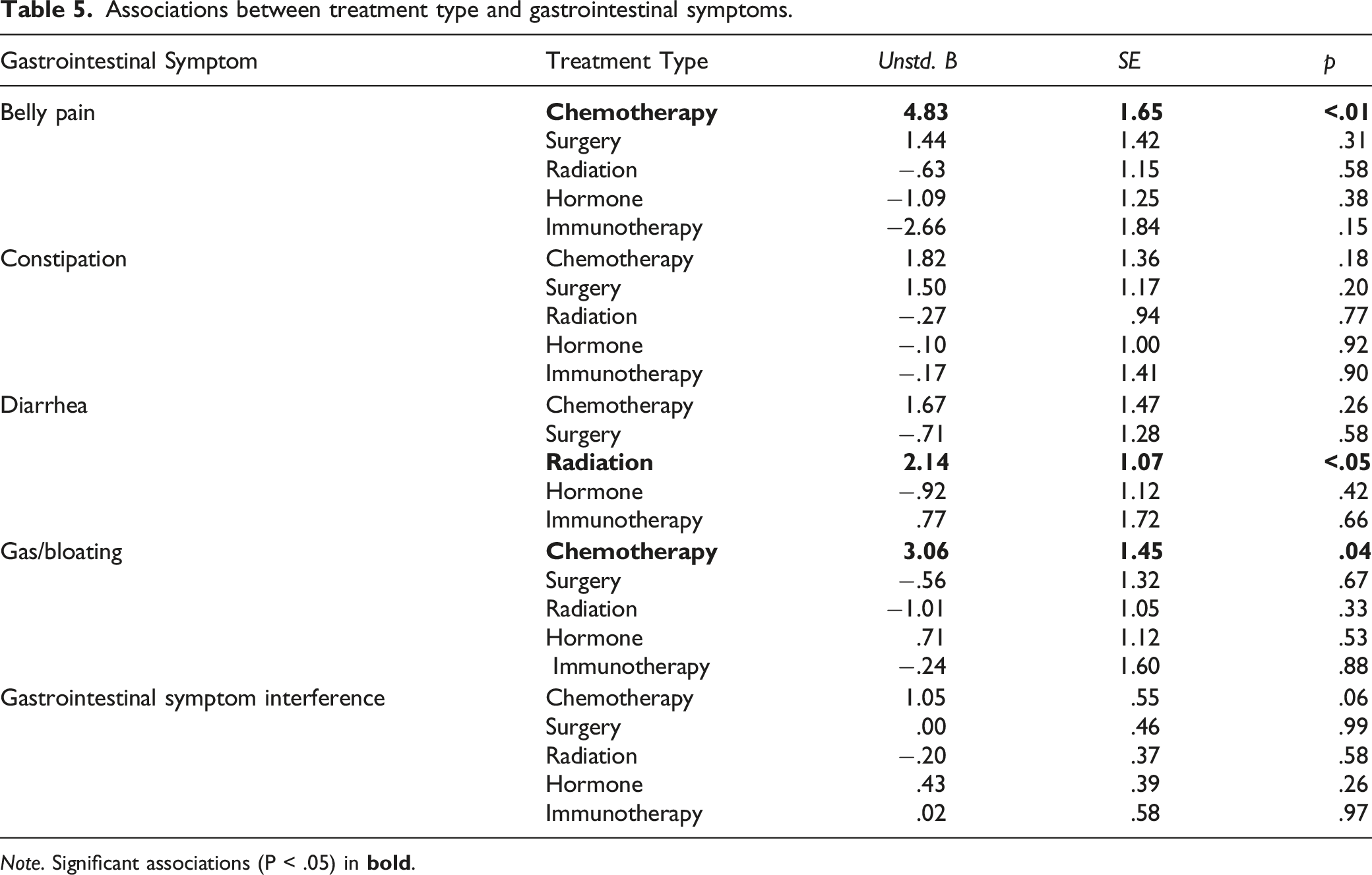
Associations between treatment type and gastrointestinal symptoms.
Note. Significant associations (P < .05) in
Discussion
This exploratory study investigated cancer treatment-related GI symptoms, and mental and physical health outcomes in adult cancer survivors in Canada. Most survivors (86.1%) received chemotherapy, and reported numerous GI symptoms after cancer treatments had ended, especially belly pain, bloating and constipation. GI symptoms lasted on average for 2.5 years in these survivors, but ranged up to 12 years post-treatment. Importantly, about half (51.9%) of the survivors rated the interference from their GI symptoms as moderate to severe. This is consistent with previous research suggesting that chronic post-treatment GI issues occur in cancer survivors. 8 However, the duration of time, type, and especially the severity of post-treatment GI symptoms experienced were previously uncharacterized in the current literature. This study provides compelling new evidence that a considerable proportion of cancer survivors do suffer from GI symptoms for several months or years post-treatment, and that for at least half of these survivors their GI symptoms are causing significant interference with their wellbeing.
Research in other patient cohorts, such as those with IBS, have found that patients tend to experience co-morbid GI symptoms and psychosocial health issues.15,16 Given this, we investigated whether GI symptoms were associated with mental and physical health in cancer survivors. Results revealed that greater severity of each GI symptom, specifically belly pain, constipation, diarrhea, gas/bloating, and GI interference, was significantly associated with poorer mental and physical health in these cancer survivors. Many cancer survivors experience psychosocial challenges such as anxiety, depression, cognitive dysfunction and fatigue after treatments for cancer have ended.4,6,7 Further, GI symptoms may exacerbate and be exacerbated by psychological distress, further compounding the symptom burden of cancer survivors. While some psychological-based therapies can help with treating psychosocial symptoms in cancer survivors, 30 underlying mechanisms that may contribute to the etiology and course of psychosocial problems require further investigation.
Mounting evidence suggests that chemotherapy is a key factor in the development of GI symptoms both during and after treatment.8-10,31 As such, the present study also investigated associations between cancer treatment type, specifically chemotherapy, and GI symptoms. Consistent with our hypothesis, treatment with chemotherapy was significantly associated with increased belly pain, and gas and bloating. Notably, the data revealed a trend towards chemotherapy treatment being associated with GI interference, although this was not statistically significant. These results suggest that chemotherapy treatment may warrant further investigation as to whether it plays a role in the development of certain GI symptoms, specifically belly pain and gas and bloating, and potentially also in the degree of GI interference survivors experience.
Chemotherapy has been shown to induce a dysbiotic state in the gut microbiota, increasing intestinal permeability and circulation of proinflammatory cytokines,12,22,23,32,33 which has implications for psychosocial health. For instance, under times of stress, illness or infection, the protective epithelial layer of the gut can be compromised leading to increased intestinal permeability, “leaky gut”, circulation of bacterial lipopolysaccharide (LPS), and endotoxemia.34,35 Subsequently, an inflammatory response is initiated, including increased systemic and central pro-inflammatory cytokine activity, dysregulation of neurotransmitters, such as serotonin, and the manifestation of sickness behaviours, including depression-like symptoms, cognitive dysfunction, and fatigue.34,35 Importantly, gut microbiota dysbiosis is associated with GI symptoms and also the onset of cytokine induced sickness behaviours, such as lethargy, social withdrawal, anxiety, cognitive dysfunction and anhedonia.34,36,37 Additionally, recent data suggest that in survivors, chemotherapy affects gut microbiota alpha diversity and taxonomic abundance after treatment has ended, and that certain bacteria are associated with psychosocial and GI symptoms. 19 Hence, chemotherapy induced dysbiosis of the gut microbiota may be one common mechanism by which patients experience co-morbid GI symptoms and psychosocial problems following treatment.
Radiation treatment was associated with symptoms of diarrhea. This finding is consistent with previous literature showing that cancer patients receiving pelvic radiotherapy frequently experience radiation-induced diarrhea and damage to the GI tract.8,10,38 Since almost one-quarter (20.5%) of participants in this study were survivors of cancers of the pelvic region (e.g. cervical, prostate, colorectal) and about two-thirds (63.4%) of participants reported receiving radiation therapy, it is likely that many survivors in this study were treated with pelvic radiotherapy, in combination with chemotherapy, and experienced diarrhea as a result of this.
Findings from the present study hold implications for clinical oncology practice and research. Specifically, in addition to screening for cancer-related and psychosocial symptoms in survivors of cancer, improved screening, and discussion with the health care team around chronic GI symptoms should also be included in clinical care. As noted by Andreyev et al., chronic GI issues are under-reported in the literature and by cancer patients, and current screening tools may fail to capture symptoms that are especially concerning to patient. 8 As shown in the present study, GI symptoms are associated with mental and physical health, and chemotherapy may be a contributing factor to some GI symptoms. Additionally, GI symptoms may exacerbate and be exacerbated by psychological distress, further compounding the symptom burden of cancer survivors. The timing and directionality of these associations are an important direction for future research. More attention should be paid to chronic GI symptoms in cancer survivors both in clinical settings and in research. The results from this study will help to inform future longitudinal research on the gut microbiota, and GI and psychosocial health in cancer survivors. Elucidating the mechanisms that contribute to chronic GI symptoms in cancer survivors may help to improve current treatments and inform the development of novel therapies.
Limitations
There are some limitations of the present study that warrant acknowledgement. Despite best efforts to recruit a diverse sample, the present study experienced limited diversity in participant demographics as most survivors were female, white and educated. Although we did have reasonable diversity in terms of cancer stage and type, a considerable proportion of survivors were those with a breast cancer diagnosis. The combination of these factors may compromise the generalizability of the findings to a certain degree. Additionally, as this study relied on self-report measures there is potential for some bias in participants reporting of symptoms and treatment history. Nevertheless, patient reported outcomes have been established as a reliable and valid way of collecting meaningful and valuable data on patients lived experiences with cancer. 39 As the data were collected during the COVID-19 pandemic, results must be interpreted with caution since periods of isolation and other pandemic-related stressors may have adversely affected patients GI and psychosocial health. Studies conducted during the pandemic suggest that people with cancer were adversely affected more than healthy peers, and particularly among younger survivors of cancer. 40 As well, it is possible that more anxiety and/or stress in survivors may be associated with GI symptoms, likely in a bidirectional manner, which would be consistent with other unpublished work our team recently completed. However, since distress and anxiety were not directly measured in this study, this is a limitation and an important area for future research. Lastly, as this survey study was anonymous there was no way to validate whether respondents were indeed cancer survivors, or confirm diagnostic or treatment information including specific types and doses of chemotherapy treatment received, and whether patients were currently taking other medications to treat GI or psychosocial concerns. However, this would be a valuable avenue for future research to better understand whether specific types and doses of chemotherapy may be especially implicated in chronic GI symptoms in survivors. Notably, since no compensation was offered for study participation and the study required about 30 minutes of participants time, it stands to reason that respondents were unlikely to participate if they were not in fact cancer survivors who wished to share their experience.
Conclusion
This study provides novel evidence that many cancer survivors experience chronic GI symptoms following cancer treatments, and that these GI symptoms may be a contributing factor in survivors compromised health, such that more severe GI symptoms may contribute to poorer physical and mental health outcomes. Moreover, chemotherapy was found to be significantly associated with specific GI outcomes, and radiation therapy with diarrhea. These findings present opportunities for better understanding the etiology of persistent GI and psychosocial issues in cancer survivors, and for the development of novel treatments that target not only the mind (i.e. psychosocial health) but also the gut. Future research would benefit from using prospective studies specifically designed to investigate relationships between cancer treatment related factors, such as chemotherapy, and the impact on GI and psychosocial health. Carefully designed studies will reduce bias and provide a more comprehensive understanding of factors related to chronic GI problems, and allow for causative associations to be revealed which may help health care providers to better prevent and/or treat these health issues in cancer survivors.
Supplemental Material
Supplemental Material - The Chemo-Gut Study: A Cross-Sectional Survey Exploring Physical, Mental, and Gastrointestinal Health Outcomes in Cancer Survivors
Supplemental Material for The Chemo-Gut Study: A Cross-Sectional Survey Exploring Physical, Mental, and Gastrointestinal Health Outcomes in Cancer Survivors by Julie M. Deleemans, Kirsti Toivonen, Raylene A. Reimer and Linda E. Carlson in Global Advances in Health and Medicine
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded by the Enbridge Psychosocial Oncology Research Chair awarded to Dr Linda Carlson, and by the Killam Foundation in the form of a scholarship awarded to Dr Deleemans.
Data Request
Research materials related to this paper can be accessed via written request to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
