Abstract
The clinical effectiveness and value of camel milk as a therapeutic agent is currently unclear. MEDLINE (1946 to March 2016), EMBASE (1974 to March 2016), and Google Scholar were searched using the following terms: milk, bodily secretions, camels, camelus, camelini, camelidae, dromedary, bactrian camel, body fluid, and bodily secretions. Articles identified were reviewed if the study was investigating the use of camel milk for the potential treatment of diseases affecting humans. Of 430 studies, 24 were included after assessment. Identified studies highlighted treatment with camel milk of diseases, including diabetes, autism, cancer, various infections, heavy metal toxicity, colitis, and alcohol-induced toxicity. Although most studies using both the human and animal model do show a clinical benefit with an intervention and camel milk, limitations of these studies must be taken into consideration before widespread use. Based on the evidence, camel milk should not replace standard therapies for any indication in humans.
Camel milk is used extensively within a variety of populations for its proposed healing properties and disease prevention mechanisms. 1 Some of the more common indications associated with its use include diabetes, allergies, immune disorders, and cancer. 2 It is also advocated as an alternative to cow’s milk for those who are allergic or intolerant to cow milk proteins. 3 Although traditionally used within Middle East, Africa, and Asia, the advent of online pharmacies and natural health product awareness has increased the availability of camel milk to nontraditional settings such as North America and Europe. Therefore, clinicians must be aware of its properties, stated claims, and clinical data when encountering patients and making therapeutic management plans.
There are 2 distinct species of camel: Camelus dromedarius (also known as the dromedary, Arabian camel, or one-hump camel), and the Camelus bactrianus (also known as the Bactrian, or two-hump camel). The Bactrian is found in the cold desert regions of Central Asia, whereas the dromedary is native to the hot deserts of North Africa and Western Asia. 2 Milk from both types of camels is composed of high mineral and vitamin content, high unsaturated fats, and protein with some differences in macronutrients that varies depending on the region. 4 The scientific rationale for the use of camel milk as a natural health product comes primarily from its purported antioxidant, immunomodulating, anti-inflammatory, insulin-like, and anti-apoptotic attributes. 4 –9 These properties were largely determined through in vitro studies and therefore only provide hypothetical mechanisms of benefit.
The clinical effectiveness and value of camel milk as a therapeutic agent is currently unclear. Although studies in animal and human populations exist, they are mostly small in nature and assess a wide variety of indications and populations. However, patient beliefs may drive use of this agent as a therapeutic alternative or supplemental health product to modern medical practices. 10 Therefore, a critical review is necessary to provide clinicians with a strong background in effectiveness data of camel milk as a health product.
The objective of this systematic review was to summarize and evaluate literature regarding the therapeutic efficacy and safety of camel milk as both a therapeutic agent and natural health product supplement.
Materials and Methods
We conducted a systematic computerized search from the inception date (1946 in MEDLINE and 1974 in EMBASE) to March 30, 2016 to identify all potentially eligible studies. We applied the following algorithm in both medical subject heading (MeSH) and free text words. In MEDLINE, the MeSH terms “milk” OR “bodily secretions” were combined with “camels.” Free text words “camel” OR “camelus” OR “camelini” OR “camelidae” OR “dromedary” OR “bactrian camel” were also included in the search as these were not MeSH terms. In EMBASE, the MeSH terms “milk” OR “camel milk” OR “body fluid” OR “bodily secretions” were combined with “camel” OR “Bactrian camel.” Free text words “camelus” OR “camelini” OR “camelidae” were also included in the search as these were not MeSH terms. International Pharmaceutical Abstracts (1970-October 2015) and Google Scholar were also searched, and references of relevant publications were reviewed to identify any articles not captured during initial search.
The articles identified were reviewed if the study was investigating the use of camel milk for the potential treatment of diseases affecting humans. Therefore, studies investigating the use of camel milk for potential veterinary uses were not included. Studies including either humans or animals were included in this review. Articles were excluded if only in vitro data were reported or if published in a language other than English.
Information extracted from studies included design, species of camel, description of study population, number of participants, intervention including quantity and frequency of use, and study outcomes (differed depending on which disease state the intervention was being used for). Adverse reactions to any interventions were also recorded if available. Data were extracted by one investigator and checked for accuracy by a second investigator.
Results
Our search resulted in 430 studies after removal of duplicate studies. Manual search of reference lists did not identify additional studies. After screening and assessing for eligibility, 24 studies 11 –34 were included with the remaining articles being excluded for reasons of irrelevance or containing only in vitro data. Among the published trials investigating the effects of camel milk, 9 studies in animals (4 studies for diabetes, 11 –14 2 studies for cancer, 15,16 1 study for colitis, 17 2 studies for various infections, 18,19 2 studies for heavy metal toxicity, 20,21 and 1 study for alcohol-induced toxicity 22 ) and 12 studies in humans (8 studies for diabetes 23 –30 and 4 studies for autism 31 –34 ) (Figure 1). No studies identified were published in a language other than English. Study characteristics and results involving animal models are summarized in Table 1 and studies involving human participants are summarized in Table 2.
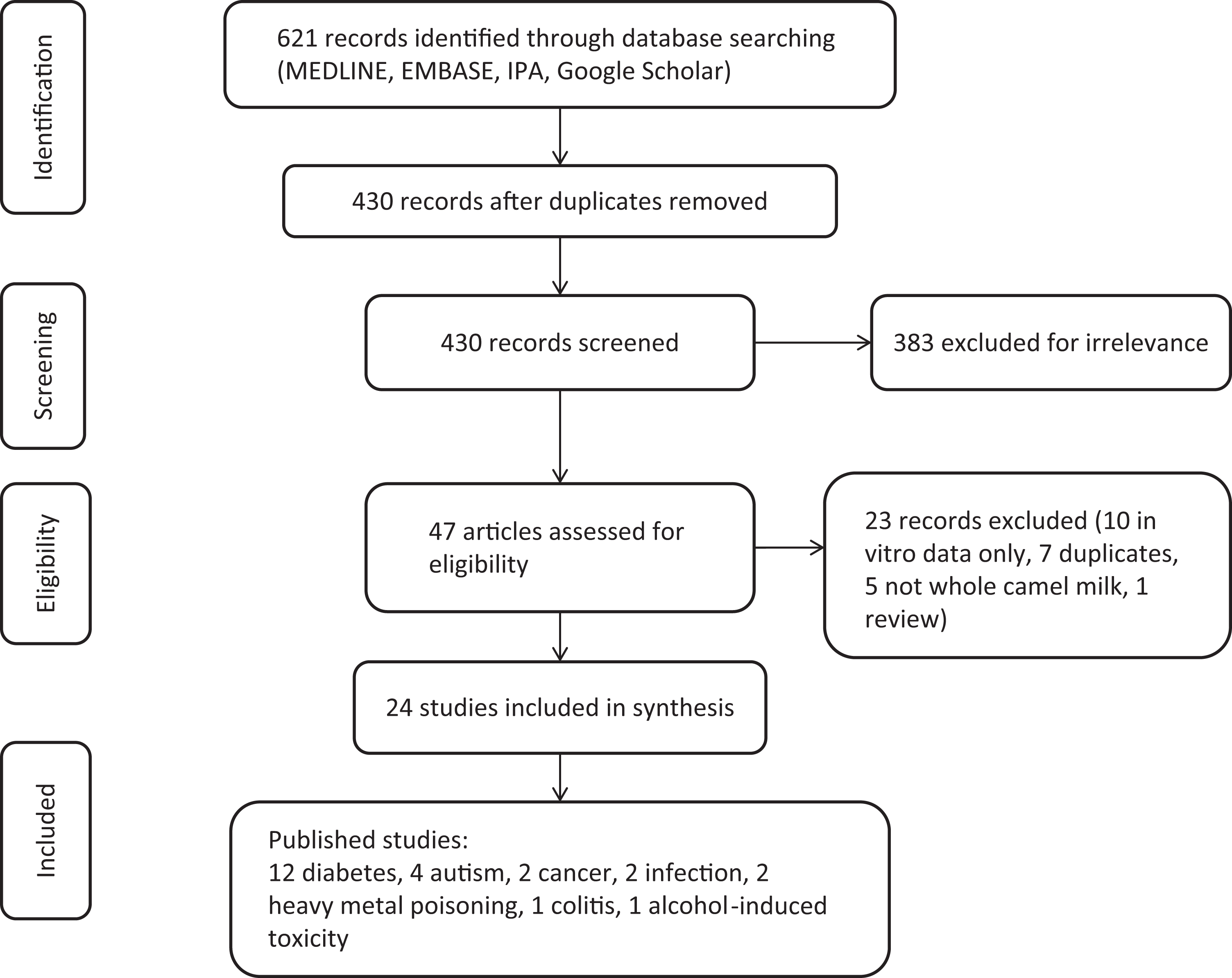
Search strategy flow diagram.
Outcomes for Studies Using Animal Models.

Abbreviations: ALP, alkaline phosphatase; ALT, alanine transaminase; AST, aspartate transaminase; AUC, area under the curve; BMI, body mass index; CAT, catalase; CM, camel milk; Col4, collagen type 4; DBP, diastolic blood pressure; DM, diabetes mellitus; ELISA, enzyme-linked immunosorbent assay; EtOH, ethanol; FBG, fasting blood glucose; GGT, gamma-glutamyl transpeptidase; GIP, gastric inhibitory peptide; GLP-1, glucagon-like peptide 1; GSH, reduced glutathione, GST, glutathione-S-transferase; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; HOMA-IR, homeostatic model assessment insulin resistance; HP, haptoglobin; IFG, impaired fasting glucose; IgG, immunoglobulin G; IGT, impaired glucose tolerance; IL, interleukin; IP, intraperitoneally; LBR, liver-to-body weight ratio; LDH, lactate dehydrogenase; LDL, low-density lipoprotein; MCP-1, monocyte chemoattractant protein 1; MCH, mean corpuscular hemoglobin; MCHC, mean corpuscular hemoglobin concentration; MCV, mean corpuscular volume; MDA, malondialdehyde; McPCE, micronucleated poly-chromatid erythrocyte; MPO, myeloperoxidase; NAG, N-acetylglucosaminidase; NO, nitric oxide; OGTT, oral glucose tolerance test; mRNA, messenger ribonucleic acid; SBG, serum blood glucose; SBP, systolic blood pressure; SC, subcutaneously; SOD, superoxide dismutase; STZ, streptozocin; SWAP, soluble worm antigen preparation; TAC, total antioxidant capacity; TBARS, thiobarbituric acid reactive substances; TC, total cholesterol; TFG, transforming growth factor; TG, triglycerides; TNBS, trinitrobenzene sulfonic acid; TNF, tumor necrosis factor; VEGF, vascular endothelial growth factor; VLDL, very low density lipoprotein; WHR, waist-to-hip ratio.
Outcomes for Studies Using Human Models.

Abbreviations: ASD, autism spectrum disorder; AUC, area under the curve; BMI, body mass index; CARS, Childhood Autism Rating Scale; DBP, diastolic blood pressure; DM, diabetes mellitus; FBG, fasting blood glucose; HbA1c, glycated hemoglobin; HDL, high-density lipoprotein; HOMA-IR, homeostatic model assessment insulin resistance; IFG, impaired fasting glucose; IGT, impaired glucose tolerance; LDL, low-density lipoprotein; OGTT, oral glucose tolerance test; SBP, systolic blood pressure; TARC, thymus and activation regulated chemokine; TC, total cholesterol; TG, triglycerides; VLDL, very low density lipoprotein; WHR, waist-to-hip ratio. *p < 0.05.
Discussion
Animal Trials
Diabetes
Presence of insulin in nanoparticles or an insulin-like substance in camel milk are theories that have been proposed to explain the traditional belief that consumption of camel milk helps prevent and control diabetes. 5 The therapeutic effect of camel milk given to animals with artificially induced diabetes was investigated in 4 studies 11 –14 (Table 1). Khan et al 20 conducted a randomized controlled trial to assess the effect of camel milk on rats with streptozocin-induced diabetes. After 30 days, they found that rats with diabetes had a 24.8% decrease in weight compared with healthy controls, but camel milk increased body weight to near normal. They also observed a significant increase in fasting plasma glucose (FPG) in diabetic rats compared with controls and lower FPG in diabetic rats given camel milk. However, FPG was still significantly higher in the camel milk group than the healthy control group and the group treated with insulin. Likewise, they found a statistically significant increase in liver enzymes, markers of renal function, and lipid panel in the diabetic group compared with the control group and a decrease in all these parameters with the administration of camel milk.
Similar parameters were assessed in a series of studies by Korish and colleagues. 12,13 Two of the studies were randomized controlled trials looking at rats with streptozocin-induced diabetes. 12,13 They found similar trends in body weight, FBG, and fasting insulin as the study by Khan et al. 20 One of the trials also looked at lipid levels and found significantly higher total cholesterol, triglycerides, low-density lipoprotein cholesterol, and very low density lipoprotein cholesterol, and significantly lower high-density lipoprotein in the streptozocin-induced diabetes group. 12 The levels of randomized controlled trial, triglycerides, low-density lipoprotein cholesterol, and very low density lipoprotein cholesterol decreased when camel milk was given to diabetic and healthy rats, and high-density lipoprotein increased in diabetic rats. This study also assessed the effect of diabetes and camel milk on glucagon-like peptide-1, glucose-dependent insulinotropic peptide, and tumor necrosis factor (TNF)-α and TNF-β. They found a significant increase in all 4 markers in diabetic rats compared with controls, and a decrease in diabetic rats given camel milk compared with those not receiving any. The administration of camel milk to healthy rats did not show any change in these markers compared with controls. The second trial also assessed the effect of camel milk on diabetic nephropathy. 13 The authors found that rats with diabetic nephropathy had a significant increase in urine volume, serum urea, serum creatinine, and proteinuria, which were lower in the group treated with camel milk. When camel milk was given to healthy rats, it decreased urine volume compared with the control group but it did not alter any of the other parameters.
The third randomized controlled trial by Korish et al 14 assessed the effect of camel milk in rats fed either a standard diet or a high-cholesterol diet. Body weight in this study showed the opposite trend compared to the results from Khan et al, 11 highlighting the pathological differences between insulin-dependent diabetes and obesity. The lipid profile and liver enzymes showed a similar trend to the studies by Khan and Korish. 11 –13 Investigators also measured serum and liver concentrations of malondialdehyde, catalase, and reduced glutathione as measures of oxidative stress on the liver. They found a statistically significant increase in malondialdehyde and decrease in catalase and reduced glutathione in rats fed a high-fat diet and a return to near-normal levels in those given camel milk.
Summary
The studies of camel milk in animals with artificially induced diabetes have shown a trend toward improved FBG and decreased insulin requirements, improvement in dyslipidemia, liver function, and diabetic nephropathy. These studies support the need for human trials of sound methodological quality in order to determine any potential clinical benefits of camel milk in diabetic patients.
Cancer
Two studies were identified that investigated the therapeutic effects of camel milk on angiogenesis and cell proliferation, as well as its potential antioxidant and antigenotoxic effects. Alhaider et al 15 used a randomized approach to determine the extent of vascularization of sponge implants between 3 groups of mice receiving increasing amounts of camel milk and controls (Table 1). Measured by the total amount of hemoglobin present in the tissue after 14 days, the study found higher amounts of camel milk administered (25 and 50 mg/kg/body weight daily) suppressed hemoglobin levels and therefore attenuated vascularization. Levels of vascular endothelial growth factor (VEGF) were also lower in the higher concentration camel milk groups.
The antigenotoxic and anticytotoxic effects of camel milk were investigated in a randomized study using seven groups of 10 mice administered different amounts and combinations of camel milk and cisplatin, as well as a control group 16 (Table 1). The objective of the study was to assess a potential protective role of camel milk against the genotoxic effects of cisplatin. Cisplatin was administered at subacute doses (0.5 mg/kg) in 3 groups and at acute doses (2.5 mg/kg) in 2 other groups. Camel milk was given orally at an amount of 33 mL/kg. Findings showed significant decreases in the frequency of micronucleated polychromatic erythrocytes and increases in the mitotic index in groups treated with camel milk, which are deemed to be markers of clastogenic and cytotoxic effects of cisplatin. However, the protective effect exhibited by camel milk did not maintain the same levels as control or camel milk alone groups.
Summary
The limited evidence from the animal studies identified suggests camel milk may have protective roles in both angiogenesis and reducing the cytotoxic and genotoxic effects of cisplatin therapy. While these findings are very preliminary, researchers should be encouraged to further explore potential oncology-related clinical applications of camel milk in human populations.
Colitis
It was anticipated that the antioxidant and anti-inflammatory properties of camel milk would provide benefit by decreasing the markers of colitis damage. Arab et al 17 randomized 32 adult rats with and without chemically induced colitis using 2,4,6-trinitrobenzene sulfonic acid (TNBS) to be treated with saline or camel milk (Table 1). The outcomes included anthropomorphic, blinded histological examination, and biochemical markers of colitis. For all results, TNBS-induced rats showed statistically significantly worse results when compared with control groups. Whereas TNBS plus camel milk showed similar outcomes to control in body weight, TNF-α, capsase-3, reduced glutathione, and total antioxidant capacity (TAC) levels but showed statistically worse results in colon weight/length, colon macroscopic damage, area of colonic lesions, myeloperoxidase activity, IL-10, malondialdehyde, and nitric oxide. Despite these results, TNBS plus camel milk showed statistically significantly favorable results when compared with TNBS.
Summary
The findings of this animal study are intriguing and warrant further study of camel milk in colitis populations.
Infections
Two studies were identified relating to the treatment of infections 18,19 (Table 1). It is hypothesized that the high lactoferrin, an antioxidant and immunomodulator, and immunoglobulin G (IgG) content of camel’s milk could protect the drinker from different infectious agents. In 2013, Cardoso et al 18 published the effects of camel milk saturated rat food compared with cow milk saturated rat food and controls on survival of mice infected with Salmonella enterica subspecies enterica. The authors found that all the controls groups worked appropriately. Two of the 10 mice treated with cow’s milk and 6 of the 10 mice treated with camel milk survived to 31 days. Camel milk–treated mice showed statistically increased survival compared with the cow’s milk group, but significantly decreased survival compared with mice not infected or those with heat killed Salmonella injections (100% survival). These results suggest improved survival outcome at 31 days in mice treated with camel milk.
A study by Maghraby et al 19 in 2005 looked at the effects of mature camel milk and camel colostrum on mice infected with Schistosoma mansoni cercariae. Mice were run in with treatment for 45 days prior to infection and followed up at 2, 4, and 6 weeks for the outcomes of total worm burden, IgG levels against soluble worm antigen, glutathione-S-transferase, aspartate transaminase, and alanine transaminase. Only numerical values were given in the results and no statistical tests reported. The authors state that there was no significant change in IgG levels at any time point between the groups without stating a P value.
Summary
Two animal model studies were identified that looked at clinically important outcomes of survival and surrogate outcomes. Limited data show that camel milk may have a protective role in Salmonella infections.
Other Indications
Two studies identified observed the effects of camel milk in rats with heavy metal induced anemia. 20,21 One study found that in cadmium-induced mice, camel milk treatment significantly improved anemia and markers of oxidative stress, except for no difference in glutathione levels. 20 The other study investigated aluminum chloride–induced kidney dysfunction, liver dysfunction, and markers of oxidative stress in each organ. 21 The camel milk–treated mice had significantly improved endpoints compared with the aluminum toxic group in all biochemical endpoints studied. No conclusions can be drawn between the camel milk–treated groups and controls in these studies as no statistical tests were reported.
It is anticipated that the antioxidant, anti-inflammatory, and antiapoptotic attributes of camel’s milk may help ameliorate damage caused by alcohol induced liver injury. One study examined these results. 22 Rats treated prophylactically (for duration of alcohol ingestion) and therapeutically (half of alcohol ingestion duration) with camel milk showed improved surrogate markers of liver injury when compared with alcohol-treated rats. Therapeutically and prophylactically treating rats resulted in similar outcomes in alanine transaminase, aspartate transaminase, alkaline phosphatase, liver weight to body weight ratio, TNF-α, and caspase-3 serum levels when compared with controls. Camel milk ingestion resulted in higher serum triglycerides in both groups compared with controls. Therapeutically treated rats had no significant difference in TAC levels whereas prophylactically treated rats had statistically lower TAC levels when compared with controls.
Summary
Animal models that investigated the beneficial effects of heavy metal and alcohol-induced toxicity found improved outcomes in many of the surrogate markers of oxidative stress and anemias. Clinical implications for humans are currently unknown.
Human Trials
Diabetes
A total of 8 studies assessed the effects of camel milk on the prevention and treatment of type 1 and 2 diabetes 23 –30 (Table 2). The effect of camel milk compared with cow milk in patients with type 2 diabetes was assessed in an randomized controlled trial conducted by Ejtahed et al. 23 After 2 months of therapy, they found a statistically significant increase in serum insulin concentration in the camel group and a nonsignificant trend toward increase in the cow milk group. There were no significant changes in any other parameters.
Mohamad et al 24 conducted another randomized controlled trial of 54 patients with type 1 diabetes randomized to receive usual care or usual care plus camel milk. The treatment groups were also compared to a group of 10 healthy patients. 24 After 16 weeks of therapy, they found that the group receiving camel milk had a significantly lower mean HbA1c, daily insulin dose, and FBG. However, this was at the expense of a significantly higher mean body mass index and c-peptide. There was no significant difference in lipid levels or renal function.
Three other studies were identified by Agrawal et al assessing various outcomes of camel milk supplementation in patients with type 1 diabetes through randomized controlled trial 25,26 and cohort designs. 27 They found a significant reduction in mean daily dose of insulin when camel milk was compared with usual care, 29 added to intensive insulin therapy for 12 months, 27 and for 24 months. 26 Two studies also found a significant decrease in mean blood glucose, 25,27 and 1 study found a reduction in HbA1c. 25 Comparatively, only 1 study found a significant reduction in mean blood glucose in the usual care group, and none showed a reduction in HbA1c or daily insulin dose. 25 Although camel milk appears to have an effect on lowering blood glucose and decreasing the dose of insulin required, there were no statistical analyses done between the 2 groups (only before and after therapy in each group); therefore, it is difficult to assess the true benefit over usual care.
Agrawal et al 28 also conducted 1 crossover trial in type 2 diabetic patients in order to assess the effect of camel milk versus cow milk on general diabetes monitoring parameters. The outcomes were measured after a 1-month run-in period (education on diet, exercise, and insulin administration), 3 months of initial therapy (camel milk in diabetic patients and cow milk in nondiabetic patients), 1 month washout, and 3 months of the other milk product. They found, in the diabetic patients, that camel milk significantly reduced FBG, HbA1c, blood glucose 2 hours after an oral glucose tolerance test, area under the curve (AUC) glucose, and AUC insulin. All of these markers returned to baseline after the patients were crossed over to cow milk. In the nondiabetic group, there were no statistically significant differences in any parameters after 3 months of cow milk consumption, but after the crossover period, where they received camel milk, there was a statistically significant decrease in HbA1c and triglycerides.
One before-and-after study was conducted by Agrawal et al 29 to assess the outcome of microalbuminuria before and after camel milk supplementation in patients with type 1 diabetes. It was noted that there was a significant decrease in daily insulin dose and microalbuminuria after 6 months of camel milk therapy. However, there was no control group in this study and all the patients were initially entered into a 1-month run-in period in which they received teaching on a standardized diet, exercise regimen, and insulin administration. Therefore, it is unclear if the positive effect they saw on microalbuminuria and daily insulin requirement was a result of the camel milk or of the new lifestyle modifications.
Finally, one study was identified that assessed the benefits of camel milk in preventing diabetes. 30 Agrawal and colleagues had previously identified that the Raica community of northern India, who consume camel milk, had low prevalence of diabetes. Subsequently, they conducted a cross-sectional study to compare the prevalence of impaired fasting glucose (IFG), impaired glucose tolerance (IGT), and overt diabetes mellitus (DM) in 1055 Raica and 1044 non-Raica Indian community consuming and not consuming camel milk. The results show a trend to lower rates of IFG, IGT, and DM independently in the Raica community and in communities consuming camel milk.
Summary
There are limited studies, with few patients, and of poor methodological quality looking at the effects of camel milk in humans with type 1 or type 2 diabetes. They consistently show a potential benefit of camel milk in reducing FPG, HbA1c, and daily insulin dose required. 23 –25,27 –30 However, this appears to come at the expense of an increase in body mass index. 27,30 One study also demonstrated a potential decrease in microalbuminuria, but this study had no control group and so conclusions are not firm. 25 Unlike the animal studies, human trials have not shown a benefit in improved lipid levels, and liver function was not assessed. All these observations are limited by a lack of statistical comparison between the camel milk group and control groups. Also, for purported randomized controlled trials, the randomization process, allocation concealment, baseline characteristics, and blinding were either not documented 27,28 or not adequately reported. 29,30 Despite the potential benefits of increasing insulin levels and decreasing serum glucose, camel milk should not be recommended as a therapeutic agent for diabetic patients. However, there does not appear to be major safety risks associated with normal dietary use based on these studies. 23 –30
Autism
Because of its unique composition, camel milk is being investigated for potential effects of oxidative stress along with therapeutic effects in patients with autism spectrum disorder (ASD). Two studies identified in the search sought to evaluate the effect of camel milk consumption on the clinical outcomes of autism and on oxidative stress markers. Al-Ayadhi et al 31 used a randomized, double blind, placebo-controlled approach to determine the effects of raw and boiled camel milk on the Childhood Autism Rating Scale (CARS) and oxidative stress biomarkers including glutathione, superoxide dismutase, and myeloperoxidase (Table 2). Study participants aged 2 to 12 years were randomized into 3 different groups (boiled camel milk, raw camel milk, cow milk [placebo]) and received 500 mL of their respective milk product daily for 14 days. Participants were assessed prior to the introduction of milk into their diet and after receiving the milk product for 14 days. Both raw and boiled camel milk groups had statistically significant reductions in the CARS and oxidative stress markers. In the placebo group, the cow milk intervention did not significantly reduce any measured outcomes. The authors stated that participants were not allowed to add or remove any interventions such as diet plans, supplements, or pharmacotherapies throughout the study period.
In a second study, Bashir et al 32 used a randomized, double blind, placebo-controlled design to assess the effects of camel milk on the CARS assessment and thymus and activation-regulated chemokine (TARC) serum levels (Table 2). Study participants aged 2 to 12 years were randomized to receive boiled or raw camel milk or cow milk 500 mL for 2 weeks. Prior to randomization a baseline CARS evaluation was performed by a child psychiatrist and blood samples were collected at an autism research and treatment center. Findings from this study showed that raw camel milk was associated with significant improvements in the CARS score compared with baseline, while both raw and boiled camel milk were associated with significant decreases in serum levels of TARC. Cow milk was not associated with any significant changes in either measurement. Camel milk in both preparations was generally well tolerated with only irritability and stomach discomfort noted, although the true numbers of children experiencing these adverse effects were not described.
Two recent reports have also been published highlighting the anecdotal benefits of camel milk in children and youth with ASD. Shabo and Yagil 33 reported that in a population of 4- to 21-year-olds with ASD, the introduction of camel milk into the regular diet was associated with a subjective improvement in behavior and functioning ability. A case report by Adams 34 in 2013 presented a mother’s experience with using camel milk in her autistic son stating improvement after a single ingestion of camel milk through behavioral changes. These beneficial effects were sustained throughout the period of camel milk use but dissipated on cessation of camel milk use.
Summary
Available studies have demonstrated significant improvements in the clinical measurement of ASD through the CARS assessment. 31,32 However, as this measurement was only completed once after the 2-week period, it is unclear whether the beneficial effects were maintained. Furthermore, it remains unclear from the current studies as to whether or not the numerical improvements identified in the CARS assessment with the use of camel milk correlated with clinically relevant improvements in patients with autism. Future studies should attempt to identify the potential long-term benefits of camel milk in ASD. Based on current evidence, camel milk should not replace current available treatment modalities.
Limitations
The current evidence is limited by the lack of well-designed trials to decrease the influence of biases. Of the animal models studied, most only reported on surrogate outcomes and many used inappropriate statistical tests increasing the risk of multiplicity errors. Of the human studies, there were incomplete efficacy and safety outcomes reported.
Footnotes
Author Contributions
All authors contributed to the design, execution, analysis, and write-up of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval was not required for this review.
