Abstract
Present study evaluates efficacy of Trifala and Ela as plaque controlling agent and compares it with chlorhexidine.
Aim.
To evaluate Antigingivitis, Antiplaque and Antihalitosis effect of Triphala and Ela decoction. A randomized sample of 60 patients with plaque induced gingivitis was enrolled and equally divided into two groups group A and group B. Group A was given Trifala and Ela decoction and Group B Chlorehexidine mouthwash for 21 days twice daily. Gingival inflammation index, plaque index and Organoleptic scoring scale was recorded at baseline, 14th day and 21st day. Comparing the plaque index for Group A with group B the reduction in from baseline to 14 day was 42.59 % and 38.62% respectively while from baseline to 21 day was 56.20% and 68.57% respectively. On comparing Gingival index for group A with group B the reduction from baseline to 14 day was 31.95% and 38.62 % respectively while from baseline to 21 day was 69.95 % and 68.57% respectively. Halitosis Percentage reduction at 14th day from base line was 33.33% and 38.18%; at 21 day from baseline 66.66% and 72.72% respectively for group A and group B. No statistical significant difference for intergroup comparison was found using paired t test. Intra group analysis using unpaired t test was significant for all the indices at different time intervals. Triphala and Ela decoction is organic, easy to prepare economical and equally effective as compared to chlorhexidine mouthwash.
Ayurveda is an ancient Indian system of medicine that offers a vast pool of herbal medication effective and often considered free of toxicity of modern allopathic preparations. In recent times, a lot of clinical trials have been conducted, evaluating the age-old science of ayurveda by testing it against the gold standard allopathic preparations used for established treatment modalities. 1 Kaval (mouthwash) is described in ayurvedic text for the treatment of various oral and dental problems.
Gingivitis is one of the most prevalent oral diseases worldwide and is caused by the bacterial biofilm surrounding the teeth. Gingivitis leads to swollen gums, bleeding gums and halitosis. 2 all these conditions results in poor oral hygiene and a cascade of events causing early loss of teeth leading to dental aesthetics and function problems.
To control the 2 most challenging problems of oral cavity, namely, gingivitis and halitosis, an array of chemical agents have been employed in the past. Chlorhexidine is one such agent, which has stood the test of time and is considered a gold standard in chemical plaque control. Chlorhexidine is available as mouthwash, topical gel, and in biodegradable form. It has a broad-spectrum antimicrobial activity and is a cationic molecule, which causes bacterial cell wall lysis and rupture. Cationic molecules bind to the negatively charged surfaces such as teeth surface, dental pellicle, and bacterial cell wall. Chlorhexidine has a substantivity of 12 hours and hence is used twice daily. 3
Triphala consists of equal parts of the Emblica officinalis, Terminalia chebula, and Terminalia belerica. 4 Ela (Elettaria cardamomum), commonly known as green or true cardamom, is an herbaceous perennial plant in ginger family native to southern India. It is described as a digestive, aromatic, deodorant, fragrant, carminative herb and chewed to prevent unpleasant odor of oral cavity. 5
The present clinical trial compares these 2 agents for their effectiveness in controlling plaque, gingivitis, and halitosis. This study explores use of herbal agent such as Triphala and Ela in combatting oral infections such as plaque-induced gingivitis. Aim of the present study is to evaluated efficacy of such herbal products and their effectiveness as a plaque-controlling agent.
Subjects and Methods
Sixty patients with plaque-induced gingivitis were enrolled, divided into 2 groups of 30 each by random computer allocation. Group A was given Triphala and Ela decoction for 21 days and group B was given chlorehexidine mouthwash for 21 days twice daily after cleaning the oral cavity with water. The decoction/ mouthwash was used for 60 seconds by vigorous swishing in mouth (mouth rinse). All the patients were kept on standardized oral hygiene protocol. Gingival index, plaque index, and organoleptic scoring scale were recorded at baseline, 14th day, and the 21st day. 6 –8
Statistical analysis was carried out using SPSS version 20.0 (SPSS, Inc, Somers, NY, USA). A comparison of the mean differences of the gingival and plaque indexes was done by a paired t test within the groups at different time intervals and unpaired t test was used when intergroup comparison was made. Level of significance was set at P ≤ .05 (95% confidence interval).
Informed consent was taken from the patients and ethical clearance was obtained by the ethical committee of Himalayan Institute of Medical Sciences vide letter no. HIHTU/HIMS/RC/2013/307. The study was conducted in accordance with Helsinki declaration. 9
The inclusion criteria were the following: Age >18 years Systemically healthy Plaque-induced gingivitis Halitosis
The exclusion criteria were the following: Mouthwash use in past 3 months Antibiotic therapy in past 3 months Orthodontic and prosthetic appliances use Systemic disorders like diabetes mellitus, renal failure, and so on Pregnancy Smoking Chemotherapy and radiotherapy Patients with established periodontitis
Standard protocol followed for preparation of Triphala and Ela decoction under in-house pharmacy of Ayurvedic Center of Swami Rama Himalayan University under expert guidance of an ayurvedic physician.
All the 4 herbs were procured from the local market after proper identification. Coarse powder of Triphala and fine powder of Ela seeds was prepared at the Ayurvedic Centre, Himalayan Institute Hospital Trust. Triphala kwath was made as per Kwath prakarana of Ayurveda Sar Sangraha and 3 g Ela churna was mixed in 100 mL of Triphala. 10 A commercial preparation of chlorhexidine—Chlorhex Plus mouthwash manufactured by Dr Reddys (batch no. BCP2067) was used.
Results
At baseline, there was no statistical difference between the 2 treatment options for gingival and plaque indexes.
Plaque Index
Both treatment groups reported significant reduction in plaque index from baseline. There was no significant statistical difference observed between the 2 treatment groups.
Triphala and Ela: Group A
The values of mean ± SD for plaque index at baseline, 14 days, and 21 days were 2.16 ± 0.324, 1.242 ± 0.246, and 0.946 ± 0.147, respectively (Table 1). There was a statistically significant difference in plaque index from baseline to 14 days and from baseline to 21 days.
Plaque Index and Gingival Index for Group A at Various Time Intervals.

Chlorhexidine group: Group B
The values of mean ± SD for plaque index at baseline, 14 days, and 21 days were 2.28 ± 0.154, 1.24 ± 0.152, and 0.916 ± 0.137, respectively (Table 1). There was a statistically significant difference in plaque index from baseline to 14 days and from baseline to 21 days. On comparison of plaque index for group a with group B, the reduction from baseline to 14 days was 42.59% and 38.62%, respectively, while from baseline to 21 days the reduction was 56.20% and 68.57%, respectively. This difference was statistically not significant (Figure 1; Table 2). Intergroup comparison of group A and group B for plaque index (mean ± SD) at baseline to 14 days was 0.92 ± 0.078 and 1.04 ± 0.002, respectively, while from baseline to 21 days, mean ± SD was 1.214 ±0.177 and 1.364 ± 0.017, respectively (Table 3).

Plaque index (PI) of group A versus group B.
Percentage Reduction in Plaque Index (PI) and Gingival Index (GI) at Various Time Intervals.

Intergroup Comparison of Plaque Index (PI) and Gingival Index (GI) (Mean ± SD) at Various Time Intervals.

Gingival Index
Both treatment groups reported significant reduction in gingival index from baseline. There was no statistical significant difference observed between the 2 treatment groups.
Triphala and Ela Group: Group A
The values of mean ± SD for gingival index at baseline, 14 days, and 21 days were 1.784 ± 0.232, 1.214 ± 0.163, and 0.536 ± 0.124, respectively (Table 4). There was a statistically significant difference in gingival index from baseline to 14 days and from baseline to 21 days.
Plaque Index and Gingival Index for Group B at Various Time Intervals.

Chlorhexidine Group: Group B
The values of mean ± SD for gingival index at baseline, 14 days, and 21 days were 1.89 ± 0.138, 1.16 ± 0.129, and 0.594 ± 0.163, respectively (Table 4). There was a statistically significant difference in gingival index from baseline to 14 days and from baseline to 21 days.
On comparison of group A with group B, the reduction in gingival index from baseline to 14 days was 31.95% and 38.62%, respectively, while from baseline to 21 days it was 69.95% and 68.57%, respectively. This difference was statistically not significant (Figure 2; Table 2). Intergroup comparison of group A and group B for gingival index (mean ± SD) at baseline to 14 days was 0.57 ± 0.069 and 0.73 ± 0.009, respectively, while from base line to 21 days it was 1.248 ± 0.108 and 1.296 ± 0.02, respectively (Table 3).
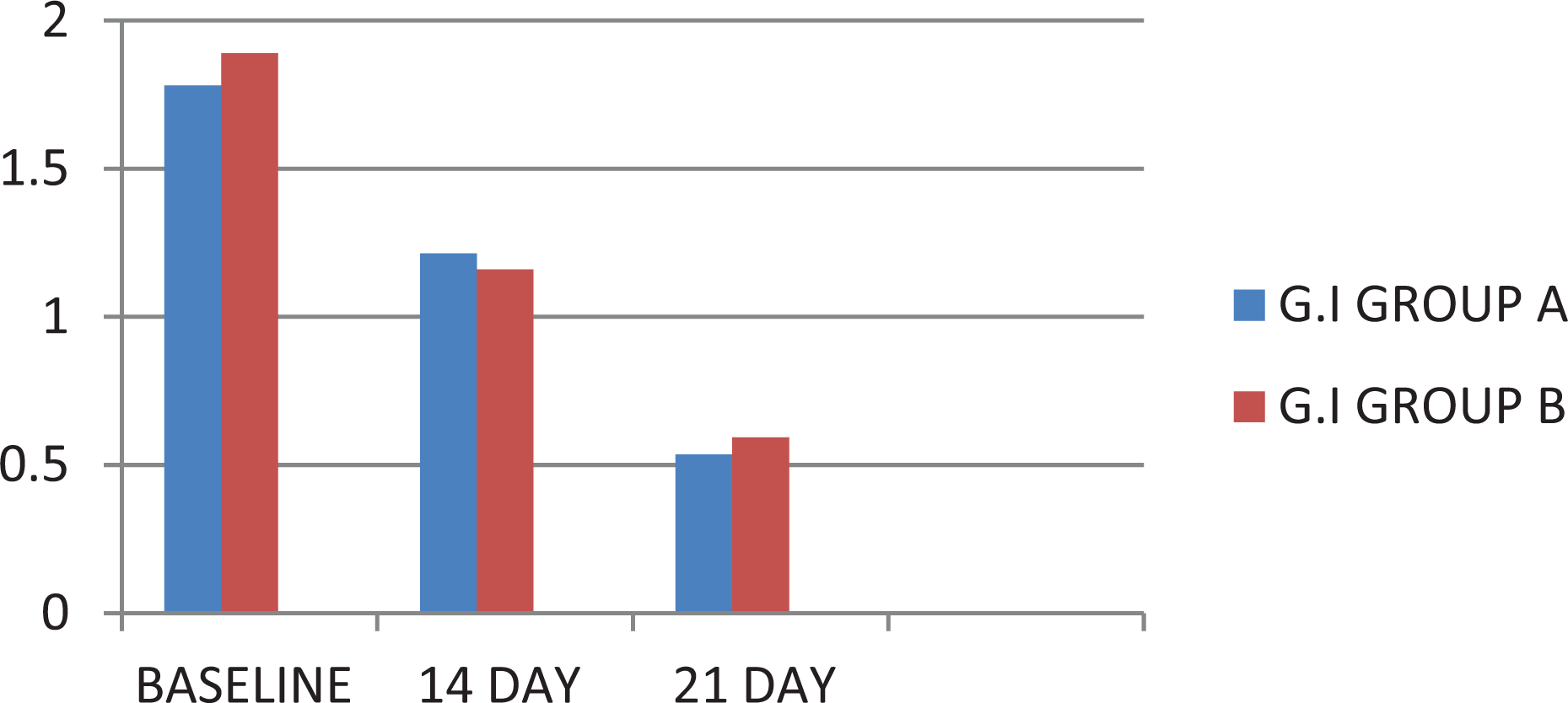
Gingival index (GI) of group A versus group B.
Significant reduction in halitosis grading mean reported in both groups. The average grading at baseline was 5.4 ± 0.342 and 5.5 ± 0.453 for group A and group B, respectively, which reduced to 3.6 ± 0.125 and 3.4 ± 0.273 on the 14th day and further to 1.8 ± 0.542 and 1.5 ± 0.491 on the 21st day, respectively. Percentage reduction at 14th day from baseline was 33.33% and 38.18% for group A and group B, respectively. At 21 days from baseline, the reduction was 66.66% and 72.72%, respectively, for group A and group B (Table 5). Within group, there was significant reduction in halitosis for both groups from baseline to 14 days and 21 days. There was no statistical difference between the 2 groups for halitosis reduction (Figure 3; Table 5).
Halitosis Grading (Mean ± SD) for Both Groups at Various Time Intervals.
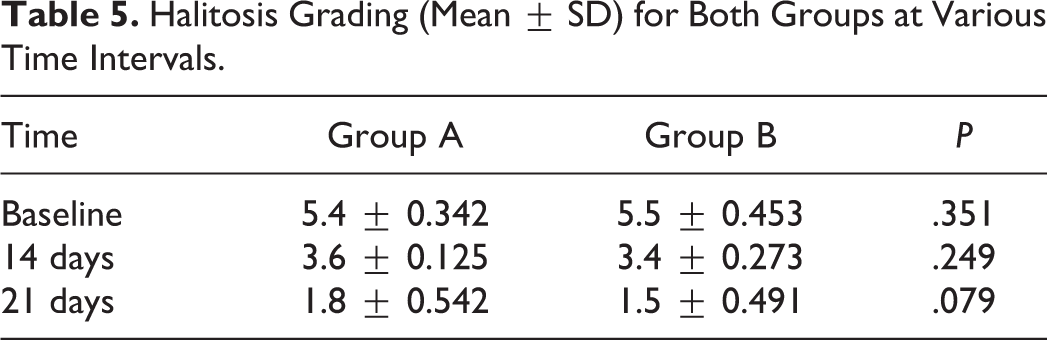

Halitosis grading of group A versus group B.
Discussion
The present study was designed to evaluate the effect of an herbal decoction in gingivitis, halitosis, and plaque inhibition. Till date there are limited studies on use of Triphala and Ela as mouth rinse to treat the aforementioned oral conditions.
Triphala has been used as decoction, powder, and in oil form to treat shitada or gingivitis in Sushruta Samhita, Chikitsa Sthan while describing treatment of mukh roga (diseases of mouth). 11 Various scientific studies on Triphala indicate its antimicrobial property and its growth inhibitory activities against common bacterial isolates. 12 –15 It has also been show to possess antioxidant properties, free radical scavenging, and thus is an appropriate formulation for further evaluation of its effect on plaque-induced gingivitis. 16,17
According to ayurveda, Triphala is tridoshahar, that is, vata, pitta, and kapha balancing in nature. Separately, Haritaki has been mentioned as tridoshahar. It has shothahar (anti-inflammatory), vranashodhan (wound cleansing), vedanasthapan (analgesic) actions, which are helpful for treating the inflammation of gingiva. Amalaki is also a tridoshhar. It is known as vedanasthapan (analgesic) that alleviates pain. It is a rich source of vitamin C, which is required for collagen maturation and is essential to maintain firm and healthy gingiva. Vibhitaki is also tridoshhar and apart from other beneficial actions it has vedanasthapan (analgesic) properties. 11 Ela churna was mixed with Triphala decoction to treat the bad odor of mouth as it is known as mukhashodhan and was found to have potential immunomodulatory action, and antimicrobial against dental caries–causing organisms. 18 –20 The earliest clinical trial on Triphala and its role in periodontal disease was conducted by Maurya et al, 21 who studied effect of Triphala alone and as an adjunct to metronidazole in treatment of periodontal disease. It was concluded that the drug Triphala or metronidazole alone is capable of providing partial relief but when both are used in a combined form, they are more effective for the treatment of periodontal disease. Similar findings were concluded in a recent trial by Tripathi and Tiwari. 22 The present study also showed effectiveness of Triphala in controlling plaque-induced gingivitis that is considered first stage of periodontal disease. An in vitro study by Prabhakar et al 23 reported that Triphala is efficient in inhibition of plaque biofilm and hence is effective in treating tooth biofilm infection. Similar outcomes were seen in present study where the plaque index improved for Triphla and Ela group at 14 and 21 days from baseline. And this improvement was similar to that of the chlorhexidine group.
A randomized trial conducted by Bajaj and Tandon 24 concluded that there is no difference between Triphala and chlorhexidine mouthwash on gingivitis and plaque formation. A concentration of 0.6% of Triphala was used against 0.1% chlorhexidine and both were efficient in controlling gingivitis and plaque biofilm formation. Also, it was seen that Triphala and chlorhexidine showed similar inhibitory effect on microbial counts, except lactobacillus, where Triphala has shown better results than chlorhexidine. 24
Similar concentration of Triphala was used in a study by Gupta et al, 25 wherein 0.6% Triphala was highly effective in preventing plaque accumulation and gingivitis as compared with 0.2% chlorhexidine mouth rinse.
Bhattacharjee et al 26 conducted a study on 57 schoolchildren and compared chlorhexidine and Triphala mouthwash. They concluded that the effectiveness of Triphala in the reduction of plaque and gingivitis was comparable to chlorhexidine and that Triphala can be used for short-term purposes without potential side effects. 26
In the present study, it was seen that Triphla and Ela group and chlorhexidine group reported similar effect on gingival index indicating that both the preparations were effective in controlling gingivitis and plaque buildup at 14 and 21 days.
None of the previous studies have evaluated improvement in halitosis grading for Triphala and Ela mouth rinse. Both groups reported reduction in halitosis and improvement in halitosis grading at 14 and 21 days compared with baseline grades.
Triphala has been recently reported to have an in vitro effect on drug modulating enzymes. Disturbance on cytochrome P450 is mentioned, thereby the likelihood of herb-drug interactions, if these are administered concomitantly, can be expected. 27 Hence care should be taken in patients under multiple drugs and more scientific relevant clinical evidence is required to advocate its use as a sole or adjunct therapy in controlling gingivitis, plaque build-up, and halitosis.
Conclusion
The present study shows that Triphala and Ela decoction has similar efficacy as that of chlorhexidine in reducing dental biofilm build-up, gingivitis, and halitosis. It is easy to prepare, organic, and relatively economical compared with chlorhexidine. Hence it can be used for effective plaque control, gingivitis control, and halitosis. Limitations of our study included small sample size and the inability to document long-term side effects, if any, of Triphala and Ela decoction. Further pharmacological studies are needed to assess for the drug interaction ability of Triphala and Ela and understand the possible drug interactions if any.
Footnotes
Author Contributions
PM contributed toward conception of design, preparation of Triphala & Ela mouthwash, interpretation of data, writing and review of manuscript. AK contributed toward conception of design, collection of dental data, gingival index and plaque index, interpretation of data, writing of manuscript, and responsibility of work. RKM contributed toward conception of design, preparation of Triphala Ela mouthwash, interpretation of data, writing, and review of manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the ethical committee of Himalayan Institute of Medical Sciences (HIHTU/HIMS/RC/2013/307).
