Abstract
Background
Traditional Indian cosmetics and Ayurvedic medicines may contain lead. Previous studies have shown a relationship between eye cosmetic use (kohl) in children and elevated blood lead levels (BLLs) > 10 µg/dL. However, an association between Ayurvedic use and elevated BLLs in children is unknown and understudied.
Methods
We assessed the feasibility of collecting BLLs in children attending Ayurvedic outpatient settings in India. Our pilot study took place over 3 days in the summer of 2010 at a large public Ayurveda hospital and a small pediatric clinic in southern India. Using a trained interpreter, we administered a standardized questionnaire in Malayalam, assessing sociodemographics, Ayurvedic medicine use, kohl use, and other potential risk factors for lead exposure, to parents of pediatric outpatients. We also analyzed BLLs using a portable lead analyzer.
Results
The study enrolled 29 children (mean age, 3.8 years). The mean BLL was 6.7 µg/dL (SD = 3.5; range, 3.5–20.2). Seventy-two percent of the children used Ayurvedic medicine in the past 2 years and 55% reported kohl use. Mean BLL of Ayurvedic users and nonusers was 6.2 µg/dL and 8.5 µg/dL, respectively (P = .08). Kohl users had a statistically significant higher BLL than nonusers (8.0 µg/dL vs 5.3 µg/dL, P = .03).
Conclusions
It is feasible to collect BLLs in pediatric Ayurvedic outpatient clinics in southern India. Collaborative relationships with community members and hospital staff were essential. Further research is needed to investigate Ayurveda and kohl use as risk factors for elevated lead burden among Indian children.
Introduction
Lead exposure is a public health problem among India’s children. Fifty-one percent of Indian children have high blood lead levels (BLLs) >10 µg/dL. 1 Lead is a well-established potent environmental toxin that damages multiple organ systems resulting in poor birth outcomes, stunted physical growth, impaired intellectual development, lowered school performance, behavioral problems, and, ultimately, decreased productivity. 2 Blood lead at the age of 2 is inversely associated with academic accomplishment at 10 years. 3 Learning disabilities, language delay, distractibility, and poor organizational skills have also been associated with elevated BLLs. 4 Other behavioral disorders including inattention, delinquency, and aggression have been linked to increased BLL. 5 Lead competes and interferes with critical micronutrients such as iron, zinc, and calcium needed for hemoglobin synthesis, mitochondrial function, nerve cell conduction, and muscle activation. 2 Compared to adults, children are at greater risk for lead toxicity due to an incomplete blood brain barrier that permits lead’s movement into the brain and an immature central nervous system. 6
Known sources of lead exposure for Indian children include lead-containing cooking utensils, spices, and religious powders.7,8 In addition, studies have found kohl, a traditional eye cosmetic, has also been used in India as mothers apply it to their infants’ eyes soon after birth. Some do this to lessen eyestrain and others believe it could prevent the child from being cursed by the “evil eye.”9,10 One study estimated that two-thirds of kohl products purchased in Saudi Arabia and India contain lead, and almost 50% of the products sampled contained levels in excess of 84%. 10 In 2004, Saper et al. found that 20% of Ayurvedic herbal medicine products (HMPs) produced in South Asia and available in Boston stores contained heavy metals. 11 Of the products that contained lead, the median concentration was 40 µg/g. 11 If used as recommended by manufacturers, both kohl and Ayurvedic HMPs could result in lead intakes above the actionable BLL.
Traditional Indian systems of medicine are sanctioned and regulated by the Government of India. Their use is very popular for all health conditions, relatively inexpensive, and sought out by an estimated 80% of India’s population. 12 More than 500 000 registered Ayurvedic practitioners provide patients of all ages preventive and treatment services for the full range of diseases. In India, there are over 2800 hospitals, 100 clinics, and 150 medical schools solely dedicated to traditional Indian medicine. 12 Although traditional Indian cosmetics such as kohl and Ayurvedic medicines are widely considered safe, case reports of lead poisoning in infants and children associated with these products raise concern about their potential risk.13–15
The relative contribution of traditional cosmetic and medicine use to India’s childhood lead burden is unknown. Although India has begun to acknowledge and address the health impact of its enormous lead burden, 1 further epidemiological studies exploring other potential risk factors for lead toxicity such as traditional cosmetic and medicine use are needed. To begin studying this question, we conducted a brief pilot study in India to assess the feasibility of an Ayurvedic clinic-based lead risk assessment.
Methods
Study Design and Setting
We conducted a pilot feasibility study to evaluate BLLs and their correlates for children receiving care in southern India. With the assistance of the National Referral Centre for Lead Poisoning in India, the research team approached 3 Ayurvedic hospitals to serve as recruitment sites for our study. We met with the heads of the hospitals, physicians, and local leaders, and gained community buy-in through presentations and discussion. Permissions from the Boston University Medical Campus Institutional Review Board and the Ayurvedic clinics were obtained prior to participant enrollment.
Recruitment
Participants were recruited by hospital staff or received a referral by their physician. Study staff were stationed in the pediatric clinics to facilitate prompt enrollment. Individuals had to be 17 years old or younger, a resident of India, and accompanied by their parent or legal guardian. Participants did not need to be currently taking Ayurvedic medicines; however, they did need to be a patient at the clinic. Individuals were excluded if their parent or guardian was unable to provide informed consent, complete the survey, or unwilling to let their child give a blood sample. Parents or guardians interested in having their child participate were administered informed consent in Malayalam by a trained interpreter under the supervision of a study staff member. Children above the age of 7 also signed an assent form. Participants and/or parents received a copy of the consent/assent forms.
Data Collection
We developed a Herbal Product Risk Assessment Survey (see Supplemental material) to identify potential sources of lead exposure (eg, lead paint, parents’ occupation, pottery, house conditions, kohl, or cosmetic use), Ayurvedic medicine use (eg, frequency, type, source), and sociodemographic information. Parents of the participants were administered the risk assessment survey through the use of one trained interpreter.
BLLs were determined through the use of the portable LeadCare© II Blood Lead Test System (ESA Biosciences, Chelmsford, MA). 16 Study staff collected 50 µL blood using a finger-stick and capillary tube after thoroughly cleaning the fingertip with alcohol. Results were displayed in micrograms per deciliter within 3 minutes and documented by study staff. The analyzer was calibrated using a blood lead control solution. The reportable range of the test was 3.3 to 65 µg/dL; the analyzer reported either “low” or “high” when the BLL detected was out of the reportable range. 16 Participants with finger-stick BLLs > 10 µg/dL were referred to the National Referral Centre for Lead Poisoning in India for further evaluation and management. 1 The interpreter explained the results of the test and answered any questions the parents or child had.
Data Analysis
Sociodemographic data, Ayurvedic medicine use, and other exposures were reported using descriptive statistics. For the variable of interest—BLL—we determined the mean, range, and standard deviation. For associated factors, we used Wilcoxon Rank-Sum to determine relationships between BLL and risk factors. We used SAS® software, version 9.1 (SAS Institute, Cary, NC) for all analyses and P value criteria of <.05 for statistical significance.
Results
One Ayurveda hospital and an Ayurvedic outpatient pediatric clinic in a region of southern India agreed to participate. Over the course of 3 days, 29 participants were recruited and enrolled in the study. All parents and children approached agreed to participate except for one (child refused finger-stick). A majority of the participants were under the age of 4 and 60% of participants were female (Table 1). Income data were incomplete, as many parents did not know their average annual income or were uncomfortable disclosing the information.
Sociodemographic Results.
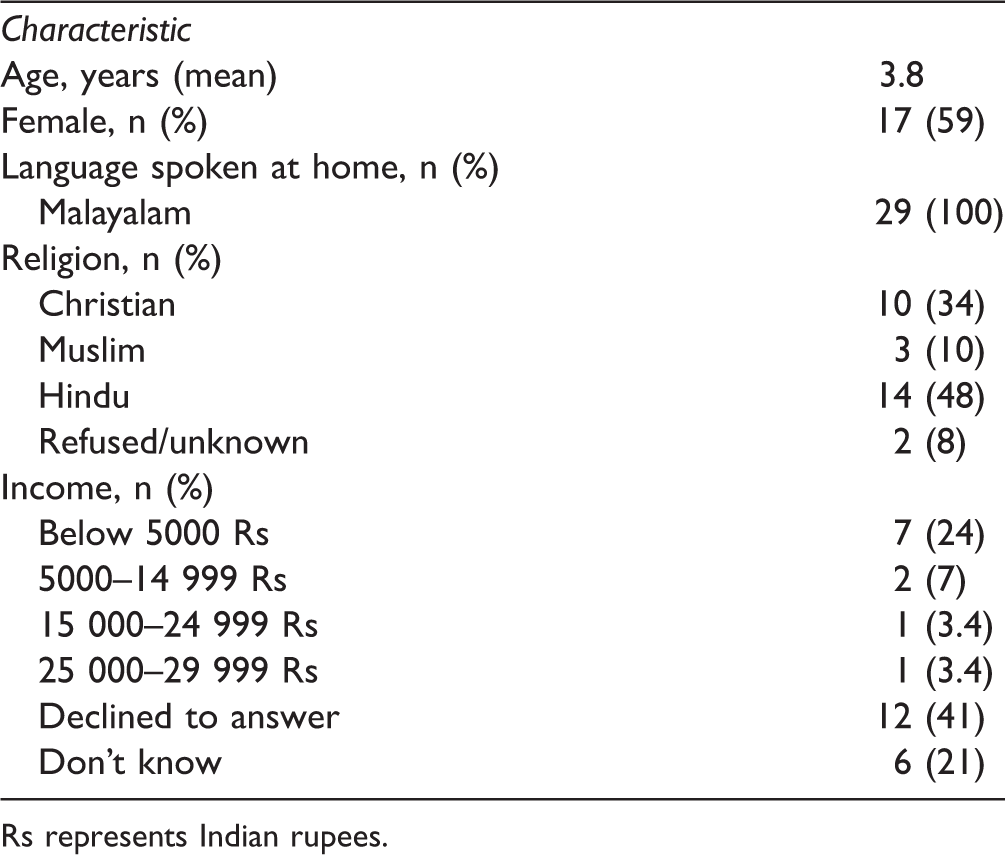
Rs represents Indian rupees.
Blood Lead Levels
The mean BLL for participants was 6.7 µg/dL (SD = 3.5; median, 5.9; and range, 3.5–20.2). Of the 29 participants, 3 had BLLs greater than 10 µg/dL and were referred for further testing and treatment through the National Referral Centre for Lead Poisoning in India. The distribution of BLLs is displayed in Figure 1.

Distribution of Blood Lead Levels Among 29 Children Attending Ayurvedic Clinics.
Prevalence of Ayurvedic Use
Results from the Herbal Product Risk Assessment Survey revealed that 75.8% (n = 22) of participants have ever used Ayurvedic medicine with 72% (n = 21) having used it in the past 2 years. Parents reported giving their children Ayurvedic medicine for a variety of health issues, detailed in Table 2. The most common reason reported for Ayurvedic use was for “cough and cold,” fever, and developmental delay. Parents reported obtaining these medicines from a variety of sources: Ayurvedic hospital (n = 16; 76%); local dispensary or pharmacy (n = 3; 14.3%); made at home (n = 1; 4.8%); and the local market (n = 1; 4.8%).
Reason for Ayurvedic Use.

Risk Assessment Associated Factors and Blood Lead Levels.
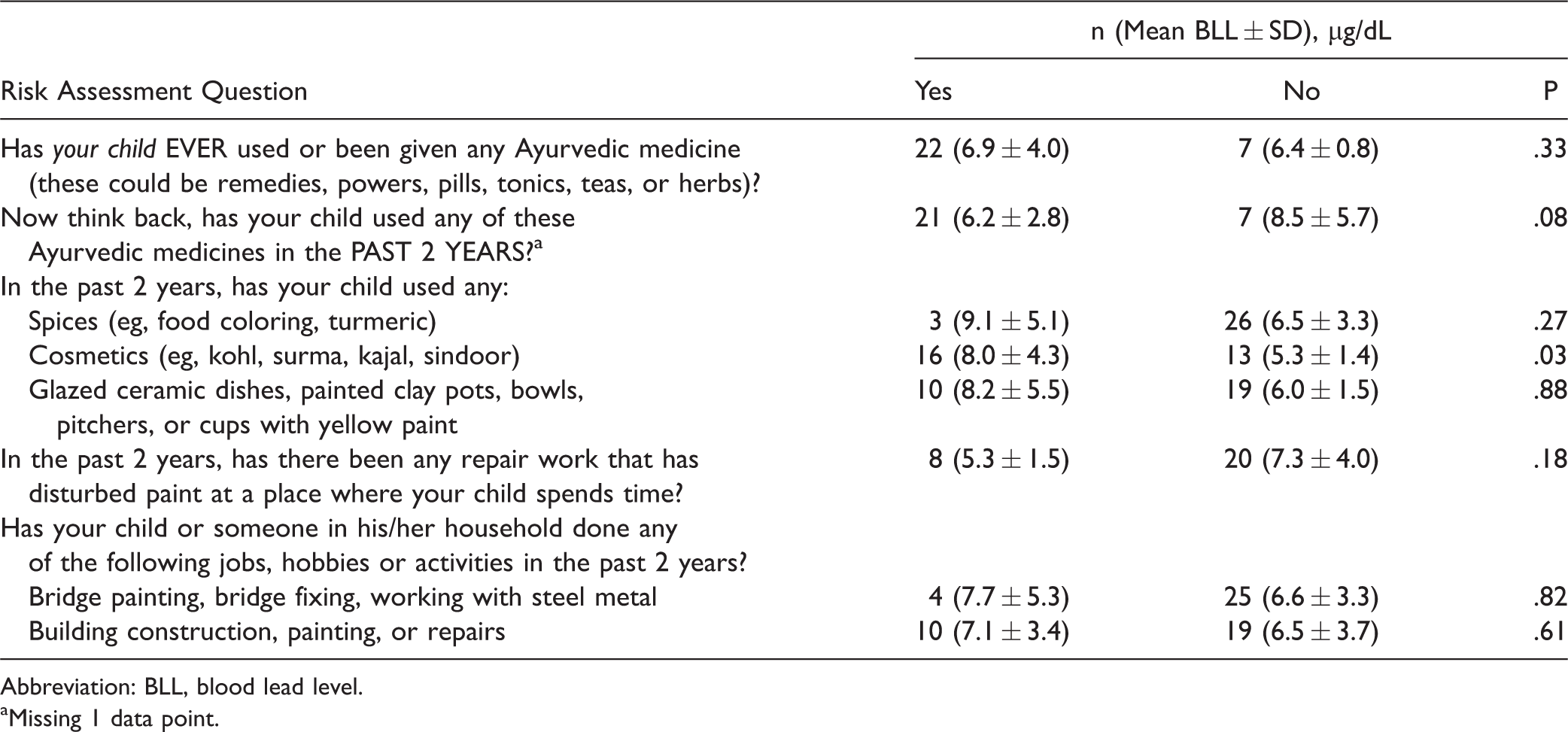
Abbreviation: BLL, blood lead level.
aMissing 1 data point.
Prevalence of Alternative Sources of Lead Exposure
Over half of the parents reported that their child wore topical kohl under their eyes (55.2%). Ten parents (34.4%) said that they cooked for their child using clay pots. Nine participants (31%) were reported to chew on pencils and 10 parents (34.4%) reported that the father’s occupation involved building construction, painting or repairs.
Associated Factors
There was a statistically significant difference (P = .03) in the mean BLL of participants who used kohl (7.9 µg/dL) compared to those who did not use kohl (5.5 µg/dL) (Table 3). Using Satterthawaite method, we assumed the population variances were unequal (F = 9.12). There was not a statistically significant difference between the BLL of participants who used Ayurvedic medicine in the past 2 years (6.2 µg/dL) and those who had not (8.4 µg/dL).
Discussion
This study demonstrated that it is feasible to collect BLLs in pediatric Ayurvedic outpatient clinics. Children who wore kohl cosmetics had statistically significant higher BLLs than nonusers. The relationship between reported Ayurvedic use and BLLs was not statistically significant. Relationships with community members and hospital staff were essential to successfully conducting the study.
BLLs were slightly lower than other prevalence data for children in India. 1 This may be a result of our small sample size or population differences (previous studies sampled children living in large cities). Analysis of associated factors of lead exposure suggests a relationship between topical kohl use and elevated BLLs, similar to other research findings.10,17
In this small study, we did not find an association between BLLs and parent-reported Ayurvedic use. This may be due to a greater emphasis on herbal Ayurvedic preparations compared to herbometallic preparations in Southern India. Rasa Shastra, a category of traditional Indian medicine where herbs are intentionally combined with metals, minerals, and gems, may be prescribed less often in the study sites.
These data underscore the need for larger studies of risk factors for lead exposure in Indian children. Presentations to the clinic administrators were key in gaining access and trust between the clinic and study team. Presenting the goal of the research study as a joint collaboration to promote Ayurvedic medicine by addressing unanswered questions regarding Ayurvedic safety and elevated lead levels appeared to be the most effective approach. The experience of identifying sites for the study highlighted the need for collaboration between Ayurvedic practitioners, stakeholders, and outside investigators wishing to do further studies in this area of research. Currently, the National Referral Centre for Lead Poisoning in India provides resource information to clinicians treating cases of lead poisoning and assists in research activities in the field of toxicology with special reference to lead. 1
Although many studies have revealed the high metal content of Ayurvedic medicine14,15,17,18,19 and prevalence of high lead levels among Indian children, 1 to the best of our knowledge, none have attempted to gather BLL from children at Ayurvedic clinics and hospitals.
A majority of the research linking elevated BLLs and Ayurvedic medicine use focuses on case studies. Our study highlights some of the successes and challenges of using this method. Basing our study at Ayurvedic clinics located within the community and easily accessible to our population contributed to participant recruitment. Getting buy-in from the clinic staff and administration proved to be the greatest challenge of the study. Leveraging our relationship with the National Referral Centre for Lead Poisoning and showing our research aims as mutual goals between our U.S.-based study team and the field of Ayurvedic medicine were both essential in creating trust.
The strengths of our study include community-based research and standard reproducible data collection methods. Limitations include low sample size. Small sample size led to unequally distributed BLLs and also precluded multivariate analyses. Despite our efforts to craft a culturally competent risk assessment, several of the questions were confusing to parents (eg, health insurance type, previous diagnosis of a learning disorder). In addition, parents seemed to suffer from survey exhaustion from the 9-page assessment. Due to lack of resources, we were unable prior to administration to do formal validity testing of the survey, reliability assessment, or back-translation. Finally, survey responses on use of Ayurvedic medicine and kohl were subject to recall bias.
Conclusions
Conducting an Ayurvedic clinic-based pilot study to gather BLLs and exposure assessment data was feasible. An association between kohl use and BLLs was found, consistent with prior research. Future studies in Ayurvedic treatment settings using improved risk assessment instruments with a larger sample size are necessary to better understand the relationship between Ayurvedic medicine use and BLL in children in India.
Supplemental Material
Supplemental material for Blood Lead Levels of Children Using Traditional Indian Medicine and Cosmetics: A Feasibility Study
Supplemental Material for Blood Lead Levels of Children Using Traditional Indian Medicine and Cosmetics: A Feasibility Study by Julia Keosaian MPH Thuppil Venkatesh PhD Salvatore D’Amico BS Paula Gardiner MD, MPH Robert Saper MD, MPH in Global Advances in Health and Medicine
Footnotes
Acknowledgments
The authors would like to thank the National Referral Centre for Lead Poisoning in India and TA Varkey.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Saper was supported by a Career Development Award (K07 AT002915-05) from the National Center for Complementary and Integrative Health, National Institutes of Health.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
