Abstract
Background
Regulatory assessment of anthroposophic medicinal products (AMPs) can be challenging due to their specific features.
Objective
The aim of this paper is therefore to provide adequate scientific information on AMPs for regulatory purposes.
Methods
A literature review was executed with database searches in PubMed, Cinahl, Merkurstab, Anthromedics, and https://iaap-pharma.org/. Search terms were: anthroposophic medicinal products, anthroposophic medicines, anthroposophic pharmacy. There was no language restriction; searches were executed from onset until June 11, 2020. In addition, experts were invited to suggest relevant literature.
Results
Eighty-seven of 660 identified publications were included. The system of anthroposophic medicine (AM) with its conceptual background and various aspects of AMPs was described: definition, pharmaceutical properties, an example of AMP development, use in clinical practice, similarities with and differences to conventional medicinal products, societal aspects, scientific and regulatory assessment.
Conclusion
AMPs are part of the integrative whole medical system of AM. AMPs are manufactured according to Good Manufacturing Practice and national drug regulations and have an excellent safety status; the limited available evidence suggests clinical benefits. Current drug regulation of AMPs in the EU and most European countries does not take the special properties of AMPs into account. Future research should focus on appropriate methodologies for the evaluation of effects of AMPs as part of the AM whole medical system, the scientific quality of its non-atomistic holistic ontological position, and the integration of AM and conventional medicine in clinical practice. Future policies should focus on appropriate ways of addressing regulatory challenges to AMPs.
Keywords
Introduction
Scientific and regulatory assessment of anthroposophic medicinal products (AMPs) can be challenging. Conceptually, historically, and in some countries legally, AMPs form a distinct group of medicinal products (MPs). But regarding ingredients and manufacturing procedures, some AMPs also overlap with herbal and homeopathic MPs. Furthermore, the pharmaceutics of AMPs is complex, involving a number of procedures rarely used elsewhere. Finally, AMP treatment is part of a larger therapy system of Anthroposophic Medicine (AM), which guides its use in clinical practice. For these reasons, there is a need for adequate information on the group of AMPs as such for the scientific community, the regulatory authorities and other stakeholders.
This article aims to provide basic information in this respect by giving an overview of AMPs within the system of AM. In order to enable an optimal understanding of the AMPs, based on a literature review, 3 we first present the AM system with its conceptual background: the holistic conception of the human organism and its relation to the natural environment, disease processes and their treatment. Subsequently, the major aspects of AMPs are presented: definition, pharmaceutics, therapy principles, an example of AMP development, use in clinical practice, similarities and differences to conventional MPs; societal aspects; scientific and regulatory assessment of AMPs and the regulation of AMPs in Europe.
Methods
Research Questions
1. What are the relevant features of the system of AM, which the AMPs are part of? a. What are the features of AM as a whole medical system? b. What is the anthroposophic holistic conception of the human organism? 2. What are the features of AMPs? a. What is the AM concept of disease and how is this related to AMP treatment? b. Which manufacturing procedures are used in AMP pharmacy? c. What are the administration forms and dosages of AMPs? d. How are therapeutic actions of AMPs conceptualized? e. What are the similarities and differences between AMPs and conventional MPs? f. How are AMPs used in clinical practice? 3. What are the specific societal aspects of anthroposophic pharmacy? 4. What is the status of the scientific and regulatory assessment of AMPs, regarding a. Pharmaceutical quality? b. Safety? c. Efficacy/effectiveness? 5. What is the status of the regulation of AMPs in Europe? a. What is the relevant background of AMP regulation for the modern drug regulation? b. What are the European legal provisions? c. What are the national provisions?
Design
A literature review was conducted. The following databases were used to identify potentially relevant literature: two generic databases (PubMed, Cinahl), two AMP-related databases (Merkurstab, Anthromedics) and one AMP-related website (https://iaap-pharma.org/). Search terms used were: anthroposophic medicinal products, anthroposophic medicines, anthroposophic pharmacy. There was no date or language restriction; searches were executed from onset until June 11, 2020. In addition, AMP experts of the ESCAMP network were invited to provide relevant literature.
Data Selection and Data Extraction
Two researchers (EB, HvW) screened titles and abstract. Two researchers (EB, HH) screened full texts of each included publications in three steps (title, abstract, full text), and extracted the relevant data (features of AM, features of AMPs, societal aspects of anthroposophic pharmacy, status of the scientific and regulatory assessment of AMPs and the status of the regulation of AMPs in Europe). Any disagreement between the authors was resolved by discussion with a third author.
Use of Data to Answer the Research Questions
The data of the included studies were used to answer the research questions in the following sections: 1. What are the relevant features of the system of AM, which the AMPs are part of? Section “Anthroposophic medicine, a whole medical system” Section “Anthroposophic holistic conception of the human organism” 2. What are the features of AMPs? Section “Anthroposophic medicinal products” The sections “Anthroposophic holistic conception of the human being,” “Disease processes, treatment with AMPs,” and “Example of AMP development: Citrus-Cydonia for hay fever” are adapted from corresponding texts in the draft ESCAMP Assessment Report on Citrus-Cydonia by author HJH, for which a systematic literature search on these issues had been conducted (141 hits, 28 sources excluded, 113 included). 3. What are the specific societal aspects of anthroposophic pharmacy? Section “Societal aspects of anthroposophic pharmacy” 4. What is the status of the scientific and regulatory assessment of AMPs? Section “Scientific and regulatory assessment of AMPs” 5. What is the status of the regulation of AMPs in Europe? Section “Regulation of AMPs in Europe”
Results
The database searches and the input from experts resulted in a total of 660 literature records for possible inclusion in the review. After assessment on relevance of these, 83 were included in the literature review to answer the five research questions (Figure 1). Flow diagram.
What are the Relevant Features of the System of Anthroposophic Medicine, Which the Anthroposophic Medicinal Products are Part of?
For an adequate understanding of AMPs, the whole medical system of AM the AMPs are part of and the anthroposophic holistic conception of the human organism are described.
Anthroposophic Medicine, a Whole Medical System
AM is an integrative whole medical system, founded in Central Europe in the early 1920s by Rudolf Steiner and Ita Wegman. AM therapy involves specific AMPs as well as non-medication therapy modalities such as rhythmical massage therapy, 4 art therapies (clay modeling, painting, music, speech exercises), 5 eurythmy therapy (an artistic movement therapy), 6 nursing techniques including compresses, oil dispersion baths 7 and rhythmical embrocation therapy, 8 biographical counseling and lifestyle modification. 9 AM is provided by physicians (counseling, AMP therapy), therapists and nurses in outpatient and inpatient settings including large university teaching hospitals. 9 AM is practiced in most European countries, the Americas, Asia, Australia, New Zealand, and South Africa. 10
AM is a complete medical system of theory and practice and can therefore be classified as a whole medical system (Box 1). Since AM is an extended form of conventional medicine in which AMPs as well as other anthroposophic treatment modalities are traditionally used alongside with conventional treatment modalities, it can also be defined as a type of Complementary and Alternative Medicine (CAM) (Box 2) or Integrative Medicine (IM) (Box 3).
2
Whole medical systems. Whole medical systems (WMSs) are complete systems of theory and practice that have evolved independently from or parallel to conventional medicine. Many are traditional medical systems that are practiced by individual cultures throughout the world (e.g., Anthroposophic Medicine, Ayurveda, Homeopathy, Naturopathy, and Traditional Chinese Medicine). Whole Medical Systems are healthcare approaches in which practitioners apply bodies of knowledge and associated practices in order to maximize the patients’ capacity to achieve mental and physical balance and restore their own health, using individualized, non-reductionist diagnostic and treatment approaches. Whole Medical System practices often involve complex interventions (CI), composed of parts that together make the whole intervention, in which the whole is more than the sum of the parts, and, in which the whole can generate the effect of the intervention. Also, in conventional medicine, there is increasing interest in the development and testing of CI (e.g., community based health promotion, stroke units).
2
Complementary and Alternative Medicine (CAM). The Cochrane Collaboration definition of complementary medicine is that it includes all such practices and ideas that are outside the domain of conventional medicine in several countries and that it is defined by its users as preventing or treating illness, or promoting health and well-being. These practices complement mainstream medicine by satisfying a demand not met by conventional practices and diversifying the conceptual framework of medicine (Manheimer and Berman, 2008). According to the National Center for Complementary and Integrative Health (NCCIH), CAM is defined as health care approaches that are not typically part of conventional medical care or that may have origins outside of usual western practice.
1
Integrative Medicine. Integrative Medicine (IM):
Reaffirms the importance of the relationship between practitioner and patient Focusses on the whole person Is informed by evidence Makes use of all appropriate therapeutic and lifestyle approaches, healthcare professionals and disciplines to achieve optimal health and healing.
1
Anthroposophic Holistic Conception of the Human Organism
AM integrates conventional medicine with the concepts, methods and therapies derived from anthroposophy. AM is founded in a non-atomistic holistic worldview with an ontological position and an epistemology that refers to the existence and an empirical and rational cognition of non-atomistic holistic formative forces. In order to assess these formative forces with corresponding organizational and functional levels of the human organism, specific methods are applied. 11 AM thus transcends the conventional paradigm that reduces organisms to molecular or cellular interactions. This paradigm difference is an underlying determinant for the differences between AMPs and MPs of conventional medicine (CMPs) regarding conception, manufacturing, and use in clinical practice.
Four organization levels.

The three functional systems.

All three systems are functionally located in all parts of the organism, but each with a predominant physical location: the nerve-sense system in the skull, the rhythmic system in the thorax, and the metabolic-limb system in the abdomen and extremities.
In these three systems, the four classes of formative forces interact differently
9
: • In the • In the • In the
Research has confirmed the existence of such a threefold organizing principle in phenomenological studies on plants, 19 animals,20,21 and human beings 16 and has in particular linked it to embryology,22,23 anatomy,16,24 immunology, 25 pharmacology, 26 and psychiatry. 27
What are the Features of Anthroposophic Medicinal Products?
In this section the following features of AMPs are described: disease processes and treatment with AMPs, the manufacturing procedures, dosage and administration forms, therapeutic actions, an example of the development of an AMP, AMP use in clinical practice, and similarities and differences between AMPs and conventional MPs. These descriptions do not aim at completeness for each feature.
Disease Processes, Treatment With Anthroposophic Medicinal Products
In AM, diseases are understood to reflect imbalances in the interactions between the four classes of formative forces and between the three functional systems (one further typology relevant for AMP therapy is not dealt with in this paper: the seven major metals 28 ). The imbalances consist either in too intensive or in too loose interpenetrations between the respective forces/organizations or functional systems. Anthroposophic therapies, including AMPs, address these imbalances.
Definition of anthroposophic medicinal products.

Direction of therapeutic action in herbal substances used in AMP pharmacy, according to the part of the plant used.

Direction of therapeutic action, according to mode of administration.

Manufacturing Procedures
In anthroposophic pharmacy a number of different pharmaceutical procedures and different pharmaceutical dosage forms are used. By means of these procedures, the substances are transformed into appropriate MPs, which can interact specifically with for example one of the three functional systems or one of the four specific organization levels or their interactions.
Specific Anthroposophic Manufacturing Procedures Include29,35:
• Cultivation of plants in soils pre-treated with diluted metal salts (“vegetabilized metals”) • Production of metal mirrors (metallicum praeparatum, deposits of metals in reduced state onto a surface by chemical vapor decompositions) • Heat and cold treatments (including specific fermentation processes) • Specific mixing processes
Through vegetabilization the physical: substance of metals is brought into the realm of the life processes. Accordingly, the therapeutic action of vegetabilized metals becomes directed at the sentient organization (see above), with the main sphere of action in the metabolic system.
For the production of metal mirrors, excessive heat is applied. Thereby the solid metal is successively transformed into the liquid and gas states, followed by condensation back to the solid state. Having gone through solid, liquid, gas, and heat, the finished metallicum praeparatum product can act on all four organization levels (cf. Table 1).
Among heat treatments, nine specific forms are used, whereby the respective substance of botanical or zoological origin is transformed, directing the therapeutic action toward one of the functional systems or parts thereof (Table 5).
An important pharmaceutical process for many of the AMPs is potentization, which implies a successive dilution, each dilution step involving a rhythmic succussion (repeated shaking of liquids) or trituration (grinding of solids within lactose monohydrate). For example, a D6 potency (also called 6X) has been potentized in a 1:9 dilution six times, resulting in a 1:10−6 dilution. 37 Anthroposophic pharmacy mainly uses decimal attenuation, rarely centesimal or vicesimal attenuation. The therapeutic action is directed toward one of the three functional systems, according to the potency used (Table 6). 36
Potencies beyond D23 are unlikely to contain any molecules of the original substance; hence effects cannot readily be explained by molecular mechanisms. Nonetheless, a systematic review of in-vitro studies found biological effects of potencies ≥D23 in nearly three-fourths of the studies and in more than two-thirds of the studies with highest quality. 38
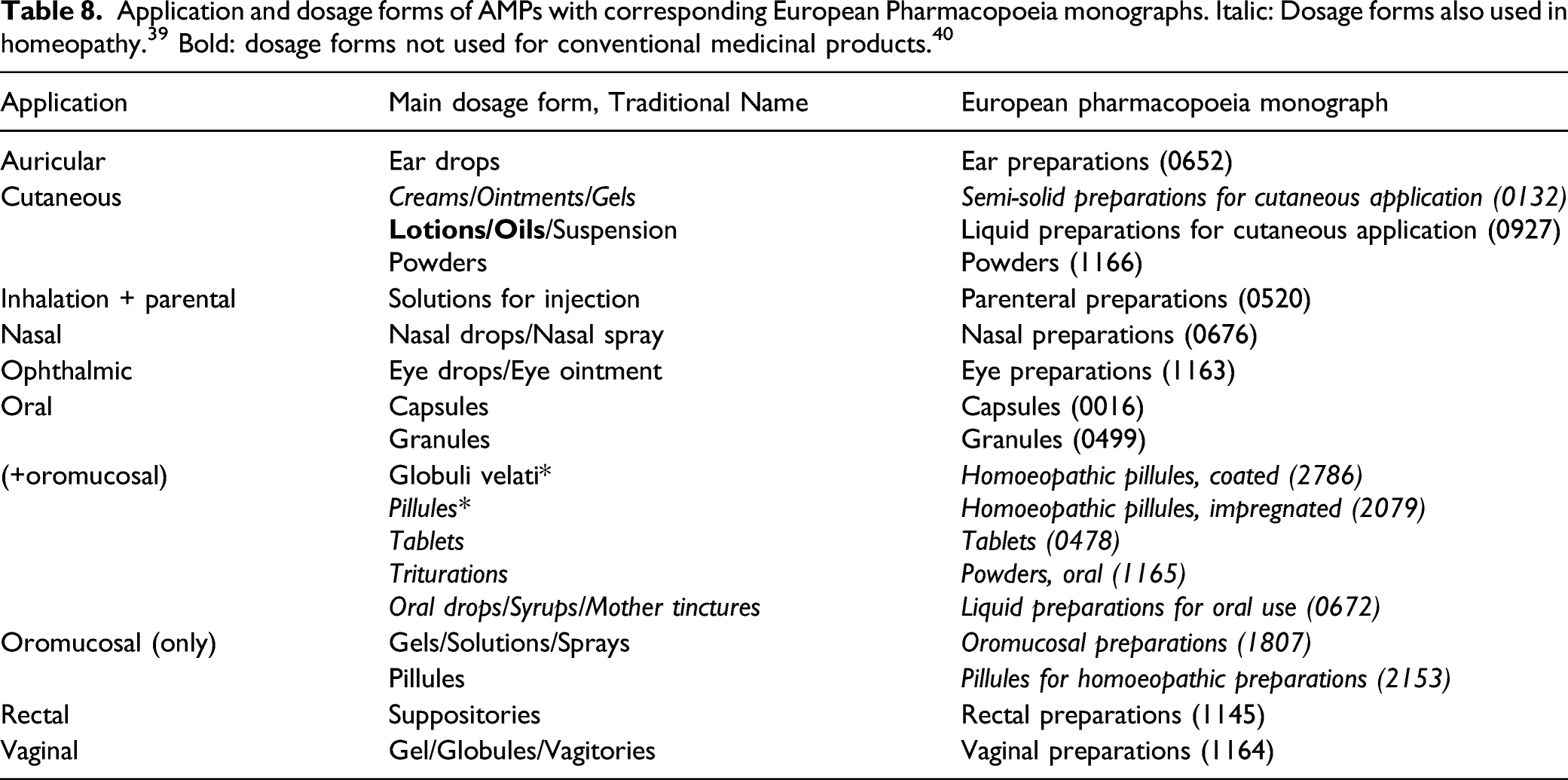
Dosage and Administration Forms
In 41 addition to the range of decimal potencies, the mode of administration of AMPs can also direct the therapeutic action toward one of the three functional systems (Table 7). 41
Anthroposophic Medicinal Product Therapeutic Actions
Anthroposophic medicinal product therapy principles include inhibition and suppression as in conventional pharmacotherapy; provocation of physiologic reactions secondary to the primary effect of the AMP; regulation and harmonizing of overarching, complex physiological processes; and transformation of physiological and psychological processes and capacities into more mature and integrated states.35,42,43 Examples are
44
: • Belladonna D3 with anti-inflammatory and analgesic effect (inhibition and suppression). • Viscum album for the treatment of cancer patients, inducing increased well-being and reduction of cancer-related fatigue (provocation of physiologic reactions secondary to the primary effects). • Hyoscyamus niger/Onopordon acanthium/Primula veris for regulation of cardio-respiratory and sleep-wake rhythms in conditions such as extrasystoles and sleep disturbances (regulation and harmonization of physiological processes). • Sulfur for preparing the physical organization in such a way that the life organization is better able to organize the life processes in the body (transforming physiological and psychological processes and capacities into more mature and integrated states).
Example of Anthroposophic Medicinal Product Development: Citrus-Cydonia for Hay Fever
The following example of the AMP Citrus-Cydonia for hay fever (seasonal allergic rhinitis or rhinoconjunctivitis) illustrates how the anthroposophic holistic conception of the human organism is used for the development of AMPs for specific indications. 1. According to the anthroposophic conception of hay fever, the I-organization and sentient organization are constitutionally too much involved in the peripheral sense processes (sense-nerve system), leaving too little formative and structuring activity towards the life and physical organizations (metabolic-limb system) (Table 1).
45
2. As a consequence, the life organization with related “water” or fluid processes dominates and the latter become insufficiently structured. In attacks of hay fever, fluid production in the affected mucosa becomes excessive and uncontrolled as an expression of both domination and insufficient structuring.
46
3. In terms of the three functional systems, the hay fever reaction is the expression of the metabolic-limb system intruding too deeply into the sense-nerve system.
45
One common trait of these features is a centrifugal dynamic:
• The I-organization and the sentient organization are too strongly “centrifugally” involved in peripheral sense processes such as vision and smell,
47
with hyper-responsivity to all types of stimuli. • In this respect they mimic processes of wind pollination of grass, which are also centrifugal: the pollen is transported away from the plant to other grass plants
47
(In the anthroposophic concepts of disease there is a strong relationship between the (patho)physiological processes in nature and the human being. The knowledge of these relationships enables the development of rational anthroposophic treatments.) • The dynamics of the metabolic system intrudes into the sense-nerve system, leading to centrifugal processes vasodilatation as a result of the interaction with the allergen, resulting in swollen mucosa with excessive mucus formation, and “centrifugal” symptoms like sneezing, rhinorrhea and lachrymation.
Another trait can be characterized as “exaggerated openness” with “insufficient discrimination.” • In spring people tend to become more intensively connected to the seasonal processes of the natural environment, like the greening and blossoming of plants. With the hay fever constitution, this connection becomes exaggerated, with the I-organization and the sentient organization “getting lost” in the periphery. As a result, the human organism is insufficiently structured and becomes too open for external processes including the pollination of grass and trees.
45
• In immunological terms, the organism is unable to discriminate between innocuous and harmful non–self-substances.
25
The pollen allergens are in themselves innocuous and would normally be washed away by small amounts of mucus ordinarily present in the nasal cavity or eye sack. • The resulting chronic inflammation in the nasal and/or conjunctival mucosa is characterized by vascular hyperpermeability, that is, vessels are too “leaky.”
Pathophysiologic features of hay fever and their counterparts in Citrus-Cydonia.

With its herbal ingredients, Citrus-Cydonia works primarily on the sentient organization, stimulating it to engage more strongly with the life organization. In addition, minerals of Citrus (eg, potassium, calcium and silica) and Cydonia (calcium) help supporting the I-organization, in order to bring about sustained healing. 48
The use of fruits from Citrus and Cydonia direct the therapeutic action towards the metabolic system (Table 4). When Citrus-Cydonia is administered parenterally, the therapeutic dynamic also activates the rhythmic system (Table 7), stimulating it to contain the metabolic system and to restore the healthy balance between sense-nerve system and metabolic-limb system.
In different ways, Citrus and Cydonia are characterized by a centripetal dynamic, which in the holistic anthroposophic conception is understood to counteract the centrifugal dynamics of hay fever:
Citrus and Cydonia have delineating and independence properties which in the anthroposophic conception are understood to counteract the organism’s exaggerated openness to the environment and to help the organism to better discriminate between innocuous and harmful substances. Most of these properties are directly in contrast with the Gramineae that provoke the hay fever attacks (pertaining to rhythmicity of blossoming, formation of aromatic substances, direction of fruit formation, and the type of pollination, cf. Table 10): • In Gramineae, blossoming and pollination occur rhythmically, at the same period each year, expressing their embeddedness in and dependence on the surroundings. In Citrus, these processes take place throughout the year, independently of seasonal rhythms.
49
• In Gramineae aromatic substances are produced only in the flowers and to a minimal extent. In Citrus there is an abundance of aromatic substances in the flowers, fruits and even the leaves.
49
• In Gramineae fruit formation takes place in a centrifugal direction, progressing from the center towards the periphery. In Citrus and Cydonia, as mentioned above, fruit formation follows a centripetal direction.
45
• Pollination of Gramineae is by wind (anemophily), for Citrus and Cydonia by self-pollination and by insects (biotic). Contrasting features of Gramineae vs Citrus + Cydonia.

With regard to another feature, the ripening process, Citrus and Cydonia are contrasted to most other fruits: When most fruits ripen, the fruit acids are converted into sugar compounds with the fruit tasting sweet. In Citrus and Cydonia fruit, acid levels remain high, with even ripe fruits having a very sour taste, 49 demonstrating the relative independency of the fruit formation from the surrounding influences (light and warmth).
The hard, leathery peel of Citrus and Cydonia contains and delineates the liquid processes. According to the anthroposophic conception, this feature can help control fluid production in hay fever. On the substance level, Citrus and Cydonia produce large amounts of pectins (slime substances) which bind water. 49
For over 90 years, Citrus-Cydonia has been prescribed for hay fever or seasonal allergic rhinitis patients. In several European countries, Citrus-Cydonia solution for injection is commercially available for the prophylaxis and treatment of hay fever and other allergic diseases, administered as subcutaneous injections or as inhalation. Additionally, the medicinal product has marketing authorization as nasal spray in France, Switzerland, and Germany and as eye drops in Switzerland and Germany.
Whereas initially the use of Citrus-Cydonia for hay fever patients was solely based on the conceptual understanding of its therapeutic action (see above) and positive individual experiences of prescribing doctors and patients, in the last decade the body of scientific evidence has increased: • Safety of Citrus-Cydonia has been investigated in in-vitro studies,50-53 a phase-I study on local tolerance,
54
documentation of experiences of prescribing doctors,44,55 cohort studies,56,57 randomized controlled trials,55,58 and a systematic evaluation of German pharmacovigilance databases,
59
demonstrating that this treatment is very safe. • Effects of Citrus-Cydonia on hay fever symptoms have been documented in a number of in-vitro studies,50-53,58 demonstrating a reduction in histamine production and the inflammatory mediator release from mast cells in a dose-dependent manner,
52
and positive immunological effects of several immunologically active compounds of Citrus and Cydonia like flavonoids and pectins on hay fever,60-66 in documentation of experiences of prescribing doctors,44,55,67 in two cohort studies56,57 and two randomized trials,68,69 showing reduction of hay fever symptoms. These results are in accordance with the AM treatment concept.
Based on current clinical knowledge and scientific evidence, it is hypothesized that Citrus-Cydonia, in conventional medical terms, works on the organizational level of the allergy-related subsystem of the immune system, promoting the regulation of the Th1/Th2 balance, reducing the degranulation and histamine release of IgE-activated basophilic cells and mast cells, and inhibiting the IgE- and PMA/A23187-induced increases in IL-8, TNF-α, and GM-CSF production in mast cells,50-53 leading to a non-allergic (normal or healthy) physiology. This formulation of the working mechanism can be seen as the conventional terminological counterpart of some aspects of the previously described AM working principles for these substances (balancing the two functional systems, counteracting the organism’s exaggerated openness to the environment and helping the organism to better discriminate between innocuous and harmful substances). 55
Although Citrus-Cydonia is a central element of anthroposophic hay fever treatment, the treatment of the individual patient can be further individualized, taking into account an additional individualized AM diagnosis, the specific symptom complex and the situation of the particular patient. As a result, individual patients with hay fever will often be prescribed additional AMPs and other non-medication AM therapies, in line with their individualized AM diagnosis. On the other hand, Citrus-Cydonia has been used for other indications with similar pathophysiological profile as that described above, including asthma, 70 rhinosinusitis, 71 otitis media with effusion and adenoid hypertrophy, 72 and Menière’s disease. 73
Anthroposophic Medicinal Product Use in Clinical Practice
Whereas CMPs are rationally indicated for specific conventional indications, AMPs are rationally indicated for specific anthroposophic indications. For example, a patient conventionally diagnosed with sleeping problems, fatigue and muscle pain in the neck region can also obtain the additional anthroposophic diagnosis that the nerve-sense system is too active, resulting in too much awareness, too much consciousness in the muscles (expressed as pain) and too little anabolic activity (resulting in a lack of vitality or fatigue). A rational AM therapy in this example would focus on reducing the activity of the nerve-sense system and/or stimulate the activity of the metabolic-limb system (to increase the anabolic activity) and/or the rhythmic system (to balance the other two systems).
Several AMPs can be used for one AM indication and also for one conventional indication, simultaneously or in different phases of the course of the disease. As a result, in a group of patients with the same conventional diagnosis, individual patients can have different anthroposophic diagnoses. As a result, each patient with the same conventional diagnosis can be treated differently with one or more different AMPs (and/or with other anthroposophic non-medication therapies). Thus, although a condition with a comparable etiological background such as in allergic rhinitis may call for a typological treatment such as Citrus-Cydonia, the anthroposophic approach to treatment is as a rule quite individualized, depending on individual differences in constitution and context influences.
This approach of prescribing several AMPs for one conventional indication is featured in standard textbooks of AMP therapy (e.g.,35,74,75). The approach is also confirmed by analyses from routine AM outpatient settings. In an analysis comprising 46,441 consultations with 22,239 patients, more than one AMP was prescribed in 40% of consultations. 76 In a study of 717 patients starting AM treatment for acute respiratory and ear infections, 87% of the patients were prescribed more than one AMP within four weeks of treatment, with a total of 256 different AMPs prescribed. 77 In 361 patients starting AMP treatment for chronic indications, 66% of the patients were prescribed more than one AMP at the first consultation. 78
Similarities and Differences Between Anthroposophic Medicinal Products and Conventional Medicinal Products
The main similarities between AMPs and CMPs are: • Both can be manufactured for external, oral, mucosal, or subcutaneous application, and • Both include active substances of vegetable and chemical origin.
The main differences are:
• CMPs are manufactured according to a biomedical, reductionist model, whereas AMPs are manufactured according to a holistic model. 11
• Manufacturing of CMPs usually starts with molecules/isolated substances, whereas AMP manufacturing in most cases starts with natural whole products (minerals, plants, etcetera).
• The development of most CMPs follow the route from pre-clinical studies, clinical studies, and application into clinical practice, whereas AMPs are developed along an opposite pathway, originating in collaboration of pharmacists and physicians working in clinical practice and subsequently being studied in clinical trials and pre-clinical studies. 79
• Most CMPs are aimed at fighting disease, whereas most AMPs are oriented at preventive or curative health promotion (pathogenetic vs salutogenetic approach).
• Most CMPs have a small number of conventional indications, whereas AMPs can be used for many conventional and AM indications (e.g., indications that describe the disease symptoms as the result of a specific imbalance in the three- or four-fold organization).
The main similarity between AMPs and other MPs from
What are the Specific Societal Aspects of Anthroposophic Pharmacy?
The two largest AMP manufacturers are Weleda and WALA. Weleda was founded in 1921 and has its headquarter in Switzerland with main production units in Germany, France and Switzerland. WALA was founded in 1935, headquarters and main production units are in Germany. Together the two companies employ more than 2,400 persons and produce more than 1,900 different MPs as well as a broad range of body care products. The products are exported to more than 50 countries.80,81 Both companies employ the principles of AMP pharmacy and use, for herbal starting materials, herbs from organic and biodynamic agriculture. Other AMP manufacturers include Abnoba, Helixor, and Iscador.
Compared to the CMP industry that in essence is characterized by business entrepreneurship, the AM pharmaceutical industry has particular features of sustainable entrepreneurship. The primary aim of AM manufacturers is not shareholder profit (as is usually the case for CMP manufacturers), but to ascertain the availability of a broad spectrum of AMPs for citizens/consumers, allowing for individualized treatment approaches described above. 82 While a business entrepreneur typically measures performance in profit and return, a social entrepreneur also measures positive returns to society. A sustainable entrepreneur measures the commitment of the business to ethical behavior and contribution to economic development while improving the quality of life of the workforce, their families, local communities, the society, nature, and the world at large as well as future generations. 83
Due to the nature of AMP therapy, marketing and sale structures of AM pharmaceutical industry differ from the mainstream of pharmaceutical business. Whereas the two largest AMP manufacturers produce many hundreds of AMPs each, thereby serving prescribing doctors and patients in an individualized treatment culture, CMP industries tend to focus on the production of a much smaller number of blockbusters in order to maximize profit. As a result of these differences in mission and product assortment structure, the AMP pharmaceutical industry is confronted with large financial demands for a large range of products (and low sale volumes per medicine), in order to comply with regulatory requirements such as licensing fees per MP and laboratory costs per MP for quality control of the manufacturing process and the products. Therefore, the AM pharmaceutical industry has had to limit the number of AMPs, leading to numerous withdrawals of AMPs from the market, not because of lack of efficacy, but because of low sale vs high costs.
What is the Status of the Scientific and Regulatory Assessment of Anthroposophic Medicinal Products?
For regulatory assessment of MPs, including AMPs, the key scientific issues are pharmaceutical quality, safety, and efficacy/effectiveness. In this section, the quality standards and the evidence for safety and effects are summarized.
Pharmaceutical Quality
Quality standards for starting materials and manufacturing procedures used in anthroposophic pharmacy are described in the European Pharmacopoeia [Ph.Eur.], in national pharmacopoeias (including the British Pharmacopoeia (B.P.), French Pharmacopoeia (Ph.fr.), German Pharmacopoeia (DAB), Swiss Pharmacopoeia (Ph.Helv.), German Homeopathic Pharmacopoeia (HAB)) and in the Anthroposophic Pharmaceutical Codex (APC). 29 All AMPs are manufactured according to Good Manufacturing Practice and national drug regulations. Toxicologically relevant starting materials (e.g., aconite, cinnabar) are highly diluted according to safety requirements of European regulations. 84
Safety
The safety of AMPs is demonstrated both historically and in several studies. AMPs were introduced in the 1920s and most AMPs in current use were developed and marketed until the 1950s. Since the start of modern drug regulation and pharmacovigilance in the 1960s, serious adverse drug reactions (ADRs) due to AMPs have been very rare, with no deaths reported. Several recent research projects have demonstrated an excellent safety status of AMPs:
EvaMed was a prospective pharmacovigilance study with 38 physicians in outpatient care in Germany. For each patient consultation, diagnoses and prescriptions were extracted from the electronic medical record. All physicians documented all ADRs of Grades III-IV and all serious ADRs, seven “prescriber physicians” also documented all non-serious ADRs of any intensity. In an analysis of 44,662 patients prescribed AMPs in the period 2001–2010, ADRs with intensity Grades III–IV occurred in .002% (n = 7/311,731) of prescriptions and .016% (n = 7/44,662) of patients. Serious ADRs occurred in .0003% (n = 1/311,731) of prescriptions and .0022% (n = 1/44,662) of patients; the one patient with a serious ADR to AMPs recovered after drug withdrawal. Among patients of the prescriber physicians, ADRs of any intensity occurred in .071% (n = 67/94,734) of AMP prescriptions and in .502% (n = 65/12,956) of patients prescribed AMPs. According to the nomenclature recommended by the Council for International Organizations of Medical Sciences, ADRs to AMP therapy in outpatient care were “rare,” and ADRs of high intensity as well as serious ADRs were “very rare.” 85
In a systematic review of 265 clinical studies of AM therapy, thereof 255 studies on AMPs, the authors concluded that this treatment had barely any risks and side effects, and if side effects were present, usually of mild to moderate intensity.17,18
Jong et al. 59 analyzed ADRs to anthroposophic and homeopathic solutions for injection in pharmacovigilance databases of eight German manufacturers. The analysis included ADR case reports from post-marketing surveillance and the literature, as well as from clinical/safety trials. Over a 10-year-period (2000–2009), a total of 303 million ampoules for injection were sold, and 1,180 ADRs were identified, yielding an overall reporting rate of less than 4 ADRs per 1 million sold ampoules, which was classified as very rare.
Efficacy/Effectiveness
The most recent comprehensive systematic review of clinical studies of AM treatment included a total of 265 studies published until the end of 2010.17,18 Of these, 74 were prospectively comparative studies (including 38 randomized clinical trials ((RCTs)), there were 90 prospective and 52 retrospective studies without comparison groups, and 49 retrospective studies with a comparison group. In 38 of the 265 studies, the AM therapy system as a whole (including AMPs and AM non-medication therapies) was assessed, 10 studies were of non-medication treatment, and 217 studies were of AMPs. The most frequent indications were cancer (treated with mistletoe AMPs: 59% of studies), acute infections (11%), pain syndromes (9%) and hepatitis B or C (6%). 17 Of the 255 studies evaluating AMP therapy, 244 (96%) showed clinical benefits, which were defined as a comparable or superior outcome to conventional treatment with regard to at least one clinically relevant parameter or a clinically significant improvement with AM. The methodological quality of the studies differed substantially, but restricting the analyses to studies with higher quality (assessed in design-specific quality rating checklists) yielded similar results. Thirty-two of 36 RCTs on single AMPs demonstrated these types of positive effects. The authors concluded that AM therapy for a broad spectrum of disorders showed predominantly good results, with few side effects, a high measure of client satisfaction and a favorable cost-effectiveness profile, compared to conventional treatment.
What is the Status of the Regulation of Anthroposophic Medicinal Products in Europe?
For an adequate understanding of the regulation status of AMPs in Europe, the following aspects are described: the background for modern drug regulation, European legal provisions, and national provisions.
Background for Modern Drug Regulation
As described previously, AMPs have been on the market in some European countries since the 1920s, with more than 2,500 different AMPs marketed since the 1950s. 86 In parallel, numerous CMPs have been introduced into the market, especially since the Second World War. One CMP, thalidomide, marketed in the years 1957–1961 as a tranquillizer without prescription and widely used by pregnant women, caused between 8,000 and 10,000 cases of congenital malformations which were often fatal, and an unknown number of stillborn offspring. 87 This catastrophe spurred the establishment of national drug regulation systems which, in the EU, have subsequently become harmonized, starting with Directive 65/65/EEC (Council directive, 1965) and onwards to Directive 2001/83/EC with subsequent amendments. Current EU drug regulation has a focus on newly developed, mostly chemically synthesized conventional MPs, which occasionally may cause very severe unexpected adverse effects. 88
European Legal Provisions
According to EU pharmaceutical legislation (Community code relating to medicinal products for human use, Directive 2001/83/EC), the sale of MPs on the market requires a marketing authorization issued by the national competent authorities or the European Medicines Agency. This also applies to AMPs,
89
with the following exceptions: • Some AMPs which are manufactured by a homeopathic procedure can be subject to a simplified registration procedure for homeopathic MPs (Directive 2004/27/EC). • Some AMPs manufactured from starting materials of herbal origin only can be regulated within the separate legal provisions for herbal MPs (Traditional-use registration: Directive 2004/24/EC; Well-established use authorization: Directive 2001/83/EC with amendments in the Directives 2003/63/EC and 2004/24/EC).
Notably, these provisions are only in part applicable to AMPs, for example, the traditional-use registration is intended for “use without the supervision of a medical practitioner” and is therefore unsuitable for AMP use requiring complex decision-making by AM expert healthcare professionals; traditional-use registration is also restricted to products of botanical origin and cannot be used for AMPs manufactured from substances of mineral or zoological origin; while well-established use authorization usually relies on clinical studies, which are only feasible for a proportion of the very large numbers of AMPs. 89
The European Commission (EC) has acknowledged the inappropriateness of current drug regulation for MPs from several medical systems or traditions, including AM, and has stated that the possibility of a separate legal framework for such MPs should be assessed.90,91
National Provisions
Germany and Switzerland have national legal provisions covering all types of AMPs. • In Germany AMPs are in part regulated as homeopathic MPs and in part according to a separate legal provision (German Medicinal Products Act, 2017)90,91 • In Switzerland AMPs are regulated within a legal provision for different types of natural MPs (Ordinance on complementary and herbal medicinal products, KPAV, 812.212.24, 2018)
82
In other European countries (e.g., The Netherlands, Sweden) AMPs are mainly regulated as other MPs, with separate provisions for AMPs to a varying and limited degree.
Discussion
The objective of this article was to provide an optimal understanding of AMPs for scientists and regulatory agencies. By executing a literature review the following topics could be described: the features of the AM system with its conceptual background (the holistic conception of the human organism and its relation to the natural environment, disease processes and their treatment), the major aspects of AMPs (definition, pharmaceutics, therapy principles, an example of AMP development, use in clinical practice, similarities and differences to conventional MPs), societal aspects, the scientific and regulatory assessment of AMPs and the regulation of AMPs in Europe. Compared to other, related articles on whole medical systems 2 and the domain of AM,9,11 this paper is focused on AMPs and goes more into detail on them.
Strengths of the article are at first the broad scope on AMPs and their background. A second strength is that it provides a good overview of the barriers to regulate AMPs within the current EU regulatory system and the issues that have to be dealt with in a future, more appropriate regulatory system (What is the Status of the Scientific and Regulatory Assessment of Anthroposophic Medicinal Products?).
Notably, in this article AMPs are not compared to MPs from other whole medical systems such as Traditional Chinese Medicine and Ayurveda (we have discussed that elsewhere 2 ). The scope of this article has other limitations: Challenges for AMPs regarding the actual regulatory requirements and quality control standards in Europe are only discussed in general and not in full detail (end of What is the Status of the Scientific and Regulatory Assessment of Anthroposophic Medicinal Products?); the regulatory status of AMPs outside Europe is not reviewed; and challenges for AMPs in the context of the current political, economic and cultural conditions are not discussed.
Future research should focus on appropriate methodologies for the evaluation of AMPs as part of the AM therapy system (for a corresponding whole-system-based research strategy for the entire AM system, see Kienle et al. 92 ), the scientific quality of its non-atomistic holistic ontological position, and the integration of AM and conventional medicine in clinical practice. Future policies should focus on appropriate ways of addressing regulatory challenges to AMPs. Hereby, one can take into account and learn from existing regulatory provisions for AMPs such as Germany, Switzerland (cf. Section “National provisions”) and Brazil (ANVISA, RDC 26/2007). 93
Conclusion
1. AMPs are part of the whole medical system of AM. 2. Conventional and anthroposophic medicinal products have several important differences. 3. All AMPs are manufactured according to good manufacturing practice and national drug regulations. 4. Several research projects have demonstrated an excellent safety status of AMPs. 5. The available evidence on effectiveness suggests that AMPs have clinical benefits. 6. Current drug regulation of AMPs in the EU and in most European countries does not sufficiently take into account the particular properties of AMPs and is therefore inappropriate.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ekhagastiftelse, Christophorus Stiftung, Mahle Stiftung, Software AG - Stiftung, Iona Stichting, and Vidarstiftelse.